Author Contributions
Conceptualization, R.Z. and Z.W.; methodology, R.Z., J.S. and Y.W.; software, R.Z., Y.Z., K.Z. and J.W.; validation, R.Z.; formal analysis, H.W.; investigation, R.Z., Y.Z. and C.Z.; data curation, Y.Z.; writing—original draft, Y.Z.; writing—review and editing, R.Z. All authors have read and agreed to the published version of the manuscript.
Figure 1.
Crystal structures: (a) Al; (b) 3C-SiC; (c) 4H-SiC.
Figure 1.
Crystal structures: (a) Al; (b) 3C-SiC; (c) 4H-SiC.
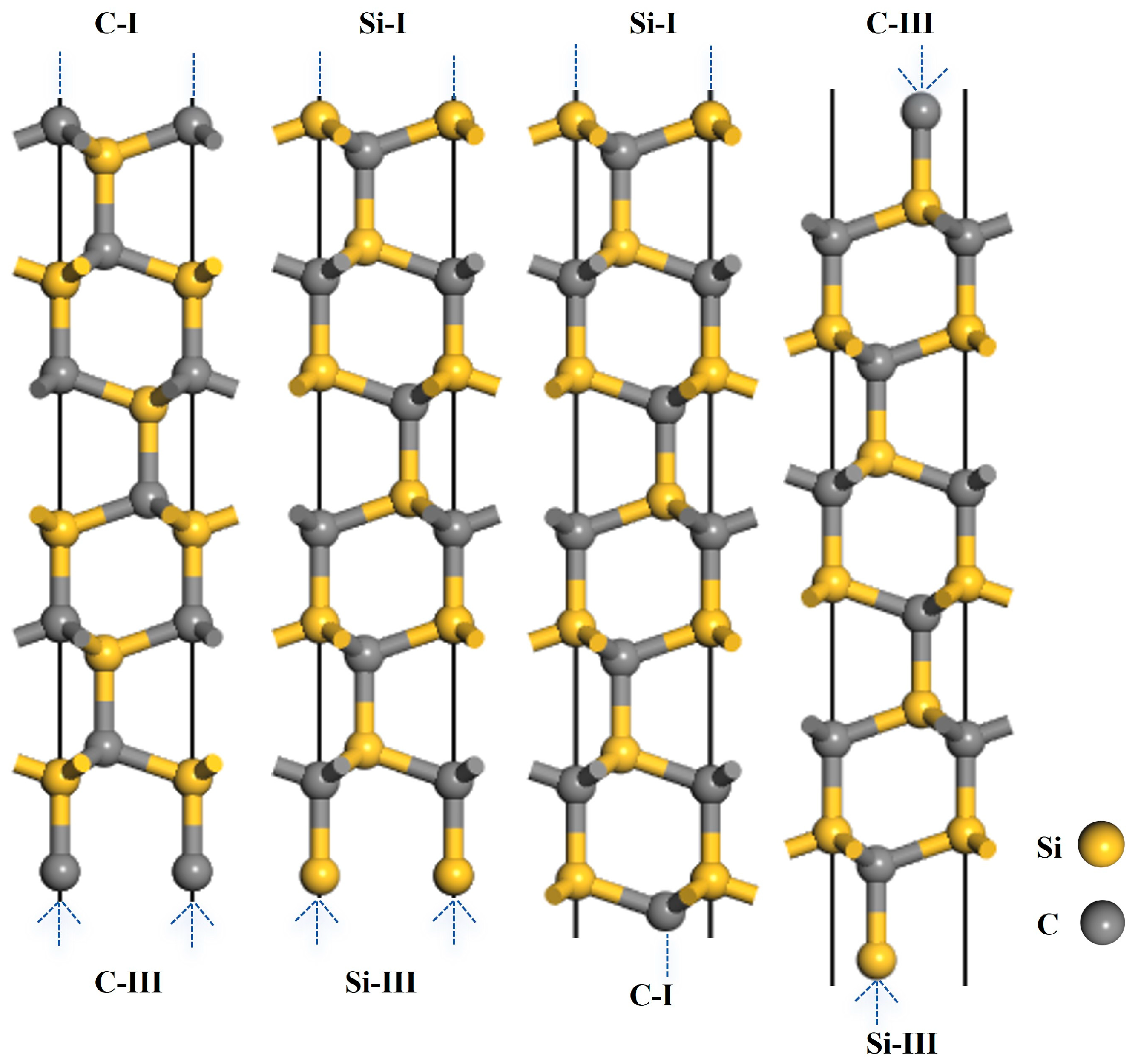
Figure 2.
Structural models of the 3C-SiC(111) slabs.
Figure 2.
Structural models of the 3C-SiC(111) slabs.
Figure 3.
Structural models of the 4H-SiC(0001) slabs.
Figure 3.
Structural models of the 4H-SiC(0001) slabs.
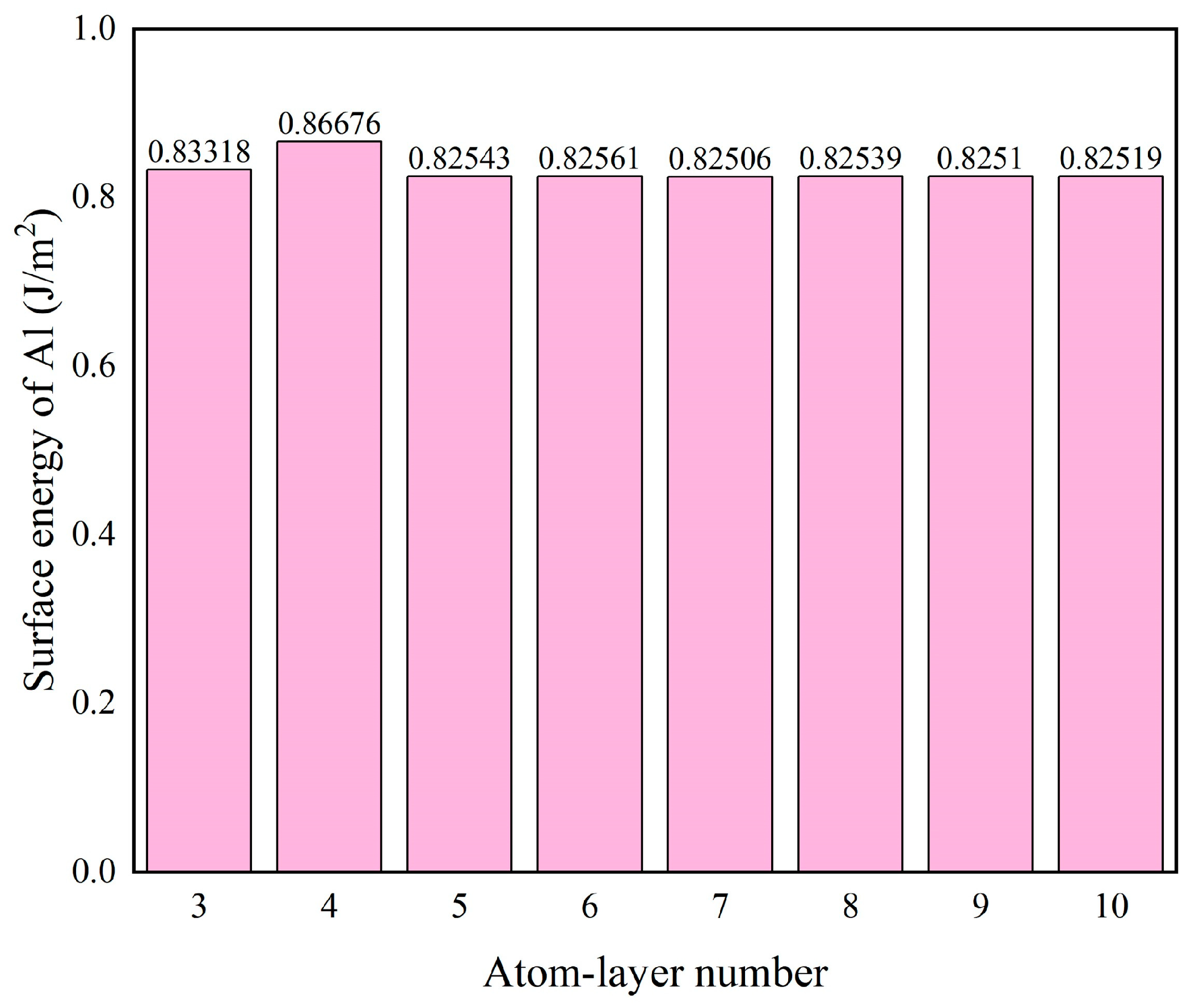
Figure 4.
Surface energy of the Al(111) surface as a function of atomic layer number.
Figure 4.
Surface energy of the Al(111) surface as a function of atomic layer number.
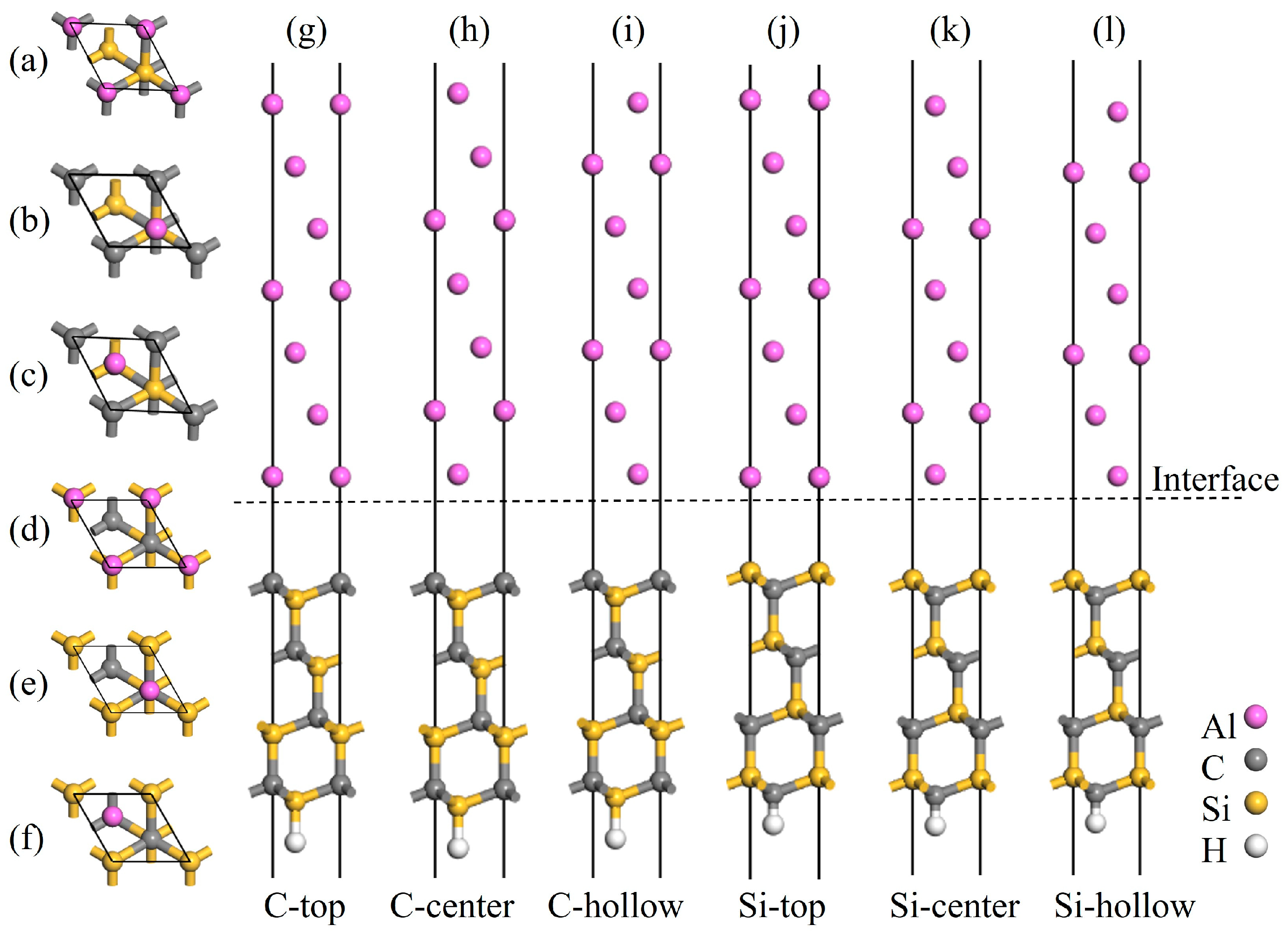
Figure 5.
Atomic structures of the C- and Si-terminated 3C-SiC(111)/Al(111) interface models: (a,g) C-top site, (b,h) C-center site, (c,i) C-hollow site, (d,j) Si-top site, (e,k) Si-center site, and (f,l) Si-hollow site.
Figure 5.
Atomic structures of the C- and Si-terminated 3C-SiC(111)/Al(111) interface models: (a,g) C-top site, (b,h) C-center site, (c,i) C-hollow site, (d,j) Si-top site, (e,k) Si-center site, and (f,l) Si-hollow site.
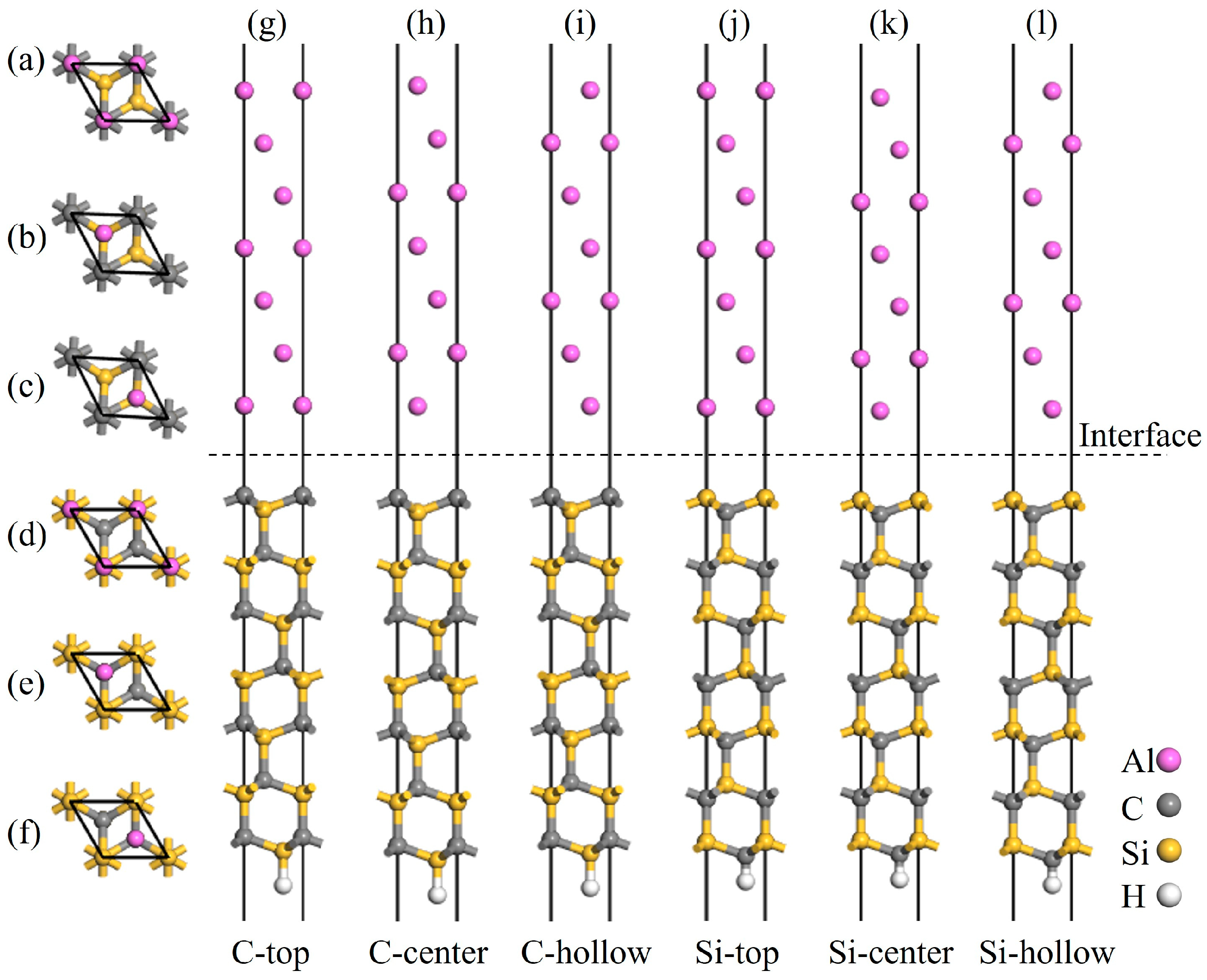
Figure 6.
Atomic structures of the C- and Si-terminated 4H-SiC(0001)/Al(111) interface models: (a,g) C-top site, (b,h) C-center site, (c,i) C-hollow site, (d,j) Si-top site, (e,k) Si-center site, and (f,l) Si-hollow site.
Figure 6.
Atomic structures of the C- and Si-terminated 4H-SiC(0001)/Al(111) interface models: (a,g) C-top site, (b,h) C-center site, (c,i) C-hollow site, (d,j) Si-top site, (e,k) Si-center site, and (f,l) Si-hollow site.
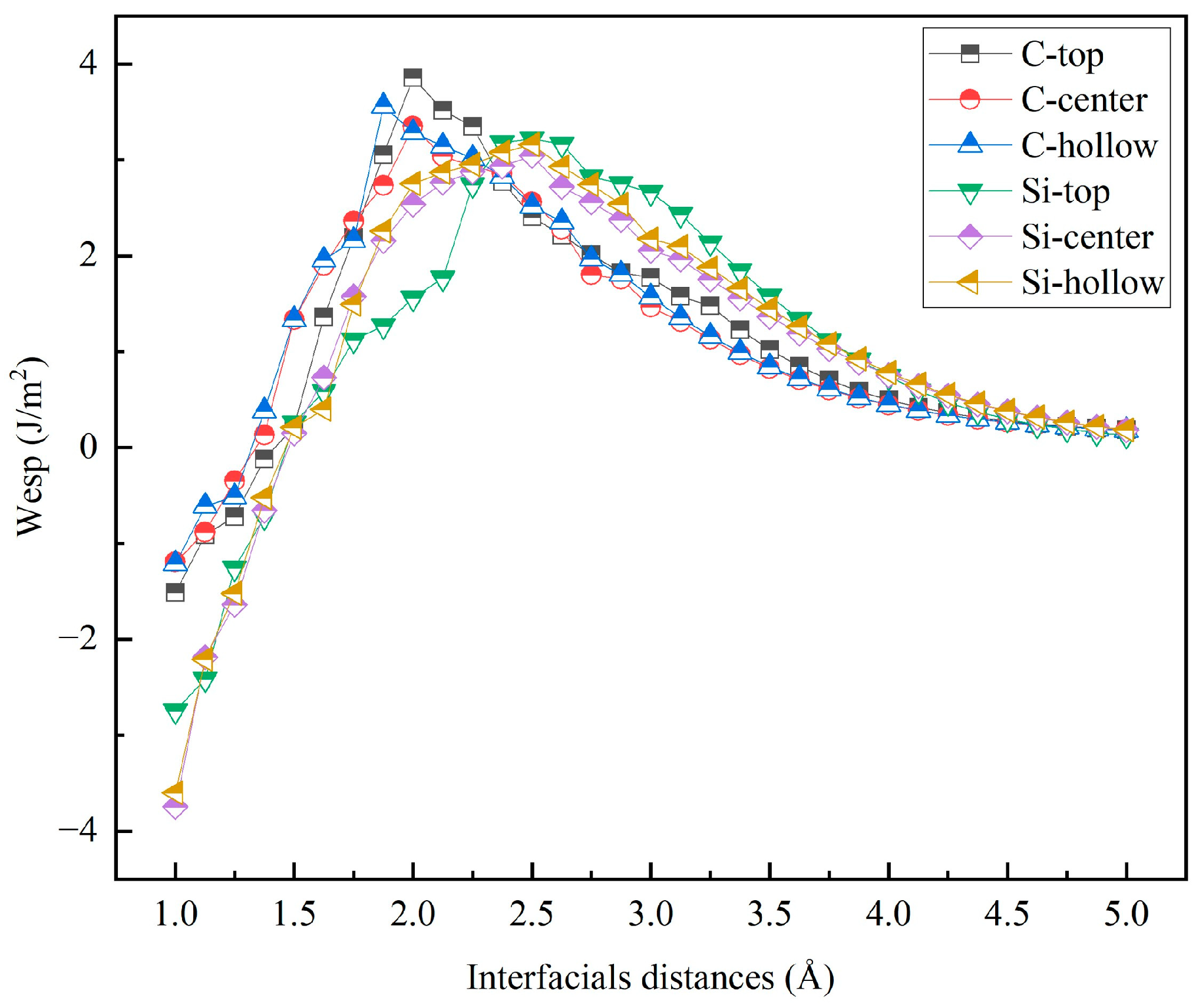
Figure 7.
Work of separation (Wsep) as a function of interfacial distance (d0) for the unrelaxed 3C-SiC(111)/Al(111) interfaces.
Figure 7.
Work of separation (Wsep) as a function of interfacial distance (d0) for the unrelaxed 3C-SiC(111)/Al(111) interfaces.
Figure 8.
Work of separation (Wsep) as a function of interfacial distance (d0) for the unrelaxed 4H-SiC(0001)/Al(111) interfaces.
Figure 8.
Work of separation (Wsep) as a function of interfacial distance (d0) for the unrelaxed 4H-SiC(0001)/Al(111) interfaces.
Figure 9.
Electronic properties at the 3C-SiC(111)/Al(111) interface for six interfacial configurations: (a) valence electron density distribution; (b) charge density difference.
Figure 9.
Electronic properties at the 3C-SiC(111)/Al(111) interface for six interfacial configurations: (a) valence electron density distribution; (b) charge density difference.
Figure 10.
Electronic properties at the 4H-SiC(0001)/Al(111) interface for six interfacial configurations: (a) valence electron density distribution; (b) charge density difference.
Figure 10.
Electronic properties at the 4H-SiC(0001)/Al(111) interface for six interfacial configurations: (a) valence electron density distribution; (b) charge density difference.
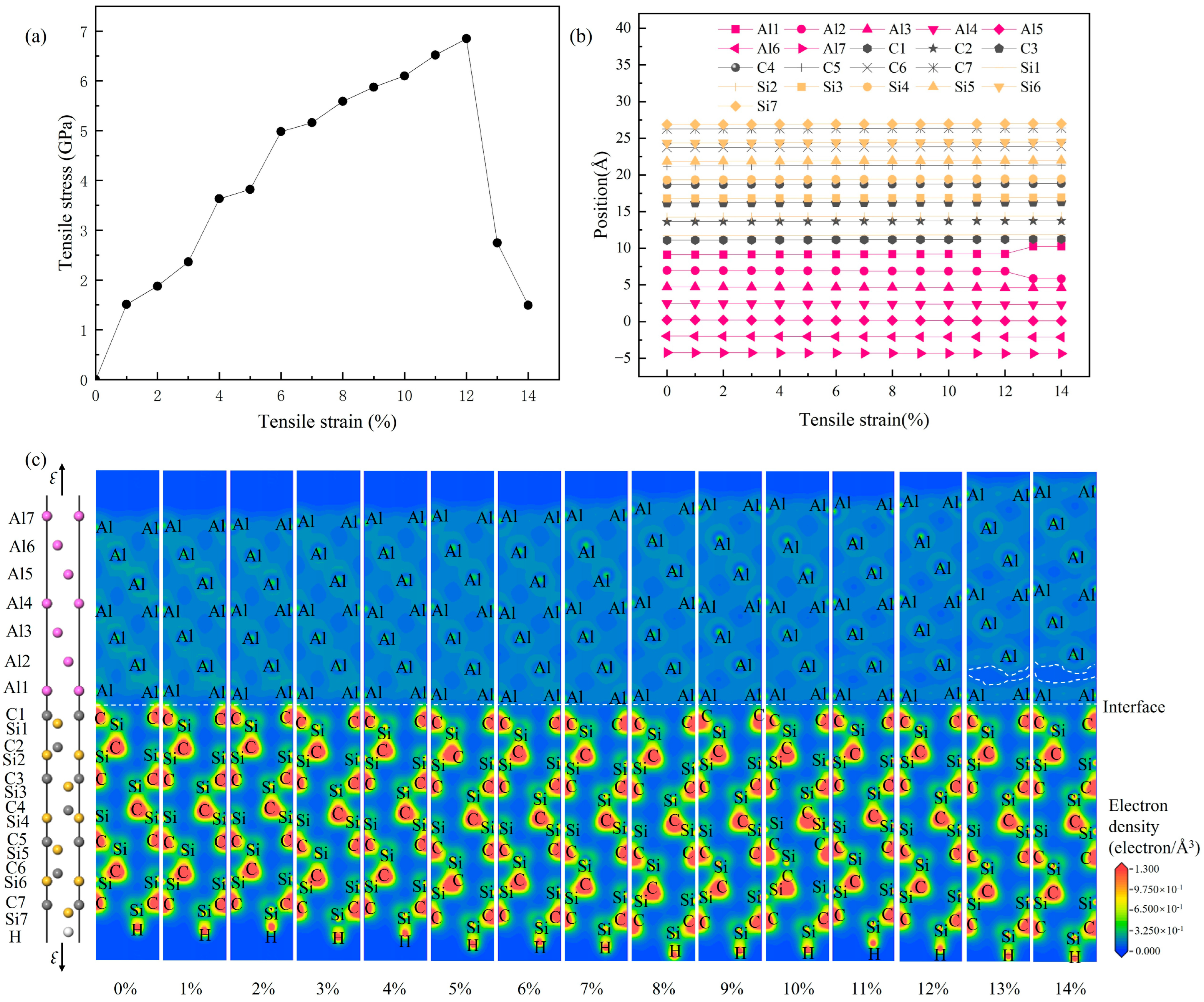
Figure 11.
Tensile failure process of the 3C-SiC(111)/Al(111) interface (C-top configuration): (a) stress–strain curves; (b) evolution of atomic positions with strain; (c) evolution of valence electron density with strain.
Figure 11.
Tensile failure process of the 3C-SiC(111)/Al(111) interface (C-top configuration): (a) stress–strain curves; (b) evolution of atomic positions with strain; (c) evolution of valence electron density with strain.
Figure 12.
Tensile failure process of the 4H-SiC(0001)/Al(111) interface (C-top configuration): (a) stress–strain curves; (b) evolution of atomic positions with strain; (c) evolution of valence electron density with strain.
Figure 12.
Tensile failure process of the 4H-SiC(0001)/Al(111) interface (C-top configuration): (a) stress–strain curves; (b) evolution of atomic positions with strain; (c) evolution of valence electron density with strain.
Table 1.
Comparison of calculated lattice constants of Al and SiC with existing theoretical and experimental data.
Table 1.
Comparison of calculated lattice constants of Al and SiC with existing theoretical and experimental data.
| | Methods | Lattice Parameters |
|---|
| a (Å) | b (Å) | c (Å) |
|---|
| Al | Present study (GGA-PBE) | 4.045 | 4.045 | 4.045 |
| Exp [27] | 4.050 | 4.050 | 4.050 |
| Calc [18] (GGA-PBE) | 4.044 | 4.044 | 4.044 |
| Calc [28] (GGA-PW91) | 4.050 | 4.050 | 4.050 |
| 3C-SiC | Present study (GGA-PBE) | 4.379 | 4.379 | 4.379 |
| Exp [29] | 4.369 | 4.369 | 4.369 |
| Calc [30] (GGA-PBE) | 4.366 | 4.366 | 4.366 |
| Calc [31] (GGA-PW91) | 4.348 | 4.348 | 4.348 |
| 4H-SiC | Present study (GGA-PBE) | 3.095 | 3.095 | 10.121 |
| Exp [32] | 3.079 | 3.079 | 10.254 |
| Calc [30] (GGA-PBE) | 3.084 | 3.084 | 10.096 |
| Calc [33] (LDA-CAPZ) | 3.089 | 3.089 | 10.113 |
Table 2.
Relative change in interlayer spacing after relaxation of the 3C-SiC (111) surface: effects of termination and slab thickness.
Table 2.
Relative change in interlayer spacing after relaxation of the 3C-SiC (111) surface: effects of termination and slab thickness.
| Termination | Change in Interlayer | Slab Thickness |
| | | 5 | 7 | 9 | 11 | 13 |
| | Si-III | Si-I | Si-III | Si-I | Si-III | Si-I | Si-III | Si-I | Si-III | Si-I |
| Si | Δd12 (%) | 2.01 | −2.20 | 1.42 | −20.7 | 0.950 | −19.6 | 0.74 | −19.6 | 0.58 | −19.1 |
| Δd23 (%) | −8.85 | 4.48 | −4.43 | 2.85 | −3.15 | 2.40 | −1.70 | 2.27 | −1.58 | 2.16 |
| Δd34 (%) | | | 1.74 | −5.06 | 1.05 | −3.80 | 0.74 | −3.48 | 0.53 | −3.16 |
| Δd45 (%) | | | | | −2.69 | 1.16 | −1.90 | 1.05 | −1.74 | 0.95 |
| Δd56 (%) | | | | | | | 0.74 | 1.74 | 0.42 | −1.74 |
| Δd67 (%) | | | | | | | | | −1.42 | 0.53 |
| Termination | Change in interlayer | Slab thickness |
| | | 5 | 7 | 9 | 11 | 13 |
| | C-III | C-I | C-III | C-I | C-III | C-I | C-III | C-I | C-III | C-I |
| C | Δd12 (%) | 0.95 | −49.7 | 1.27 | −42.6 | 1.21 | −42.9 | 1.16 | −42.9 | 1.11 | −42.9 |
| Δd23 (%) | −13.1 | 11.3 | −1.27 | 5.17 | −0.16 | 5.29 | 0.16 | 5.60 | 0.16 | 5.64 |
| Δd34 (%) | | | 1.53 | −7.74 | 0.31 | −6.31 | 0.16 | −6.16 | 0 | −5.84 |
| Δd45 (%) | | | | | −0.94 | −0.89 | 0.31 | 0.90 | 0 | 1.00 |
| Δd56 (%) | | | | | | | 0.21 | −0.31 | 0 | −0.16 |
| Δd67 (%) | | | | | | | | | 0.63 | 0.21 |
Table 3.
Relative change in interlayer spacing after relaxation of the 4H-SiC(0001) surface: effects of termination and slab thickness.
Table 3.
Relative change in interlayer spacing after relaxation of the 4H-SiC(0001) surface: effects of termination and slab thickness.
| Termination | Change in Interlayer | Slab Thickness |
| | | 9 | 11 | 13 | 15 | 17 |
| | Si-III | Si-I | Si-III | Si-I | Si-III | Si-I | Si-III | Si-I | Si-III | Si-I |
| Si | Δd12 (%) | 0.89 | −9.0 | 0.31 | −8.03 | 0.68 | −7.06 | 0.16 | −7.38 | 0.53 | −7.22 |
| Δd23 (%) | −2.53 | 2.36 | −1.12 | 2.05 | −1.58 | 2.05 | −0.64 | 1.89 | −1.11 | 1.89 |
| Δd34 (%) | 0.73 | −3.32 | 0.53 | −2.37 | 0.42 | −2.37 | 0.37 | −2.06 | 0.37 | −2.06 |
| Δd45 (%) | −2.40 | 1.00 | −1.42 | 0.84 | −1.28 | 0.74 | −0.95 | 0.63 | −0.64 | 0.58 |
| Δd56 (%) | | | 0.37 | −3.01 | 0.263 | −1.28 | 0.21 | −0.96 | 0.21 | −0.80 |
| Δd67 (%) | | | | | −1.27 | 0.31 | −0.80 | 0.21 | −0.79 | 0.21 |
| Δd78 (%) | | | | | | | 0.21 | −0.79 | 0.26 | −0.79 |
| Termination | Change in interlayer | Slab thickness |
| | | 9 | 11 | 13 | 15 | 17 |
| | C-III | C-I | C-III | C-I | C-III | C-I | C-III | C-I | C-III | C-I |
| C | Δd12 (%) | 1.16 | −38.3 | 0.79 | −44.9 | 1.11 | −45.1 | 0.68 | −45.1 | 1.05 | −44.9 |
| Δd23 (%) | 0.96 | 5.51 | 0.31 | 5.67 | 1.44 | 5.67 | −0.16 | 5.61 | 1.28 | 5.72 |
| Δd34 (%) | 0.52 | −11.2 | 0.26 | −9.63 | 0.16 | −9.47 | 0.05 | −9.63 | 0.05 | −9.63 |
| Δd45 (%) | −1.10 | 1.16 | 0.80 | 1.21 | 0.47 | 1.16 | 0.80 | 1.10 | 0.47 | 1.26 |
| Δd56 (%) | | | 0.37 | −0.16 | −42.6 | −0.47 | 0.05 | 0.96 | −0.05 | −0.31 |
| Δd67 (%) | | | | | 0.48 | 0.26 | 0.79 | 0.05 | 0.48 | 0.31 |
| Δd78 (%) | | | | | | | 0.05 | −0.79 | 0 | 0.64 |
Table 4.
Calculated surface energies for various Al crystal planes.
Table 4.
Calculated surface energies for various Al crystal planes.
| Alsurf | Al(001) | Al(110) | Al(111) |
|---|
| Esurf (J/m2) | 0.954 | 1.035 | 0.825 |
| Calc [36] | 0.903 | 1.044 | 0.839 |
| Calc [20] | 1.024 | 1.073 | 0.936 |
Table 5.
Calculated parameters (Wsep and d0) for 3C-SiC(111)/Al(111) interfaces.
Table 5.
Calculated parameters (Wsep and d0) for 3C-SiC(111)/Al(111) interfaces.
| Termination | Unrelaxed | Relaxed |
|---|
| | d0 (Å) | Wsep (J/m2) | d0 (Å) | Wsep (J/m2) |
|---|
| C-top-SiC(111)/Al(111) | 2.000 | 3.427 | 1.984 | 3.640 |
| C-center-SiC(111)/Al(111) | 2.000 | 2.540 | 1.983 | 2.711 |
| C-hollow-SiC(111)/Al(111) | 2.000 | 2.424 | 1.659 | 2.536 |
| Si-top-SiC(111)/Al(111) | 2.750 | 2.439 | 2.539 | 2.513 |
| Si-center-SiC(111)/Al(111) | 2.500 | 2.569 | 2.261 | 2.584 |
| Si-hollow-SiC(111)/Al(111) | 2.500 | 2.480 | 2.308 | 2.537 |
Table 6.
Calculated parameters (Wsep and d0) for 4H-SiC(0001)/Al(111) interfaces.
Table 6.
Calculated parameters (Wsep and d0) for 4H-SiC(0001)/Al(111) interfaces.
| Interface | Unrelaxed | Relaxed |
|---|
| | d0 (Å) | Wsep (J/m2) | d0 (Å) | Wsep (J/m2) |
|---|
| C-top-SiC(111)/Al(111) | 2.000 | 3.855 | 1.988 | 3.971 |
| C-center-SiC(111)/Al(111) | 2.000 | 3.347 | 1.985 | 3.581 |
| C-hollow-SiC(111)/Al(111) | 1.875 | 3.561 | 1.713 | 3.751 |
| Si-top-SiC(111)/Al(111) | 2.500 | 3.221 | 2.533 | 3.422 |
| Si-center-SiC(111)/Al(111) | 2.500 | 3.042 | 2.294 | 3.103 |
| Si-hollow-SiC(111)/Al(111) | 2.500 | 3.162 | 2.288 | 3.275 |
Table 7.
Mulliken population analysis of atoms for the C- and Si-top terminations of the 3C-SiC(111)/Al(111) interface.
Table 7.
Mulliken population analysis of atoms for the C- and Si-top terminations of the 3C-SiC(111)/Al(111) interface.
| Type | Atoms | s | p | d | Total | Charge | Populations |
|---|
| C-top | Al | 1.01 | 1.70 | 0.00 | 2.72 | +0.28 | 0.25 |
| C | 1.45 | 3.80 | 0.00 | 5.25 | −1.25 |
| Si-top | Al | 1.16 | 1.89 | 0.00 | 3.05 | −0.05 | 0.33 |
| Si | 1.08 | 1.99 | 0.00 | 3.07 | +0.93 |
Table 8.
Mulliken population analysis of atoms for the C- and Si-top terminations of the 4H-SiC(0001)/Al(111) interface.
Table 8.
Mulliken population analysis of atoms for the C- and Si-top terminations of the 4H-SiC(0001)/Al(111) interface.
| Type | Atoms | s | p | d | Total | Charge | Populations |
|---|
| C-top | Al | 1.02 | 1.70 | 0.00 | 2.72 | +0.28 | 0.26 |
| C | 1.45 | 3.80 | 0.00 | 5.25 | −1.25 |
| Si-top | Al | 1.15 | 1.89 | 0.00 | 3.05 | −0.05 | 0.35 |
| Si | 1.09 | 1.98 | 0.00 | 3.07 | +0.93 |
Table 9.
Comparison of work of separation, ultimate tensile strength, and ultimate tensile strain for the 3C- and 4H-SiC/Al interfaces.
Table 9.
Comparison of work of separation, ultimate tensile strength, and ultimate tensile strain for the 3C- and 4H-SiC/Al interfaces.
| Interface | Methods | Work of Separation (J/m2) | Ultimate Tensile Strength (GPa) | Ultimate Tensile Strain (%) |
|---|
| 3C-SiC(111)/Al(111) | Present study (GGA-PBE) | 3.640 (C-top) | 6.603 (C-top) | 10 (C-top) |
| 4H-SiC(0001)/Al(111) | Present study (GGA-PBE) | 3.971 (C-top) | 6.851 (C-top) | 12 (C-top) |
| 3C-SiC(111)/Al(111) | Calc [19] (GGA-PBE) | 3.576 (C-top) | 6.33 (C-top) | 10 (C-top) |
| 4H-SiC(0001)/Al(111) | Calc [18] (GGA-PBE) | 3.44 (C-top) | - | - |
| 6H-SiC(0001)/Al(111) | Calc [40] (GGA-PBE) | 5.09 (C-top) | 3.90 (C-top) | 10 (C-top) |
| 6H-SiC(0001)/Al(111) | Calc [20] (GGA-PBE) | 2.689 (C-top) | 5.60 (C-top) | 9 (C-top) |
| 6H-SiC(0001)/Al(111) | Calc [41] (GGA-PBE) | - | 4.27 (C-top) | 11 (C-top) |
Table 10.
Mechanical properties of SiC/Al composites.
Table 10.
Mechanical properties of SiC/Al composites.
| Materials | Yielding Strength (MPa) | Ultimate Tensile Strength (MPa) |
|---|
| 9vol%SiCp/2009Al [42] | 346.3 ± 3.0 | ≈510 |
| 17vol%SiCp/2009Al [43] | 141.1 | 214.4 |
| 25vol%SiCp/2009Al [42] | 395.8 ± 3.2 | ≈550 |