Novel Ti6Al4V Surface Treatment for Subperiosteal Dental Implants (Part II): Matrix Deposition and Osteogenic Markers
Abstract
1. Introduction
2. Materials and Methods
2.1. Atomic Force Microscope (AFM)
2.2. Experimental Design
2.3. Cell Culture
2.4. Scanning Electron Microscopy (SEM) and Energy Dispersive X-Ray Spectroscopy (EDS)
2.5. Western Blot Analyses
2.6. Fluorescence Microscopy
2.7. Statistical Analysis
3. Results
3.1. Atomic Force Microscope (AFM)
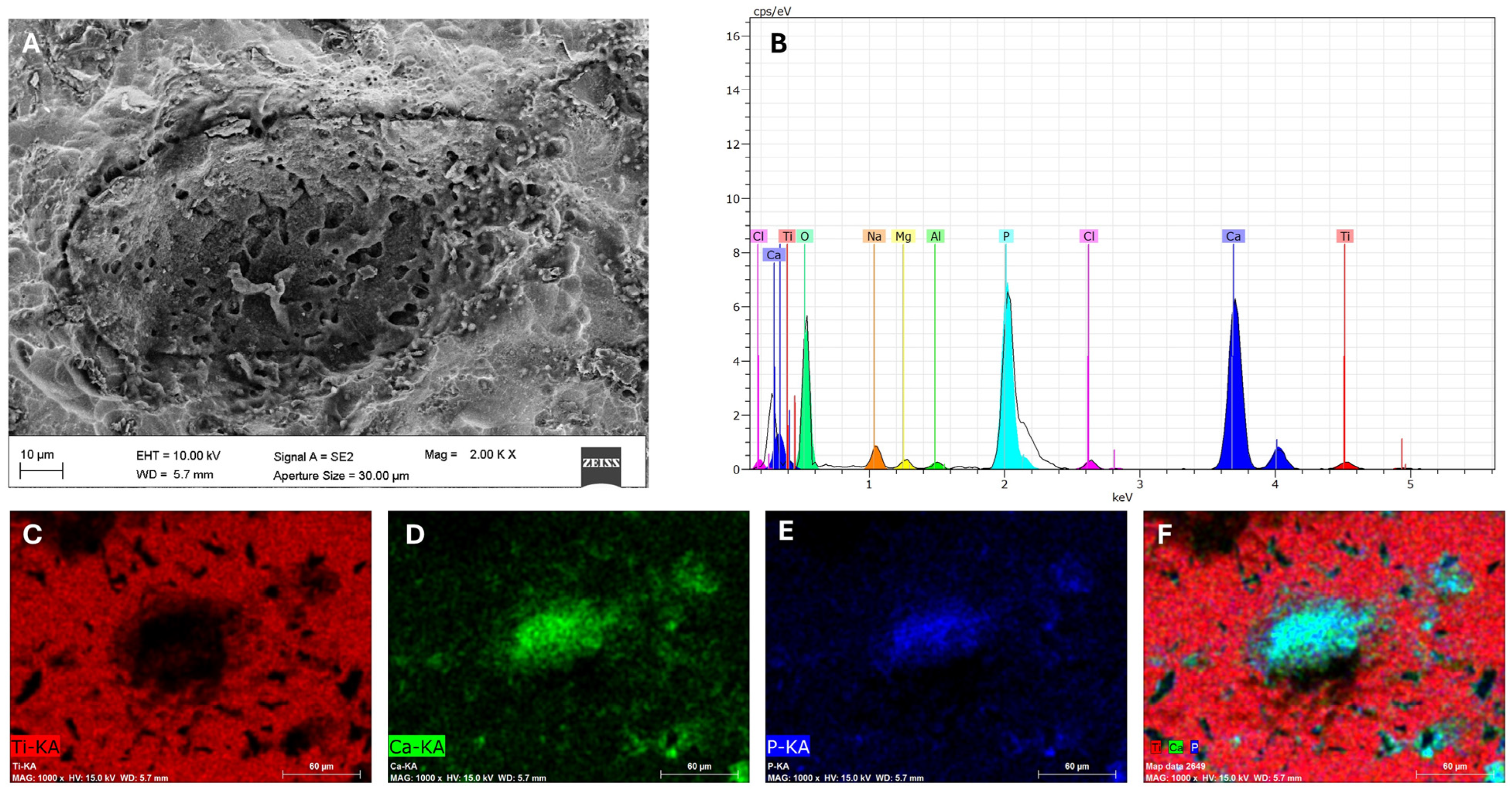
3.2. Scanning Electron Microscopy (SEM) and Energy Dispersive X-Ray Spectroscopy (EDS)
3.3. Evaluation of Markers of Osteogenic Differentiation and Extracellular Matrix (ECM) Formation
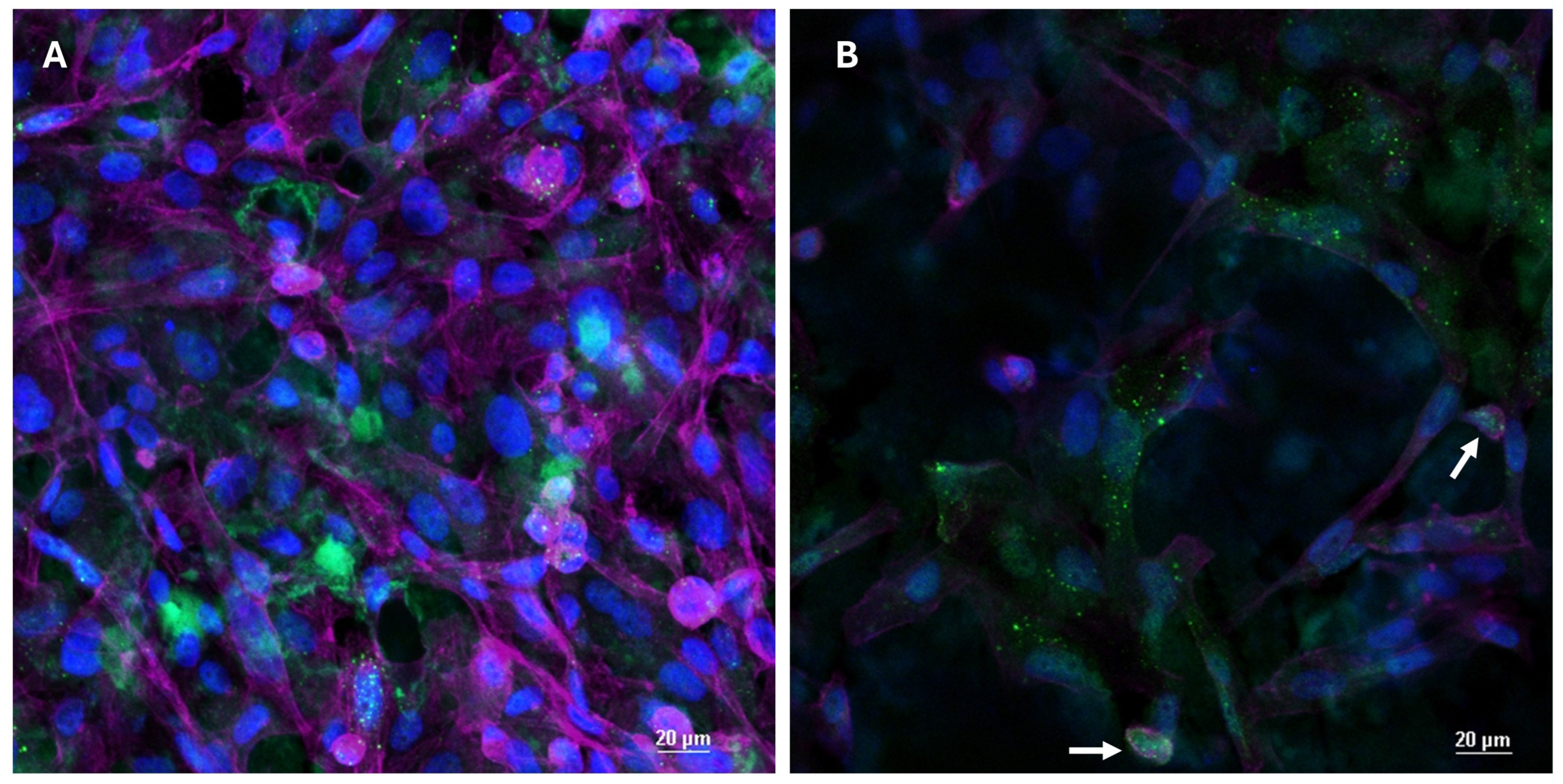
3.4. DMP1 Immunofluorescence and Confocal Analysis for Subcellular Localization
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AFM | Atomic Force Microscope |
| ATcs | Al Ti Color surface |
| CA | Colour Anodizing |
| CBCT | Cone-beam computed tomography |
| COL1A1 | Collagen Type I Alpha 1 Chain |
| CTRL | Control |
| DMP1 | Dentin Matrix Protein 1 |
| ECM | Extracellular Matrix |
| EE | Polished via Electroerosion |
| ES | Etching and Sandblasting |
| PMSF | Phenylmethylsulfonyl fluoride |
| PVDF | Polyvinylidene fluoride |
| RMS | Root Mean Square |
| SD | Standard Deviation |
| SDS-PAGE | Sodium dodecyl sulfate–polyacrylamide gels |
| SEM-EDS | Scanning Electron Microscopy coupled with Energy-Dispersive Spectroscopy |
| SPARC | Secreted Protein Acidic and Rich in Cysteine |
References
- Javed, F.; Ahmed, H.B.; Crespi, R.; Romanos, G.E. Role of primary stability for successful osseointegration of dental implants: Factors of influence and evaluation. Interv. Med. Appl. Sci. 2013, 5, 162–167. [Google Scholar] [CrossRef] [PubMed]
- Albrektsson, T.; Chrcanovic, B.; Ostman, P.O.; Sennerby, L. Initial and long-term crestal bone responses to modern dental implants. Periodontol. 2000 2017, 73, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Kupka, J.R.; Konig, J.; Al-Nawas, B.; Sagheb, K.; Schiegnitz, E. How far can we go? A 20-year meta-analysis of dental implant survival rates. Clin. Oral Investig. 2024, 28, 541. [Google Scholar] [CrossRef]
- Di Stefano, D.A.; Arosio, P.; Cappare, P.; Barbon, S.; Gherlone, E.F. Stability of Dental Implants and Thickness of Cortical Bone: Clinical Research and Future Perspectives. A Systematic Review. Materials 2021, 14, 7183. [Google Scholar] [CrossRef] [PubMed]
- Steigenga, J.T.; Al-Shammari, K.F.; Nociti, F.H.; Misch, C.E.; Wang, H.L. Dental implant design and its relationship to long-term implant success. Implant Dent. 2003, 12, 306–317. [Google Scholar] [CrossRef]
- El-Sawy, M.A.; Hegazy, S.A. Subperiosteal implants constructed with digital technology: A systematic review. Oral Maxillofac. Surg. 2024, 28, 1063–1075. [Google Scholar] [CrossRef]
- Goh, R.; Vaquette, C.; Breik, O.; Ivanovski, S.; Batstone, M. Subperiosteal Implants: A Lost Art Worth Revisiting? Clin. Implant Dent. Relat. Res. 2025, 27, e70025. [Google Scholar] [CrossRef]
- Gellrich, N.C.; Korn, P.; Neuhaus, M.; Lentge, F.; Jehn, P.; Rahlf, B. Long-Term Survival of Subperiosteal Implants: Meta-Analysis and Current Status of Subperiosteal Implants for Dental Rehabilitation. Oral Maxillofac. Surg. Clin. N. Am. 2025, 37, 163–177. [Google Scholar] [CrossRef]
- Ayhan, M.; Cankaya, A.B. Custom-made Subperiosteal Implants: A Finite Element Analysis on Monoblock and Dual Implant Systems in Atrophic Maxilla. Int. J. Med. Sci. 2023, 20, 1755–1762. [Google Scholar] [CrossRef]
- Strappa, E.M.; Meme, L.; Cerea, M.; Roy, M.; Bambini, F. Custom-made additively manufactured subperiosteal implant. Minerva Dent. Oral Sci. 2022, 71, 353–360. [Google Scholar] [CrossRef]
- Anitua, E.; Eguia, A.; Staudigl, C.; Alkhraisat, M.H. Clinical performance of additively manufactured subperiosteal implants: A systematic review. Int. J. Implant Dent. 2024, 10, 4. [Google Scholar] [CrossRef] [PubMed]
- Azar, R.-R.; Azar, E.-R.; Ardelean, A.-V.; Stoian, A.-D.; Boici, G.; Sinescu, C.; Negruțiu, M.-L. Revisiting Subperiosteal Implants: A Narrative Review of the Contemporary Literature. Proceedings 2025, 127, 29. [Google Scholar] [CrossRef]
- Bordbar-Khiabani, A.; Gasik, M. Electrochemical behavior of additively manufactured patterned titanium alloys under simulated normal, inflammatory, and severe inflammatory conditions. J. Mater. Res. Technol. 2023, 26, 356–370. [Google Scholar] [CrossRef]
- Loginoff, J.; Majos, A.; Elgalal, M. The Evolution of Custom Subperiosteal Implants for Treatment of Partial or Complete Edentulism in Patients with Severe Alveolar Ridge Atrophy. J. Clin. Med. 2024, 13, 3582. [Google Scholar] [CrossRef]
- Kouhi, M.; de Souza Araujo, I.J.; Asa’ad, F.; Zeenat, L.; Bojedla, S.S.R.; Pati, F.; Zolfagharian, A.; Watts, D.C.; Bottino, M.C.; Bodaghi, M. Recent advances in additive manufacturing of patient-specific devices for dental and maxillofacial rehabilitation. Dent. Mater. 2024, 40, 700–715. [Google Scholar] [CrossRef]
- Vishnu, J.; Kesavan, P.; Shankar, B.; Dembinska, K.; Swiontek Brzezinska, M.; Kaczmarek-Szczepanska, B. Engineering Antioxidant Surfaces for Titanium-Based Metallic Biomaterials. J. Funct. Biomater. 2023, 14, 344. [Google Scholar] [CrossRef]
- Li, J.; Zheng, Y.; Yu, Z.; Kankala, R.K.; Lin, Q.; Shi, J.; Chen, C.; Luo, K.; Chen, A.; Zhong, Q. Surface-modified titanium and titanium-based alloys for improved osteogenesis: A critical review. Heliyon 2024, 10, e23779. [Google Scholar] [CrossRef]
- Campagna, R.; Schiavoni, V.; Rao, L.; Bambini, F.; Frontini, A.; Sampalmieri, F.; Salvolini, E.; Meme, L. Novel Ti6Al4V Surface Treatment for Subperiosteal Dental Implants: Evaluation of Osteoblast-like Cell Proliferation and Osteogenic Response. Materials 2025, 18, 1234. [Google Scholar] [CrossRef]
- Nečas, D.; Klapetek, P. Gwyddion: An open-source software for SPM data analysis. Cent. Eur. J. Phys. 2012, 10, 181–188. [Google Scholar] [CrossRef]
- Serritelli, E.N.; Sartini, D.; Campagna, R.; Pozzi, V.; Martin, N.I.; van Haren, M.J.; Salvolini, E.; Cecati, M.; Rubini, C.; Emanuelli, M. Targeting nicotinamide N-methyltransferase decreased aggressiveness of osteosarcoma cells. Eur. J. Clin. Investig. 2024, 54, e14185. [Google Scholar] [CrossRef]
- Eliaz, N.; Metoki, N. Calcium Phosphate Bioceramics: A Review of Their History, Structure, Properties, Coating Technologies and Biomedical Applications. Materials 2017, 10, 334. [Google Scholar] [CrossRef] [PubMed]
- Addison, W.N.; Nelea, V.; Chicatun, F.; Chien, Y.C.; Tran-Khanh, N.; Buschmann, M.D.; Nazhat, S.N.; Kaartinen, M.T.; Vali, H.; Tecklenburg, M.M.; et al. Extracellular matrix mineralization in murine MC3T3-E1 osteoblast cultures: An ultrastructural, compositional and comparative analysis with mouse bone. Bone 2015, 71, 244–256. [Google Scholar] [CrossRef] [PubMed]
- Nitiputri, K.; Ramasse, Q.M.; Autefage, H.; McGilvery, C.M.; Boonrungsiman, S.; Evans, N.D.; Stevens, M.M.; Porter, A.E. Nanoanalytical Electron Microscopy Reveals a Sequential Mineralization Process Involving Carbonate-Containing Amorphous Precursors. ACS Nano 2016, 10, 6826–6835. [Google Scholar] [CrossRef] [PubMed]
- Nollet, M.; Santucci-Darmanin, S.; Breuil, V.; Al-Sahlanee, R.; Cros, C.; Topi, M.; Momier, D.; Samson, M.; Pagnotta, S.; Cailleteau, L.; et al. Autophagy in osteoblasts is involved in mineralization and bone homeostasis. Autophagy 2014, 10, 1965–1977. [Google Scholar] [CrossRef]
- Hasegawa, T.; Yamamoto, T.; Tsuchiya, E.; Hongo, H.; Tsuboi, K.; Kudo, A.; Abe, M.; Yoshida, T.; Nagai, T.; Khadiza, N.; et al. Ultrastructural and biochemical aspects of matrix vesicle-mediated mineralization. Jpn. Dent. Sci. Rev. 2017, 53, 34–45. [Google Scholar] [CrossRef]
- Selvaraj, V.; Sekaran, S.; Dhanasekaran, A.; Warrier, S. Type 1 collagen: Synthesis, structure and key functions in bone mineralization. Differentiation 2024, 136, 100757. [Google Scholar] [CrossRef]
- Huang, W.; Yang, S.; Shao, J.; Li, Y.P. Signaling and transcriptional regulation in osteoblast commitment and differentiation. Front. Biosci. 2007, 12, 3068–3092. [Google Scholar] [CrossRef]
- Twine, N.A.; Chen, L.; Pang, C.N.; Wilkins, M.R.; Kassem, M. Identification of differentiation-stage specific markers that define the ex vivo osteoblastic phenotype. Bone 2014, 67, 23–32. [Google Scholar] [CrossRef]
- Kalajzic, I.; Kalajzic, Z.; Hurley, M.M.; Lichtler, A.C.; Rowe, D.W. Stage specific inhibition of osteoblast lineage differentiation by FGF2 and noggin. J. Cell Biochem. 2003, 88, 1168–1176. [Google Scholar] [CrossRef]
- Rosset, E.M.; Bradshaw, A.D. SPARC/osteonectin in mineralized tissue. Matrix Biol. 2016, 52–54, 78–87. [Google Scholar] [CrossRef]
- Zhu, Y.S.; Gu, Y.; Jiang, C.; Chen, L. Osteonectin regulates the extracellular matrix mineralization of osteoblasts through P38 signaling pathway. J. Cell. Physiol. 2020, 235, 2220–2231. [Google Scholar] [CrossRef] [PubMed]
- Mundlos, S.; Schwahn, B.; Reichert, T.; Zabel, B. Distribution of osteonectin mRNA and protein during human embryonic and fetal development. J. Histochem. Cytochem. 1992, 40, 283–291. [Google Scholar] [CrossRef] [PubMed]
- Delany, A.M.; Kalajzic, I.; Bradshaw, A.D.; Sage, E.H.; Canalis, E. Osteonectin-null mutation compromises osteoblast formation, maturation, and survival. Endocrinology 2003, 144, 2588–2596. [Google Scholar] [CrossRef] [PubMed]
- Gittens, R.A.; Olivares-Navarrete, R.; Cheng, A.; Anderson, D.M.; McLachlan, T.; Stephan, I.; Geis-Gerstorfer, J.; Sandhage, K.H.; Fedorov, A.G.; Rupp, F.; et al. The roles of titanium surface micro/nanotopography and wettability on the differential response of human osteoblast lineage cells. Acta Biomater. 2013, 9, 6268–6277. [Google Scholar] [CrossRef]
- Riedel, R.; Perez-Amodio, S.; Cabo-Zabala, L.; Velasco-Ortega, E.; Maymo, J.; Gil, J.; Monsalve-Guil, L.; Ortiz-Garcia, I.; Perez-Perez, A.; Sanchez-Margalet, V.; et al. Influence of the Surface Topography of Titanium Dental Implants on the Behavior of Human Amniotic Stem Cells. Int. J. Mol. Sci. 2024, 25, 7416. [Google Scholar] [CrossRef]
- Narayanan, K.; Ramachandran, A.; Hao, J.; He, G.; Park, K.W.; Cho, M.; George, A. Dual functional roles of dentin matrix protein 1. Implications in biomineralization and gene transcription by activation of intracellular Ca2+ store. J. Biol. Chem. 2003, 278, 17500–17508. [Google Scholar] [CrossRef]
- Kalajzic, I.; Braut, A.; Guo, D.; Jiang, X.; Kronenberg, M.S.; Mina, M.; Harris, M.A.; Harris, S.E.; Rowe, D.W. Dentin matrix protein 1 expression during osteoblastic differentiation, generation of an osteocyte GFP-transgene. Bone 2004, 35, 74–82. [Google Scholar] [CrossRef]
- Zhang, S.; Wan, H.; Wang, P.; Liu, M.; Li, G.; Zhang, C.; Sun, Y. Extracellular matrix protein DMP1 suppresses osteogenic differentiation of Mesenchymal Stem Cells. Biochem. Biophys. Res. Commun. 2018, 501, 968–973. [Google Scholar] [CrossRef]
- Ye, L.; MacDougall, M.; Zhang, S.; Xie, Y.; Zhang, J.; Li, Z.; Lu, Y.; Mishina, Y.; Feng, J.Q. Deletion of dentin matrix protein-1 leads to a partial failure of maturation of predentin into dentin, hypomineralization, and expanded cavities of pulp and root canal during postnatal tooth development. J. Biol. Chem. 2004, 279, 19141–19148. [Google Scholar] [CrossRef]


| Sample | Average RMS (nm) | SD | Average Mean Roughness (nm) | SD |
|---|---|---|---|---|
| CTRL | 93.9 | 19.7 | 71.6 | 15.6 |
| EE | 57.3 | 15.7 | 43.8 | 11.5 |
| ES | 31.8 | 3.1 | 23.4 | 3.2 |
| ATcs | 59.5 | 15.5 | 45.4 | 10.8 |
| CA | 61.8 | 9.2 | 44.6 | 4.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Schiavoni, V.; Memé, L.; Tossetta, G.; Marzioni, D.; Bambini, F.; Frontini, A.; Santoni, C.; Moretti, P.; Vignini, A.; Campagna, R.; et al. Novel Ti6Al4V Surface Treatment for Subperiosteal Dental Implants (Part II): Matrix Deposition and Osteogenic Markers. Materials 2026, 19, 1522. https://doi.org/10.3390/ma19081522
Schiavoni V, Memé L, Tossetta G, Marzioni D, Bambini F, Frontini A, Santoni C, Moretti P, Vignini A, Campagna R, et al. Novel Ti6Al4V Surface Treatment for Subperiosteal Dental Implants (Part II): Matrix Deposition and Osteogenic Markers. Materials. 2026; 19(8):1522. https://doi.org/10.3390/ma19081522
Chicago/Turabian StyleSchiavoni, Valentina, Lucia Memé, Giovanni Tossetta, Daniela Marzioni, Fabrizio Bambini, Andrea Frontini, Chiara Santoni, Paolo Moretti, Arianna Vignini, Roberto Campagna, and et al. 2026. "Novel Ti6Al4V Surface Treatment for Subperiosteal Dental Implants (Part II): Matrix Deposition and Osteogenic Markers" Materials 19, no. 8: 1522. https://doi.org/10.3390/ma19081522
APA StyleSchiavoni, V., Memé, L., Tossetta, G., Marzioni, D., Bambini, F., Frontini, A., Santoni, C., Moretti, P., Vignini, A., Campagna, R., & Salvolini, E. (2026). Novel Ti6Al4V Surface Treatment for Subperiosteal Dental Implants (Part II): Matrix Deposition and Osteogenic Markers. Materials, 19(8), 1522. https://doi.org/10.3390/ma19081522













