Controllable Preparation of Si3N4@MgSiN2 Core–Shell Powders via a “Template Growth” Mechanism in NaCl-KCl Mixed Molten Salt
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Si3N4@MgSiN2 Powders
2.2. Characterizations
3. Results and Discussion
3.1. Synthesis Mechanism of Si3N4@MgSiN2 Powder
3.2. Controlled Synthesis and Microstructural Analysis of MgSiN2 Shells
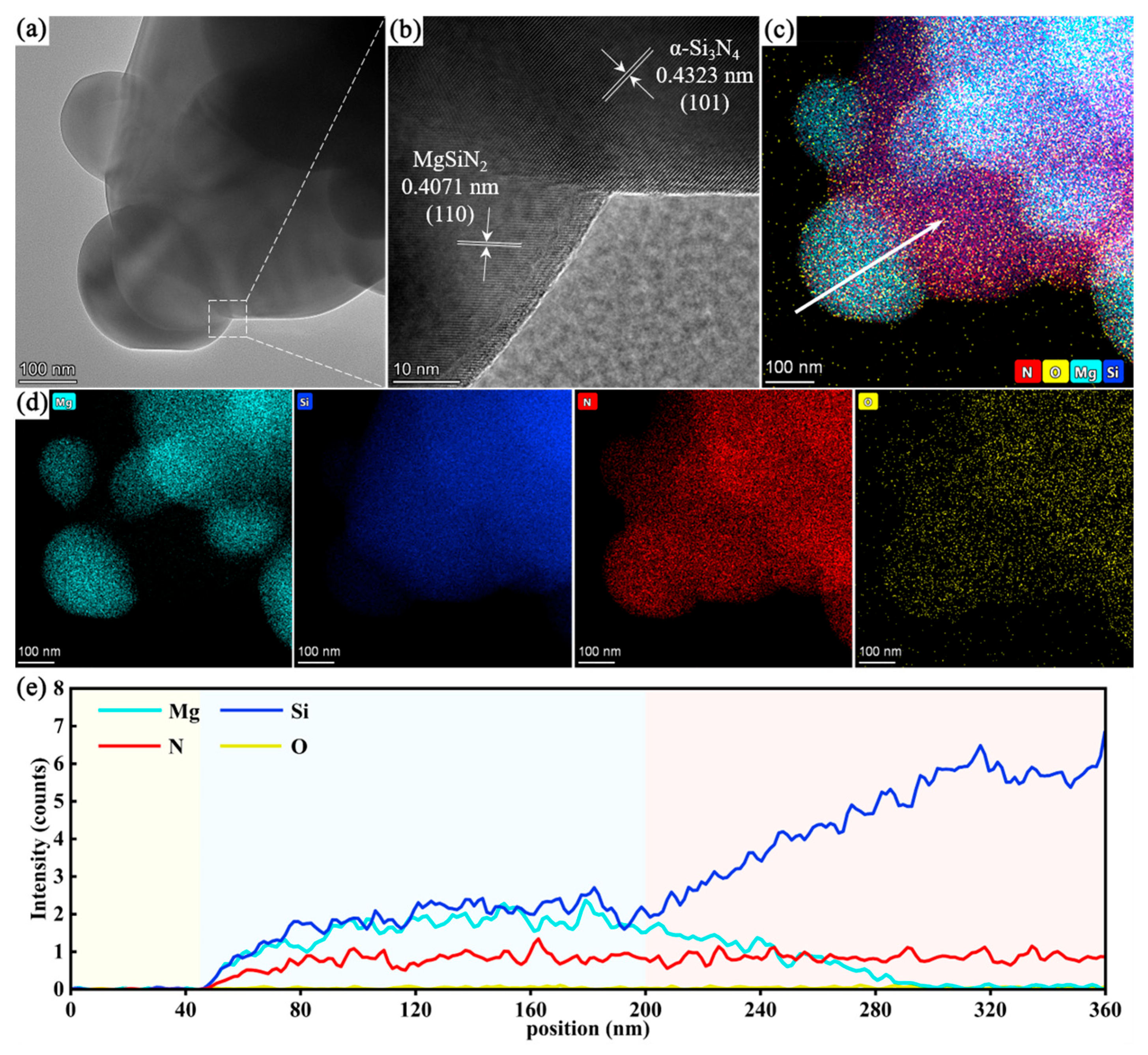
3.3. Surface Structure of Si3N4@MgSiN2 Particles
3.4. Sintering Properties of Si3N4@MgSiN2
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Tang, S.J.; Guo, W.M.; Sun, S.K.; Lin, H.T. Design Strategy of phase and microstructure of Si3N4 ceramics with simultaneously high hardness and toughness. J. Adv. Ceram. 2023, 12, 122–131. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, G.H.; Cui, W.; Ge, Y.Y.; Du, S.M.; Gao, Y.X.; Zhang, Y.Y.; Li, F.; Chen, Z.L.; Du, S.X.; et al. Plastic deformation in silicon nitride ceramics via bond switching at coherent interfaces. Science 2022, 378, 371–376. [Google Scholar] [CrossRef]
- Zhan, Z.; Deng, B.; Chen, Y.R.; Zhang, X.H.; Song, J.Z. Synergistic improvement of the mechanical and biological performance of Si3N4 by incorporating nanostructured graphene. J. Adv. Ceram. 2015, 14, 9221026. [Google Scholar] [CrossRef]
- Hirosaki, N.; Ogata, S.; Kocer, C.; Kitagawa, H.; Nakamura, Y. Molecular dynamics calculation of the ideal thermal conductivity of single-crystal α- and β-Si3N4. Phys. Rev. B Condens. Matter Mater. Phys. 2002, 65, 134110. [Google Scholar] [CrossRef]
- Wang, W.M.; Wang, W.D.; Liu, Y.M.; Zeng, K.H.; Mo, Y.; Ma, Q.S. Synergistic improvement of flexural strength and fracture toughness in Si3N4 ceramics featuring high-entropy grain boundary phase. J. Adv. Ceram. 2025, 15, 9221230. [Google Scholar] [CrossRef]
- Li, S.S.; Chen, H.B.; Wang, W.D.; Yao, D.X.; Xia, Y.F.; Zeng, Y.P. Effects of Y2O3/MgO ratio on mechanical properties and thermal conductivity of silicon nitride ceramics. Int. J. Appl. Ceram. Technol. 2022, 19, 2873–2882. [Google Scholar] [CrossRef]
- Han, Z.Q.; Liu, J.; Wang, W.W.; Zhou, Q.X.; Zhao, Z.C.; Han, Y.N.; Wang, Y.Y.; Wang, Y.; Liu, F.T.; Li, L. Effect of Er2O3/MgO ratio on mechanical and thermal properties of Si3N4 ceramics. Int. J. Appl. Ceram. Technol. 2024, 21, 2915–2922. [Google Scholar] [CrossRef]
- Hampshire, S.; Pomeroy, M.J. Oxynitride Glasses. Int. J. Appl. Ceram. Technol. 2008, 5, 155–163. [Google Scholar] [CrossRef]
- Wang, L.J.; Liu, X.J.; Qi, Q.; Ni, D.W.; Zhang, H.; Yin, J.; Qiao, Z.H.; Huang, Z.R. A new type of dense silicon nitride-based metamaterial. J. Adv. Ceram. 2025, 14, 9221037. [Google Scholar] [CrossRef]
- Kitayama, M.; Hirao, K.; Tsuge, A.; Watari, K.; Toriyama, M.; Kanzaki, S. Thermal Conductivity of β-Si3N4: II, Effect of Lattice Oxygen. J. Am. Ceram. Soc. 2000, 83, 1985–1992. [Google Scholar] [CrossRef]
- Kitayama, M.; Hirao, K.; Tsuge, A.; Toriyama, M.; Kanzaki, A. Oxygen content in β-Si3N4 crystal lattice. J. Am. Ceram. Soc. 1999, 82, 3263–3265. [Google Scholar] [CrossRef]
- Kitayama, M.; Hirao, K.; Toriyama, M.; Kanzaki, S. Thermal Conductivity of β-Si3N4: I, Effects of various microstructural factors. J. Am. Ceram. Soc. 1999, 82, 3105–3112. [Google Scholar] [CrossRef]
- Wang, W.D.; Liu, Y.M.; Pan, Y.; Chen, S.A.; Guo, L.; Peng, Z.; Yao, D.X.; Ma, Q.S.; Zeng, Y.P. The effects of silicon additive content on thermal conductivity and mechanical properties of Si3N4 ceramics. J. Am. Ceram. Soc. 2025, 108, e20534. [Google Scholar] [CrossRef]
- Wang, W.D.; Yao, D.X.; Chen, H.B.; Xia, Y.F.; Zuo, K.H.; Yin, J.W.; Liang, H.Q.; Zeng, Y.P. ZrSi2–MgO as novel additives for high thermal conductivity of β-Si3N4 ceramics. J. Am. Ceram. Soc. 2020, 103, 2090–2100. [Google Scholar] [CrossRef]
- Zhuang, Y.H.; Sun, F.; Zhou, L.J.; Jiang, C.X.; Wang, J.J.; Li, S.; Liao, S.J. The influence of magnesium compounds on the properties of silicon nitride ceramics. Int. J. Appl. Ceram. Technol. 2024, 21, 2273–2287. [Google Scholar] [CrossRef]
- Peng, A.Y.; Li, J.G.; Chen, Y.; Li, M.J.; Shen, Q. Low-temperature fabrication of Si3N4 ceramics with high thermal conductivities using a single Mg2Si sintering additive. Ceram. Int. 2023, 49, 39473–39478. [Google Scholar] [CrossRef]
- Lee, H.M.; Lee, E.B.; Kim, D.L.; Kim, D.K. Comparative study of oxide and non-oxide additives in high thermal conductive and high strength Si3N4 ceramics. Ceram. Int. 2016, 42, 17466–17471. [Google Scholar] [CrossRef]
- Lee, H.M.; Tatami, J.; Kim, D.K. Microstructural evolution of Si3N4 ceramics from starting powders with different α-to-β ratios. J. Ceram. Soc. Jpn. 2016, 124, 800–807. [Google Scholar] [CrossRef]
- Lia, S.J.; Zhou, L.J.; Jiang, C.X.; Wang, J.J.; Zhuan, Y.H.; Li, S. Thermal conductivity and mechanical properties of Si3N4 ceramics with binary fluoride sintering additives. J. Eur. Ceram. Soc. 2021, 41, 6971–6982. [Google Scholar] [CrossRef]
- Yang, C.P.; Ye, F.; Ma, J.; Ding, J.J.; Zhang, B.; Liu, Q.; Zhang, H.Q. Comparative study of fluoride and non-fluoride additives in high thermal conductive silicon nitride ceramics fabricated by spark plasma sintering and post-sintering heat treatment. Ceram. Int. 2018, 44, 23202–23207. [Google Scholar] [CrossRef]
- Hu, F.; Zhao, L.; Xie, Z.P. Silicon nitride ceramics with high thermal conductivity and excellent mechanical properties fabricated with MgF2 sintering aid and post-sintering heat treatment. J. Ceram. Sci. Technol. 2016, 7, 423–428. [Google Scholar] [CrossRef]
- Ratzker, B.; Sokol, M.; Kalabukhov, S.; Frage, N. High-pressure spark plasma sintering of silicon nitride with LiF additive. J. Eur. Ceram. Soc. 2018, 38, 1271–1277. [Google Scholar] [CrossRef]
- Luo, C.X.; Zhang, Y.X.; Deng, T.F. Pressureless sintering of high performance silicon nitride ceramics at 1620 °C. Ceram. Int. 2021, 47, 29371–29378. [Google Scholar] [CrossRef]
- Wang, W.D.; Yao, D.X.; Liang, H.Q.; Xia, Y.F.; Zuo, K.H.; Yin, J.W.; Zeng, Y.P. Improved thermal conductivity of β-Si3N4 ceramics by lowering SiO2/Y2O3 ratio using YH2 as sintering additive. J. Am. Ceram. Soc. 2020, 103, 5567–5572. [Google Scholar] [CrossRef]
- Wang, W.D.; Yao, D.X.; Liang, H.Q.; Xia, Y.F.; Zuo, K.H.; Yin, J.W.; Zeng, Y.P. Improved thermal conductivity of β-Si3N4 ceramics through the modification of the liquid phase by using GdH2 as a sintering additive. Ceram. Int. 2021, 47, 5631–5638. [Google Scholar] [CrossRef]
- Wang, W.D.; Yao, D.X.; Liang, H.Q.; Xia, Y.F.; Zuo, K.H.; Yin, J.W.; Zeng, Y.P. Enhanced thermal conductivity in Si3N4 ceramics prepared by using ZrH2 as an oxygen getter. J. Alloys Compd. 2021, 855, 157451. [Google Scholar] [CrossRef]
- Li, Y.; Kim, H.N.; Wu, H.B.; Kim, M.J.; Ko, J.W.; Park, Y.J.; Huang, Z.; Kim, H.D. Enhanced thermal conductivity in Si3N4 ceramic with the addition of Y2Si4N6C. J. Am. Ceram. Soc. 2018, 101, 4128–4136. [Google Scholar] [CrossRef]
- Fan, B.B.; Sun, W.; Hu, Z.L.; Zhao, D.K.; Wang, B.H.; Zhao, S.; Zhang, S.J.; Li, F.; Xi, Z.P.; Chen, K.C.; et al. Preparation of Si3N4 ceramics with enhanced thermal conductivity and mechanical properties using nitride sintering additives. Int. J. Appl. Ceram. Technol. 2025, 22, e15182. [Google Scholar] [CrossRef]
- Fu, S.; Yang, Z.C.; Li, H.H.; Wang, L.; Li, Y.; Li, J.T. Effects of Gd2O3 and MgSiN2 sintering additives on the thermal conductivity and mechanical properties of Si3N4 ceramics. Int. J. Appl. Ceram. Technol. 2022, 20, 1855–1864. [Google Scholar] [CrossRef]
- Li, J.B.; Jiang, Q.G.; Pan, Z.F.; Lv, D.L.; Wu, S.H. Fabrication of silicon nitride with high thermal conductivity and flexural strength by hot-pressing flowing sintering. Int. J. Appl. Ceram. Technol. 2023, 21, 2841–2849. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, R.X.; Zheng, Y.H.; Zhao, J.X.; Sui, T.Y.; Li, X.L.; Lin, B. Effect of the ratio of Y2O3 and MgSiN2 sintering additives on the microstructure, thermal and mechanical properties of Si3N4 ceramic. Ceram. Int. 2023, 49, 36490–36496. [Google Scholar] [CrossRef]
- Hayashi, H.; Hirao, K.; Toriyama, M.; Kanzaki, S.; Itatani, K. MgSiN2 Addition as a Means of Increasing the Thermal Conductivity of β-Silicon Nitride. J. Am. Ceram. Soc. 2001, 84, 3060–3062. [Google Scholar] [CrossRef]
- Zhou, M.; Liu, Y.; Li, X.L.; Sui, T.Y.; Su, D.; Ji, H.M. Microstructure evolution of Si3N4 ceramics with high thermal conductivity by using Y2O3 and MgSiN2 as sintering additives. Ceram. Int. 2023, 49, 26331–26337. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, R.X.; Zheng, Y.H.; Zhao, J.X.; Sui, T.Y.; Li, X.L.; Lin, B. Densification, microstructure, thermal and mechanical properties of Si3N4 ceramics: Effect of Y2Si4N6C and MgSiN2 content. Ceram. Int. 2024, 50, 38507–38513. [Google Scholar] [CrossRef]
- Li, M.; Zhou, X.B.; Yang, H.; Du, S.Y.; Huang, Q. The critical issues of SiC materials for future nuclear systems. Scr. Mater. 2018, 143, 149–153. [Google Scholar] [CrossRef]
- Lu, D.L.; Yang, P.; Huang, Y.; Huang, N.S.; Wu, S.H. Enhanced thermal conductivity in Si3N4 ceramics by carbonizing polydopamine coatings. Ceram. Int. 2022, 48, 18615–18624. [Google Scholar] [CrossRef]
- Ge, M.Z.; Kan, H.H.; Yang, H.; Qiao, J.M.; Jiang, Z.H. Additive-coated Si3N4 powder prepared by the sol-gel method. MRS Online Proc. Libr. 1994, 346, 667. [Google Scholar] [CrossRef]
- Shao, J.Q.; Li, M.; Chang, K.K.; Huang, Y.; Ren, D.L.; Wang, J.; Zhou, X.B.; He, L.; Huang, F.; Du, S.Y.; et al. Fabrication and characterization of SPS sintered SiC-based ceramic from Y3Si2C2-coated SiC powders. J. Eur. Ceram. Soc. 2018, 38, 4833–4841. [Google Scholar] [CrossRef]
- Wan, P.; Li, M.; Huang, Q. Molten salt assisted synthesis of Dy3Si2C2 coated SiC powders and sintering behavior of SiC ceramics. J. Inorg. Mater. 2021, 36, 49. [Google Scholar] [CrossRef]
- Li, W.X.; Pang, H.J.; Zhang, Z.X.; Cheng, L.Y.; Wang, Y.F.; Zhang, X.L.; Mu, J.B.; Dong, Y.; Wang, Y.M.; Zhang, X.R. Core–shell SiCw@TiC composite whisker-reinforced Al2O3 ceramics: Preparation, properties, and toughening mechanisms. J. Adv. Ceram. 2025, 14, 9221093. [Google Scholar] [CrossRef]
- Zhang, G.G.; Li, G.S.; Lan, Z.A.; Lin, L.H.; Savateev, A.; Heil, T.; Zafeiratos, S.; Wang, X.C.; Antonietti, M. Optimizing optical absorption, exciton dissociation, and charge transfer of a polymeric carbon nitride with ultrahigh solar hydrogen production activity. Angew. Chem. Int. Ed. 2017, 56, 13445–13449. [Google Scholar] [CrossRef]
- Li, Y.; Gong, F.; Zhou, Q.; Feng, X.H.; Fan, J.J.; Xiang, Q.J. Crystalline isotype heptazine-/triazine-based carbon nitride heterojunctions for an improved hydrogen evolution. Appl. Catal. B-Environ. 2020, 268, 118381. [Google Scholar] [CrossRef]
- Liu, D.; Fu, Q.G.; Chu, Y.H. Molten salt synthesis, formation mechanism, and oxidation behavior of nanocrystalline HfB2 powders. J. Adv. Ceram. 2020, 9, 35–44. [Google Scholar] [CrossRef]
- Zong, F.J.; Meng, C.Z.; Guo, Z.M.; Ji, F.; Xiao, H.D.; Zhang, X.J.; Ma, J.; Ma, H.L. Synthesis and characterization of magnesium nitride powder formed by Mg direct reaction with N2. J. Alloys Compd. 2010, 508, 172–176. [Google Scholar] [CrossRef]
- Hu, J.J.; Qin, J.; Zhou, Z.Y.; Shi, Z.Q.; Peng, K.; Gao, K.; Xia, H.Y. Facile synthesis of BP powder via molten salt method and their photoelectrochemical properties. Ceram. Int. 2023, 49, 14596–14606. [Google Scholar] [CrossRef]
- Lenčéš, Z.; Pentráková, L.; Hrabalová, M.; Šajgalík, P.; Hirao, K. Decomposition of MgSiN2 in nitrogen atmosphere. J. Eur. Ceram. Soc. 2011, 31, 1473–1480. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, C.Y.; Luo, W.; Bai, L.; Xu, Y.; Hao, X.D.; Zhu, J.F.; Guo, S.W. Facile synthesis of hollow Ti3AlC2 microrods in molten salts via Kirkendall effect. J. Adv. Ceram. 2022, 11, 1491–1497. [Google Scholar] [CrossRef]
- Kim, J.M.; Ko, S.I.; Kim, H.N.; Ko, J.W.; Lee, J.W.; Kim, H.D.; Park, Y.J. Effects of microstructure and intergranular glassy phases on thermal conductivity of silicon nitride. Ceram. Int. 2017, 43, 5441–5449. [Google Scholar] [CrossRef]
- Ye, J.; Zhang, S.; Lee, W.E. Molten salt synthesis and characterization of SiC coated carbon black particles for refractory castable applications. J. Eur. Ceram. Soc. 2013, 33, 2023–2029. [Google Scholar] [CrossRef]
- Li, Y.J.; Xu, L.Q.; Wang, L.C.; Ju, Z.C.; Yang, L.S.; Xing, Z.; Qian, Y.T. Synthesis of MgSiN2 cuboids by a solid-state reaction. Chem. Lett. 2010, 39, 888–889. [Google Scholar] [CrossRef]
- Sarin, V.K. On the α-to-β phase transformation in silicon nitride. Mater. Sci. Eng. A 1988, 105–106, 151–159. [Google Scholar] [CrossRef]







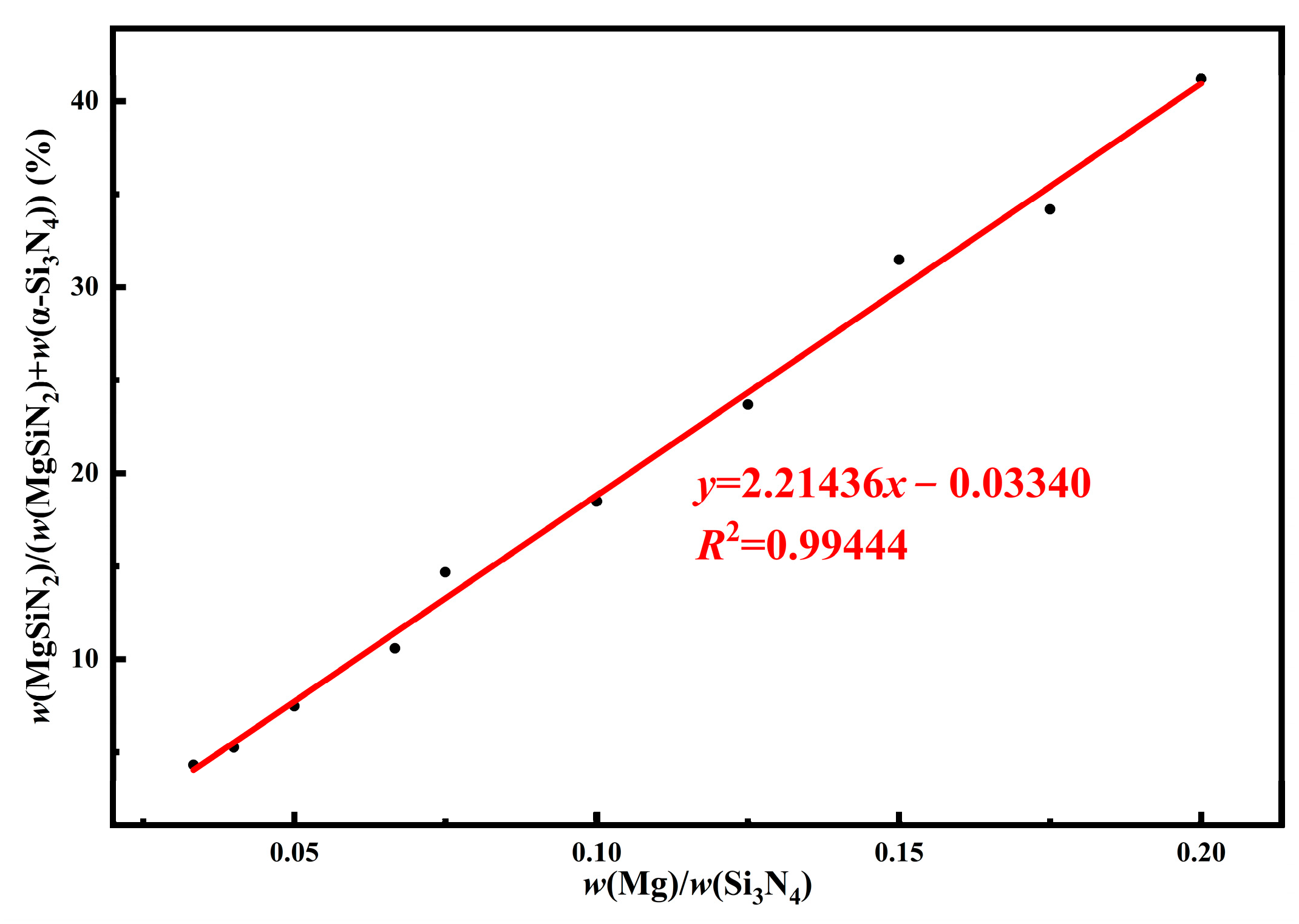


| Sample | Types of Salt | Content of Each Phase/wt.% | ||
|---|---|---|---|---|
| α-Si3N4 | β-Si3N4 | MgSiN2 | ||
| 1 | KCl | 77.922 | 6.094 | 15.894 |
| 2 | NaCl | 76.076 | 6.206 | 17.718 |
| 3 | NaCl + KCl | 72.228 | 5.794 | 21.978 |
| Sample | Mg/Si3N4 Mass Ratio | Content of Each Phase/wt.% | w(MgSiN2)/(w(MgSiN2) + w(α-Si3N4)) | |||
|---|---|---|---|---|---|---|
| α-Si3N4 | β-Si3N4 | MgSiN2 | MgO | |||
| MS1 | 0.200 | 57.207 | 1.849 | 40.103 | 0.841 | 41.212 |
| MS2 | 0.175 | 62.258 | 2.963 | 32.332 | 2.447 | 34.181 |
| MS3 | 0.150 | 65.034 | 3.465 | 29.872 | 1.629 | 31.475 |
| MS4 | 0.125 | 72.228 | 2.788 | 22.417 | 2.567 | 23.685 |
| MS5 | 0.100 | 78.991 | 2.334 | 17.915 | 0.760 | 18.487 |
| MS6 | 0.075 | 80.732 | 3.720 | 13.884 | 1.664 | 14.674 |
| MS7 | 0.067 | 85.987 | 2.925 | 10.185 | 0.903 | 10.590 |
| MS8 | 0.050 | 89.385 | 2.610 | 7.228 | 0.777 | 7.481 |
| MS9 | 0.040 | 91.801 | 2.604 | 5.098 | 0.497 | 5.261 |
| MS10 | 0.033 | 90.809 | 3.296 | 4.096 | 1.798 | 4.316 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Liu, Y.; Wang, W.; Mo, Y.; Guo, L.; Peng, Z.; Wang, W.; Ma, Q. Controllable Preparation of Si3N4@MgSiN2 Core–Shell Powders via a “Template Growth” Mechanism in NaCl-KCl Mixed Molten Salt. Materials 2026, 19, 1475. https://doi.org/10.3390/ma19071475
Liu Y, Wang W, Mo Y, Guo L, Peng Z, Wang W, Ma Q. Controllable Preparation of Si3N4@MgSiN2 Core–Shell Powders via a “Template Growth” Mechanism in NaCl-KCl Mixed Molten Salt. Materials. 2026; 19(7):1475. https://doi.org/10.3390/ma19071475
Chicago/Turabian StyleLiu, Yiming, Weiming Wang, Yong Mo, Lei Guo, Zheng Peng, Weide Wang, and Qingsong Ma. 2026. "Controllable Preparation of Si3N4@MgSiN2 Core–Shell Powders via a “Template Growth” Mechanism in NaCl-KCl Mixed Molten Salt" Materials 19, no. 7: 1475. https://doi.org/10.3390/ma19071475
APA StyleLiu, Y., Wang, W., Mo, Y., Guo, L., Peng, Z., Wang, W., & Ma, Q. (2026). Controllable Preparation of Si3N4@MgSiN2 Core–Shell Powders via a “Template Growth” Mechanism in NaCl-KCl Mixed Molten Salt. Materials, 19(7), 1475. https://doi.org/10.3390/ma19071475







