Enhancement of the Phase Transition Enthalpy of an Organic Phase Change Material Through the Use of Clinoptilolite
Highlights
- Prevention of phase change material leakage through the microstructure of clinoptilolite.
- The microstructure of clinoptilolite mitigates leakage of phase change materials.
- Increase in the phase change enthalpy of the composite.
Abstract
1. Introduction
1.1. Limited Applicability of PCMs
1.2. Properties of Microcapsules and Their Shells
1.3. Limitations in the Use of Inorganic PCMs Due to Congruency Phenomena
1.4. Phase Change Materials in Thermal Management Applications
1.5. Ensuring the Integrity of Composites with Organic PCMs
2. Materials and Methods
2.1. Materials
- Phase change material RT21HC, characterized by a melting/freezing enthalpy of 190 J/g and a phase change temperature range of 19–23 °C (Rubiterm GmbH, Berlin, Germany).
- Clinoptilolite (powder with a median particle diameter of 50 μm, Nanga, Blękwit, Poland).
2.2. Apparatus and Experimental Methods
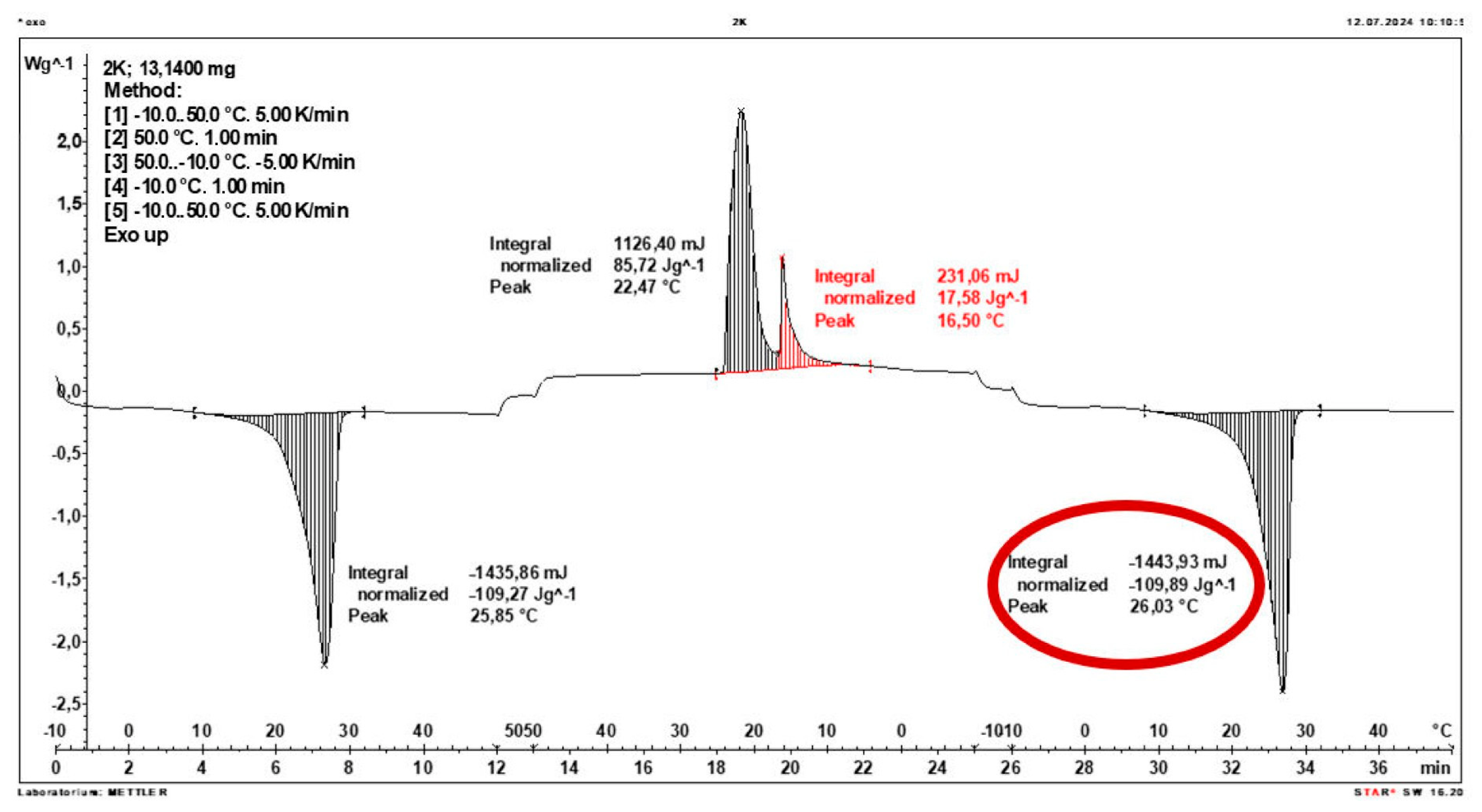
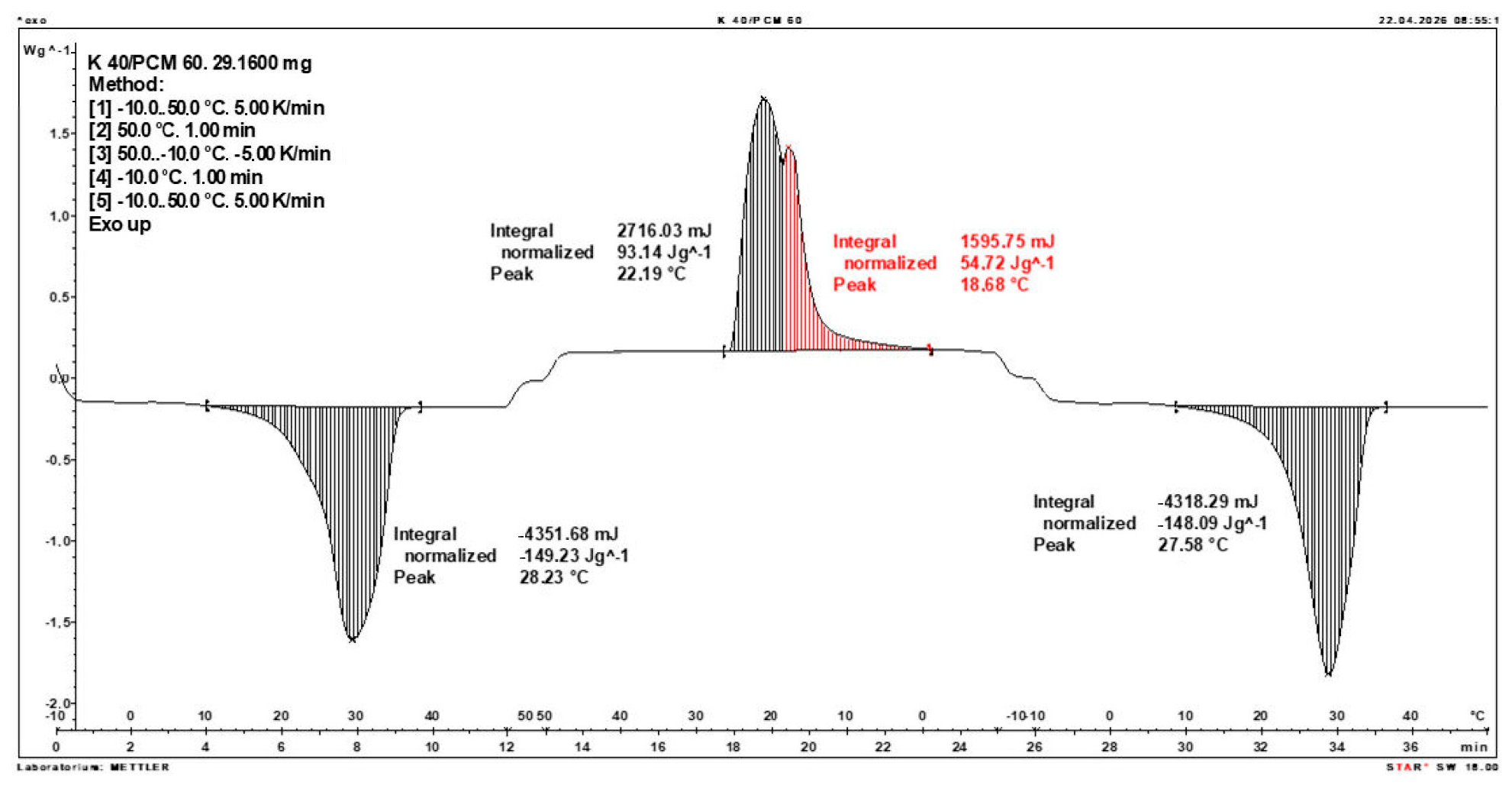
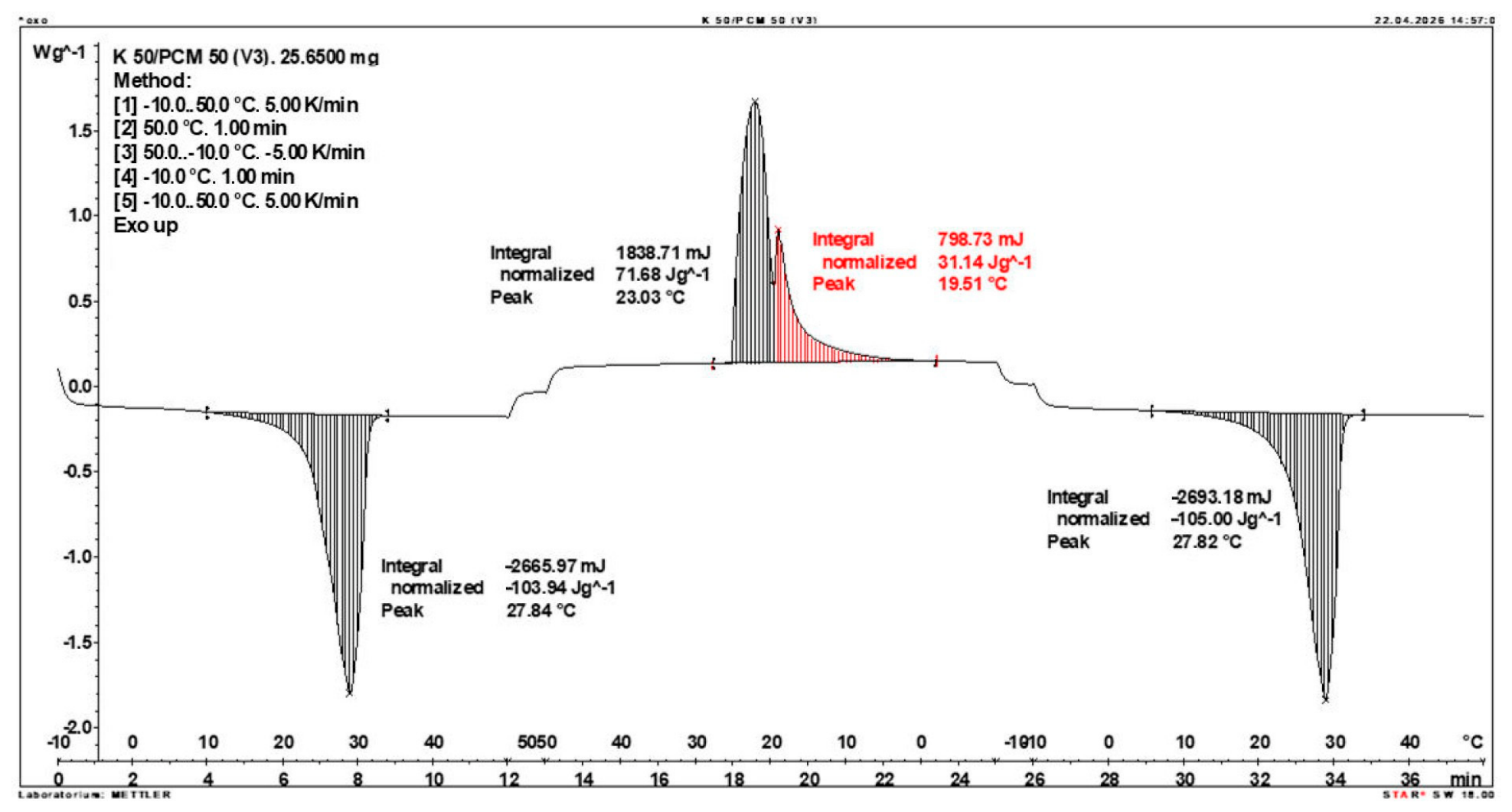
- The enthalpy of phase transition of the composite and the corresponding temperature range were determined using a DSC1 instrument (from Mettler Toledo, Greifensee, Switzerland), using STARe System software, Versions 19.00, (Mettler Toledo). The DSC traces were recorded at a heating and cooling rate of 5 °C/min under a nitrogen flow of 60 mL/min. The DSC instrument was calibrated using indium and zinc standards supplied by Mettler Toledo.
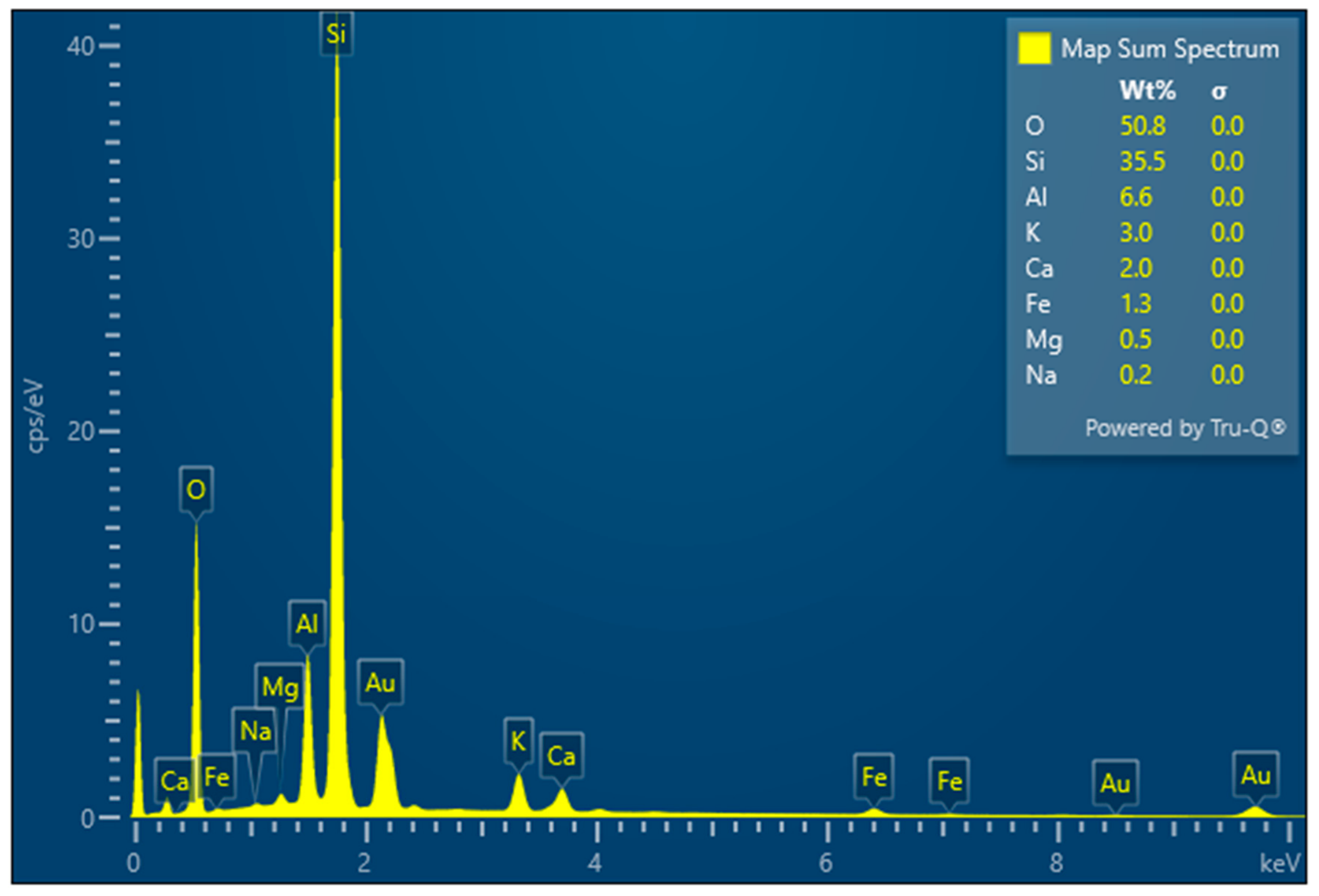
- Clinoptilolite chemical composition analysis was performed using a wavelength-dispersive X-ray fluorescence spectrometer, Ecublens, Switzerland, ARL Perform’X (WDXRF), Ecublens, Switzerland, equipped with OXSAS, Version 2.8.2 (Optical Emission Analytical Software) for data acquisition and analysis.
- The macroscopic examination of the composites was carried out using a stereoscopic microscope (Olympus SZX-7), Hamburg, Germany, with a Galilean optical system and distortion-free plan apochromatic objectives, coupled with a high-sensitivity sCMOS microscope camera (Moticam PRO-S5), Hong Kong, and software for image acquisition and measurement.
- Structural and textural characteristics, as well as surface elemental distribution, were investigated using a MIRA3 scanning electron microscope (SEM) (Tescan), Brno Czech Republic. Natural clinoptilolite samples were used for these analyses.
2.3. Experimental Procedure
3. Results
3.1. Structural and Textural Characteristics
3.2. Calorimetric Results of Composites
3.3. Chemical Composition
4. Discussion and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PAN | Polyacrylonitrile |
| PCM | Phase-change materials |
| DSC | Differential Scanning Calorimetry |
| EDS | Energy Dispersive X-ray Spectroscopy |
| WDXRF | Wavelength Dispersive X-ray Fluorescence |
| SEM | Scanning Electron Microscopy |
References
- Musiał, M.; Pochwat, K.; Lichołai, L.; Katunsky, D. Assessment of the interaction between forced convection and a phase-change battery. Appl. Therm. Eng. 2025, 278, 127229. [Google Scholar] [CrossRef]
- Musiał, M.; Lichołai, L. Multi-Faceted Analysis of Phase-Change Composite Intended for Autonomous Buildings. Materials 2024, 17, 2604. [Google Scholar] [CrossRef]
- Hedau, A.; Singal, S.K. Heat transfer and fluid flow analysis of PCM-based thermal energy storage concept for double pass solar air heater. Int. Commun. Heat Mass Transf. 2024, 157, 107813. [Google Scholar] [CrossRef]
- Roy, A.; Venkitaraj, K.P.; Vigneshwaran, P.; Saboor, S.; Cuce, E.; Saxena, K.K. Enhanced convective heat transfer with Al2O3-water nanofluid in a PCM-based thermal energy storage system. J. Energy Storage 2024, 97, 112853. [Google Scholar] [CrossRef]
- Sun, J.; Zhao, J.; Zhang, W.; Xu, J.; Wang, B.; Wang, X.; Zhou, J.; Guo, H.; Liu, Y. Composites with a Novel Core–shell Structural Expanded Perlite/Polyethylene glycol Composite PCM as Novel Green Energy Storage Composites for Building Energy Conservation. Appl. Energy 2023, 330, 120363. [Google Scholar] [CrossRef]
- Pękala, A.; Musiał, M.; Lichołai, L. Carpathian Diatomites and Their Applications in Phase-Change Composites. Materials 2025, 18, 2097. [Google Scholar] [CrossRef] [PubMed]
- Musiał, M.; Lichołai, L.; Pękala, A. Analysis of the Thermal Performance of Isothermal Composite Heat Accumulators Containing Organic Phase-Change Material. Energies 2023, 16, 1409. [Google Scholar] [CrossRef]
- Öztürk, B.; Gölbaşı, Z.; Yazıcı, M.Y. Experimental investigation of the melting performance of a low porosity metal foam/PCM composite heat sink in various configurations. Int. Commun. Heat Mass Transf. 2023, 149, 107169. [Google Scholar] [CrossRef]
- Parvate, S.; Singh, J.; Dixit, P.; Vennapusa, J.R.; Maiti, T.K.; Chattopadhyay, S. Titanium Dioxide Nanoparticle-Decorated Polymer Microcapsules Enclosing Phase Change Material for Thermal Energy Storage and Photocatalysis. ACS Appl. Polym. Mater. 2021, 3, 1866–1879. [Google Scholar] [CrossRef]
- Soni, K.; Panwar, N.L. Revolutionizing thermal energy storage: An overview of porous support materials for advanced composite Phase Change Materials (PCMs). Prog. Eng. Sci. 2024, 1, 100023. [Google Scholar] [CrossRef]
- Nikolaev, P.; Jivkov, A.P.; Fifre, M.; Sedighi, M. Peridynamic analysis of thermal behaviour of PCM composites for heat storage. Comput. Methods Appl. Mech. Eng. 2024, 424, 116905. [Google Scholar] [CrossRef]
- Raj, V.K.; Baiju, V.; Sha, A.A.; Priya, K.L.; Vishnu, S.B. Performance of hybrid nano-PCM composite: Insights from T-history method and numerical analysis. J. Energy Storage 2024, 90, 111708. [Google Scholar] [CrossRef]
- Acikök, F.; Ardoǧa, M.K.; Şahmaran, M. Performance of the phase-change material (PCM) to reduce freeze–thaw effect in cementitious composites. Adv. Cem. Res. 2024, 36, 150–163. [Google Scholar] [CrossRef]
- Agarwal, P.; Prabhakar, A. Energy and thermo-economic analysis of PCM integrated brick in composite climatic condition of Jaipur—A numerical study. Sustain. Cities Soc. 2023, 88, 104294. [Google Scholar] [CrossRef]
- Ates, F.; Woo, B.H.; Jang, C.; Kim, H.G. Enhancing cementitious composites with PCM-impregnated cork granules for sustainable and energy-efficient building elements. Constr. Build. Mater. 2024, 416, 135071. [Google Scholar] [CrossRef]
- Erdogmus, E.; Yaras, A.; Ustaoglu, A.; Hekimoğlu, G.; Sarı, A.; Gencel, O. Thermal performance analysis of novel foam concrete composites with PCM for energy storage and environmental benefits in buildings. Energy Build. 2023, 296, 113413. [Google Scholar] [CrossRef]
- Jin, X.; Haider, M.Z.; Ahn, J.; Fang, G.; Hu, J.W. Development of nanomodified eco-friendly thermal energy storing cementitious composite using PCM microencapsulated in biosourced encapsulation shell. Case Stud. Constr. Mater. 2023, 19, e02447. [Google Scholar] [CrossRef]
- Katish, M.; Allen, S.; Squires, A.; Ferrandiz-Mas, V. Experimental study of phase change material (PCM) biochar composite for net-zero built environment applications. Clean. Mater. 2024, 14, 100274. [Google Scholar] [CrossRef]
- Aksoy, S.A.; Yılmaz, D.; Maleki, H.; Rahbar, R.S.; Barani, H. Fabrication and characterization of nanoencapsulated PCM-doped cotton/PAN nanofiber based composite yarns for thermoregulation. J. Energy Storage 2024, 101, 113849. [Google Scholar] [CrossRef]
- He, Z.; Ma, H.; Lu, S. Preparation and thermal performance study of a novel hydrated salt composite PCM for space heating. J. Energy Storage 2024, 90, 111906. [Google Scholar] [CrossRef]
- Akula, R.; Minnikanti, A.; Balaji, C. Pin fin-PCM composite heat sink solution for thermal management of cylindrical Li-ion battery. Appl. Therm. Eng. 2024, 248, 123146. [Google Scholar] [CrossRef]
- Elshaer, A.M.; Soliman, A.M.A.; Kassab, M.; Hawwash, A.A. Experimental and numerical investigations of an open-cell copper foam (OCCF)/phase change material (PCM) composite-based module for satellite avionics thermal management in a thermal vacuum chamber (TVC). J. Energy Storage 2024, 75, 109572. [Google Scholar] [CrossRef]
- Kumar, P.; Thomas, S.; Sobhan, C.B.; Peterson, G.P. Activated carbon foam composite derived from PEG400/Terminalia Catappa as form stable PCM for sub-zero cold energy storage. J. Clean. Prod. 2024, 434, 139993. [Google Scholar] [CrossRef]
- Cheng, F.; Xu, Y.; Lv, Z.; Huang, Z.; Fang, M.; Liu, Y.; Wu, X.; Min, X. Form-stable and tough paraffin-Al2O3/high density polyethylene composites as environment-friendly thermal energy storage materials: Preparation, characterization and analysis. J. Therm. Anal. Calorim. 2021, 146, 2089–2099. [Google Scholar] [CrossRef]
- Gado, M.G. Thermal management and heat transfer enhancement of electronic devices using integrative phase change material (PCM) and triply periodic minimal surface (TPMS) heat sinks. Appl. Therm. Eng. 2025, 258, 124504. [Google Scholar] [CrossRef]
- Ryms, M.; Januszewicz, K.; Haustein, E.; Kazimierski, P.; Lewandowski, W.M. Thermal properties of a cement composite containing phase change materials (PCMs) with post-pyrolytic char obtained from spent tyres as a carrier. Energy 2022, 239, 121936. [Google Scholar] [CrossRef]
- Al-Yasiri, Q.; Alktranee, M.; Szabó, M. Metal fibers-enhanced PCM thermal energy storage unit: An experimental approach on a composite roof application. Int. J. Thermofluids 2024, 23, 100812. [Google Scholar] [CrossRef]
- Sheikh, Y.; Fatih Orhan, M.; Kanoglu, M. Heat transfer enhancement of a bio-based PCM/metal foam composite heat sink. Therm. Sci. Eng. Prog. 2022, 36, 101536. [Google Scholar] [CrossRef]
- Elshaer, A.M.; Soliman, A.M.A.; Kassab, M.; Mori, S.; Hawwash, A.A. Experimental investigations on copper foam/PCM composite-based thermal control hardware (TCH) using foam samples with different pore sizes under intermittent thermal conditions. J. Energy Storage 2023, 72, 108320. [Google Scholar] [CrossRef]
- Das, D.; Masek, O.; Paul, M.C. Development of novel form-stable PCM-biochar composites and detailed characterization of their morphological, chemical and thermal properties. J. Energy Storage 2024, 84, 110995. [Google Scholar] [CrossRef]
- Ma, F.; Liang, Y.; Tao, Z.; Guo, X.; Guo, Q.; Liu, Z. A novel PCM/expanded graphite composite sphere with high thermal conductivity and excellent shape stability used for a packed-bed thermal energy system. Diam. Relat. Mater. 2024, 145, 111102. [Google Scholar] [CrossRef]
- Mombeki Pea, H.J.; An, Z.; Du, X.; Hou, W.; Zhang, D.; Liu, X. Experimental and analytical studies on latent heat of hydrated salt/modified EG-based form-stable composite PCMs for energy storage application. Renew. Energy 2024, 222, 119978. [Google Scholar] [CrossRef]
- Chen, Y.; Yao, Z.; Chen, B.; Liu, Z.; Yang, C. Numerical and experimental study of the ocean thermal energy capture process utilizing metal foam–phase-change material (PCM) composites. J. Energy Storage 2023, 67, 107600. [Google Scholar] [CrossRef]
- Hekimoğlu, G.; Sarı, A.; Gencel, O.; Tyagi, V.V.; Sharma, R.K. Activated carbon/expanded graphite hybrid structure for development of nonadecane based composite PCM with excellent shape stability, enhanced thermal conductivity and heat charging-discharging performance. Therm. Sci. Eng. Prog. 2023, 44, 102081. [Google Scholar] [CrossRef]
- Lin, J.; Feng, X.; Huang, J.; Liu, Y.; Xiao, Y.; Li, Y.; Min, Y.; Tang, B.Z. Flexible AIE/PCM composite fiber with biosensing of alcohol, fluorescent anti-counterfeiting and body thermal management functions. Biosens. Bioelectron. 2025, 267, 116799. [Google Scholar] [CrossRef]
- Ortega, E.A.; Cheeseman, C.; Knight, J.; Loizidou, M. Properties of alkali activated clinoptilolite. Cem. Concr. Res. 2000, 30, 1641. [Google Scholar] [CrossRef]
- Luke, K. Mocrostructure of Early Age Hydration of Natural Zeolite with Pure Cement Phases; ICMA: Denver, CO, USA, 2006. [Google Scholar]
- Bundyra-Oracz, G.; Siemaszko-Lotkowska, D. Zeolite—A pozzolanic additive to cement. Constr. Technol. Archit. 2010, 52, 72–74. [Google Scholar]
- Pietras, M.; Konkol, J. Zeolite as an Additive to Mortars and Concretes; Scientific Papers; Częstochowa University of Technology: Częstochowa, Poland, 2020; pp. 149–155. [Google Scholar] [CrossRef]
- Żygadło, M.; Seweryn, A.; Woźniak, M. Synthesis of zeolites based on fly ash from selected heat recovery installations. Arch. Waste Manag. Environ. Prot. 2010, 1, 15–26. [Google Scholar]
- Barnat-Hunek, D.; Klimek, B.; Franus, W. The influence of zeolite addition on the physicomechanical properties of renovation plasters. Insulation 2014, 9, 72–78. [Google Scholar]
- Małolepszy, J.; Grabowska, E. The influence of zeolites on the hydration process of mineral binders. Constr. Archit. 2016, 3, 185–192. [Google Scholar] [CrossRef]
- ChemTube3D. Available online: https://www.chemtube3d.com/ss-z-clinoptilolite (accessed on 21 March 2026).
- Rakkappan, S.R.; Sivian, S.; Ahmed, S.N.; Naarendharan, M.; Sudhir, P.S. Preparation, characterisation and energy storage performance study on 1-Decanol-Expanded graphite composite PCM for air-conditioning cold storage system. Int. J. Refrig. 2021, 123, 91–101. [Google Scholar] [CrossRef]
- Sardari, P.T.; Grant, D.; Giddings, D.; Walker, G.S.; Gillott, M. Composite metal foam/PCM energy store design for dwelling space air heating. Energy Convers. Manag. 2019, 201, 112151. [Google Scholar] [CrossRef]
- Tang, J.; Xie, Y.; Chang, S.; Yan, Z.; Wu, H.; Zhang, H. Performance analysis of acceleration effect on paraffin melting in finned copper foam. Appl. Therm. Eng. 2022, 202, 117826. [Google Scholar] [CrossRef]
- Yang, Y.; Liu, X.; Zheng, J.; Fu, J.; Lyu, Y.; Cheng, Z.; Li, F.; Zhang, W. Zeolite pore confinement in adsorption and skeletal isomerization of n-hexane. Chem. Eng. Sci. 2025, 317, 122072. [Google Scholar] [CrossRef]
- Soualah, A.; Lemberton, J.L.; Pinard, L.; Chater, M.; Magnoux, P.; Moljord, K. Hydroisomerization of long-chain n-alkanes on bifunctional Pt/zeolite catalysts: Effect of the zeolite structure on the product selectivity and on the reaction mechanism. Appl. Catal. A Gen. 2008, 336, 23. [Google Scholar] [CrossRef]
- Farjoo, A.; Sawada, J.A.; Kuznicki, S.M. Manipulation of the pore size of clinoptilolite for separation of ethane from ethylene. Chem. Eng. Sci. 2015, 138, 685. [Google Scholar] [CrossRef]
- Chica, A.; Corma, A.; Miguel, P.J. Isomerization of C5–C7 n-alkanes on unidirectional large pore zeolites: Activity, selectivity and adsorption features. Catal. Today 2001, 65, 101. [Google Scholar] [CrossRef]
- Ono, Y. A survey of the mechanism in catalytic isomerization of alkanes. Catal. Today 2003, 81, 3. [Google Scholar] [CrossRef]
















Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Musiał, M.; Pękala, A.; Lichołai, L.; Mossety-Leszczak, B. Enhancement of the Phase Transition Enthalpy of an Organic Phase Change Material Through the Use of Clinoptilolite. Materials 2026, 19, 1888. https://doi.org/10.3390/ma19091888
Musiał M, Pękala A, Lichołai L, Mossety-Leszczak B. Enhancement of the Phase Transition Enthalpy of an Organic Phase Change Material Through the Use of Clinoptilolite. Materials. 2026; 19(9):1888. https://doi.org/10.3390/ma19091888
Chicago/Turabian StyleMusiał, Michał, Agnieszka Pękala, Lech Lichołai, and Beata Mossety-Leszczak. 2026. "Enhancement of the Phase Transition Enthalpy of an Organic Phase Change Material Through the Use of Clinoptilolite" Materials 19, no. 9: 1888. https://doi.org/10.3390/ma19091888
APA StyleMusiał, M., Pękala, A., Lichołai, L., & Mossety-Leszczak, B. (2026). Enhancement of the Phase Transition Enthalpy of an Organic Phase Change Material Through the Use of Clinoptilolite. Materials, 19(9), 1888. https://doi.org/10.3390/ma19091888








