Infrared Stealth Characteristics of WO3-Based Electrochromic Devices Mediated by Zn2+-Al3+ Gel Electrolyte
Abstract
1. Introduction
2. Materials and Methods
2.1. Device Fabrication
2.1.1. Pretreatment of Nickel Mesh
2.1.2. Fabrication of WO3 Thin Films
2.1.3. Preparation of Zn2+-Doped Al3+ Gel Electrolyte
2.1.4. Preparation of Electrochromic Device
2.2. Characterization and Performance Measurements
2.2.1. Microstructural Characterization
2.2.2. Electrochemical Characterization
2.2.3. Infrared Stealth Performance Measurement
2.2.4. Cycling Stability Measurement
3. Results and Discussion
3.1. Microstructural Characterization of the WO3/Nickel Mesh Electrode
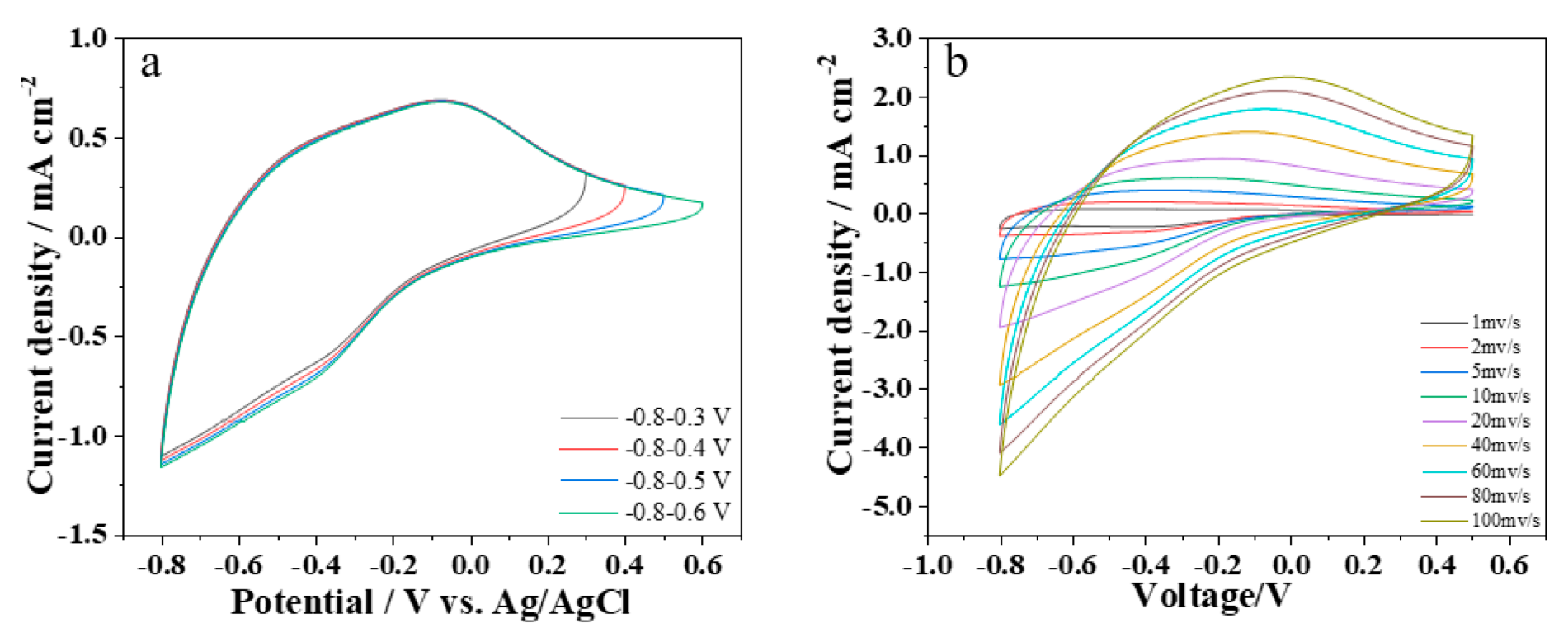
3.2. Electrochemical Performance Analysis of the WO3/Nickel Mesh Electrode
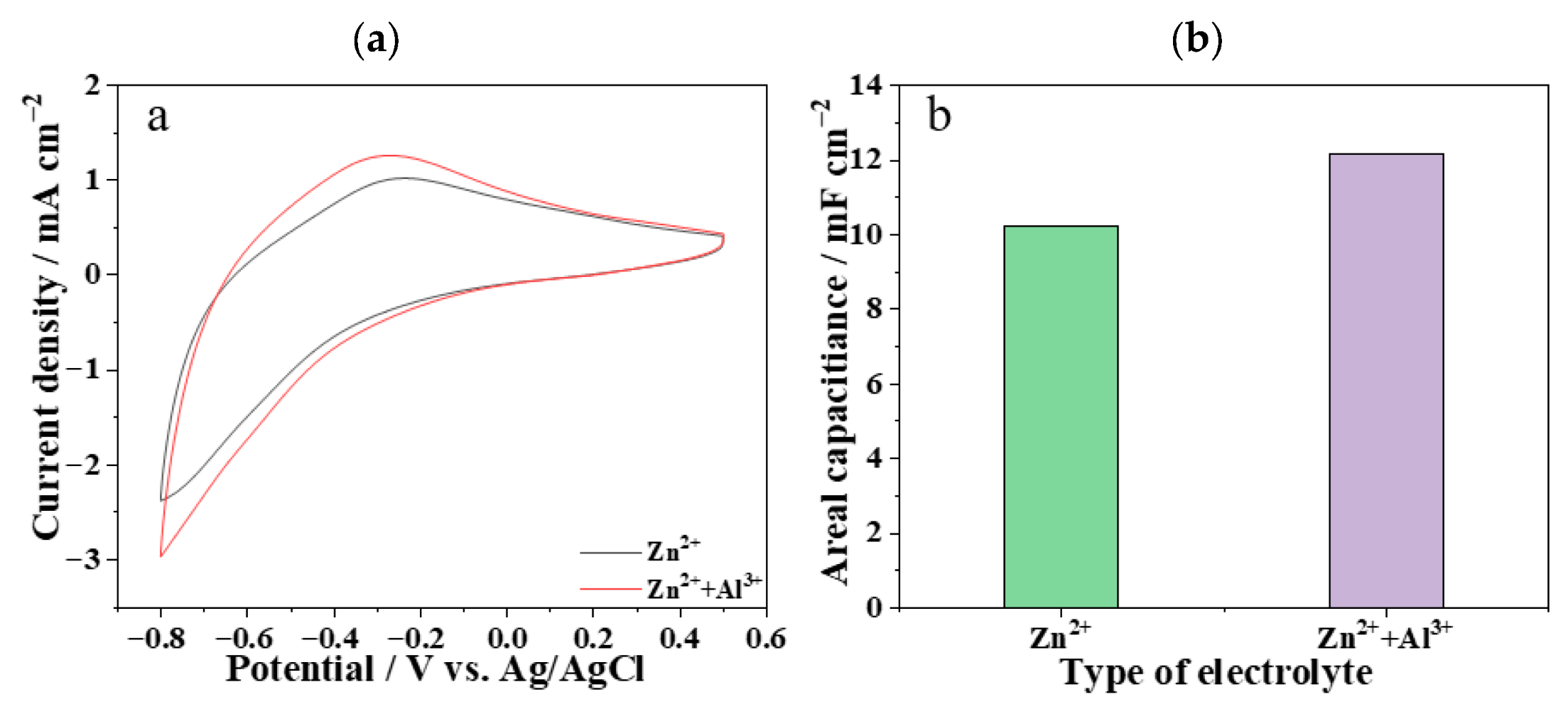
3.3. Electrochemical Performance of the WO3/Ni Mesh/Zn2+-Al3+/Zn Foil Electrochromic Device
3.4. Infrared Modulation Performance of the WO3/Ni Mesh/Zn2+-Al3+/Zn Foil Electrochromic Device
3.5. Analysis of Modulation Mechanism
- (1)
- Ion Synergistic Regulation Mechanism
- (2)
- Interface and Stability Enhancement Mechanism
- (3)
- Ion–Electron Matching Transport Mechanism of the Whole Structure
4. Conclusions
- (1)
- The as-prepared WO3 thin film exhibited a dense spherical morphology, indicating excellent uniformity and compactness. The nickel mesh substrate improved the conductivity and mechanical strength of the film. The device showed good cycling stability: after 1000 cycles, the areal capacitance of the electrode remained 83.7% of its initial value. The doping of Al3+ in the electrolyte enhanced the areal capacitance of the electrochromic electrode, further demonstrating that the synergistic effect between Zn2+ and Al3+ improved the cycling stability of the device.
- (2)
- Under voltage modulation between −0.1 V and 1.1 V, at a typical mid-wave infrared wavelength of 4.0 μm, the emissivity ε decreased from 0.89 (−0.1 V) to 0.67 (1.1 V), with an absolute modulation amplitude Δε of 0.22. At a typical long-wave infrared wavelength of 8.7 μm, ε decreased from 0.96 (−0.1 V) to 0.69 (1.1 V), with an absolute modulation amplitude Δε of 0.29. The coloring/bleaching response times were 10.1 s and 2.44 s, respectively, revealing fast and efficient infrared stealth modulation capability.
- (3)
- In the temperature range of 30–40 °C, the surface temperature difference ΔT between the colored state and the bleached state increased from 4.4 °C to 4.6 °C. When the temperature exceeded 40 °C, ΔT gradually decreased to 3.4 °C at an ambient temperature of 60 °C. The device reached the maximum modulation amplitude of 4.6 °C at 40 °C. These results confirm that the device possesses effective infrared radiation modulation ability at various ambient temperatures, which provides promising application potential in the field of infrared stealth.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shchegolkov, A.V.; Lipkin, M.S.; Korbova, E.V.; Lipkina, T.V.; Lipkin, V.M. On the Mechanism of Formation of Electrochromic WO3 Films on the Surface of Sn, Ti, ITO Electrodes in the Process of Cathodic Electrodeposition. Inorg. Mater. Appl. Res. 2022, 13, 1081–1086. [Google Scholar] [CrossRef]
- Vasireddy, S.; Pillem, R. Invisibility system using image processing and optical camouflage technology. Int. J. Eng. Trends Technol. 2013, 4, 1874. [Google Scholar]
- Jiang, X.; Wang, X.; Nong, J.; Zhu, G.; He, X.; Du, T.; Ma, H.; Zhang, Z.; Chen, H.; Yu, Y.; et al. Bicolor Regulation of an Ultrathin Absorber in the Mid-Wave Infrared and Long-Wave Infrared Regimes. ACS Photonics 2024, 11, 218–229. [Google Scholar] [CrossRef]
- Zhang, X.; Hao, X.; Wei, G.; Zhang, H.; Chen, W.; Shan, F.; Liu, K.; Tan, S.; Ji, G. Artificial Neural Network-Assisted Design of 3d Printed Moth-Eye Inspired Metastructure for Broadband Microwave Absorption and Infrared Stealth. Compos. Part B Eng. 2026, 313, 113391. [Google Scholar] [CrossRef]
- Zhao, J.; Zhou, B.; Wang, G.; Ying, J.; Liu, J.; Chen, Q. Spectral camouflage characteristics and recognition ability of targets based on visible/near-infrared hyperspectral images. Photonics 2022, 9, 957. [Google Scholar] [CrossRef]
- Ma, Q.; Shen, C.; Cheng, Z.; Liu, T.X.; Han, Z.; Lu, T.J.; Meng, H. Multistage Porous Hybrid Aerogel: A Multifunctional Material for Acoustic-Electromagnetic-Infrared Multispectral Stealth. Adv. Compos. Hybrid Mater. 2026, 9, 74. [Google Scholar] [CrossRef]
- Nguyen, D.; Lee, H.; Kim, I.-T. Synthesis and thermochromic properties of Cr-doped Al2O3 for a reversible thermochromic sensor. Materials 2017, 10, 476. [Google Scholar] [CrossRef]
- Zhou, J.; Han, Y.; Ren, D.; Lin, Q. Adaptive Infrared Radiation Modulator Using Reversible Metal Electrodeposition with Silicon Carbide-Based Fabry-Perot Cavity. J. Alloys Compd. 2024, 1002, 175418. [Google Scholar] [CrossRef]
- Wei, H.; Chen, S.; Chen, Z.; Tang, L.; Xue, J.; Wang, C.; Wang, Z.; Li, Y. Hetero-Interface Engineering of Biomass Carbon Foam for Broadband Microwave Absorption and Thermal Insulation Properties. Carbon 2025, 241, 120385. [Google Scholar] [CrossRef]
- Liu, L.; Liu, C.; Wang, M.; Li, B.; Wang, K.; Fan, X.; Wang, L.; Wang, H.; Hu, S.; Diao, X. Anti-Self-Discharge Ultrathin All-Inorganic Electrochromic Asymmetric Supercapacitors Enabling Intelligent and Effective Energy Storage. Rare Met. 2023, 42, 2957–2971. [Google Scholar] [CrossRef]
- Deng, C.; Zhang, K.; Liu, L.; He, Z.; Huang, J.; Wang, T.; Liu, Y.; He, X.; Du, K.; Yi, Y. High-Performance All-Solid-State Electrochromic Asymmetric Zn-Ion Supercapacitors for Visualization of Energy Storage Devices. J. Mater. Chem. A 2022, 10, 17326–17337. [Google Scholar] [CrossRef]
- Zhao, J.; Zhang, S.; Chang, S.; Li, C.; Fang, C.; Xia, X.; Shen, L.; Yang Lee, J.; Cao, C.; Zhang, X.; et al. A Flexible Electrochromic Device with Variable Infrared Emissivity Based on W18o49 Nanowire Cathode and Mxene Infrared Transparent Conducting Electrode. Chem. Eng. J. 2024, 480, 148010. [Google Scholar] [CrossRef]
- Meng, Y.; Li, Z.; Wang, L.; Yang, X.; Yang, Y.; Li, X.; Jiang, Y.; Gao, Y.; Lü, W. Flexible Solid-State Zinc-Ion Electrochromic Energy Storage Device with Self-Healing Electrolyte for Wearable Electronics. J. Energy Storage 2024, 99, 113221. [Google Scholar] [CrossRef]
- Hernández Sierra, J.; Cortez Elizalde, J.; Torres Torres, J.G.; Silahua Pavón, A.A.; Cervantes Uribe, A.; Cordero García, A.; Guerra Que, Z.; Córdova Pérez, G.E.; Rangel Vázquez, I.; Arevalo Perez, J.C. Effects of Wo3 Amount and Treatment Temperature on Tio2-Zro2-Wo3 Photocatalysts Used in the Solar Photocatalytic Oxidation of Sildenafil. Catalysts 2026, 16, 82. [Google Scholar] [CrossRef]
- Luo, Z.; Liu, L.; Yang, X.; Luo, X.; Bi, P.; Fu, Z.; Pang, A.; Li, W.; Yi, Y. Revealing the Charge Storage Mechanism of Nickel Oxide Electrochromic Supercapacitors. ACS Appl. Mater. Interfaces 2020, 12, 39098–39107. [Google Scholar] [CrossRef]
- Zhang, M.; Zhou, W.; Zhuang, B.; Zheng, Z.; Zhang, Q.; Wang, H. Theoretical investigation of electrochromic mechanism in D-A conjugated polymers in visible and infrared bands. RSC Adv. 2023, 13, 11337–11345. [Google Scholar] [CrossRef]
- Wu, Q.; Wang, J.; Yang, X.; Tong, Z.; Ji, H.; Qu, H.Y. Visible and near-infrared dynamic electrochromic modulation with the plasmon-enhanced two-dimensional ordered macroporous Au/PProDot-Me2 thin film. Adv. Opt. Mater. 2023, 11, 2202624. [Google Scholar] [CrossRef]
- Kang, M.; Wang, X.; Wei, Y.; Yu, Z.; Liu, Z. Latest research progress of infrared stealth textiles. Infrared Phys. Technol. 2024, 139, 105313. [Google Scholar] [CrossRef]
- Wang, W.; Wang, L.; Wang, L.; Liu, G.; Cui, Y.; Wang, B.; Ge, C.; Xu, K.; Huang, J.; Lei, Z. Construction of wear-resistant visible-infrared-compatible camouflage film and its spectral control mechanisms. Infrared Phys. Technol. 2024, 140, 105383. [Google Scholar] [CrossRef]
- Wang, J.; Zeng, L.; Xia, M.; Ji, S.; Zhang, Z.; Liang, J.; Bao, W.; Xie, G.; Li, J. Vanadium dioxide/aluminum composites for adaptive infrared stealth. Ceram. Int. 2024, 50, 25034–25040. [Google Scholar] [CrossRef]
- Xu, C.; Stiubianu, G.T.; Gorodetsky, A.A. Adaptive infrared-reflecting systems inspired by cephalopods. Science 2018, 359, 1495–1500. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.; Gao, L.; Gu, X.; Li, Z.; Huang, M.; Su, G. Improvement of electrochromic performance of nickel oxide porous films by Sn doping. Mater. Chem. Phys. 2023, 306, 128079. [Google Scholar] [CrossRef]
- Ling, H.; Wu, J.; Su, F.; Tian, Y.; Liu, Y.J. Automatic light-adjusting electrochromic device powered by perovskite solar cell. Nat. Commun. 2021, 12, 1010. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Mei, Z.; Wu, X.; Zhang, W.; Zhang, Y.; Xi, A.; Gao, D.; Lan, F.; Xu, J.; Diao, X.; et al. Integrated Multispectral Modulator with Efficient Radiative Cooling for Innovative Thermal Camouflage. Adv. Funct. Mater. 2025, 35, 2500122. [Google Scholar] [CrossRef]
- Li, C.; Zhen, M.; Wang, K.; Liu, L.; Zhang, W.; Wang, Y.; Fan, X.; Hou, W.; Xiong, J. Temperature Sensors Integrated with an Electrochromic Readout toward Visual Detection. ACS Appl. Mater. Interfaces 2023, 15, 40772–40780. [Google Scholar] [CrossRef]
- Wang, M.; Li, X.; Liu, L.; Li, B.; Xun, J.; Wang, L.; Wang, H.; Hu, S.; Li, C. Carbon Dots as Multifunctional Electrolyte Additives toward Multicolor and Low Self-Discharge Electrochromic Energy Storage Devices. Energy Storage Mater. 2024, 65, 103110. [Google Scholar] [CrossRef]
- Farouk, M.; Samir, A.; Ibrahim, A.; Farag, M.A.; Solieman, A. Raman, Ftir Studies and Optical Absorption of Zinc Borate Glasses Containing Wo3. Appl. Phys. A 2020, 126, 696. [Google Scholar] [CrossRef]
- Yan, D.; Liu, L.; Guo, T.; Zhang, Z.; Wang, M.; Li, C.; Li, N.; Xue, C.; Diao, X.; Hu, S. High-Performance Reflective-Type Zn-Ions Electrochromic Devices toward Visible-to-Infrared Broadband Dynamic Regulation. Chem. Eng. J. 2025, 504, 159117. [Google Scholar] [CrossRef]
- Liu, L.; Zhen, M.; Wang, L.; Li, B.; Deng, C.; Zhang, Z.; Fan, X.; Li, N.; Wang, H.; Li, C.; et al. Full-temperature all-solid-state dendrite-free Zn-ion electrochromic energy storage devices for intelligent applications. Chem. Eng. J. 2023, 468, 143837. [Google Scholar] [CrossRef]
- Wu, C.; Shi, H.; Zhao, L. High-performance aqueous Zn2+/Al3+ electrochromic batteries based on niobium tungsten oxides. Adv. Energy Mater. 2023, 13, 2300872. [Google Scholar] [CrossRef]






Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Wang, K.; Yang, X.; Liu, T.; Zhang, W. Infrared Stealth Characteristics of WO3-Based Electrochromic Devices Mediated by Zn2+-Al3+ Gel Electrolyte. Materials 2026, 19, 1506. https://doi.org/10.3390/ma19081506
Wang K, Yang X, Liu T, Zhang W. Infrared Stealth Characteristics of WO3-Based Electrochromic Devices Mediated by Zn2+-Al3+ Gel Electrolyte. Materials. 2026; 19(8):1506. https://doi.org/10.3390/ma19081506
Chicago/Turabian StyleWang, Ke, Xiaoting Yang, Tongyu Liu, and Wei Zhang. 2026. "Infrared Stealth Characteristics of WO3-Based Electrochromic Devices Mediated by Zn2+-Al3+ Gel Electrolyte" Materials 19, no. 8: 1506. https://doi.org/10.3390/ma19081506
APA StyleWang, K., Yang, X., Liu, T., & Zhang, W. (2026). Infrared Stealth Characteristics of WO3-Based Electrochromic Devices Mediated by Zn2+-Al3+ Gel Electrolyte. Materials, 19(8), 1506. https://doi.org/10.3390/ma19081506




