Realizing 303 ps Ultrafast Scintillation Time in 2-Inch CsPbCl3 Single Crystals Grown Under Br2 Overpressure
Abstract
1. Introduction
2. Materials and Methods
2.1. Material Preparation
2.2. Crystal Growth
3. Results and Discussion
3.1. Structural and Optical Characterization
3.2. Charge Transport Property
3.3. Steady-State and Time-Resolved Transient Photoluminescence Spectra
3.4. Power-Dependent Photoluminescence
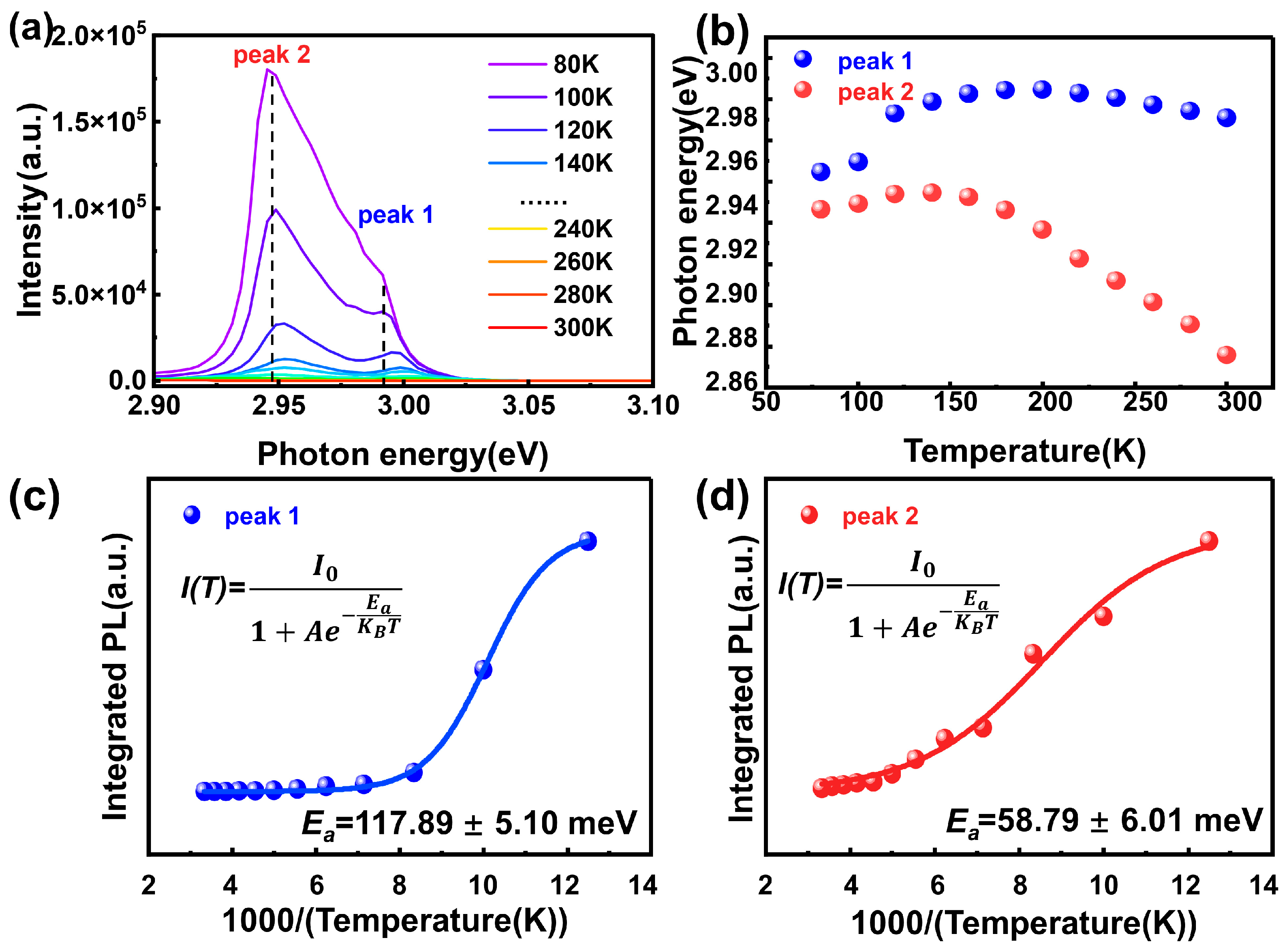
3.5. Temperature-Dependent Photoluminescence
3.6. Alpha Particle Excitation
3.7. X-Ray Imaging
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lecoq, P. Pushing the Limits in Time-of-Flight PET Imaging. IEEE Trans. Radiat. Plasma Med. Sci. 2017, 1, 473–485. [Google Scholar] [CrossRef]
- Lecoq, P.; Morel, C.; Prior, J.O.; Visvikis, D.; Gundacker, S.; Auffray, E.; Križan, P.; Turtos, R.M.; Thers, D.; Charbon, E.; et al. Roadmap toward the 10 Ps Time-of-Flight PET Challenge. Phys. Med. Biol. 2020, 65, 21RM01. [Google Scholar] [CrossRef] [PubMed]
- Silver, C.S.; Beeman, J.; Piccirillo, L.; Timbie, P.T.; Zhou, J.W. Optimization of a Bolometric Neutron Detector. Nucl. Instrum. Methods Phys. Res. Sect. A 2002, 485, 615–623. [Google Scholar] [CrossRef]
- Faderl, N. Fast Decay Solid-State Scintillators for High-Speed x-Ray Imaging. In Electro-Optical and Infrared Systems: Technology and Applications XVI; Hickman, D.L., Bürsing, H., Eds.; SPIE: Strasbourg, France, 2019; p. 10. [Google Scholar] [CrossRef]
- Valdes, D.J.; Rendon, L.; Miller, S.; Castaneda, J.J.; Leak, C.; Haque, S.; Gunthoti, K.; Wender, S.A.; Winkelbauer, J.; Paneru, S.N.; et al. Gallium Nitride (GaN) Scintillation Response to Fast Neutrons and Flash Gamma Rays and Associated Broadband Photoluminescence. AIP Adv. 2025, 15, 055037. [Google Scholar] [CrossRef]
- Kurudirek, M.; Kurudirek, S.V.; Erickson, A.; Sellin, P.J.; Duce, M.; Gouws, J.; Lawrie, B.J.; Melcher, C.L.; Hertel, N.E. Vertically Self-Oriented, Ultrafast 1D ZnO:Li Nanorods as Scintillators for Thermal Neutron Detection. ACS Appl. Nano Mater. 2025, 8, 20697–20712. [Google Scholar] [CrossRef]
- Wan, P.; Zhang, A.; Du, X.; Bao, Z.; Zhu, Z.; He, S.; Sui, X.; Han, C.; Niu, G.; Tang, J.; et al. High Spatial Resolution Perovskite Fast Neutron Imaging Screen by35 Cl (n, p) Nuclear Reaction. Small 2025, 21, e02360. [Google Scholar] [CrossRef]
- Lin, R.; Zhu, Y.; Chen, L.; Zheng, W.; Xu, M.; Ruan, J.; Li, R.; Li, T.; Lin, Z.; Cheng, L.; et al. Ultrafast (600 ps) α-Ray Scintillators. PhotoniX 2022, 3, 9. [Google Scholar] [CrossRef]
- Mykhaylyk, V.B.; Kraus, H.; Kapustianyk, V.; Kim, H.J.; Mercere, P.; Rudko, M.; Da Silva, P.; Antonyak, O.; Dendebera, M. Bright and Fast Scintillations of an Inorganic Halide Perovskite CsPbBr3 Crystal at Cryogenic Temperatures. Sci. Rep. 2020, 10, 8601. [Google Scholar] [CrossRef]
- Kang, J.; Wang, L.-W. High Defect Tolerance in Lead Halide Perovskite CsPbBr3. J. Phys. Chem. Lett. 2017, 8, 489–493. [Google Scholar] [CrossRef]
- Mosquera-Lois, I.; Huang, Y.-T.; Lohan, H.; Ye, J.; Walsh, A.; Hoye, R.L.Z. Multifaceted Nature of Defect Tolerance in Halide Perovskites and Emerging Semiconductors. Nat. Rev. Chem. 2025, 9, 287–304. [Google Scholar] [CrossRef]
- He, Y.; Matei, L.; Jung, H.J.; McCall, K.M.; Chen, M.; Stoumpos, C.C.; Liu, Z.; Peters, J.A.; Chung, D.Y.; Wessels, B.W.; et al. High Spectral Resolution of Gamma-Rays at Room Temperature by Perovskite CsPbBr3 Single Crystals. Nat. Commun. 2018, 9, 1609. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Stoumpos, C.C.; Hadar, I.; Luo, Z.; McCall, K.M.; Liu, Z.; Chung, D.Y.; Wessels, B.W.; Kanatzidis, M.G. Demonstration of Energy-Resolved γ-Ray Detection at Room Temperature by the CsPbCl3 Perovskite Semiconductor. J. Am. Chem. Soc. 2021, 143, 2068–2077. [Google Scholar] [CrossRef] [PubMed]
- Pan, L.; Liu, Z.; Welton, C.; Klepov, V.V.; Peters, J.A.; De Siena, M.C.; Benadia, A.; Pandey, I.; Miceli, A.; Chung, D.Y.; et al. Ultrahigh-Flux X-ray Detection by a Solution-Grown Perovskite CsPbBr3 Single-Crystal Semiconductor Detector. Adv. Mater. 2023, 35, 2211840. [Google Scholar] [CrossRef]
- Voloshynovskii, A.; Savchyn, P.; Karbovnyk, I.; Myagkota, S.; Cestelli Guidi, M.; Piccinini, M.; Popov, A.I. CsPbCl3 Nanocrystals Dispersed in the Rb0,8Cs0,2Cl Matrix Studied by Far-Infrared Spectroscopy. Solid State Commun. 2009, 149, 593–597. [Google Scholar] [CrossRef]
- Zhang, Y.; Shen, M.; Cheng, B.; Ma, W.; Huang, X.; Zhang, L.; Chai, Z.; Lin, W. Ultrafast Scintillation at Room Temperature Achieved in CsPbCl3-Based Single Crystals through Br over-Doping. J. Mater. Chem. C 2024, 12, 7169–7175. [Google Scholar] [CrossRef]
- Liu, W.; Peng, X.; Ge, B.; Zhang, X.; Hao, Y.; Tang, J.; Bai, R.; Xu, M.; Jie, W.; Xu, Y. Stoichiometric Engineering for Large-Size CsPbBr3 Crystal Growth and Gamma-Ray Detection Optimization. J. Mater. Chem. C 2026, 14, 3126–3135. [Google Scholar] [CrossRef]
- Badawi, R.D.; Shi, H.; Hu, P.; Chen, S.; Xu, T.; Price, P.M.; Ding, Y.; Spencer, B.A.; Nardo, L.; Liu, W.; et al. First Human Imaging Studies with the EXPLORER Total-Body PET Scanner*. J. Nucl. Med. 2019, 60, 299–303. [Google Scholar] [CrossRef]
- Shoji, Y.; Kurosawa, S.; Yokota, Y.; Hayasaka, S.; Kamada, K.; Yoshino, M.; Yamaji, A.; Chani, V.; Ohashi, Y.; Sakuragi, S.; et al. Growth and Scintillation Properties of Two-Inch-Diameter SrI2(Eu) Single Crystals. Cryst. Growth Des. 2018, 18, 3747–3752. [Google Scholar] [CrossRef]
- Peng, J.; Xu, Y.; Yao, F.; Lin, Q. Thick-Junction Perovskite X-Ray Detectors: Processing and Optoelectronic Considerations. Nanoscale 2022, 14, 9636–9647. [Google Scholar] [CrossRef]
- Rudolph, P. (Ed.) Handbook of Crystal Growth; Elsevier: Amsterdam, The Netherlands, 2015. [Google Scholar] [CrossRef]
- Derby, J.J. Macroscopic Transport Processes During the Growth of Single Crystals from the Melt. In Science and Technology of Crystal Growth; Van Der Eerden, J.P., Bruinsma, O.S.L., Eds.; Springer: Dordrecht, The Netherlands, 1995; pp. 97–110. [Google Scholar] [CrossRef]
- Müller, G. Convection in Melts and Crystal Growth. Adv. Space Res. 1983, 3, 51–60. [Google Scholar] [CrossRef]
- Ding, S.; Huang, X.; Yin, Q.; Dong, Y.; Bai, Y.; Wang, T.; Hao, H. Heat Transfer and Its Effect on Growth Behaviors of Crystal Layers during Static Layer Melt Crystallization. Chem. Eng. Sci. 2021, 233, 116390. [Google Scholar] [CrossRef]
- Brik, M.G.; Srivastava, A.M.; Popov, A.I. A Few Common Misconceptions in the Interpretation of Experimental Spectroscopic Data. Opt. Mater. 2022, 127, 112276. [Google Scholar] [CrossRef]
- Maddalena, F.; Mahyuddin, M.H.; Kowal, D.; Witkowski, M.E.; Makowski, M.; Kuddus Sheikh, M.A.; Mahato, S.; Jȩdrzejewski, R.; Drozdowski, W.; Dujardin, C.; et al. Lattice Expansion in Rb-Doped Hybrid Organic–Inorganic Perovskite Crystals Resulting in Smaller Band-Gap and Higher Light-Yield Scintillators. Inorg. Chem. 2023, 62, 8892–8902. [Google Scholar] [CrossRef]
- Huang, X.; Cheng, B.; Ma, W.; Qi, S.; Zhang, Y.; Shen, M.; Bo, T.; Zhang, L.; Lin, W. CsPbBrxCl3–x Solid Solutions: Understanding the Relationship between Lattice Expansion and Optical Properties. Inorg. Chem. 2024, 63, 11924–11929. [Google Scholar] [CrossRef] [PubMed]
- Prasanna, R.; Gold-Parker, A.; Leijtens, T.; Conings, B.; Babayigit, A.; Boyen, H.-G.; Toney, M.F.; McGehee, M.D. Band Gap Tuning via Lattice Contraction and Octahedral Tilting in Perovskite Materials for Photovoltaics. J. Am. Chem. Soc. 2017, 139, 11117–11124. [Google Scholar] [CrossRef]
- Kresse, G.; Furthmüller, J. Efficient Iterative Schemes for Ab Initio Total-Energy Calculations Using a Plane-Wave Basis Set. Phys. Rev. B 1996, 54, 11169–11186. [Google Scholar] [CrossRef]
- Blöchl, P.E. Projector Augmented-Wave Method. Phys. Rev. B 1994, 50, 17953–17979. [Google Scholar] [CrossRef]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized Gradient Approximation Made Simple. Phys. Rev. Lett. 1996, 77, 3865–3868. [Google Scholar] [CrossRef]
- Chadi, D.J. Special Points for Brillouin-Zone Integrations. Phys. Rev. B 1977, 16, 1746–1747. [Google Scholar] [CrossRef]
- Pela, R.R.; Hsiao, C.-L.; Hultman, L.; Birch, J.; Gueorguiev, G.K. Electronic and Optical Properties of Core–Shell InAlN Nanorods: A Comparative Study via LDA, LDA-1/2, mBJ, HSE06, G0W0 and BSE Methods. Phys. Chem. Chem. Phys. 2024, 26, 7504–7514. [Google Scholar] [CrossRef]
- Dou, L.; Wong, A.B.; Yu, Y.; Lai, M.; Kornienko, N.; Eaton, S.W.; Fu, A.; Bischak, C.G.; Ma, J.; Ding, T.; et al. Atomically Thin Two-Dimensional Organic-Inorganic Hybrid Perovskites. Science 2015, 349, 1518–1521. [Google Scholar] [CrossRef]
- Gabelloni, F.; Biccari, F.; Andreotti, G.; Balestri, D.; Checcucci, S.; Milanesi, A.; Calisi, N.; Caporali, S.; Vinattieri, A. Recombination Dynamics in CsPbBr3 Nanocrystals: Role of Surface States. Opt. Mater. Express 2017, 7, 4367. [Google Scholar] [CrossRef]
- Mykhaylyk, V.B.; Rudko, M.; Kraus, H.; Kapustianyk, V.; Kolomiets, V.; Vitoratou, N.; Chornodolskyy, Y.; Voloshinovskii, A.S.; Vasylechko, L. Ultra-Fast Low Temperature Scintillation and X-Ray Luminescence of CsPbCl3 Crystals. J. Mater. Chem. C 2023, 11, 656–665. [Google Scholar] [CrossRef]
- Lin, W.; He, J.; McCall, K.M.; Stoumpos, C.C.; Liu, Z.; Hadar, I.; Das, S.; Wang, H.; Wang, B.; Chung, D.Y.; et al. Inorganic Halide Perovskitoid TlPbI3 for Ionizing Radiation Detection. Adv. Funct. Mater. 2021, 31, 2006635. [Google Scholar] [CrossRef]
- Schmidt, T.; Lischka, K.; Zulehner, W. Excitation-Power Dependence of the near-Band-Edge Photoluminescence of Semiconductors. Phys. Rev. B 1992, 45, 8989–8994. [Google Scholar] [CrossRef] [PubMed]
- Wright, A.D.; Verdi, C.; Milot, R.L.; Eperon, G.E.; Pérez-Osorio, M.A.; Snaith, H.J.; Giustino, F.; Johnston, M.B.; Herz, L.M. Electron–Phonon Coupling in Hybrid Lead Halide Perovskites. Nat. Commun. 2016, 7, 11755. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Yuan, X.; Jing, P.; Li, J.; Wei, M.; Hua, J.; Zhao, J.; Tian, L. Temperature-Dependent Photoluminescence of Inorganic Perovskite Nanocrystal Films. RSC Adv. 2016, 6, 78311–78316. [Google Scholar] [CrossRef]
- Yi, J.; Ge, X.; Liu, E.; Cai, T.; Zhao, C.; Wen, S.; Sanabria, H.; Chen, O.; Rao, A.M.; Gao, J. The Correlation between Phase Transition and Photoluminescence Properties of CsPbX3 (X = Cl, Br, I) Perovskite Nanocrystals. Nanoscale Adv. 2020, 2, 4390–4394. [Google Scholar] [CrossRef]
- Baranowski, M.; Plochocka, P.; Su, R.; Legrand, L.; Barisien, T.; Bernardot, F.; Xiong, Q.; Testelin, C.; Chamarro, M. Exciton Binding Energy and Effective Mass of CsPbCl3: A Magneto-Optical Study. Photon. Res. 2020, 8, A50. [Google Scholar] [CrossRef]
- Zhang, Q.; Su, R.; Liu, X.; Xing, J.; Sum, T.C.; Xiong, Q. High-Quality Whispering-Gallery-Mode Lasing from Cesium Lead Halide Perovskite Nanoplatelets. Adv. Funct. Mater. 2016, 26, 6238–6245. [Google Scholar] [CrossRef]
- Wang, H.; Sun, Q.; Yang, F.; Li, Z.; Sun, L.; Zhang, X.; Zhao, Z.; Hu, W. Ultrahigh Resolution X-Ray Imaging with Thin-Film Scintillators Based on Aggregation-Induced Delayed Fluorescence Luminogens. SmartMat 2025, 6, e70002. [Google Scholar] [CrossRef]








Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Yang, J.; Wang, F.; Chen, L.; Bo, T.; Chai, Z.; Lin, W. Realizing 303 ps Ultrafast Scintillation Time in 2-Inch CsPbCl3 Single Crystals Grown Under Br2 Overpressure. Materials 2026, 19, 1479. https://doi.org/10.3390/ma19081479
Yang J, Wang F, Chen L, Bo T, Chai Z, Lin W. Realizing 303 ps Ultrafast Scintillation Time in 2-Inch CsPbCl3 Single Crystals Grown Under Br2 Overpressure. Materials. 2026; 19(8):1479. https://doi.org/10.3390/ma19081479
Chicago/Turabian StyleYang, Jingwei, Fangbao Wang, Liang Chen, Tao Bo, Zhifang Chai, and Wenwen Lin. 2026. "Realizing 303 ps Ultrafast Scintillation Time in 2-Inch CsPbCl3 Single Crystals Grown Under Br2 Overpressure" Materials 19, no. 8: 1479. https://doi.org/10.3390/ma19081479
APA StyleYang, J., Wang, F., Chen, L., Bo, T., Chai, Z., & Lin, W. (2026). Realizing 303 ps Ultrafast Scintillation Time in 2-Inch CsPbCl3 Single Crystals Grown Under Br2 Overpressure. Materials, 19(8), 1479. https://doi.org/10.3390/ma19081479




