Figure 1.
Overall process scheme for lithium recovery from LiFePO4 cathode powder via hydrogen reduction, CaCl2-assisted chlorination roasting, water leaching/vacuum filtration, and Ca2+ removal by ion exchange, including photographs of the starting LiFePO4 powder and the final recovered LiCl powder prepared in this study. Blue arrows indicate the process flow direction, and red circles denote the heating zone.
Figure 1.
Overall process scheme for lithium recovery from LiFePO4 cathode powder via hydrogen reduction, CaCl2-assisted chlorination roasting, water leaching/vacuum filtration, and Ca2+ removal by ion exchange, including photographs of the starting LiFePO4 powder and the final recovered LiCl powder prepared in this study. Blue arrows indicate the process flow direction, and red circles denote the heating zone.
Figure 2.
X-ray diffraction (XRD) pattern of the as-received LiFePO4 (LFP) powder (reference: triphylite, PDF #01-081-1173).
Figure 2.
X-ray diffraction (XRD) pattern of the as-received LiFePO4 (LFP) powder (reference: triphylite, PDF #01-081-1173).
Figure 3.
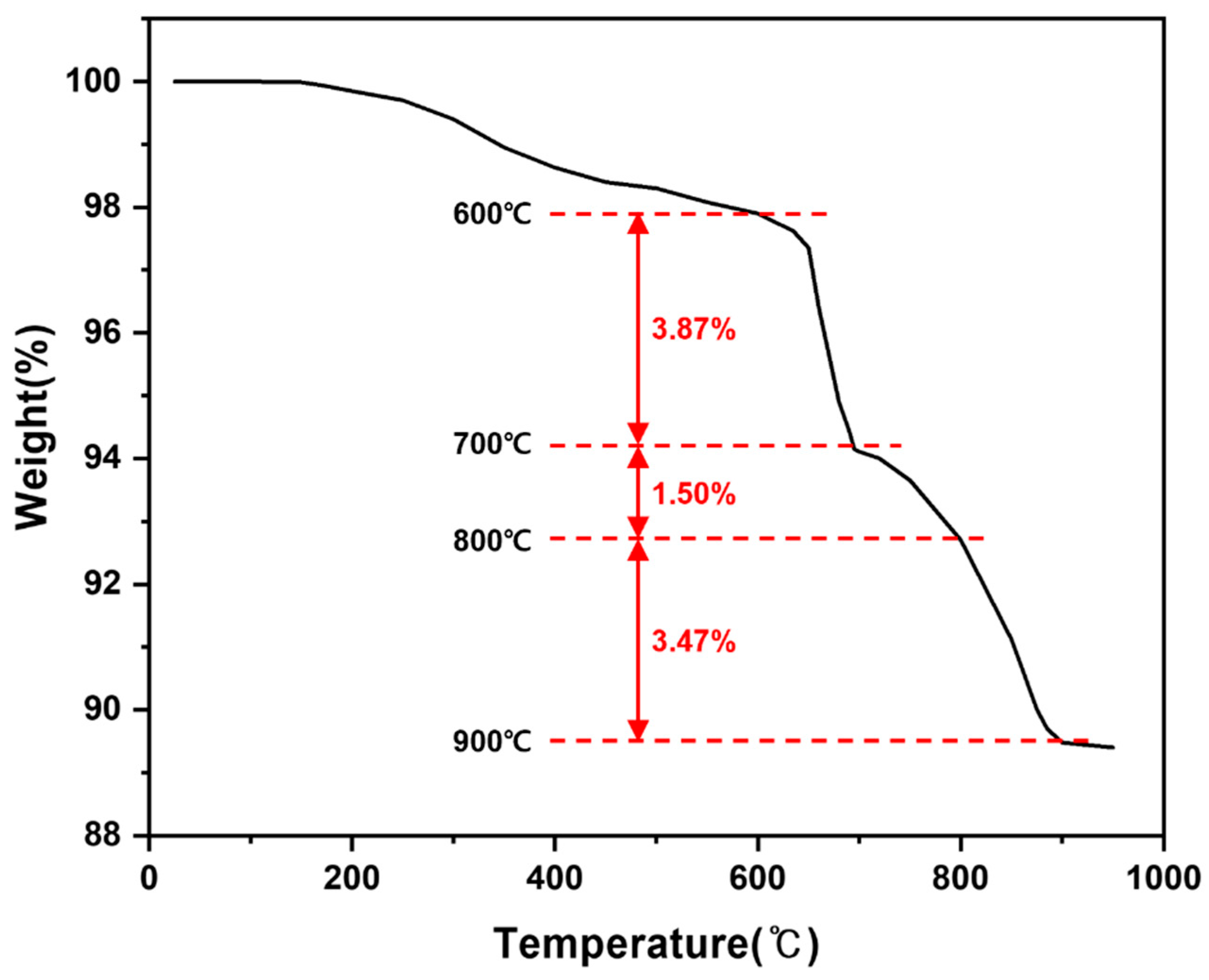
Thermogravimetric analysis (TGA) curve of the as-received LiFePO4 powder measured under Ar atmosphere.
Figure 3.
Thermogravimetric analysis (TGA) curve of the as-received LiFePO4 powder measured under Ar atmosphere.
Figure 4.
Comparison of XRD patterns between raw LiFePO4 powder and calcined LiFePO4 powder.
Figure 4.
Comparison of XRD patterns between raw LiFePO4 powder and calcined LiFePO4 powder.
Figure 5.
Horizontal tube furnace used for drying, H2 reduction, and dehydration of the LiCl-containing product: (a) photograph of the furnace system; (b) schematic diagram showing the main components: (i) thermocouple, (ii) alumina refractories, (iii) water-cooling line, (iv) gas line, (v) quartz tube, (vi) SiC heater, (vii) flowmeter, and (viii) controller.
Figure 5.
Horizontal tube furnace used for drying, H2 reduction, and dehydration of the LiCl-containing product: (a) photograph of the furnace system; (b) schematic diagram showing the main components: (i) thermocouple, (ii) alumina refractories, (iii) water-cooling line, (iv) gas line, (v) quartz tube, (vi) SiC heater, (vii) flowmeter, and (viii) controller.
Figure 6.
Vertical tube furnace used for CaCl2-assisted chlorination roasting: (a) schematic diagram of the vertical tube furnace; (b) photograph of the experimental setup. The main components are: (i) thermocouple, (ii) gas line, (iii) water-cooling line, (iv) alumina tube, (v) Kanthal heater, (vi) protection crucible, and (vii) alumina refractories.
Figure 6.
Vertical tube furnace used for CaCl2-assisted chlorination roasting: (a) schematic diagram of the vertical tube furnace; (b) photograph of the experimental setup. The main components are: (i) thermocouple, (ii) gas line, (iii) water-cooling line, (iv) alumina tube, (v) Kanthal heater, (vi) protection crucible, and (vii) alumina refractories.
Figure 7.
Equilibrium composition calculated using HSC Chemistry 6.0 (Outotec Research Oy, Pori, Finland) for the system 10H2 (g) + 5MgO + Li3PO4 + FeP + Fe2P as a function of temperature, showing negligible MgO consumption.
Figure 7.
Equilibrium composition calculated using HSC Chemistry 6.0 (Outotec Research Oy, Pori, Finland) for the system 10H2 (g) + 5MgO + Li3PO4 + FeP + Fe2P as a function of temperature, showing negligible MgO consumption.
Figure 8.
Equilibrium composition calculated using HSC Chemistry 6.0 (Outotec Research Oy, Pori, Finland) for the system 10H2 (g) + 5Al2O3 + Li3PO4 + FeP + Fe2P as a function of temperature, showing alumina consumption and the formation of Li–Al–O and Fe–Al–O phases.
Figure 8.
Equilibrium composition calculated using HSC Chemistry 6.0 (Outotec Research Oy, Pori, Finland) for the system 10H2 (g) + 5Al2O3 + Li3PO4 + FeP + Fe2P as a function of temperature, showing alumina consumption and the formation of Li–Al–O and Fe–Al–O phases.
Figure 9.
Water-leaching and purification setup for LiCl recovery: (a) photographs of the magnetic stirrer/hotplate, vacuum filtration unit with vacuum pump, and resin-based Ca2+ removal setup; (b) schematic illustration of the overall sequence (water leaching → vacuum filtration → Ca2+ removal using a cation-exchange resin). Arrows indicate the process flow direction, and the white circles represent resin beads in the cation-exchange column.
Figure 9.
Water-leaching and purification setup for LiCl recovery: (a) photographs of the magnetic stirrer/hotplate, vacuum filtration unit with vacuum pump, and resin-based Ca2+ removal setup; (b) schematic illustration of the overall sequence (water leaching → vacuum filtration → Ca2+ removal using a cation-exchange resin). Arrows indicate the process flow direction, and the white circles represent resin beads in the cation-exchange column.
Figure 10.
Thermal decomposition and phase evolution of LiCl·H2O from 0 to 1000 °C, calculated using HSC Chemistry 6.0 (Outotec Research Oy, Pori, Finland). The red line denotes Li2Cl2 (g), and the blue line denotes LiCl (g).
Figure 10.
Thermal decomposition and phase evolution of LiCl·H2O from 0 to 1000 °C, calculated using HSC Chemistry 6.0 (Outotec Research Oy, Pori, Finland). The red line denotes Li2Cl2 (g), and the blue line denotes LiCl (g).
Figure 11.
Thermogravimetric response of LiFePO4 under H2 atmosphere during stepwise heating (100–950 °C; 50 °C increments; 30 min hold at each step; 5 °C min−1), indicating a pronounced mass loss region between 600 and 900 °C.
Figure 11.
Thermogravimetric response of LiFePO4 under H2 atmosphere during stepwise heating (100–950 °C; 50 °C increments; 30 min hold at each step; 5 °C min−1), indicating a pronounced mass loss region between 600 and 900 °C.
Figure 12.
Mass-retention ratio of hydrogen-reduced products as a function of reaction time at 700–900 °C.
Figure 12.
Mass-retention ratio of hydrogen-reduced products as a function of reaction time at 700–900 °C.
Figure 13.
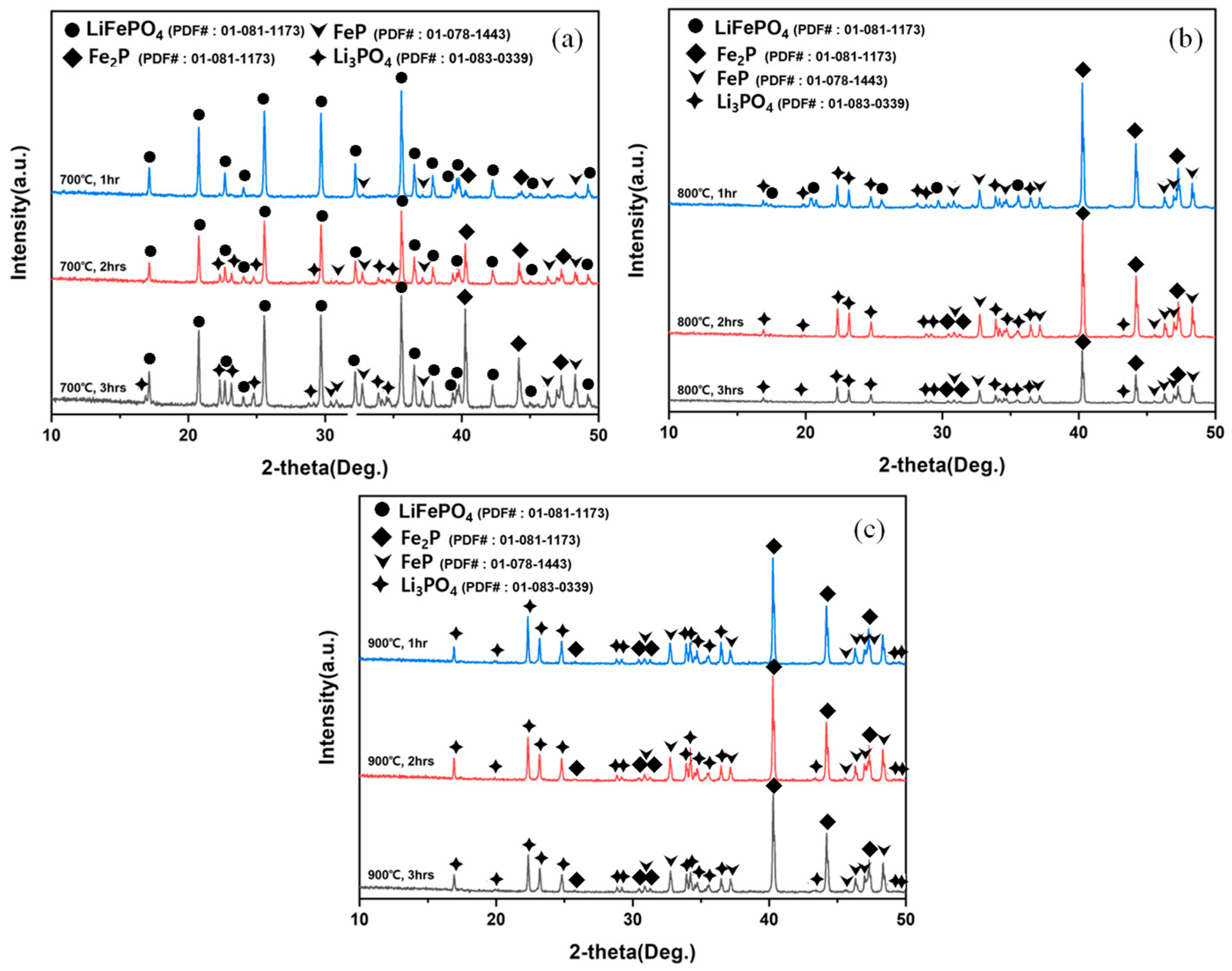
XRD patterns of hydrogen-reduction products obtained at (a) 700 °C, (b) 800 °C, and (c) 900 °C for 1–3 h, showing the progressive disappearance of LiFePO4 peaks and the formation of Li3PO4 and iron phosphides (FeP/Fe2P).
Figure 13.
XRD patterns of hydrogen-reduction products obtained at (a) 700 °C, (b) 800 °C, and (c) 900 °C for 1–3 h, showing the progressive disappearance of LiFePO4 peaks and the formation of Li3PO4 and iron phosphides (FeP/Fe2P).
Figure 14.
Phase evolution of LiFePO4 through hydrogen reduction, illustrated by XRD patterns of the raw material (a) and the reduced product (b) obtained under the selected condition (900 °C, 1 h), confirming the formation of Li3PO4 and iron phosphides (FeP/Fe2P).
Figure 14.
Phase evolution of LiFePO4 through hydrogen reduction, illustrated by XRD patterns of the raw material (a) and the reduced product (b) obtained under the selected condition (900 °C, 1 h), confirming the formation of Li3PO4 and iron phosphides (FeP/Fe2P).
Figure 15.
Temperature dependence of the standard Gibbs free energy change (ΔG°) and equilibrium constant (K) for the chlorination roasting reaction.
Figure 15.
Temperature dependence of the standard Gibbs free energy change (ΔG°) and equilibrium constant (K) for the chlorination roasting reaction.
Figure 16.
XRD patterns of chlorination-roasted products obtained at 700–1000 °C for 3 h. Major phases include LiCl, chlorapatite (Ca5(PO4)3Cl), FeP, and Fe2P; residual CaCl2 and/or phosphate-related phases may remain at lower temperatures (e.g., 700 °C) due to incomplete melting/reaction of CaCl2.
Figure 16.
XRD patterns of chlorination-roasted products obtained at 700–1000 °C for 3 h. Major phases include LiCl, chlorapatite (Ca5(PO4)3Cl), FeP, and Fe2P; residual CaCl2 and/or phosphate-related phases may remain at lower temperatures (e.g., 700 °C) due to incomplete melting/reaction of CaCl2.
Figure 17.
XRD patterns after water leaching and filtration: (a) dried residue and (b) dried filtrate.
Figure 17.
XRD patterns after water leaching and filtration: (a) dried residue and (b) dried filtrate.
Figure 18.
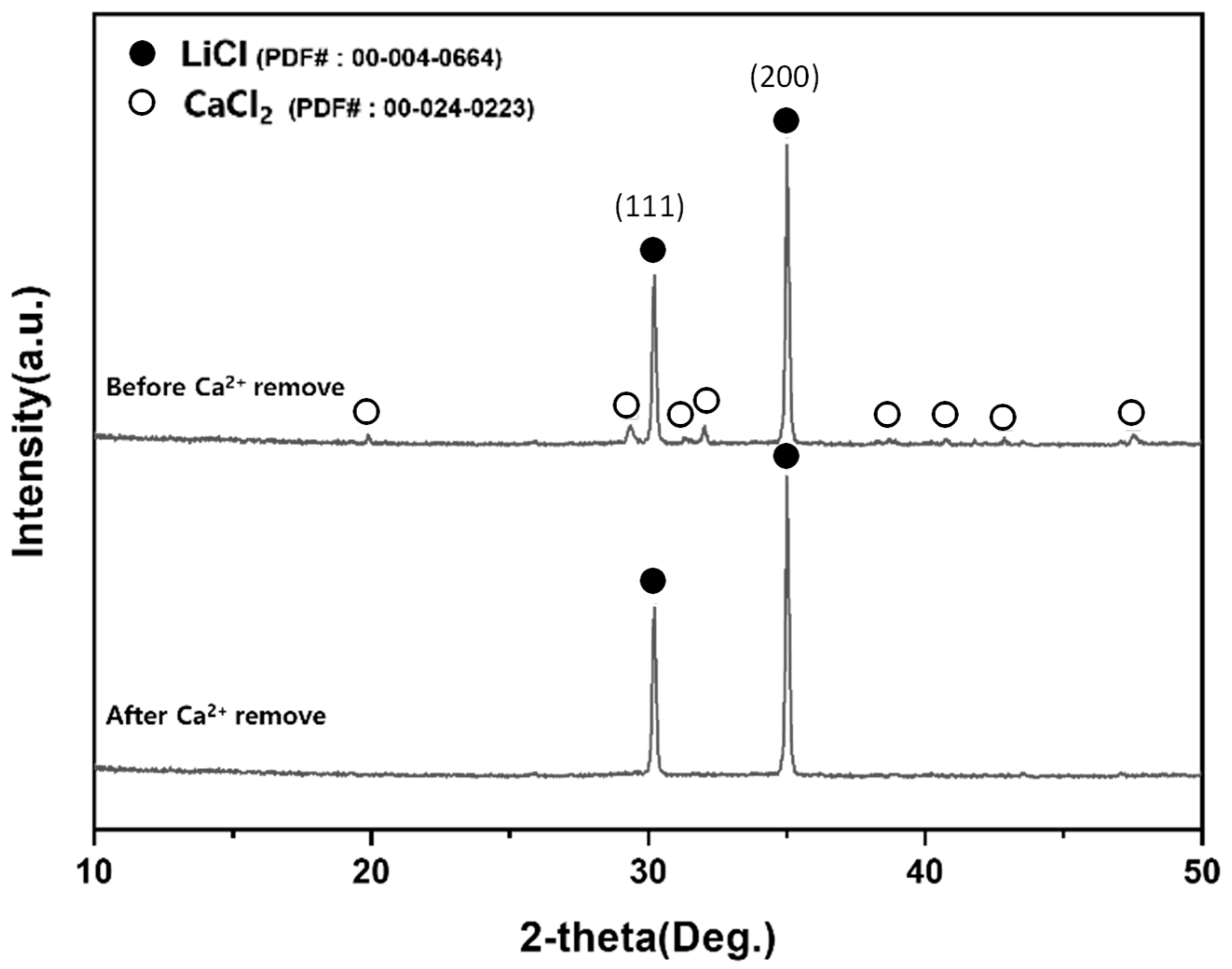
XRD comparison of the dried filtrate before and after Ca2+ removal by ion-exchange resin.
Figure 18.
XRD comparison of the dried filtrate before and after Ca2+ removal by ion-exchange resin.
Figure 19.
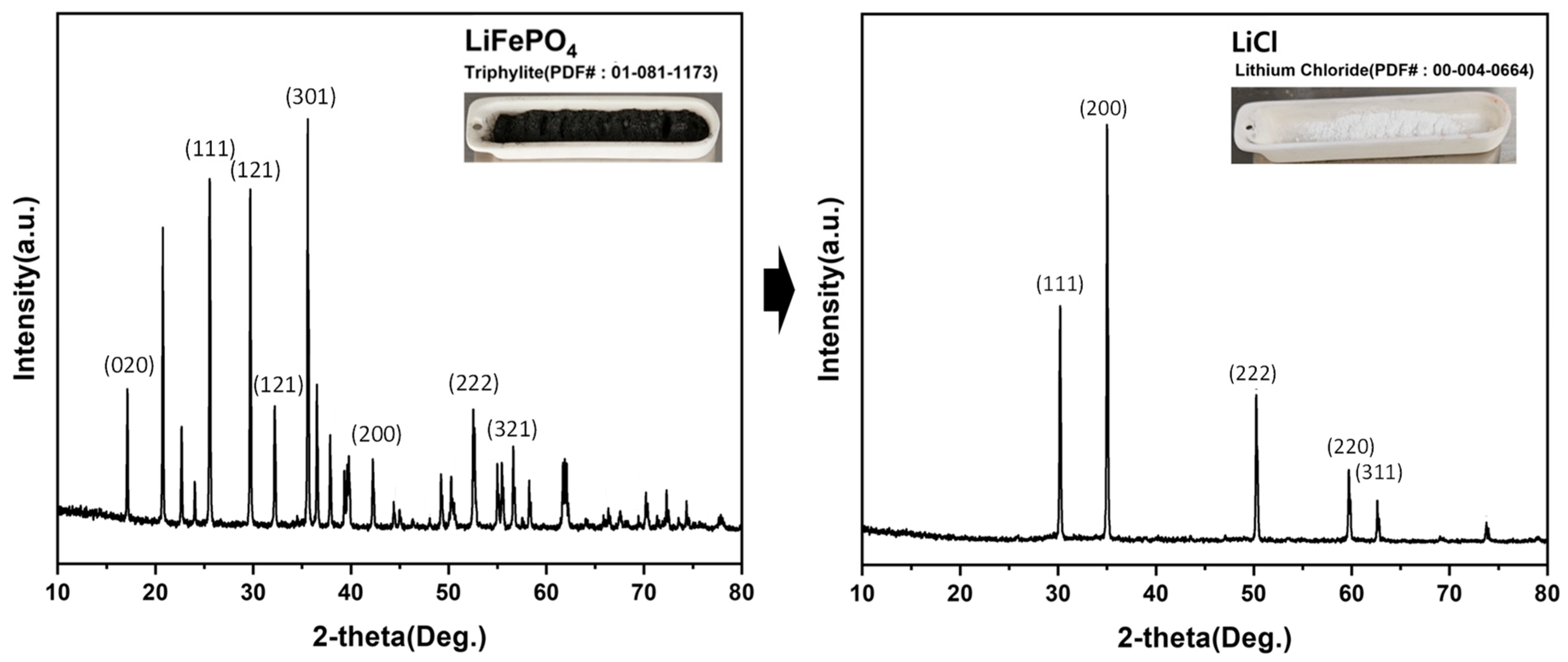
XRD patterns of the initial LiFePO4 powder and the recovered LiCl powder, confirming phase transformation and LiCl formation.
Figure 19.
XRD patterns of the initial LiFePO4 powder and the recovered LiCl powder, confirming phase transformation and LiCl formation.
Table 1.
Chemical composition of the LiFePO4 powder determined by bulk elemental analysis (sample mass: 10.0 g), compared with theoretical values.
Table 1.
Chemical composition of the LiFePO4 powder determined by bulk elemental analysis (sample mass: 10.0 g), compared with theoretical values.
| Sample Weight (g) |
|---|
| 10.0 |
|---|
| Element | Theoretical wt.% | Analyzed wt.% |
|---|
| Li | 4.40 | 4.56 |
| Fe | 35.4 | 35.1 |
| P | 19.6 | 18.4 |
Table 2.
Hydrogen content of LiFePO4 powder before and after calcination pretreatment measured by elemental analysis (EA).
Table 2.
Hydrogen content of LiFePO4 powder before and after calcination pretreatment measured by elemental analysis (EA).
| Element | Raw Material (wt.%) | Calcined LFP (wt.%) |
|---|
| H | 0.017 | - |
Table 3.
Equilibrium phase distribution of MgO (unit: mol) in the MgO crucible case under hydrogen-reduction conditions, showing Total MgO = 5.0000 mol over 300–900 °C (unit: mol).
Table 3.
Equilibrium phase distribution of MgO (unit: mol) in the MgO crucible case under hydrogen-reduction conditions, showing Total MgO = 5.0000 mol over 300–900 °C (unit: mol).
| | 300 °C | 400 °C | 500 °C | 600 °C | 700 °C | 800 °C | 900 °C |
|---|
| MgO | 3.24100 | 3.09050 | 2.97070 | 2.87290 | 2.79140 | 2.72210 | 2.66240 |
| MgO(M) | 1.75900 | 1.90950 | 2.02930 | 2.12710 | 2.20860 | 2.27790 | 2.33760 |
| Total MgO | 5.00000 | 5.00000 | 5.00000 | 5.00000 | 5.00000 | 5.00000 | 5.00000 |
Table 4.
Equilibrium phase distribution of Al2O3 (unit: mol) in the Al2O3 crucible case under hydrogen-reduction conditions, showing Total Al2O3 < 5.0000 mol over 300–900 °C, indicating alumina participation in side reactions (unit: mol).
Table 4.
Equilibrium phase distribution of Al2O3 (unit: mol) in the Al2O3 crucible case under hydrogen-reduction conditions, showing Total Al2O3 < 5.0000 mol over 300–900 °C, indicating alumina participation in side reactions (unit: mol).
| | 300 °C | 400 °C | 500 °C | 600 °C | 700 °C | 800 °C | 900 °C |
|---|
| Al2O3(C) | 1.91080 | 1.85730 | 1.81200 | 1.75090 | 1.66090 | 1.54440 | 1.41280 |
| Al2O3 | 1.82340 | 1.75890 | 1.71500 | 1.65800 | 1.57440 | 1.46590 | 1.34290 |
| Al2O3(D) | 0.28459 | 0.38192 | 0.47762 | 0.56278 | 0.62835 | 0.66995 | 0.68888 |
| Al2O3(K) | 0.16561 | 0.25114 | 0.34310 | 0.43206 | 0.50785 | 0.56396 | 0.59923 |
| Al2O3(G) | 0.04775 | 0.08801 | 0.13989 | 0.19903 | 0.25886 | 0.31327 | 0.35854 |
| Total Al2O3 | 4.23215 | 4.33727 | 4.48761 | 4.60277 | 4.63036 | 4.55748 | 4.40235 |
Table 5.
Solubility of LiCl in water as a function of temperature.
Table 5.
Solubility of LiCl in water as a function of temperature.
| Temperature (°C) | Solubility in Water (LiCl) |
|---|
| 0 | 68.29 |
| 10 | 74.48 |
| 25 | 84.25 |
| 40 | 88.7 |
| 100 | 123.44 |
Table 6.
Calculated equilibrium composition for LiCl·H2O thermal decomposition at 200–800 °C (unit: mol).
Table 6.
Calculated equilibrium composition for LiCl·H2O thermal decomposition at 200–800 °C (unit: mol).
| | 200 °C | 300 °C | 400 °C | 500 °C | 600 °C | 700 °C | 800 °C |
|---|
| H2O (g) | 0.78663 | 0.98152 | 0.99706 | 0.99925 | 0.99973 | 0.99988 | 0.99989 |
| LiCl | 0.78663 | 0.98152 | 0.99706 | 0.99924 | 0.99965 | 0.9991 | 0.99527 |
| LiCl·H2O | 0.21337 | 0.01848 | 0.00294 | 0.00075 | 0.00026 | 0.00009 | 0.00003 |
| H2O | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
Table 7.
Hydrogen-reduction conditions and mass-change results for LiFePO4 (H2 flow rate: 300 cm3 min−1; initial mass: 10.0 g; heating rate: 5 °C min−1).
Table 7.
Hydrogen-reduction conditions and mass-change results for LiFePO4 (H2 flow rate: 300 cm3 min−1; initial mass: 10.0 g; heating rate: 5 °C min−1).
| Sample | Temperature (°C) | Reaction Time (h) | H2 Flow Rate (cm3 min−1) | Initial Mass (g) | Product Mass (g) | Mass-Retention Ratio (%) |
|---|
| #1 | 700 | 1 | 300 | 10 | 9.685 | 96.85 |
| #2 | 700 | 2 | 300 | 10 | 8.638 | 86.38 |
| #3 | 700 | 3 | 300 | 10 | 8.513 | 85.13 |
| #4 | 800 | 1 | 300 | 10 | 7.667 | 76.67 |
| #5 | 800 | 2 | 300 | 10 | 7.606 | 76.06 |
| #6 | 800 | 3 | 300 | 10 | 7.518 | 75.18 |
| #7 | 900 | 1 | 300 | 10 | 7.292 | 72.92 |
| #8 | 900 | 2 | 300 | 10 | 7.274 | 72.74 |
| #9 | 900 | 3 | 300 | 10 | 7.256 | 72.56 |
Table 8.
Stoichiometric calculation of theoretical mass change associated with oxygen removal during hydrogen reduction of LiFePO4 based on Equation (3) (basis: 10.0 g of LiFePO4).
Table 8.
Stoichiometric calculation of theoretical mass change associated with oxygen removal during hydrogen reduction of LiFePO4 based on Equation (3) (basis: 10.0 g of LiFePO4).
| Basis (10.0 g LiFePO4) | Li | Fe | P | O | Total |
|---|
| Amount in raw material (mol) | 0.063388 | 0.063388 | 0.063388 | 0.253554 | 0.443719 |
| Amount in raw material (g) | 0.439979 | 3.539929 | 1.963382 | 4.05671 | 10 |
| Amount in reduced product (mol) | 0.063388 | 0.063388 | 0.063388 | 0.084518 | 0.274683 |
| Amount in reduced product (g) | 0.439979 | 3.539929 | 1.963382 | 1.352237 | 7.295527 |
| Oxygen removed (mol) | — | — | — | 0.169036 | — |
| Oxygen removed (g) | — | — | — | 2.704473 | — |
| Theoretical mass-retention ratio (%) | | | | | 72.96 |
Table 9.
Chemical composition of the hydrogen-reduction product obtained under the selected condition (900 °C, 1 h), compared with theoretical values (sample mass: 7.29 g).
Table 9.
Chemical composition of the hydrogen-reduction product obtained under the selected condition (900 °C, 1 h), compared with theoretical values (sample mass: 7.29 g).
| Sample Weight (g) |
|---|
| 7.29 |
|---|
| Element | Theoretical wt.% | Analyzed wt.% |
|---|
| Li | 6.03 | 6.11 |
| Fe | 48.5 | 47.1 |
| P | 26.9 | 25.1 |
Table 10.
Experimental conditions for chlorination roasting of the H2-reduced product with CaCl2 (Ar: 300 cm3 min−1; holding time: 3 h).
Table 10.
Experimental conditions for chlorination roasting of the H2-reduced product with CaCl2 (Ar: 300 cm3 min−1; holding time: 3 h).
| Sample | H2 Reduction | Raw Mat. | Sample Input (g) |
|---|
| Temp. | Reaction Time | Ar (cc/min) | H2 Reduction Product | CaCl2 |
|---|
| (°C) | (h) |
|---|
| #1 | 700 | 3 | 300 | 7.292 | 3.562 |
| #2 | 800 |
| #3 | 900 |
| #4 | 1000 |
Table 11.
Chemical composition of the dried product after Ca2+ removal (ICP-OES, wt.%).
Table 11.
Chemical composition of the dried product after Ca2+ removal (ICP-OES, wt.%).
| Sample Weight (g) |
|---|
| 2.009 |
|---|
| Element | Analyzed wt.% |
|---|
| Li | 16.3 |
| Ca | 0.17 |
Table 12.
Lithium content and mass balance by experimental step and calculated lithium recovery.
Table 12.
Lithium content and mass balance by experimental step and calculated lithium recovery.
| Experimental Step | Sample wt.% | Li Content | Li wt.% | Recovery Rate |
|---|
| (g) | (%) | (g) | (%) |
|---|
| Raw material (LiFePO4) | 10 | 4.56 | 0.456 | - |
| After H2 reduction | 7.292 | 6.11 | 0.446 | - |
| Recovered LiCl (powder) | 2.009 | 16.3 | 0.327 | 71.70% |
Table 13.
Comparison of representative LFP recycling routes and key outcomes reported in the literature versus this work.
Table 13.
Comparison of representative LFP recycling routes and key outcomes reported in the literature versus this work.
| Study | Route Type/
Key Steps | Main Recovered Li Form | Purification (Key) | Reported Li Recovery/Extraction
(as Defined in Each Reference) | Notes/Limitation |
|---|
Barbosa de Mattos
et al. [2] | Review (hydrometallurgy + hybrid/thermal) | Li2CO3/LiOH/others (varies) | Various | – | Highlights reagent/wastewater burden and flowsheet complexity |
| Zhao et al. [3] | Review (direct regeneration) | Re-lithiated LFP | – | – | Feed variability and scale-up challenges |
| Cao et al. [12] | CaCl2 chlorination roasting of LFP | LiCl (leaching) | Limited/depends | (reported in ref.) | Phosphate byproducts may form; condition-sensitive |
| Chang et al. [13] | CaCl2-assisted roasting (LFP) | Li salts (leaching) | Fluorine stabilization focus | (reported in ref.) | Emphasizes stabilization/impurity control |
| Kim et al. [9] | Chlorination roasting + leaching (spent LIBs) | LiCl/Li salts | Process-dependent | (reported in ref.) | Chlorination can benefit leaching; impurity management important |
| This work | H2 reduction → Li3PO4 + FeP/Fe2P (phase separation) → CaCl2 chlorination → water leaching/vacuum filtration → Ca2+ ion exchange → drying/dehydration | LiCl (powder) | Ca2+ removed by strong-acid cation exchange (H-form) | 71.70% | Addresses Ca impurity in LiCl stream; discusses chlorapatite formation and LiCl retention mechanisms |