Redox Mechanisms of Silica-Supported Ni Particles: An X-Ray Absorption Fine Structure Investigation
Abstract
1. Introduction
2. Experimental Procedure
2.1. Sample Preparation and Characterization
2.2. In Situ XAFS Measurement
3. Results and Discussion
3.1. Sample Characterization
3.2. Reduction Process of SiO2-Supported NiO Particles
3.3. Oxidation Process of SiO2-Supported Ni Particles
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kawasaki, T.; Matsuda, J.; Tachikawa, Y.; Lyth, S.M.; Shiratori, Y.; Taniguchi, S.; Sasaki, K. Oxidation-induced degradation and performance fluctuation of solid oxide fuel cell Ni anodes under simulated high fuel utilization conditions. Fuel Cells 2019, 44, 9386–9399. [Google Scholar] [CrossRef]
- Sarantaridis, D.; Atkinson, A. Redox cycling of Ni-based solid oxide fuel cell anodes: A review. Int. J. Hydrogen Energy 2007, 7, 246–258. [Google Scholar] [CrossRef]
- Pan, Y.; Tursun, Y.; Abduhani, H.; Turap, Y.; Abulizi, A.; Talifua, D. Chemical looping gasification of cotton stalk with bimetallic Cu/Ni/olivine as oxygen carrier. Int. J. Energy Res. 2020, 44, 7268–7282. [Google Scholar] [CrossRef]
- Liu, B.; You, C.; Wang, H. Chemical looping gasification of waste plastics for syngas production using NiO/Al as dual functional material. Chem. Eng. J. 2024, 501, 157728. [Google Scholar] [CrossRef]
- Nam, H.; Wang, Z.; Shanmugam, S.R.; Adhikari, S.; Abdoulmoumine, N. Chemical looping dry reforming of benzene as a gasification tar model compound with Ni- and Fe-based oxygen carriers in a fluidized bed reactor. Int. J. Hydrogen Energy 2018, 43, 18790–18800. [Google Scholar] [CrossRef]
- Ghoniem, A.F.; Zhao, Z.; Uddi, M. Redox kinetics of NiO/YSZ for chemical-looping combustion and the effect of support on reducibility. Proc. Combust. Inst. 2023, 39, 4477–4487. [Google Scholar] [CrossRef]
- Nordness, O.; Han, L.; Zhou, Z.; Bollas, G.M. High-pressure chemical-looping of methane and synthesis gas with Ni and Cu oxygen carriers. Energy Fuels 2016, 30, 504–514. [Google Scholar] [CrossRef]
- Tijani, M.M.; Aqsha, A.; Mahinpey, N. Synthesis and study of metal-based oxygen carriers (Cu, Co, Fe, Ni) and their interaction with supported metal oxides (Al2O3, CeO2, TiO2, ZrO2) in a chemical looping combustion system. Energy 2017, 138, 873–882. [Google Scholar] [CrossRef]
- Erri, P.; Varma, A. Diffusional effects in nickel oxide reduction kinetics. Ind. Eng. Chem. Res. 2009, 48, 4–6. [Google Scholar] [CrossRef]
- Richardson, J. X-ray diffraction study of nickel oxide reduction by hydrogen. Appl. Catal. A Gen. 2003, 246, 137–150. [Google Scholar] [CrossRef]
- Sá, J.; Kayser, Y.; Milne, C.J.; Fernandes, D.L.A.; Szlachetko, J. Temperature-programmed reduction of NiO nanoparticles followed by time-resolved RIXS. Phys. Chem. Chem. Phys. 2014, 16, 7692–7696. [Google Scholar] [CrossRef]
- Jeangros, Q.; Hansen, T.; Wagner, J.B.; Dunin-Borkowski, R.E.; Hébert, C.; Herle, J.V.; Hessler-Wyser, A. Oxidation mechanism of nickel particles studied in an environmental transmission electron microscope. Acta Mater. 2014, 67, 362–372. [Google Scholar] [CrossRef]
- Jeangros, Q.; Hansen, T.W.; Wagner, J.B.; Damsgaard, C.D.; Dunin-Borkowski, R.E.; Hébert, C.; Herle, J.V.; Hessler-Wyser, A. Reduction of nickel oxide particles by hydrogen studied in an environmental TEM. J. Mater. Sci. 2013, 48, 2893–2907. [Google Scholar] [CrossRef]
- Shindo, D.; Akase, Z. Direct observation of electric and magnetic fields of functional materials. Mater. Sci. Eng. R. 2020, 142, 100564. [Google Scholar] [CrossRef]
- Iwasawa, Y. In situ characterization of supported metal catalysts and model surfaces by time-resolved and three-dimensional XAFS techniques. J. Catal. 2003, 216, 165–177. [Google Scholar] [CrossRef]
- Bolin, T.B.; Wu, T.; Schweitzer, N.; Lobo-Lapidus, R.; Kropf, A.J.; Wang, H.; Hu, Y.; Miller, J.T.; Heald, S.M. In situ intermediate-energy X-ray catalysis research at the advanced photon source beamline 9-BM. Catal. Today 2013, 205, 141–147. [Google Scholar] [CrossRef]
- Yamashita, S.; Yamamoto, Y.; Katayama, M.; Inada, Y. Kinetic study on solid-phase reduction of silica-supported nickel oxide species. Bull. Chem. Soc. Jpn. 2015, 88, 1629–1635. [Google Scholar] [CrossRef]
- Chotiwan, S.; Tomiga, H.; Katagiri, M.; Yamamoto, Y.; Yamashita, S.; Katayama, M.; Inada, Y. Particle size effect of redox reactions for Co species supported on silica. J. Solid State Chem. 2016, 241, 212–218. [Google Scholar] [CrossRef]
- Dann, E.K.; Gibson, E.K.; Catlow, R.A.; Collier, P.; Erden, T.E.; Gianolio, D.; Hardacre, C.; Kroner, A.; Raj, A.; Goguet, A.; et al. Combined in situ XAFS/DRIFTS studies of the evolution of nanoparticle structures from molecular precursors. Chem. Mater. 2017, 29, 7515–7523. [Google Scholar] [CrossRef]
- Yamashita, S.; Yamamoto, Y.; Kawabata, H.; Niwa, Y.; Katayama, M.; Inada, Y. Dynamic chemical state conversion of nickel species supported on silica under CO-NO reaction conditions. Catal. Today 2018, 303, 33–39. [Google Scholar] [CrossRef]
- Mizuno, S.; Horiuchi, J.; Sharma, K.P.; Saida, T.; Maruyama, T. In situ XAFS study on the chemical states of transition-metal nanoparticle catalysts during single-walled carbon nanotube growth under conventional CVD conditions. J. Nanopart. Res. 2025, 27, 155. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Yamashita, S.; Afiza, N.; Katayama, M.; Inada, Y. Oxidation property of SiO2-supported small nickel particle prepared by the sol-gel method. J. Phys. Conf. Ser. 2016, 712, 012075. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Suzuki, A.; Tsutsumi, N.; Katagiri, M.; Yamashita, S.; Niwa, Y.; Katayama, M.; Inada, Y. In situ X-ray absorption fine structure analysis of redox reactions of nickel species with variable particle sizes supported on silica. J. Solid State Chem. 2018, 258, 264–270. [Google Scholar] [CrossRef]
- Novitasari, E.; Azuma, A.; Ohta, K.; Katayama, M.; Niwa, Y.; Kimura, M.; Inada, Y. Chemical state analysis of size-controlled particles during redox reactions between NiO and metallic Ni. Next Mater. 2025, 8, 100738. [Google Scholar] [CrossRef]
- Tauster, S.J.; Fung, S.C.; Garten, R.L. Strong metal-support interactions. group 8 noble metals supported on TiO2. J. Am. Chem. Soc. 1978, 100, 170–175. [Google Scholar] [CrossRef]
- Tauster, S.J.; Fung, S.C.; Baker, R.T.K.; Horsley, J.A. Strong interactions in supported-metal catalysts. Science 1981, 211, 1121–1125. [Google Scholar] [CrossRef]
- Tauster, S.J. Strong metal-support interactions. Acc. Chem. Res. 1987, 20, 389–394. [Google Scholar] [CrossRef]
- Ueckert, T.; Lamber, R.; Jaeger, N.I.; Schubert, U. Strong metal-support interactions in a Ni/SiO2 catalyst prepared via sol-gel synthesis. Appl. Catal. A 1997, 155, 75–85. [Google Scholar] [CrossRef]
- Tikekar, N.M.; Armstrong, T.J.; Virkar, A.V. Reduction and reoxidation kinetics of nickel-based SOFC anodes. J. Electrochem. Soc. 2006, 153, A654–A663. [Google Scholar] [CrossRef]
- Kulyk, V.V.; Vasyliv, B.D.; Duriagina, Z.A.; Kovbasiuk, T.M.; Lemishka, I.A. The effect of water vapor containing hydrogenous atmospheres on the micro-structure and tendency to brittle fracture of anode materials of YSZ-NiO(Ni) system. Arch. Mater. Sci. Eng. 2021, 108, 49–67. [Google Scholar] [CrossRef]
- Ravel, B.; Newville, M. ATHENA, ARTEMIS, HEPHAESTUS: Data analysis for X-ray absorption spectroscopy using IFEFFIT. J. Synchrotron Rad. 2005, 12, 537–541. [Google Scholar] [CrossRef] [PubMed]
- Cai, W.; Ye, L.; Zhang, L.; Ren, Y.; Yue, B.; Chen, X.; He, H. Highly dispersed nickel-containing mesoporous silica with superior stability in carbon dioxide reforming of methane: The effect of anchoring. Materials 2014, 7, 2340–2355. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Das, S.; Hongmanorom, P.; Dewangan, N.; Wai, M.H.; Kawi, S. Silica-based micro- and mesoporous catalysts for dry reforming of methane. Catal. Sci. Technol. 2018, 8, 2763–2778. [Google Scholar] [CrossRef]
- Lensveld, D.J.; Mesu, J.G.; van Dillen, A.J.; de Jong, K.P. The application of well-dispersed nickel nanoparticles inside the mesopores of MCM-41 by use of a nickel citrate chelate as precursor. Stud. Surf. Sci. Catal. 2000, 143, 647–657. [Google Scholar] [CrossRef]
- van Dillen, A.J.; Terörde, R.J.A.M.; Lensveld, D.J.; Geus, J.W.; de Jong, K.P. Synthesis of supported catalysts by impregnation and drying using aqueous chelated metal complexes. J. Catal. 2003, 216, 257–264. [Google Scholar] [CrossRef]
- Bentaleb, F.; Che, M.; Dubreuil, A.C.; Thomazeau, C.; Marceau, E. Influence of organic additives on the properties of impregnation solutions and on nickel oxide particle size for Ni/Al2O3 catalysts. Catal. Today 2014, 235, 250–255. [Google Scholar] [CrossRef]
- Lv, Y.; Xin, Z.; Meng, X.; Tao, M.; Bian, Z.; Gu, J.; Gao, W. Essential role of organic additives in preparation of efficient Ni/KIT-6 catalysts for CO methanation. Appl. Catal. A Gen. 2018, 558, 99–108. [Google Scholar] [CrossRef]
- Medford, J.A.; Johnston-Peck, A.C.; Tracy, J.B. Nanostructural transformations during the reduction of hollow and porous nickel oxide nanoparticles. Nanoscale 2013, 5, 155–159. [Google Scholar] [CrossRef]
- Cabrera, N.; Mott, N.F. Theory of the oxidation of metals. Rep. Prog. Phys. 1949, 12, 163–184. [Google Scholar] [CrossRef]
- Railsback, J.G.; Johnston-Peck, A.C.; Wang, J.; Tracy, J.B. Size-dependent nanoscale Kirkendall effect during the oxidation of nickel nanoparticles. ACS Nano 2010, 4, 1913–1920. [Google Scholar] [CrossRef]
- Flege, J.I.; Meyer, A.; Falta, J.; Krasovskii, E.E. Self-limited oxide formation in Ni(111) oxidation. Phys. Rev. B 2011, 84, 1913–1920. [Google Scholar] [CrossRef]

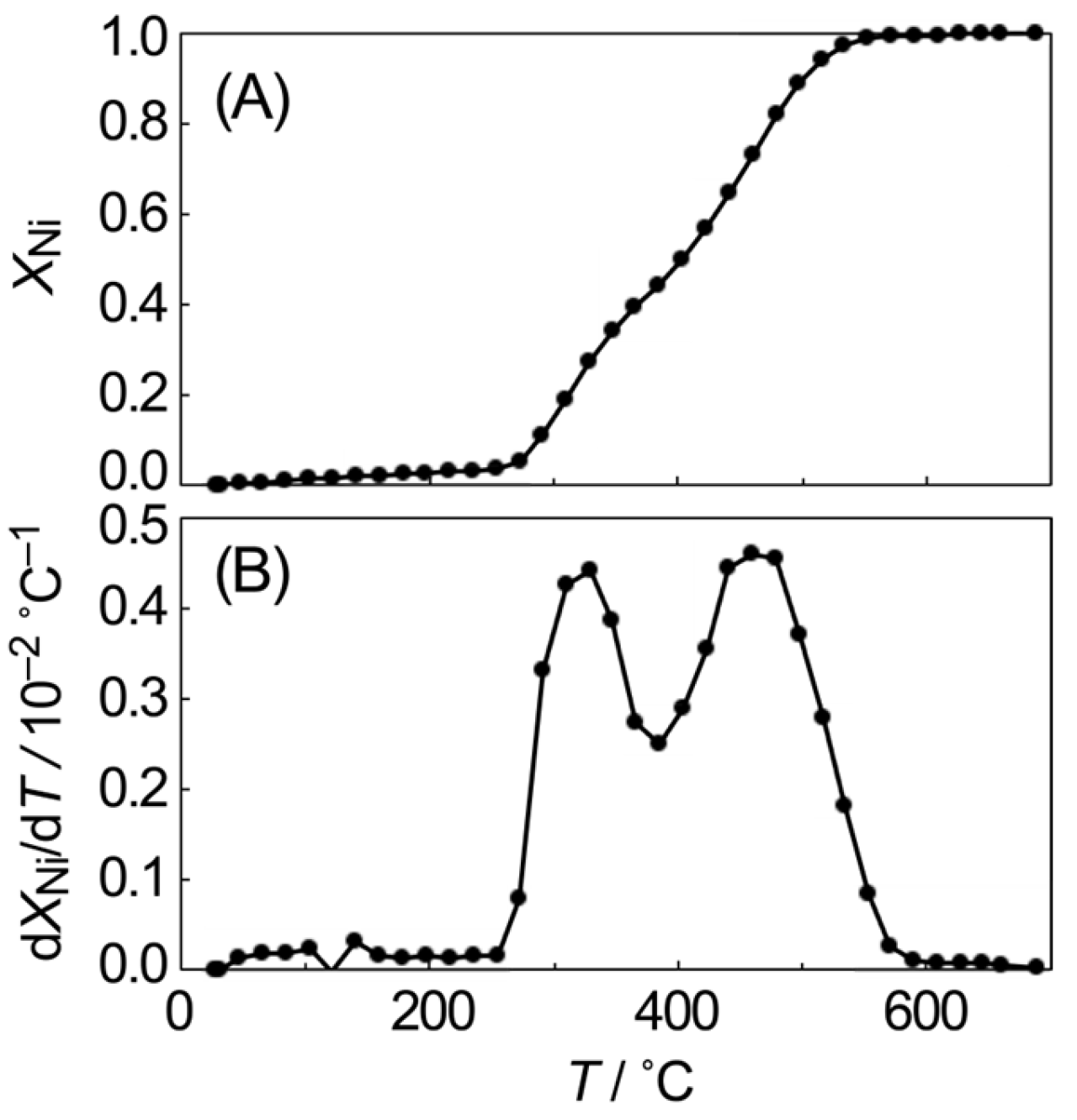


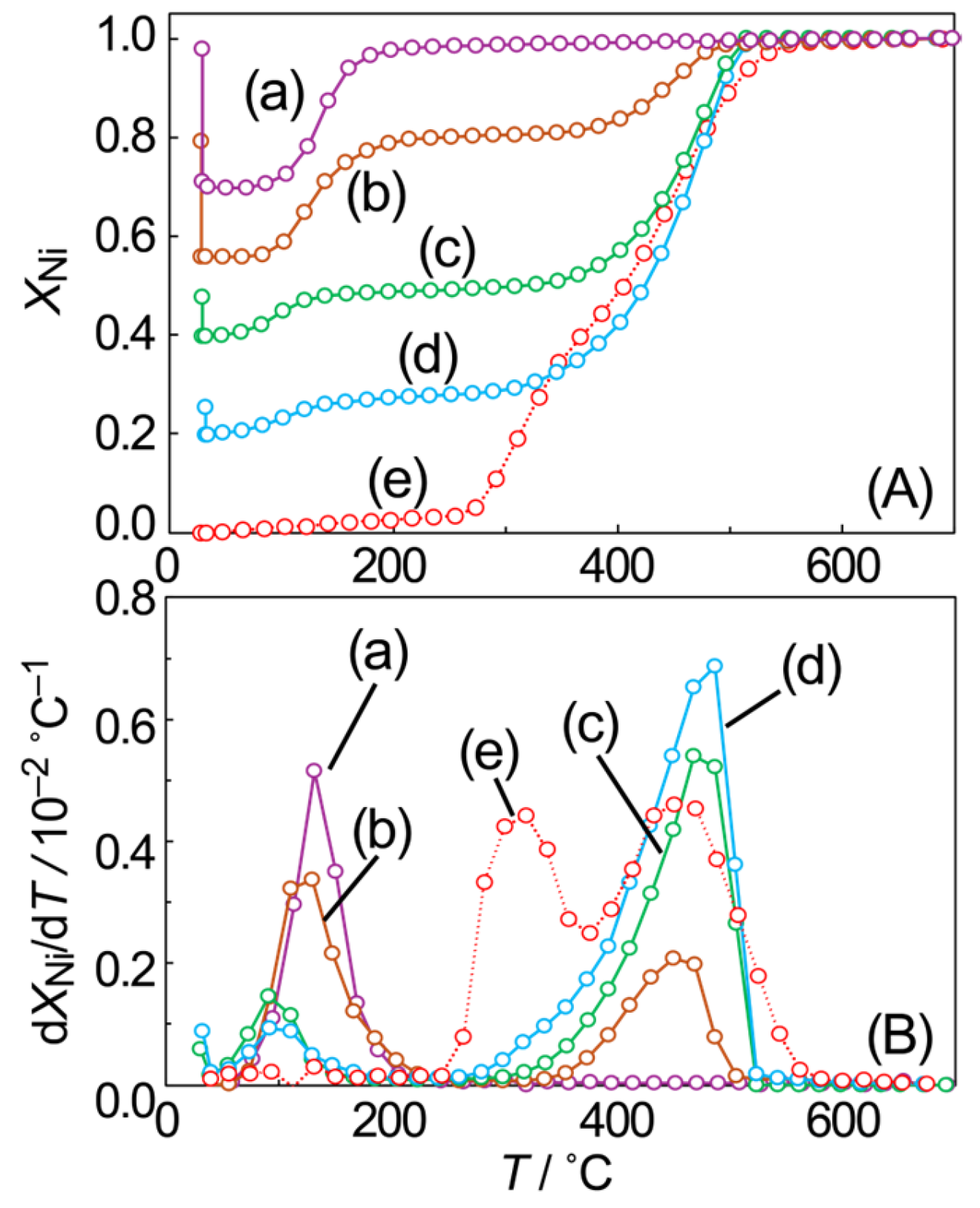


| Before | After | Change with O2 Exposure |
|---|---|---|
| 0.25 | 0.20 | 0.05 |
| 0.48 | 0.39 | 0.11 |
| 0.79 | 0.55 | 0.24 |
| 1.00 | 0.71 | 0.29 |
| XNiO | d/Å | Tred/°C |
|---|---|---|
| 0.28 | 4 | 70 |
| 0.44 | 7 | 87 |
| 0.60 | 10 | 116 |
| 0.81 | 17 | 178 |
| 1.00 | 31 * | 358 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Novitasari, E.; Ohta, K.; Azuma, A.; Niwa, Y.; Kimura, M.; Inada, Y. Redox Mechanisms of Silica-Supported Ni Particles: An X-Ray Absorption Fine Structure Investigation. Materials 2026, 19, 1509. https://doi.org/10.3390/ma19081509
Novitasari E, Ohta K, Azuma A, Niwa Y, Kimura M, Inada Y. Redox Mechanisms of Silica-Supported Ni Particles: An X-Ray Absorption Fine Structure Investigation. Materials. 2026; 19(8):1509. https://doi.org/10.3390/ma19081509
Chicago/Turabian StyleNovitasari, Eka, Kodai Ohta, Asaka Azuma, Yasuhiro Niwa, Masao Kimura, and Yasuhiro Inada. 2026. "Redox Mechanisms of Silica-Supported Ni Particles: An X-Ray Absorption Fine Structure Investigation" Materials 19, no. 8: 1509. https://doi.org/10.3390/ma19081509
APA StyleNovitasari, E., Ohta, K., Azuma, A., Niwa, Y., Kimura, M., & Inada, Y. (2026). Redox Mechanisms of Silica-Supported Ni Particles: An X-Ray Absorption Fine Structure Investigation. Materials, 19(8), 1509. https://doi.org/10.3390/ma19081509






