Biophysical Cueing and Vascular Endothelial Cell Behavior
Abstract
:1. Introduction
2. Vascular Endothelial Cells
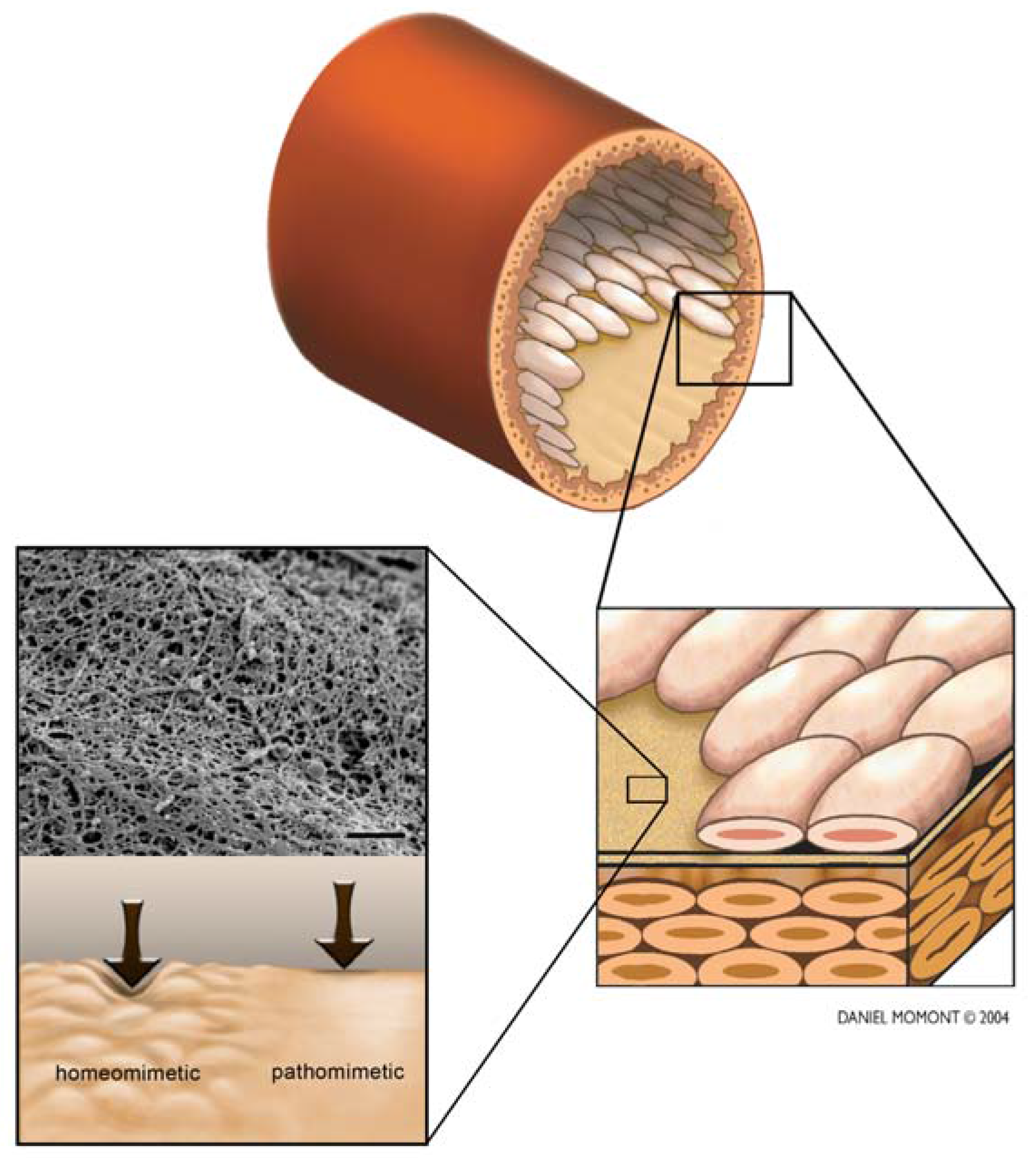
3. Basmenet Membrane
4. Biomaterials
5. Topography
6. Compliance
7. Conclusions
Acknowledgements
References and Notes
- Derham, C.; Yow, H.; Ingram, J.; Fisher, J.; Ingham, E.; Korrosis, S.A.; Homer-Vanniasinkam, S. Tissue engineering small-diameter vascular grafts: Preparation of a biocompatible porcine ureteric scaffold. Tissue Eng. A 2008, 14, 1871–1882. [Google Scholar]
- Sun, G.; Gerecht, S. Vascular regeneration: Engineering the stem cell microenvironment. Regen. Med. 2009, 4, 435–447. [Google Scholar]
- Lu, J.; Rao, M.P.; MacDonald, N.C.; Khang, D.; Webster, T.J. Improved endothelial cell adhesion and proliferation on patterned titanium surfaces with rationally designed, micrometer to nanometer features. Acta Biomater. 2008, 4, 192–201. [Google Scholar]
- Mackay, J.; Mensah, G.A. The Atlas of Heart Disease and Stroke; World Health Organization: Geneva, Switzerland, 2004. [Google Scholar]
- Shaw, L.J.; Bugiardini, R.; Merz, C.N.B. Women and ischemic heart disease. J. Am. Coll. Cardiol. 2009, 54, 1561–1575. [Google Scholar]
- Muthurangu, V.; Atkinson, D.; Sermesant, M.; Miquel, M.E.; Hegde, S.; Johnson, R.; Andriantsimiavona, R.; Taylor, A.M.; Baker, E.; Tulloh, R.; Hill, D.; Razavi, R.S. Measurement of total pulmonary arterial compliance using invasive pressure monitoring and MR flow quantification during MR-guided cardiac catheterization. Am. J. Physiol. Heart. Circ. Physiol. 2005, 289, H1301–H1306. [Google Scholar]
- Libby, P.; Ridker, P.M.; Maseri, A. Inflammation and atherosclerosis. Circulation 2002, 105, 1135–1143. [Google Scholar]
- Hlatky, M.A.; Boothroyd, D.B.; Bravata, D.M.; Boersma, E.; Booth, J.; Brooks, M.M.; Carrie, D.; CLayton, T.C.; Danchin, N.; Flather, M.; Hamm, C.W.; Hueb, W.A.; Kahler, J.; Kelsey, S.F.; King, S.B.; Kosinski, A.S.; Lopes, N.; McDonald, K.M.; Rodriguez, A.; Serruys, P.; Sigwart, U.; Stables, R.H.; Owens, D.K.; Pocock, S.J. Coronary bypass surgery compared with percutaneous coronary interventions for multivessel disease: A collaborative analysis of individual patient data from ten randomised trials. Lancet 2009, 373, 1190–1197. [Google Scholar]
- Rosamond, W.; Flegal, K.; Friday, G.; Furie, K.; Go, A.; Greenlund, K.; Haase, N.; Ho, M.; Howard, V.; Kissela, B.; Kittner, S.; Lloyd-Jones, D.; McDermott, M.; Meigs, J.; Moy, C.; Nichol, G.; O'Donnell, C.J.; Roger, V.; Rumsfeld, J.; Sorlie, P.; Steinberger, J.; Thom, T.; Wasserthiel-Smoller, S.; Hong, Y.; Committee, f.t.A.H.A.S. Stroke statistics subcommittee, heart disease and stroke statistics−2007 update: A report from the american heart association statistics committee and stroke statistics subcommittee. Circulation 2007, 115, e69–e171. [Google Scholar]
- Campbell, G.R.; Campbell, J.H. Development of tissue engineered vascular grafts. Curr. Pharm. Biotechnol. 2007, 8, 43–50. [Google Scholar]
- Piccone, V. Modern Vascular Grafts; McGraw Hill: New York, NY, USA, 1987. [Google Scholar]
- Darling, R.C.; Linton, R.R. Durability of femoropopliteal reconstructions. Endarterectomy versus vein bypass grafts. Am. J. Surg. 1972, 123, 472. [Google Scholar]
- Michaels, A.D.; Chatteriee, K. Cardiology patient pages. Angioplasty versus bypass surgery for coronary artery disease. Circulation 2002, 106, e187. [Google Scholar]
- L'Heureux, N.; Dusserre, N.; Konig, G.; Victor, B.; Keire, P.; Wight, T.N.; Chronos, N.A.F.; Kyles, A.E.; Gregory, C.R.; Hoyt, G.; Robbins, R.C.; McAllister, T.N. Human tissue-engineered blood vessels for adult arterial revascularization. Nat. Med. 2006, 12, 361–365. [Google Scholar]
- Faries, P.L.; LoGerfo, F.W.; Arora, S.; Hook, S.; Pulling, M.C.; Akbari, C.M.; Campbell, D.R.; Pomposelli, F.B. A comparative study of alternative conduits for lower extremity revascularization: All-autogenous conduit versus prosthetic grafts. J. Vasc. Surg. 2000, 32, 1080–1090. [Google Scholar]
- Kirklin, J.W.; Barratt-Bowers, B.G. Ventricular septal defect and pulmonary stenosis or atresia. In Cardiac Surgery; Churchill Livingstone: New York, NY, USA, 1993; pp. 861–1612. [Google Scholar]
- Pomposelli, J.F.B.; Arora, S.; Gibbons, G.W.; Frykberg, R.; Smakowski, P.; Campbell, D.R.; Freeman, D.V.; LoGerfo, F.W. Lower extremity arterial reconstruction in the very elderly: Successful outcome preserves not only the limb but also residential status and ambulatory function. J. Vasc. Surg. 1998, 28, 215–225. [Google Scholar]
- Yang, J.; Motlagh, D.; Webb, A.R.; Ameer, G.A. Novel biphasic elastomeric scaffold for small-diameter blood vessel tissue engineering. Tiss. Eng. 2005, 11, 1876–1886. [Google Scholar]
- Kapadia, M.R.; Popowich, D.A.; Kibbe, M.R. Modified prosthetic vascular conduits. Circulation 2008, 117, 1873–1882. [Google Scholar]
- Peterson, L.H.; Jensen, R.E.; Parnell, J. Mechanical properties of arteries in vivo. Circ. Res. 1960, 8, 622–639. [Google Scholar]
- Alvarado, J.; Murphy, C.; Juster, R. Age-related-changes in the basement-membrane of the human corneal epithelium. Invest. Ophthalmol. Vis. Sci. 1983, 24, 1015–1028. [Google Scholar]
- Candiello, J.; Balasubramani, M.; Schreiber, E.M.; Cole, G.J.; Mayer, U.; Halfter, W.; Lin, H. Biomechanical properties of native basement membranes. FEBS. J. 2007, 274, 2897–2908. [Google Scholar]
- Jacot, J.G.; Dianis, S.; Schnall, J.; Wong, J.Y. A simple microindentation technique for mapping the microscale compliance of soft hydrated materials and tissues. J. Biomed. Mater. Res. A 2006, 79A, 485–494. [Google Scholar]
- Bayless, K.J.; Davis, G.E. Microtubule depolymerization rapidly collapses capillary tube networks in vitro and angiogenic vessels in vivo through the small GTPase rho. J. Biol. Chem. 2004, 279, 11686–11695. [Google Scholar]
- Hayashi, K.; Naiki, T. Adaptation and remodeling of the vascular wall; Biochemical response to hypertension. J. Mech. Beh. Biomed. Mater. 2008, 2, 3–19. [Google Scholar]
- Kasaj, A.; Reichert, C.; Gotz, H.; Rohrig, B.; Smeets, R.; Willerhausen, B. In vitro evaluation of various bioasbsorbable and nonresorbable barrier membranes for guided tissue regeneration. Head Face Med. 2008, 4. [Google Scholar] [CrossRef]
- Abrams, G.A.; Goodman, S.L.; Nealey, P.F.; Franco, M.; Murphy, C.J. Nanoscale topography of the basement membrane underlying the corneal epithelium of the rhesus macaque. Cell Tissue Res. 2000, 299, 39–46. [Google Scholar]
- Liliensiek, S.J.; Nealey, P.; Murphy, C.J. Characterization of endothelial basement membrane nanotopography in rhesus macaque as a guide for vessel tissue engineering. Tiss. Eng. A 2009, 15, 2643–2651. [Google Scholar]
- Ausprunk, D.; Folkman, J. Migration and proliferation of endothelial cells in preformed and newly formed blood vessels during angiogenesis. Microvasc. Res. 1977, 14, 53–65. [Google Scholar]
- Ingber, D.E. Mechanical signaling and the cellular response to extracellular matrix in angiogenesis and cardiovascular physiology. Circ. Res. 2002, 91, 877–887. [Google Scholar]
- Deroanne, C.F.; Lapiere, C.M.; Nusgens, B.V. In vitro tubulogenesis of endothelial cells by relaxation of the coupling extracellular matrix-cytoskeleton. Cardiovasc. Res. 2001, 49, 647–658. [Google Scholar]
- Engler, A.; Bacakova, L.; Newman, C.; Hategan, A.; Griffin, M.; Discher, D. Substrate compliance versus ligand density in cell on gel responses. Biophys. J. 2004, 86, 617–628. [Google Scholar]
- Engler, A.J.; Griffin, M.A.; Sen, S.; Bonnemann, C.G.; Sweeney, H.L.; Discher, D.E. Myotubes differentiate optimally on substrates with tissue-like stiffness: Pathological implications for soft or stiff microenvironments. J. Cell Biol. 2004, 166, 877–887. [Google Scholar]
- Wong, G.A.; Tang, V.; El-Sabeawy, F.; Weiss, R.H. BMP-2 inhibits proliferation of human aortic smooth muscle cells via p21Cip1/Waf1. Am. J. Physiol. Endocrinol. Metab. 2003, 284, E972–E979. [Google Scholar]
- Gasiorowski, J.Z.; Russell, P.; Liliensiek, S.J.; Stephan, D.A.; Nealey, P.F.; Murphy, C.J. Comparison of Gene Expression Differences Between Endothelial Cells Grown on Flat and Nanopatterned Surfaces; In The American Society for Cell Biology's 47th Annual Meeting, Washington, DC, USA, December 2007.
- Ferreira, L.S.; Gerecht, S.; Shieh, H.F.; Watson, N.; Rupnick, M.A.; Dallabrida, S.M.; Vunjak-Novakovic, G.; Langer, R. Vascular progenitor cells isolated from human embryonic stem cells give rise to endothelial and smooth muscle like cells and form vascular networks in vivo. Circ. Res. 2007, 101, 286–294. [Google Scholar]
- Cliff, W.J. Blood Vessels; Cambridge: New York, NY, USA, 1976. [Google Scholar]
- Rhodin, J.A.G. Blood Vessel. In An Atlas of Ultrastructure; W B Suanders Co: Philedelphia, PA, USA, 1963; pp. 50–53. [Google Scholar]
- Simionescu, M. Implications of early structural-functional changes in the endothelium for vascular disease. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 266–274. [Google Scholar]
- Ingram, D.A.; Mead, L.E.; Tanaka, H.; Meade, V.; Fenoglio, A.; Mortell, K.; Pollok, K.; Ferkowicz, M.J.; Gilley, D.; Yoder, M.C. Identification of a novel hierarchy of endothelial progenitor cells using human peripheral and umbilical cord blood. Blood 2004, 104, 2752–2760. [Google Scholar]
- Yoder, M.C. Hemangioblasts: Of mice and men. Blood 2007, 109, 2667–2668. [Google Scholar]
- Mathur, A.B.; Collinsworth, A.M.; Reichert, W.M.; Kraus, W.E.; Truskey, G.A. Endothelial, cardiac muscle and skeletal muscle exhibit different viscous and elastic properties as determined by atomic force microscopy. J. Biomech. 2001, 34, 1545–1553. [Google Scholar]
- Cuerrier, C.M.; Gagner, A.; Lebel, R.; Gobeil, F., Jr.; Grandbois, M. Effect of thrombin and bradykinin on endothelial cell mechanical properties monitored through membrane deformation. J. Mol. Recognit. 2009, 22, 389–396. [Google Scholar]
- Hoang, M.V.; Whelan, M.C.; Senger, D.R. Rho activity critically and selectively regulates endothelial cell organization during angiogenesis. Proc. Natl. Acad. Sci. USA 2004, 101, 1874–1879. [Google Scholar]
- Silva, C.C.D.; Lamerant-Fayel, N.; Paprocka, M.; Mitterrand, M.; Gosset, D.; Dus, D.; Kieda, C. Selective human endothelial cell activation by chemikines as a guide to cell homing. Immunology 2008, 126, 394–404. [Google Scholar]
- Ribatti, D. The discovery of endothelial progenitor cells: An historical review. Leuk. Res. 2007, 31, 439–444. [Google Scholar]
- Kozulin, P.; Natoli, R.; O'Brian, K.M.B.; Madigan, M.C.; Provis, J.M. Differential expression of anti-angiogenic factors and guidance genes in teh developing macula. Mol. Vis. 2009, 15, 45–59. [Google Scholar]
- Chung, E.-S.; Saban, D.R.; Chauhan, S.K.; Dana, R. Regulation of blood vessel versus lymphatic vessel growth in the cornea. Invest. Ophthalmol. Vis. Sci. 2009, 50, 1613–1618. [Google Scholar]
- Ling, S.; Qi, C.; Li, W.; Xu, J.; Kuang, W. The expression of vascular endothelial growth factor c in transplanted corneas. Curr. Eye Res. 2009, 34, 553–561. [Google Scholar]
- Fan, Y.; Gong, Y.; Ghosh, P.K.; Graham, L.M.; Fox, P.L. Spatial coordination of actin polymerization and ILK-Akt2 activity during endothelial cell migration. Dev. Cell 2009, 16, 661–674. [Google Scholar]
- Skarie, J.M.; Link, B.A. FoxC1 is essential for vascular basement membrane integrity and hyaloid vessel morphogenesis. Invest. Ophthalmol. Vis. Sci. 2009. [Google Scholar] [CrossRef]
- Hida, K.; Hida, Y.; Shindoh, M. Understanding tumor endothelial cell abnormalities to develop ideal anti-angiogenic therapies. Cancer Sci. 2008, 99, 459–466. [Google Scholar]
- Mendonça, R.J.; Maurício, V.B.; Teixeira, L.d.B.; Lachat, J.J.; Coutinho-Netto, J. Increased vascular permeability, angiogenesis and wound healing induced by the serum of natural latex of the rubber tree Hevea brasiliensis. Phytother. Res. 2009. [Google Scholar] [CrossRef]
- Rae, M.; Mohamad, A.; Price, D.; Hadoke, P.W.F.; Walker, B.R.; Mason, J.I.; Hillier, S.G.; Critchley, H.O.D. Cortisol inactivation by 11{beta}-Hydroxysteroid dehydrogenase-2 may enhance endometrial angiogenesis via reduced thrombospondin-1 in heavy menstruation. J. Clin. Endocrinol. Metab. 2009, 94, 1443–1450. [Google Scholar]
- Kniazeva, E.; Putnam, A.J. Endothelial cell traction and ECM density influence both capillary morphogenesis and maintenance in 3-D. Am. J. Physiol. Cell Physiol. 2009, 297, C179–C187. [Google Scholar]
- Ohashi, T.; Ishii, Y.; Ishikawa, Y.; Matsumoto, T.; Sato, M. Experimental and numerical analyses of local mechanical properties measured by atomic force microscopy for sheared endothelial cells. Biomed. Mat. Eng. 2002, 12, 319–327. [Google Scholar]
- Lu, L.; Oswald, S.J.; Ngu, H.; Yin, F.C.P. Mechanical properties of actin stress fibers in living cells. Biophys. J. 2008, 95, 6060–6071. [Google Scholar]
- Satcher, R.; Jr, C.F.D.; Hartwig, J.H. Mechanical remodeling of the endothelial surface and actin cytoskeleton induced by fluid flow. Microcirculation 1997, 4, 439–453. [Google Scholar]
- Hochmuth, R.M. Micropipette aspiration of living cells. J. Biomech. 2000, 33, 15–22. [Google Scholar]
- Hanjaya-Putra, D.; Gerecht, S. Vascular engineering using human embryonic stem cells. Biotechnol. Prog. 2009, 25, 2–9. [Google Scholar]
- Alamo, J.C.d.; Norwich, G.N.; Li, Y.-s.J.; Lasheras, J.C.; Chien, S. Anisotropic rheology and directional mechanotransduction in vascular endotheilial cells. Proc. Natl. Acad. Sci. USA 2008, 105, 15411–15416. [Google Scholar]
- Pesen, D.; Hoh, J.H. Micromechanical architecture of the endothelial cell cortex. Biophys. J. 2005, 88, 670–679. [Google Scholar]
- Mathur, A.B.; Truskey, G.A.; Reichert, W.M. Atomic force and total internal reflection fluorescence microscopy for the study of force transmission in endothelial cells. Biophys. J. 2000, 78, 1725–1735. [Google Scholar]
- Oberleithner, H.; Riethmuller, C.; Ludwig, T.; Shahin, V.; Stock, C.; Schwab, A.; Hausberg, M.; Kusche, K.; Schillers, H. Differential action of steroid hormones on human endothelium. J. Cell Sci. 2006, 119, 1926–1932. [Google Scholar]
- Chouinard, J.A.; Grenier, G.; Khalil, A.; Vermette, P. Oxidized-LDL induce morphological changes and increas stiffness of endothelial cells. Exp. Cell Res. 2008, 314, 3007–3016. [Google Scholar]
- Sinionescu, M.; Antohe, F. Functional Unstructure of the Vascular Endothelium: Changes in Various Pathologies. In Handbook of Experimental Pharmacology; Springer-Verlag: Berlin Heidelberg, Germany, 2006; Volume 176, pp. 41–69. [Google Scholar]
- Campbell, G.R.; Chamleycampbell, J.H. Smooth-muscle phenotypic modulation—Role in atherogenesis. Med. Hypotheses 1981, 7, 729–735. [Google Scholar]
- Kataoka, N.; Iwaki, K.; Hashimoto, K.; Mochizuki, S.; Ogasawara, Y.; Sato, M.; Tsujioka, K.; Kajiya, F. Measurements of endothelial cell-to-cell and cell-to-substrate gaps and micromechanical propeties of endothelial cells during moncyte adhesion. Proc. Natl. Acad. Sci. USA 2002, 99, 15638–15643. [Google Scholar]
- Hayashi, K. Experimental approaches on measuring the mechanical properties and constitutive laws of arterial walls. J. Biomed. Eng. 1993, 115, 481–488. [Google Scholar]
- Hillebrand, U.; Hausber, M.; Stock, C.; Shahin, V.; Nikova, D.; Riethmuller, C.; Kliche, K.; Ludwig, T.; Schillers, H.; Schneider, S.W.; Oberleithmer, H. 17B-estradiol increases volume, apical surface and elasticity of human endothelium mediated by Na+/H+ exchange. Cardiovasc. Res. 2006, 69, 916–924. [Google Scholar]
- Hirschi, K.K.; Ingram, D.A.; Yoder, M.C. Assessing identity, phenotype, and fate of endothelial progenitor cells. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 1584–1595. [Google Scholar]
- Xu, Q. Stem cell and transplant arteriosclerosis. Circ. Res. 2008, 102, 1011–1024. [Google Scholar]
- Dunn, P.F.; Newman, K.D.; Jones, M.; Yamada, I.; Shayani, V.; Virmani, R.; Dichek, D.A. Seeding of vascular grafts with genetically modified endothelial cells : Secretion of recombinant TPA results in decreased seeded cell retention in vitro and in vivo. Circulation 1996, 93, 1439–1446. [Google Scholar]
- Discher, D.E.; Janmey, P.; Wang, Y.L. Tissue cells feel and respond to the stiffness of their substrate. Science 2005, 310, 1139–1143. [Google Scholar]
- Last, J.A.; Liliensiek, S.J.; Nealey, P.F.; Murphy, C.J. Determining the mechanical properties of human corneal basement membranes with atomic force microscopy. J. Struct. Biol. 2009, 167, 19–24. [Google Scholar]
- Abrams, G.A.; Teixeira, A.I.; Nealey, P.F.; Murphy, C.J. Effects of Substratum Topography on Cell Behavior. In Biomimetic Materials and Design: Biointerfacial Strategies, Tissue Engineering and Targeted Drug Delivery; Dillow, A.K., Lowman, A.M., Eds.; CRC Press: New York, NY, USA, 2002; pp. 91–137. [Google Scholar]
- LeBleu, V.S.; MacDonald, B.; Kalluri, R. Structure and function of basement membranes. Exp. Biol. Med. 2007, 232, 1121–1129. [Google Scholar]
- Brody, S.; Anilkumar, T.; Liliensiek, S.; Last, J.A.; Murphy, C.J.; Pandit, A. Characterizing nanoscale topography of the aortic heart valve basement membrane for tissue engineering heart valve scaffold design. Tissue Eng. 2006, 12, 413–421. [Google Scholar]
- Timpl, R. Macromolecular organization of basement membranes. Curr. Opin. Cell Biol. 1996, 8, 618–624. [Google Scholar]
- Kolega, J.; Manabe, M.; Sun, T.-T. Basement membrane heterogeneity and variation in corneal epithelial differentiation. Differentiation 1989, 42, 54–63. [Google Scholar]
- Ekblom, P.; Timpl, R. Cell-to-cell contact and extracellular matrix A multifaceted approach emerging. Curr. Opin. Cell Biol. 1996, 8, 599–601. [Google Scholar]
- Merker, H.-J. Morphology of the basement membrane. Microsc. Res. Tech. 1994, 28, 95–124. [Google Scholar]
- Yurchenco, P.D.; O'Rear, J. Supramolecular organization of basment membranes. In Molecular and Cellular Aspects of Basment Membranes; Rohrbach, D.H., Timpl, R., Eds.; Academic Press: New York, NY, USA, 1993; pp. 19–47. [Google Scholar]
- Simionescu, M.; Antohe, F. Functional Unstructure of the Vascular Endothelium: Changes in Various Pathologies. In Handbook of Experimental Pharmacology; Springer-Verlag: Berlin Heidelberg, Germany, 2006; Volume 176, pp. 41–69. [Google Scholar]
- Rhodin, J.A.G. Cardiovascular System. In Histology: A Text and Atlas; Oxford University Press: New York, NY, USA, 1977; pp. 331–370. [Google Scholar]
- Saux, O.L.; Teeters, K.; Miyasato, S.; Choi, J.; Nakamatsu, G.; Richardson, J.A.; Starcher, B.; Davis, E.C.; Tam, E.K.; Saux, C.J.-L. The role of caveolin-1 in pulmonary matrix remodeling and mechanical properties. Am. J. Physiol. Lung Cell Mol. Physiol. 2008, 295, L1007–L1017. [Google Scholar]
- Godier, A.F.; Marolt, D.; Gerecht, S.; Tajnsek, U.; Martens, T.P.; Vunjak-Novakovic, G. Engineered microenvironments for human stem cells. Birth Defects Res. C, Embryo Today 2008, 84, 335–347. [Google Scholar]
- Figallo, E.; Cannizzaro, C.; Gerecht, S.; Burdick, J.A.; Langer, R.; Elvassore, N.; Vunjak-Novakovic, G. Micro-bioreactor array for controlling cellular microenvironments. Lab Chip 2007, 7, 710–719. [Google Scholar]
- Chan, F.L.; Inoue, S. Lamina lucida of basement membrane: An artefact. Microsc. Res. Tech. 1994, 28, 48–59. [Google Scholar]
- Ghadially, F.N. Cell Membrane and Coat. In Ultrastructural Pathology of the Cell and Matrix; Butterworth-Heinemann: Boston, MA, USA, 1997; Volume 2, pp. 1124–1158. [Google Scholar]
- Tanner, G.A.; Evan, A.P.; Williams, J.C. Reply to miner. Am. J. Renal Physiol. 2009, 297, F551. [Google Scholar]
- Mobjerg, N.; Jespersen, A.; Wilkinson, M. Morphology of the kidney in the west african caecilian, geotrypetes seraphini (Amphibia, Gymnophiona, Caeciliidae). J. Morphol. 2004, 262, 583–607. [Google Scholar]
- Ridley, A.J.; Hall, A. The small GTP-binding protein rho regulates the assembly of focal adhesions and actin stress fibers in response to growth factors. Cell 1992, 70, 389–399. [Google Scholar]
- Bentley, E.; Murphy, C.J. Topical therapeutic agents that modulate corneal wound healing. Vet Clin. North Am. Small Anim. Pract. 2004, 34, 623–638. [Google Scholar]
- Davis, G.E.; Senger, D.R. Endothelial extracellular matrix: Biosynthesis, remodeling, and functions during vascular morphogenesis and neovessel stabilization. Circ. Res. 2005, 97, 1093–1107. [Google Scholar]
- Abrams, G.A.; Schaus, S.S.; Goodman, S.L.; Nealey, P.F.; Murphy, C.J. Nanoscale topography of the corneal epithelial basment membrane and descemet's membrane of the human. Cornea 2000, 19, 57–64. [Google Scholar]
- Ebenstein, D.M.; Pruitt, L.A. Nanoindentation of soft hydrated materials of application to vascular tissues. J. Biomed. Mater. Res. 2004, 69A, 222–232. [Google Scholar]
- Oie, T.; Murayama, Y.; Fukuda, T.; Nagai, C.; Omata, S.; Kanda, K.; Yaku, H.; Nakayama, Y. Local elasticity imaging of vascular tissues using a tactile mapping system. J. Artif. Organs 2009, 12, 40–46. [Google Scholar]
- Lundkvist, A.; Lilleodden, E.; Siekhaus, W.; Kinney, J.; Pruitt, L.; Balooch, M. Viscoelastic Proerties of Healthy Human Artery Measured in Saline Solution by Afm-Based Indentation Technique. In Thin Films: Stresses and Mechnical Properties VI; Gerberich, W.W., Gao, H., Sundgren, J.E., Baker, S.P., Eds.; Materials Research Society: Pittsburg, PA, USA, 1997; Volume 436, pp. 353–357. [Google Scholar]
- Engler, A.J.; Richert, L.; Wong, J.Y.; Picart, C.; Discher, D.E. Surface probe measurements of the elasticity of sectioned tissue, thin gels and polyelectrolyte multilayer films: Correlations between substrate stiffness and cell adhesion. Surf. Sci. 2004, 570, 142–154. [Google Scholar]
- Form, D.M.; Pratt, B.M.; Madri, J.A. Endothelial cell proliferation duirng angiogenesis. Lab Invest. 1986, 55, 521–530. [Google Scholar]
- Iivanainen, E.; Kähäri, V.-M.; Heino, J.; Elenius, K. Endothelial cell-Matrix interactions. Microsc. Res. Tech. 2003, 60, 13–22. [Google Scholar]
- Soofi, S.S.; Last, J.A.; Liliensiek, S.J.; Nealey, P.F.; Murphy, C.J. The elastic modulus of Matrigel(TM) as determined by atomic force microscopy. J. Struct. Biol. 2009, 167, 216–219. [Google Scholar]
- Zhang, X.; Wang, X.; Keshav, V.; Wang, X.; Johanas, J.T.; Leisk, G.G.; Kaplan, D.L. Dynamic culture conditions to generate silk-based tissue-engineered vascular grafts. Biomaterials 2009, 30, 3213–3223. [Google Scholar]
- Tsai, S.-H.; Liu, Y.-W.; Tang, W.-C.; Zhou, Z.-W.; Hwang, C.-Y.; Hwang, G.-Y.; Ou, B.-R.; Hu, C.-P.; Yang, V.C.; Chen, J.-K. Characterization of porcine arterial endothelial cells cultured on amniotic membrane, a potential matrix for vascular tissue engineering. Biochem. Biophys. Res. Commun. 2007, 357, 984–990. [Google Scholar]
- Plant, A.L.; Bhadriraju, K.; Spurlin, T.A.; Elliot, J.T. Cell response to matix mechanics: Focus on collagen. Bioch. Biophy. Acta 2009, 1793, 893–902. [Google Scholar]
- Liu, Y.; Vrana, N.E.; Cahill, P.A.; McGuinness, G.B. Physically crosslinked composite hydrogels of PVA with natural macromolecules: Structure, mechanical properties, and endothelial cell compatability. J. Biomed. Mater. Res. B 2009, 90, 492–502. [Google Scholar]
- Abrams, G.A.; Bentley, E.; Nealey, P.F.; Murphy, C.J. Electron microscopy of the canine corneal basement membranes. Cells Tissues Organs 2002, 170, 251–257. [Google Scholar]
- Abrams, G.A.; Murphy, C.J.; Wang, Z.Y.; Nealey, P.F.; Bjorling, D.E. Ultrastructural basement membrane topography of the bladder epithelium. Urol. Res. 2003, 31, 341–346. [Google Scholar]
- Biela, S.A.; Su, Y.; Spatz, J.P.; Kemkemer, R. Different sensitivity of human endothelial cells, smooth muscle cells and fibroblasts to topography in the nano-micro range. Acta Biomater. 2009, 5, 2460–2466. [Google Scholar]
- Uttayarat, P.; Chen, M.; Li, M.; Allen, F.D.; Composto, R.J.; Lelkes, P.I. Microtopography and flow modulate the direction of endothelial cell migration. Am. J. Physiol. Heart Circ. Physiol. 2008, 294, H1027–H1035. [Google Scholar]
- Barbucci, R.; Lamponi, S.; Magnani, A.; Pasqui, D. Micropatterned surfaces for the control of endothelial cell behaviour. Biomol. Eng. 2002, 19, 161–170. [Google Scholar]
- Lin, X.; Helmke, B.P. Micropatterned structural control suppresses mechanotaxis of endothelial cells. Biphys. J. 2008, 95, 3066–3078. [Google Scholar]
- Flemming, R.G.; Murphy, C.J.; Abrams, G.A.; Goodman, S.L.; Nealey, P.F. Effects of synthetic micro- and nano-structured surfaces on cell behavior. Biomaterials 1999, 20, 573–588. [Google Scholar]
- Karuri, N.W.; Porri, T.J.; Albrecht, R.M.; Murphy, C.J.; Nealey, P.F. Nano- and microscale holes modulate cell-substrate adhesion, cytoskeletal organization, and -beta1 integrin localization in SV40 human corneal epithelial cells. IEEE Trans. Nanobioscience. 2006, 5, 273–280. [Google Scholar]
- Teixeira, A.I.; Nealey, P.F.; Murphy, C.J. Responses of human keratocytes to micro- and nanostructured substrates. J. Biomed. Mater. Res. A 2004, 71, 369–376. [Google Scholar]
- Haydarkhan-Hagvall, S.; Choi, C.-H.; Dunn, H.; Heydarkhan, S.; Schenke-Layland, K.; Maclellan, W.R.; Beygui, R.E. Influence of systemically varied nano-scale topography on cell morphology and adhesion. Cell Commun. Adhes. 2007, 14, 181–194. [Google Scholar]
- Bettinger, C.J.; Langer, R.; Borenstein, J.T. Engineering substrate topography at the micro- and nanoscale to control cell function. Angew. Chem. Int. Ed. 2009, 48, 5406–5415. [Google Scholar]
- Cavalcanti-Adam, E.A.; Aydin, D.; Hischfield-Warneken, V.C.; Spatz, J.P. Cell adhesion and response to synthetic nanopatterned environments by steering receptor clustering and spatial location. HFSP J. 2008, 2, 276–295. [Google Scholar]
- Chai, C.; Leong, K.W. Biomaterials approach to expang and direct differentiation of stem cells. Mol. Ther. 2007, 15, 467–480. [Google Scholar]
- Yim, E.K.; Pang, S.W.; Leong, K.W. Synthetic nanostructures inducing differentiation of human mesenchymal stem cell into neuronal lineage. Exp. Cell Res. 2007, 313, 1820–1829. [Google Scholar]
- Foley, J.D.; Grunwald, E.W.; Nealey, P.F.; Murphy, C.J. Cooperative modulation of neuritogenesis by PC12 cells by topography and nerve growth factor. Biomaterials 2005, 26, 3639–3644. [Google Scholar]
- Tajima, S.; Chu, J.S.F.; Li, S.; Komvopoulos, K. Differential regulation of endothelial cell adhesion, spreading, and cytoskeleton on low-density polyethylene by nanotopography and surface chemistry modification induced by argon plasma treatment. J. Biomed. Mater. Res. A 2008, 84A, 828–836. [Google Scholar]
- Samaroo, H.D.; Lu, J.; Webster, T.J. Enhanced endothelial cell density on NiTi surfaces with sub-micron to nanometer roughness. Int. J. Nanomed. 2008, 3, 75–82. [Google Scholar]
- Khang, D.; Lu, J.; Yao, C.; Haberstroh, K.M.; Webster, T.J. The role of nanometer and sub-micron surface features on vascular and bone cell adhesion on titanium. Biomaterials 2008, 29, 970–983. [Google Scholar]
- Chung, T.-W.; Liu, D.-Z.; Wang, S.-Y.; Wang, S.-S. Enhancement of the growth of human endothelial cells by surface roughness at nanometer scale. Biomaterials 2003, 24, 4655–4661. [Google Scholar]
- Foley, J.D.; Grunwald, E.W.; Nealey, P.F.; Murphy, C.J. Cooperative modulation of neuritogenesis by PC12 cells by topography and nerve growth factor. Biomaterials 2005, 26, 3639–3644. [Google Scholar]
- Karuri, N.W.; Liliensiek, S.; Teixeira, A.I.; Abrams, G.; Campbell, S.; Nealey, P.F.; Murphy, C.J. Biological length scale topography enhances cell-substratum adhesion of human corneal epithelial cells. J. Cell Sci. 2004, 117, 3153–3164. [Google Scholar]
- Bettinger, C.J.; Zhang, Z.; Gerecht, S.; Borenstein, J.T.; Langer, R. Enhancement of in vitro capillary tube formation by substrate nanotopography. Adv. Mater. Deerfield 2008, 20, 99–103. [Google Scholar]
- Diehl, K.A.; Foley, J.D.; Nealey, P.F.; Murphy, C.J. Nanoscale topography modulates corneal epithelial cell migration. J. Biomed. Mater. Res. A 2005, 75, 603–611. [Google Scholar]
- Liliensiek, S.J.; Campbell, S.; Nealey, P.F.; Murphy, C.J. The scale of substratum topographic features modulates proliferation of corneal epithelial cells and corneal fibroblasts. J Biomed. Mater. Res. A 2006, 79, 185–192. [Google Scholar]
- Teixeira, A.I.; McKie, G.A.; Foley, J.D.; Bertics, P.J.; Nealey, P.F.; Murphy, C.J. The effect of environmental factors on the response of human corneal epithelial cells to nanoscale substrate topography. Biomaterials 2006, 27, 3945–3954. [Google Scholar]
- Silva, T.S.N.; Machado, D.C.; Viezzer, C.; Jr, A.N.S.; Oliveira, M.G.d. Effect of titanium surface roughness on human bone marrow cell proliferation and differentiation. An experimental study. Acta Cir. Bra. 2009, 24, 200–205. [Google Scholar]
- Brammer, K.; Oh, S.; Gallagher, J.; Jin, S. Enhanced cellular mobility guided by TiO2 nantube surfaces. Nano Lett. 2008, 8, 786–793. [Google Scholar]
- Park, J.; Bauer, S.; Schlegel, K.A.; Neukam, F.W.; Mark, K.v.d.; Schmuki, P. TiO2 Nanotube surfaces: 15nm—An optima length scale of surface topography for cell adhesion and differentiation. Small 2009, 5, 666–671. [Google Scholar]
- Szivek, J.A.; Margolis, D.S.; Schnepp, A.B.; Grana, W.A.; William, S.K. Selective cell proliferation can be controlled with CPC paritcle coatings. J. Biomed. Mater. Res. A 2007, 81, 939–947. [Google Scholar]
- Stankus, J.J.; Soletti, L.; Fujimoto, K.; Hong, Y.; Vorp, D.A.; Wagner, W.R. Fabrication of cell microintegrated blood vessel constructs through electrohydrodynamic atomization. Biomaterials 2007, 28, 2738–2746. [Google Scholar]
- Teixeira, A.I.; Abrams, G.A.; Murphy, C.J.; Nealey, P.F. Cell behavior on lithographically defined nanostructured substrates. J.Vac. Sci. Technol. B 2003, 21, 683–687. [Google Scholar]
- Teixeira, A.I.; Abrams, G.A.; Bertics, P.J.; Murphy, C.J.; Nealey, P.F. Epithelial contact guidance on well-defined micro- and nanostructured substrates. J. Cell Sci. 2003, 116, 1881–1892. [Google Scholar]
- Ebenstein, D.M.; Pruitz, L.A. Nonoindentation fo biological materials. Nanotoday 2006, 1, 26–33. [Google Scholar]
- Cao, Y.; Ma, D.; Raabe, D. The use of flat punch indentation to determine the viscoelastic properties in the time and frequency domains of soft layer bonded to a rigid substrate. Acta Biomater. 2009, 5, 240–248. [Google Scholar]
- Dahl, S.L.M.; Rhim, C.; Song, Y.C.; Niklason, L.E. Mechanical properties and compositions of tissue engineered and native arteries. Ann. Biomed. Eng. 2007, 35, 348–355. [Google Scholar]
- Brown, X.Q.; Ookawa, K.; Wong, J.Y. Evaluation of polydimethylsiloxane scaffolds with physiologically-relevant elastic moduli: interplay of substrate mechanics and surface chemistry effects on vascular smooth muscle cell response. Biomaterials 2005, 26, 3123–3129. [Google Scholar]
- Lee, J.C.; Discher, D.E. Deformation-enhanced fluctuations in the red cell skeleton with theoretical relations to elasticity, connectivity, and spectrin unfolding. Biophys. J. 2001, 81, 3178–3192. [Google Scholar]
- Ra, H.J.; Picart, C.; Feng, H.; Sweeney, H.L.; Discher, D.E. Muscle cell peeling from micropatterned collagen: Direct probing of focal and molecular properties of matrix adhesion. J. Cell Sci. 1999, 112, 1425–1436. [Google Scholar]
- Engler, A.; Bacakova, L.; Newman, C.; Sheehan, M.; Discher, D. Mechanical role of cytoskeletal components in vascular smoothmuscle cell adhesion in vitro. In Proceedings of the IEEE 28th Annual Northeast, Philadelphia, PA, USA, 20–21 April 2002; pp. 23–24.
- Engler, A.J.; Sen, S.; Sweeney, H.L.; Discher, D.E. Matrix elasticity directs stem cell lineage specification. Cell 2006, 126, 677–689. [Google Scholar]
- Ghajar, C.M.; Chen, X.; Harris, J.W.; Suresh, V.; Hughes, C.C.W.; Jeon, N.L.; Putnam, A.J.; George, S.C. The effect of matrix density on the regulation of 3-D capillary morphogenesis. Biophys. J. 2008, 94, 1930–1941. [Google Scholar]
- Griffith, L.G.; Swartz, M.A. Capturing complex 3D tissue physiology in vitry. Nat. Rev. Mol. Cell Biol. 2006, 7, 211–223. [Google Scholar]
- Shin, H.; Zygourakis, K.; Farach-Carson, M.C.; Yaszemski, M.J.; Mikos, A.G. Modulation of differentiation and mineralization of marrow stromal cells cultured on biomimetic hydrogels modified with Arg-Gly-Asp containing peptides. J. Biomed. Mater. Res. A 2004, 69, 535–543. [Google Scholar]
- Kasper, G.; Dankert, N.; Tuischer, J.; Hoeft, M.; Gaber, T.; Glaeser, J.D.; Zander, D.; Tschirschmann, M.; Thompson, M.; Matziolis, G.; Duda, G.N. Mesenchymal stem cells regulate angiogenesis according to their mechanical environment. Stem Cells 2007, 25, 903–910. [Google Scholar]
- Ghajar, C.M.; Blevins, K.S.; Hughes, C.C.W.; George, S.C.; Putnam, A.J. Mesenchymal stem cells enhance angiogenesis in mechanically viable prevascularized tissues via early matrix metalloproteinase upregulation. Tissue Eng. 2006, 12, 2875–2888. [Google Scholar]
- Peetla, C.; Labhasetwar, V. Biophysical characterization of nanoparticles-endothelial model of cell membrane interactions. Mol. Pharm. 2008, 5, 418–429. [Google Scholar]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wood, J.A.; Liliensiek, S.J.; Russell, P.; Nealey, P.F.; Murphy, C.J. Biophysical Cueing and Vascular Endothelial Cell Behavior. Materials 2010, 3, 1620-1639. https://doi.org/10.3390/ma3031620
Wood JA, Liliensiek SJ, Russell P, Nealey PF, Murphy CJ. Biophysical Cueing and Vascular Endothelial Cell Behavior. Materials. 2010; 3(3):1620-1639. https://doi.org/10.3390/ma3031620
Chicago/Turabian StyleWood, Joshua A., Sara J. Liliensiek, Paul Russell, Paul F. Nealey, and Christopher J. Murphy. 2010. "Biophysical Cueing and Vascular Endothelial Cell Behavior" Materials 3, no. 3: 1620-1639. https://doi.org/10.3390/ma3031620
APA StyleWood, J. A., Liliensiek, S. J., Russell, P., Nealey, P. F., & Murphy, C. J. (2010). Biophysical Cueing and Vascular Endothelial Cell Behavior. Materials, 3(3), 1620-1639. https://doi.org/10.3390/ma3031620




