From Farm to Table and Back Again: Circular Valorization of Biomass Ash and Sewage Sludge into Sustainable Material Blends
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Materials
2.2. Justification of the Mixture Ratios
2.3. Methods
3. Results
3.1. Elemental and Chemical Composition
3.1.1. Characterization of Raw Materials
3.1.2. Characterization of Mixtures
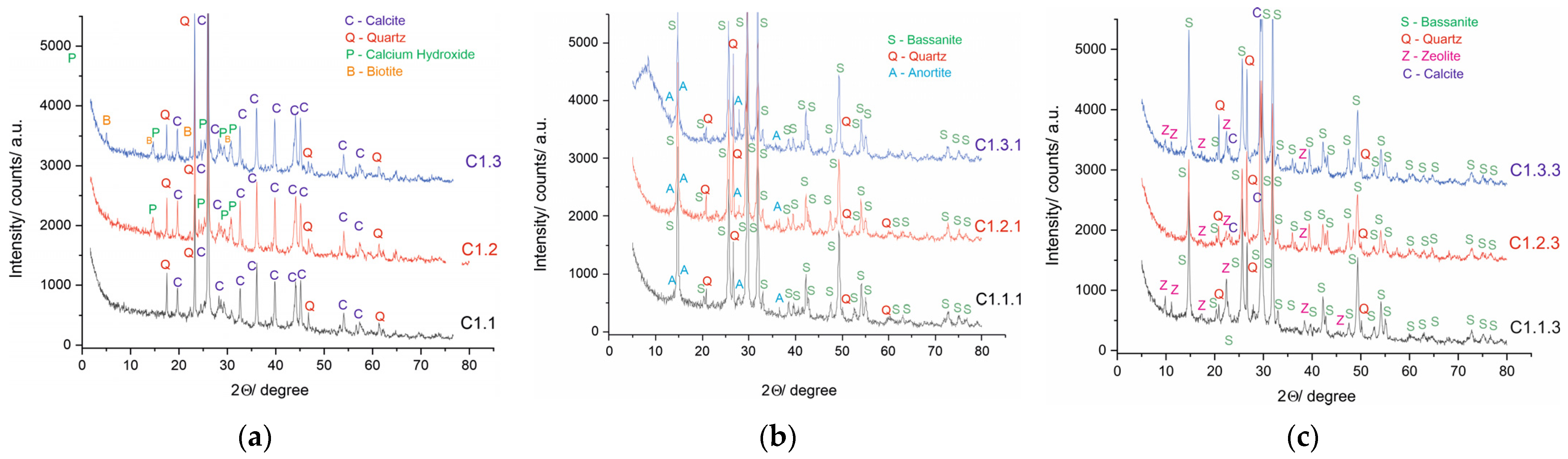
3.2. Phase Composition and Mineralogical Development
3.2.1. XRD of Raw Materials
3.2.2. XRD of Mixtures
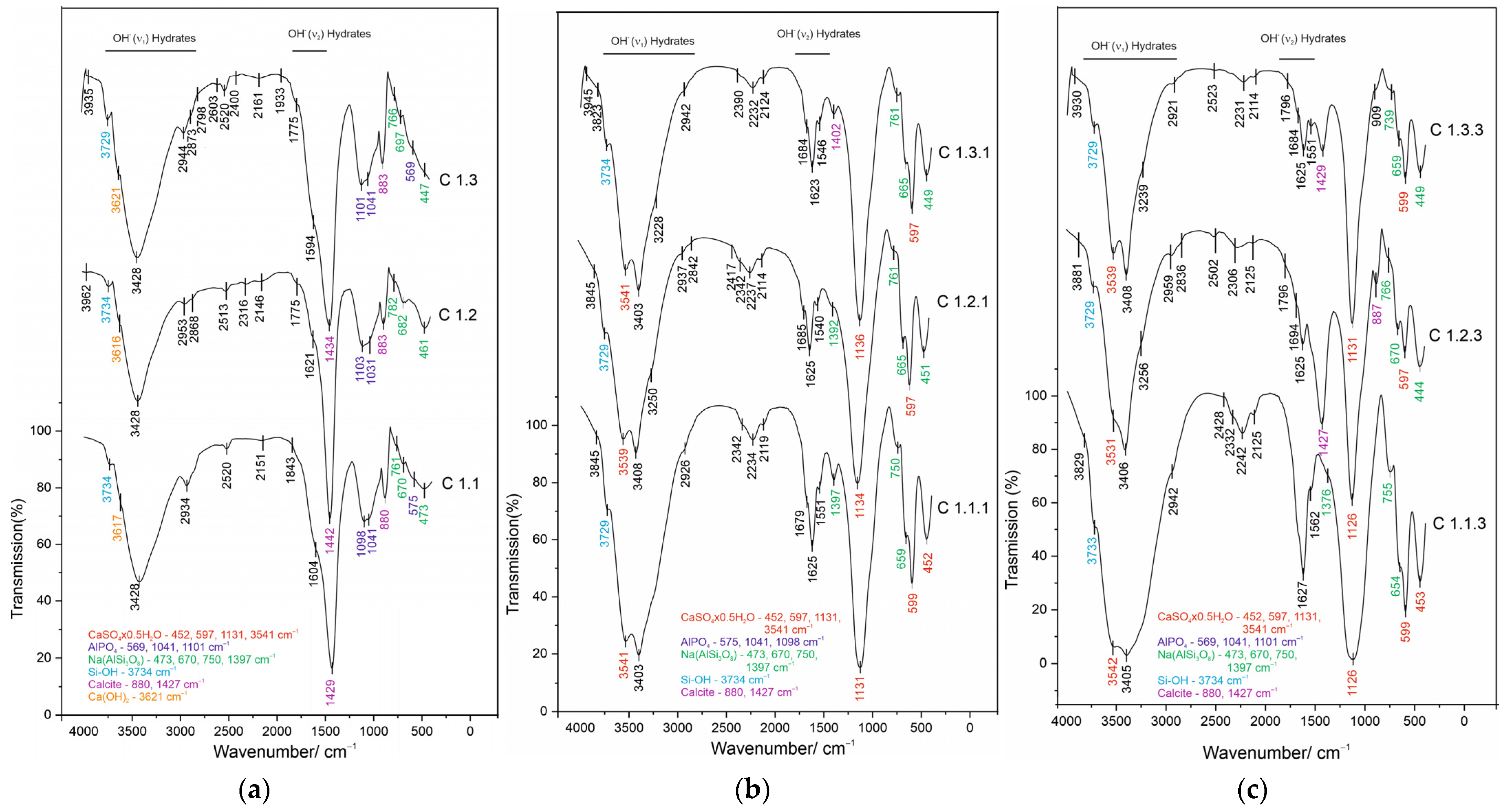
3.3. FTIR Analysis
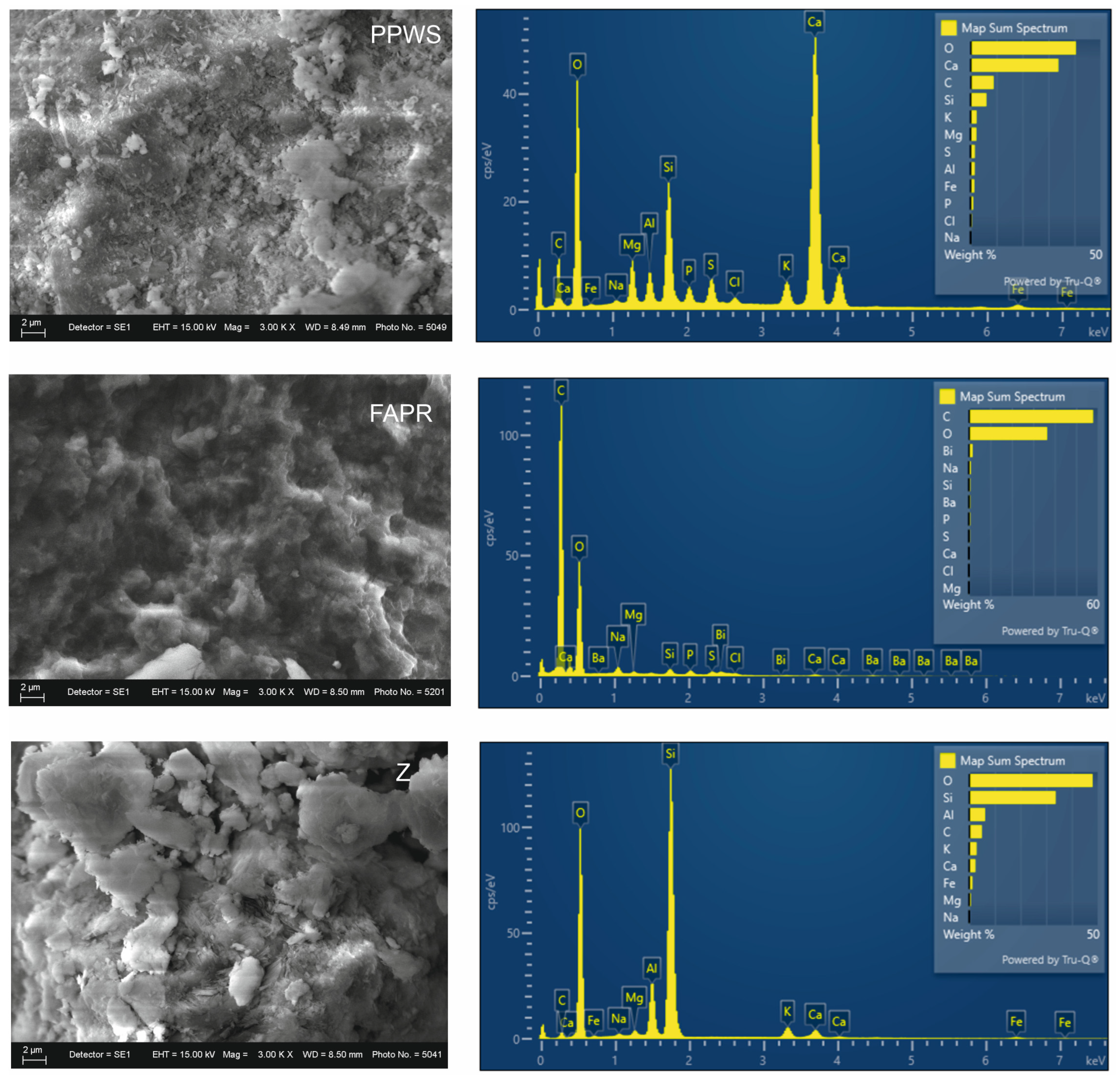
3.4. Microstructural Characterization (SEM–EDS)
3.4.1. Microstructural Characterization of Raw Materials
3.4.2. Mixtures
4. Discussion
4.1. Role of Raw Materials and Initial Structure of the Base Mixtures
4.2. Chemical Behavior and Interaction Mechanisms
4.3. Mineral Transformation in Acid-Activated Mixtures (C1.1.1, C1.2.1, C1.3.1)
4.4. Effect of Zeolite Modification on the Development of the Mineral Structure (C1.1.3, C1.2.3, C1.3.3)
4.5. Significance for Pozzolanic Activity and Material Stability
4.6. Confirmation of the Working Hypothesis
4.7. Compared to Existing Literature
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cherian, C.; Siddiqua, S. Pulp and Paper Mill Fly Ash: A Review. Sustainability 2019, 11, 4394. [Google Scholar] [CrossRef]
- Velenturf, A.P.M.; Purnell, P. Principles for a Sustainable Circular Economy. Sustain. Prod. Consum. 2021, 27, 1437–1457. [Google Scholar] [CrossRef]
- Gusiatin, M.Z.; Kulikowska, D.; Bernat, K. Municipal Sewage Sludge as a Resource in the Circular Economy. Energies 2024, 17, 2474. [Google Scholar] [CrossRef]
- Lehmusto, J.; Tesfaye, F.; Karlström, O.; Hupa, L. Ashes from challenging fuels in the circular economy. Waste Manag. 2024, 177, 211–231. [Google Scholar] [CrossRef] [PubMed]
- Donatello, S.; Cheeseman, C.R. Recycling and recovery routes for incinerated sewage sludge ash (ISSA): A review. Waste Manag. 2013, 33, 2328–2340. [Google Scholar] [CrossRef] [PubMed]
- Demeyer, A.; Voundi Nkana, J.C.; Verloo, M.G. Characteristics of wood ash and influence on soil properties and nutrient uptake: An overview. Bioresour. Technol. 2001, 77, 287–295. [Google Scholar] [CrossRef]
- Pitman, R.M. Wood ash use in forestry—A review of the environmental impacts. Forestry 2006, 79, 563–588. [Google Scholar] [CrossRef]
- Fytili, D.; Zabaniotou, A. Utilization of sewage sludge in EU application of old and new methods—A review. Renew. Sustain. Energy Rev. 2008, 12, 116–140. [Google Scholar] [CrossRef]
- Barrera-Díaz, C.; Martínez-Barrera, G.; Gencel, O.; Bernal-Martínez, L.A.; Brostow, W. Processed wastewater sludge for improvement of mechanical properties of concretes. J. Hazard. Mater. 2011, 192, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Snellings, R.; Mertens, G.; Elsen, J. Supplementary cementitious materials. Rev. Mineral. Geochem. 2012, 74, 211–278. [Google Scholar] [CrossRef]
- Moolchandani, K. Industrial byproducts in concrete: A state-of-the-art review. Next Mater. 2025, 8, 100593. [Google Scholar] [CrossRef]
- Jwaida, Z.; Dulaimi, A.; Alyhya, W.; Algretawee, H.; Al-Busaltan, S. Recycling and utilization of paper sludge ash-current status review and future perspectives. Sustain. Mater. Technol. 2024, 40, e00960. [Google Scholar] [CrossRef]
- Justo, F.; Faria, L. Use of industrial pulp and paper waste as agricultural inputs: A systematic literature review. Braz. J. Agric. Environ. Eng. 2025, 29, e287587. [Google Scholar] [CrossRef]
- Ahmaruzzaman, M. A review on utilization of fly ash. Prog. Energy Combust. Sci. 2010, 36, 327–363. [Google Scholar] [CrossRef]
- Fernández, R.; Martirena, F.; Scrivener, K. The Origin of the Pozzolanic Activity of Calcined Clay Minerals: A Comparison Between Kaolinite, Illite and Montmorillonite. Cem. Concr. Res. 2011, 41, 113–122. [Google Scholar] [CrossRef]
- Zheng, X.; Liu, K.; Gao, S.; Wang, F.; Wu, Z. Effect of pozzolanic reaction of zeolite on its internal curing performance in cement-based materials. J. Build. Eng. 2023, 63, 105503. [Google Scholar] [CrossRef]
- Petkova, V.; Stoyanov, V.; Kostova, B.; Mihaylova, K. Effects of zeolite incorporation and inert fillers on the curing of cement mortars. J. Chem. Technol. Metall. 2024, 59, 313–322. [Google Scholar] [CrossRef]
- Liang, C.; Le, X.; Fang, W.; Zhao, J.; Fang, L.; Hou, S. The Utilization of Recycled Sewage Sludge Ash as a Supplementary Cementitious Material in Mortar: A Review. Sustainability 2022, 14, 4432. [Google Scholar] [CrossRef]
- Kominko, H.; Gorazda, K.; Wzorek, Z. Sewage sludge: A review of its risks and circular raw material potential. J. Water Process Eng. 2024, 63, 105522. [Google Scholar] [CrossRef]
- Chojnacka, K.; Gorazda, K.; Witek-Krowiak, A.; Moustakas, K. Recovery of fertilizer nutrients from materials—Contradictions, mistakes and future trends. Renew. Sustain. Energy Rev. 2019, 110, 485–498. [Google Scholar] [CrossRef]
- Świerczek, L.; Cieślik, M.; Konieczka, P. The potential of raw sewage sludge in construction industry—A review. J. Clean. Prod. 2018, 200, 342–356. [Google Scholar] [CrossRef]
- Maj, I.; Niesporek, K.; Plaza, P.; Maier, J.; Loj, P. Biomass Ash: A Review of Chemical Compositions and Management Trends. Sustainability 2025, 17, 4925. [Google Scholar] [CrossRef]
- Olatoyan, O.; Kareem, M.; Adebanjo, A.; Olawale, O.; Alao, K. Potential use of biomass ash as a sustainable alternative for fly ash in concrete production: A review. Hybrid Adv. 2023, 1, 100076. [Google Scholar] [CrossRef]
- Rajamma, R.; Ball, R.J.; Tarelho, L.A.C.; Allen, G.C.; Labrincha, J.A.; Ferreira, V.M. Biomass fly ash in concrete. J. Hazard. Mater. 2009, 172, 1049–1060. [Google Scholar] [CrossRef]
- Świerczek, L.; Cieślik, B.; Konieczka, P. Challenges and opportunities related to the use of sewage sludge ash in cement-based building materials—A review. J. Clean. Prod. 2021, 287, 125054. [Google Scholar] [CrossRef]
- Berra, M.; Mangialardi, T.; Paolini, A.E. Reuse of woody biomass fly ash. Constr. Build. Mater. 2015, 76, 286–296. [Google Scholar] [CrossRef]
- Taylor, H.F.W. Cement Chemistry, 2nd ed.; Thomas Telford: London, UK, 1997. [Google Scholar]
- Lothenbach, B.; Scrivener, K.; Hooton, R.D. Supplementary cementitious materials. Cem. Concr. Res. 2011, 41, 1244–1256. [Google Scholar] [CrossRef]
- Liguori, B.; Aprea, P.; Gennaro, B.D.; Iucolano, F.; Colella, A.; Caputo, D. Pozzolanic Activity of Zeolites: The Role of Si/Al Ratio. Materials 2019, 12, 4231. [Google Scholar] [CrossRef] [PubMed]
- Mumpton, F.A. La roca magica: Uses of natural zeolites in agriculture and industry. Proc. Natl. Acad. Sci. USA 1999, 96, 3463–3470. [Google Scholar] [CrossRef]
- Wang, S.; Peng, Y. Natural zeolites as effective adsorbents in water and wastewater treatment. Chem. Eng. J. 2010, 156, 11–24. [Google Scholar] [CrossRef]
- EN 14899:2005; Characterization of Waste—Sampling of Waste Materials. BSI: London, UK, 2005.
- Serafimova, E.; Petkova, V.; Manoylova, A. The effects of sulfur-acid treatment on the thermal properties of wood biomass and chicken litter mixtures. J. Therm. Anal. Calorim. 2025, 150, 19963–19979. [Google Scholar] [CrossRef]
- Powder Diffraction File. International Centre for Diffraction Data (ICDD) Newtown Square. 2001. Available online: https://www.icdd.com/ (accessed on 7 April 2026).
- Lafuente, B.; Downs, R.T.; Yang, H.; Stone, N. 1. The Power of Databases: The RRUFF Project. In Highlights in Mineralogical Crystallography; Armbruster, T., Danisi, R.M., Eds.; De Gruyter: Berlin, Germany, 2015; pp. 1–30. [Google Scholar]
- Gražulis, S.; Daškevič, A.; Merkys, A.; Chateigner, D.; Lutterotti, L.; Quiros, M.; Serebryanaya, N.R.; Moeck, P.; Downs, R.T.; Le Bail, A. Crystallography Open Database (COD): An Open-Access Collection of Crystal Structures and Platform for World-Wide Collaboration. Nucleic Acids Res. 2012, 40, D420–D427. [Google Scholar] [CrossRef]
- Rintoul, L.; Wentrup-Byrne, E.; Suzuki, S.; Grondahl, L. FT-IR Spectroscopy of Fluoro-Substituted Hydroxyapatite: Strengths and Limitations. J. Mater. Sci. Mater. Med. 2007, 18, 1701–1709. [Google Scholar] [CrossRef] [PubMed]
- Chukanov, N.V. Infrared Spectra of Mineral Species: Extended Library; Springer Geochemistry/Mineralogy; Springer: Dordrecht, The Netherlands, 2014. [Google Scholar]
- Coates, J. Interpretation of Infrared Spectra. A Practical Approach. Encyclopedia of Analytical Chemistry; Meyers, R.A., Ed.; John Wiley & Sons Ltd.: Chichester, UK, 2000; pp. 10815–10837. [Google Scholar]
- Derrik, M.R.; Stulik, D.; Landry, J.M. Infrared Spectroscopy in Conservation Science; Scientific Tools for Conservation; Getty Publications: Los Angeles, CA, USA, 1999. [Google Scholar]
- Hesse, M.; Meier, H.; Zeeh, B. Spektroskopische Methoden in der Organischen Chemie; Georg Thieme Verlag: Stuttgart, Germany; New York, NY, USA, 1987. [Google Scholar]
- Lee, D.C.; Chapman, D. Infrared Spectroscopic Studies of Biomembranes and Model Membranes. Biosci. Rep. 1986, 6, 235–256. [Google Scholar] [CrossRef] [PubMed]
- EU Regulation (EU) 2019/1009. Regulation of the European Parliament and of the Council on EU fertilising products. Off. J. Eur. Union 2019, L170, 1–114.





| Mixtures | PPWS (wt.%) | FAPR (wt.%) | H2SO4 (wt.%) | Z (wt.%) |
|---|---|---|---|---|
| C 1.1 | 75 | 25 | 0 | 0 |
| C 1.2 | 59 | 41 | 0 | 0 |
| C 1.3 | 67 | 33 | 0 | 0 |
| C 1.1.1 | 67 | 23 | 10 | 0 |
| C 1.2.1 | 52 | 38 | 10 | 0 |
| C 1.3.1 | 60 | 30 | 10 | 0 |
| C 1.1.3 | 56 | 27 | 10 | 7 |
| C 1.2.3 | 48 | 35 | 10 | 7 |
| C 1.3.3 | 55 | 28 | 10 | 7 |
| Parameter | Unit | FARP | PPWS | Z |
|---|---|---|---|---|
| Ca | % | 30.48 | 0.29 | 2.48 |
| Al | % | 1.20 | 0.02 | 4.02 |
| Fe | % | 1.05 | 0.02 | 0.48 |
| K | % | 1.11 | 0.02 | 0.93 |
| Mg | % | 0.83 | 0.03 | 0.44 |
| P | % | 0.41 | 0.10 | 0.004 |
| S | % | 0.06 | 2.47 | 0.01 |
| Na | mg/kg | 2838 | 984 | 3175 |
| Zn | mg/kg | 122 | 13 | 34 |
| B | mg/kg | 90 | 12 | 5 |
| Ba | mg/kg | 466 | 57 | 18 |
| Cd | mg/kg | 1 | <0.1 | <0.1 |
| Co | mg/kg | 4 | <0.5 | <0.5 |
| Cr | mg/kg | 25 | 4 | <0.5 |
| Cu | mg/kg | 60 | 6 | 2 |
| Mn | mg/kg | 1617 | 391 | 479 |
| Mo | mg/kg | 1 | <0.5 | <0.5 |
| Ni | mg/kg | 19 | <0.5 | <0.5 |
| Pb | mg/kg | 19 | <0.5 | 53 |
| V | mg/kg | 26 | <1 | 2 |
| Parameters (Unit) | C 1.1 | C 1.1.1 | C 1.1.3 | C 1.2 | C 1.2.1 | C 1.2.3 | C 1.3 | C 1.3.1 | C 1.3.3 |
|---|---|---|---|---|---|---|---|---|---|
| Ca (%) | 30.21 | 13.2 | 11.6 | 31.31 | 24.44 | 13.68 | 33.32 | 23.83 | 14.6 |
| Al (%) | 1.12 | 0.70 | 0.90 | 1.24 | 0.91 | 1.31 | 1.20 | 0.87 | 1.21 |
| Fe (%) | 0.97 | 0.65 | 0.58 | 1.03 | 0.79 | 0.74 | 1.02 | 0.74 | 0.72 |
| K (%) | 1.03 | 0.78 | 0.79 | 1.08 | 0.90 | 0.91 | 1.13 | 0.88 | 0.88 |
| Mg (%) | 0.76 | 0.59 | 0.52 | 0.85 | 0.67 | 0.62 | 0.88 | 0.65 | 0.65 |
| P (%) | 0.53 | 0.36 | 0.25 | 0.50 | 0.38 | 0.26 | 0.56 | 0.40 | 0.30 |
| S (%) | 0.05 | 5.40 | 4.62 | 0.05 | 3.01 | 2.52 | 0.04 | 8.77 | 7.20 |
| Na (mg/kg) | 3662 | 3005 | 2894 | 3548 | 2866 | 2884 | 3648 | 2969 | 3006 |
| Zn (mg/kg) | 124 | 72 | 63 | 132 | 102 | 88 | 143 | 93 | 86 |
| B (mg/kg) | 91 | 67 | 54 | 95 | 77 | 64 | 100 | 73 | 70 |
| Ba (mg/kg) | 466 | 256 | 253 | 505 | 334 | 194 | 514 | 218 | 333 |
| Cd (mg/kg) | 2 | 0.98 | 0.86 | 2 | 1 | 0.99 | 2 | 1 | 1 |
| Co (mg/kg) | 4 | 3 | 2 | 4 | 3 | 3 | 4 | 3 | 3 |
| Cr (mg/kg) | 29 | 19 | 15 | 31 | 22 | 19 | 30 | 21 | 18 |
| Cu (mg/kg) | 61 | 45 | 36 | 70 | 47 | 43 | 71 | 46 | 43 |
| Mn (mg/kg) | 1894 | 1265 | 1090 | 1810 | 1473 | 1228 | 1951 | 1456 | 1321 |
| Mo (mg/kg) | 2 | 2 | 1 | 2 | 1 | 1 | 2 | 1 | 1 |
| Ni (mg/kg) | 19 | 11 | 9 | 21 | 14 | 12 | 21 | 13 | 12 |
| Pb (mg/kg) | 21 | 18 | 20 | 27 | 18 | 24 | 30 | 18 | 22 |
| V (mg/kg) | 21 | 14 | 11 | 23 | 17 | 15 | 22 | 16 | 15 |
| Parameters | Nitrogen N, % | Carbon, C, % | Hydrogen H, % | C:H (Atomic) | C:N (Atomic) | C:S (Atomic) |
|---|---|---|---|---|---|---|
| C 1.1 | 0.27 | 10.74 | 0.95 | 0.95 | 46.39 | 573.44 |
| C 1.1.1 | 0.28 | 9.46 | 1.12 | 0.71 | 39.40 | 4.68 |
| C 1.1.3 | 0.23 | 8.73 | 0.98 | 0.75 | 44.26 | 5.04 |
| C 1.2 | 0.28 | 11.04 | 1.01 | 0.92 | 45.98 | 589.46 |
| C 1.2.1 | 0.25 | 9.23 | 1.55 | 0.50 | 43.06 | 8.19 |
| C 1.2.3 | 0.22 | 10.76 | 1.28 | 0.71 | 57.04 | 11.40 |
| C 1.3 | 0.19 | 9.36 | 1.18 | 0.67 | 57.45 | 624.69 |
| C 1.3.1 | 0.27 | 8.46 | 0.82 | 0.87 | 36.54 | 2.58 |
| C 1.3.3 | 0.23 | 9.65 | 1.35 | 0.60 | 48.93 | 3.58 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Serafimova, E.; Petkova, V.; Petkova, V. From Farm to Table and Back Again: Circular Valorization of Biomass Ash and Sewage Sludge into Sustainable Material Blends. Materials 2026, 19, 1552. https://doi.org/10.3390/ma19081552
Serafimova E, Petkova V, Petkova V. From Farm to Table and Back Again: Circular Valorization of Biomass Ash and Sewage Sludge into Sustainable Material Blends. Materials. 2026; 19(8):1552. https://doi.org/10.3390/ma19081552
Chicago/Turabian StyleSerafimova, Ekaterina, Vilma Petkova, and Veneta Petkova. 2026. "From Farm to Table and Back Again: Circular Valorization of Biomass Ash and Sewage Sludge into Sustainable Material Blends" Materials 19, no. 8: 1552. https://doi.org/10.3390/ma19081552
APA StyleSerafimova, E., Petkova, V., & Petkova, V. (2026). From Farm to Table and Back Again: Circular Valorization of Biomass Ash and Sewage Sludge into Sustainable Material Blends. Materials, 19(8), 1552. https://doi.org/10.3390/ma19081552








