Engineering Design Strategies for Boosting Photocatalytic Activity: Theory-to-Data-Driven Perspective
Abstract
1. Introduction
2. Fundamentals of Photocatalysis
3. Synthesis Approaches and Particle Growth Mechanisms
4. Morphological and Structural Design
5. Nanostructures Across Dimensions
5.1. Quantum Dots and 1D Nanostructures
5.2. Two-Dimensional (2D) Nanostructures
5.3. Three-Dimensional (3D) Nanostructures
6. Design Strategies for Heterojunctions

7. In Situ and Operando Techniques to Characterize the Photocatalyst
8. Computational Modeling and DFT Simulations, and Machine Learning Design
9. Challenges and Future Perspectives
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ralphs, K.; Liu, J.; Lan, L.; Hardacre, C.; Skillen, N.; Robertson, P.K.J. Photocatalytic valorisation of real-world substrates. RSC Sustain. 2025, 3, 2079–2110. [Google Scholar] [CrossRef]
- Naz, R.; Tahir, M. Recent Developments in Metal-free Materials for Photocatalytic and Electrocatalytic Carbon Dioxide Conversion into Value Added Products. Energy Fuels 2025, 39, 6127–6150. [Google Scholar] [CrossRef]
- Zhang, Z.; Qian, Z.; Chen, M.; Zhu, R.; Zhang, F.; Zhong, T.; Lin, J.; Ning, L.; Xie, W.; Creutzig, F.; et al. Worldwide rooftop photovoltaic electricity generation may mitigate global warming. Nat. Clim. Chang. 2025, 15, 393–402. [Google Scholar] [CrossRef]
- Jia, P.; Yu, Y.; Chen, T.; Huang, H. “Electricity”-Assisted Catalytic Solar-to-Fuel Processes. Angew. Chem. Int. Ed. 2025, 64, e202508809. [Google Scholar] [CrossRef]
- Xi, Z.; Liu, M. Advancing photoelectrochemical systems for sustainable energy and chemical production: Challenges and opportunities. npj Mater. Sustain. 2025, 3, 18. [Google Scholar] [CrossRef]
- Zaman, A.; Kausar, A.; Asim, S.; Mansha, A. Modified TiO2 and ZnO photocatalysts for microplastic degradation: Mechanisms, challenges, and recent advances. Photochem. Photobiol. Sci. 2026. [Google Scholar] [CrossRef]
- Kook, M.; Peterson, C.; Bhat, A.S.; Nefedova, A.; Vanetsev, A.; Ivask, A.; Kisand, V. Artificial aging induced changes in ZnO- and TiO2-based polyacrylic surface coatings. npj Mater. Degrad. 2026, 10, 28. [Google Scholar] [CrossRef]
- Atanacio-Sánchez, X.; Pech-Rodríguez, W.J.; Armendáriz-Mireles, E.N.; Castillo-Robles, J.A.; Meléndez-González, P.C.; Rocha-Rangel, E. Improving performance of ZnO flexible dye sensitized solar cell by incorporation of graphene oxide. Microsyst. Technol. 2020, 26, 3591–3599. [Google Scholar] [CrossRef]
- Chen, C.; Shenoy, S.; Chuaicham, C.; Trakulmututa, J.; Tian, Q.; Zhang, H.; Sasaki, K. Enhancing photocatalytic efficiency: Geopolymer-supported TiO2 composites for sustainable organic pollutant degradation. Opt. Mater. 2024, 150, 115165. [Google Scholar] [CrossRef]
- Singh, K.; Jhajharia, P.; Kumari, V.; Kumar, R.; Rawat, D.; Singh, P.P.; Joshi, R.; Sahu, P.K.; Kumar, V. Exploration of engineered ZnO-based photocatalysts for green hydrogen production and carbon dioxide conversion. J. Mol. Struct. 2025, 1346, 143139. [Google Scholar] [CrossRef]
- Pech-Rodríguez, W.J.; Şahin, N.E.; Suarez-Velázquez, G.G.; Meléndez-González, P.C. Semiconductor-Based Photoelectrocatalysts in Water Splitting: From the Basics to Mechanistic Insights—A Brief Review. Materials 2025, 18, 1952. [Google Scholar] [CrossRef]
- Hedrich, C.; Petit, R.R.; Minjauw, M.M.; Burson, A.R.; Haugg, S.; Furlan, K.P.; Detavernier, C.; Dendooven, J.; Blick, R.H.; Zierold, R. Impact of surface treatments on the photocatalytic performance of anodic aluminum oxide templates. Sci. Rep. 2025, 15, 15030. [Google Scholar] [CrossRef]
- Zhanfeng, Q.; Guancheng, L.; Xiuli, G. Multi-element synergy in photocatalytic materials integrated mechanism-design-preparation strategies. Mater. Res. Express 2025, 12, 062001. [Google Scholar] [CrossRef]
- Taneja, Y.; Dube, D.; Singh, R. Recent advances in elemental doping and simulation techniques: Improving structural, photophysical and electronic properties of titanium dioxide. J. Mater. Chem. C 2024, 12, 14774–14808. [Google Scholar] [CrossRef]
- Sohail, M.; Rauf, S.; Irfan, M.; Hayat, A.; Alghamdi, M.M.; El-Zahhar, A.A.; Ghernaout, D.; Al-Hadeethi, Y.; Lv, W. Recent developments, advances and strategies in heterogeneous photocatalysts for water splitting. Nanoscale Adv. 2024, 6, 1286–1330. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Xiong, Y.; Jin, C.; Chen, C.; Zhang, L.; Jiang, J.; Xie, J. Cu-Ce MOF-based heterojunction catalysts for the electrooxidation of 5-hydroxymethylfurfural. Ind. Crops Prod. 2025, 226, 120714. [Google Scholar] [CrossRef]
- Xu, K.; Liu, Z.; Qi, S.; Yin, Z.; Deng, S.; Zhang, M.; Sun, Z. Construction of Ag-modified TiO2/ZnO heterojunction nanotree arrays with superior photocatalytic and photoelectrochemical properties. RSC Adv. 2020, 10, 34702–34711. [Google Scholar] [CrossRef]
- Ke, S.; Naghizadeh, M.; Sun, L.; Jin, H.; Dong, S.; Huang, T. Highly reactive ZnFe2O4/TiO2 p-n heterojunction photocatalyst accelerates interfacial charge transfer for boosted photodegradation of ammonia nitrogen. Chem. Eng. Sci. 2025, 307, 121361. [Google Scholar] [CrossRef]
- Ashu Abey, S.; Reis, N.M.; Emanuelsson, E.A.C.; Expósito, A.J. Harnessing visible light: Advanced photocatalytic strategies for sustainable environmental reactions. Chem. Eng. J. 2025, 519, 164951. [Google Scholar] [CrossRef]
- Foo, J.J.; Chiah, Z.-J.; Ng, S.-F.; Ong, W.-J. Strategic facet engineering of bismuth-based photocatalysts for the applications in solar-to-chemical conversion. InfoScience 2024, 1, e12023. [Google Scholar] [CrossRef]
- Wang, S.; Mo, P.; Li, D.; Syed, A. Intelligent Algorithms Enable Photocatalyst Design and Performance Prediction. Catalysts 2024, 14, 217. [Google Scholar] [CrossRef]
- Li, K.; Du, H.; Liu, L.; Yang, H.; Fang, J.; Li, D. Research progress of machine learning in the field of photocatalysis applications. J. Ind. Eng. Chem. 2025, 151, 146–166. [Google Scholar] [CrossRef]
- Zhang, C.; Ahmad, I.; Ahmed, S.B.; Ali, M.D.; Karim, M.R.; Bayahia, H.; Khasawneh, M.A. A review of rare earth oxides-based photocatalysts: Design strategies and mechanisms. J. Water Process Eng. 2024, 63, 105548. [Google Scholar] [CrossRef]
- Chen, Q.; Wang, H.; Gu, M.; Zhang, X.; Wang, G. Reasonable design pentamerous artificial photosynthesis system for efficient overall CO2 reduction. Chem. Eng. J. 2024, 481, 148656. [Google Scholar] [CrossRef]
- Bao, T.; Xi, Y.; Zhang, C.; Du, P.; Xiang, Y.; Li, J.; Yuan, L.; Yu, C.; Liu, C. Highly efficient nitrogen fixation over S-scheme heterojunction photocatalysts with enhanced active hydrogen supply. Natl. Sci. Rev. 2024, 11, nwae093. [Google Scholar] [CrossRef]
- Zhang, K.; Wang, X.; Su, Y. Investigation of Two Novel Heterojunction Photocatalysts with Boosted Hydrogen Evolution Performance. Nanomaterials 2024, 14, 1947. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Jiang, Y.-n.; Zhang, M.; Zhang, X.; Ma, Y. Non-Noble-Metal-Doped Carbon Nitride Photocatalysts for Water Splitting Screened Out by Empty Defect States and the d-Band Center. ACS Appl. Mater. Interfaces 2024, 16, 12455–12466. [Google Scholar] [CrossRef]
- Zhang, M.; Liu, R.; Zhang, F.; Zhao, H.; Li, X.; Lang, X.; Guo, Z. Interfacial design of pyrene-based covalent organic framework for overall photocatalytic H2O2 synthesis in water. J. Colloid Interface Sci. 2025, 678, 1170–1180. [Google Scholar] [CrossRef] [PubMed]
- Musa, E.N.; Yadav, A.K.; Smith, K.T.; Jung, M.S.; Stickle, W.F.; Eschbach, P.; Ji, X.; Stylianou, K.C. Boosting Photocatalytic Hydrogen Production by MOF-Derived Metal Oxide Heterojunctions with a 10.0% Apparent Quantum Yield. Angew. Chem. Int. Ed. 2024, 63, e202405681. [Google Scholar] [CrossRef]
- Shao, Y.; Zhang, Y.; Chen, C.; Dou, S.; Lou, Y.; Dong, Y.; Zhu, Y.; Pan, C. Enhancement of H2O2 generation rate in porphyrin photocatalysts via crystal facets regulation to create strong internal electric field. Chin. J. Catal. 2024, 61, 205–214. [Google Scholar] [CrossRef]
- Saber, A.F.; Liao, H.-T.; Li, P.-J.; Chen, Y.-F.; Mabuti, L.; Kuo, S.-W.; Lüder, J.; El-Mahdy, A.F.M. Synergistic structural engineering of donor–acceptor–acceptor type conjugated microporous polymers as photocatalysts for boosting sunlight-driven hydrogen evolution. J. Colloid Interface Sci. 2025, 699, 138156. [Google Scholar] [CrossRef]
- Zhang, T.; Wang, P.; Yue, S.; Li, F.; Zhao, Z.; Zhan, S. Tailoring the surface and interface structures of photocatalysts to enhance hydrogen production. Sci. China Chem. 2024, 67, 1161–1174. [Google Scholar] [CrossRef]
- Luo, Y.; Wang, X.; Gao, F.; Jiang, L.; Wang, D.; Pan, H. From Single Atom Photocatalysts to Synergistic Photocatalysts: Design Principles and Applications. Adv. Funct. Mater. 2025, 35, 2418427. [Google Scholar] [CrossRef]
- Velasco-Garcia, L.; Casadevall, C. Bioinspired photocatalytic systems towards compartmentalized artificial photosynthesis. Commun. Chem. 2023, 6, 263. [Google Scholar] [CrossRef]
- Mohammadzadeh Kakhki, R. Beyond photosynthesis: Engineering self-healing photocatalytic systems for sustainability. Colloid Interface Sci. Commun. 2025, 67, 100842. [Google Scholar] [CrossRef]
- Chakravorty, A.; Roy, S. A review of photocatalysis, basic principles, processes, and materials. Sustain. Chem. Environ. 2024, 8, 100155. [Google Scholar] [CrossRef]
- Mandade, P. Chapter 5—Introduction, basic principles, mechanism, and challenges of photocatalysis. In Handbook of Nanomaterials for Wastewater Treatment; Bhanvase, B., Sonawane, S., Pawade, V., Pandit, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2021; pp. 137–154. [Google Scholar]
- Sakar, M.; Mithun Prakash, R.; Do, T.-O. Insights into the TiO2-Based Photocatalytic Systems and Their Mechanisms. Catalysts 2019, 9, 680. [Google Scholar] [CrossRef]
- Ishaq, T.; Ehsan, Z.; Qayyum, A.; Abbas, Y.; Irfan, A.; Al-Hussain, S.A.; Irshad, M.A.; Zaki, M.E.A. Recent Strategies to Improve the Photocatalytic Efficiency of TiO2 for Enhanced Water Splitting to Produce Hydrogen. Catalysts 2024, 14, 674. [Google Scholar] [CrossRef]
- Zhang, J.; Shi, T.; Liu, T.; Gao, F.; Cai, D.; Liu, P.; Yang, S.; Zhang, Y. Design and synthesis of a UV–vis-NIR response heterostructure system: For efficient solar energy conversion and BPA photocatalytic degradation. Appl. Surf. Sci. 2024, 653, 159346. [Google Scholar] [CrossRef]
- Vinukonda, A.; Bolledla, N.; Jadi, R.K.; Chinthala, R.; Devadasu, V.R. Synthesis of nanoparticles using advanced techniques. Next Nanotechnol. 2025, 8, 100169. [Google Scholar] [CrossRef]
- Goswami, Y.C.; Bisauriya, R.; Goswami, R.; Hlaing, A.A.; Moe, T.T. Hydrothermal synthesis of SnO2/cellulose nanocomposites: Optical, Structural, and morphological characterization. Sci. Rep. 2025, 15, 9752. [Google Scholar] [CrossRef]
- Tazim, T.Q.; Kawsar, M.; Sahadat Hossain, M.; Bahadur, N.M.; Ahmed, S. Hydrothermal synthesis of nano-metal oxides for structural modification: A review. Next Nanotechnol. 2025, 7, 100167. [Google Scholar] [CrossRef]
- Ghamarpoor, R.; Fallah, A.; Jamshidi, M. A Review of Synthesis Methods, Modifications, and Mechanisms of ZnO/TiO2-Based Photocatalysts for Photodegradation of Contaminants. ACS Omega 2024, 9, 25457–25492. [Google Scholar] [CrossRef]
- Koozegar Kaleji, B.; Gorgani, M. Comparison of sol-gel and hydrothermal synthesis methods on the structural, optical and photocatalytic properties of Nb/Ag codoped TiO2 mesoporous nanoparticles. Int. J. Environ. Anal. Chem. 2022, 102, 3357–3372. [Google Scholar] [CrossRef]
- Rafiq, U.; Mehraj, O.; Lone, S.; Wahid, M.; Majid, K. Solvothermal synthesis of Ag2WO4/Sb2WO6 heterostructures for enhanced charge transfer properties and efficient visible-light-driven photocatalytic activity and stability. J. Environ. Chem. Eng. 2020, 8, 104301. [Google Scholar] [CrossRef]
- Wang, Y.; Yang, C.; Liu, Y.; Fan, Y.; Dang, F.; Qiu, Y.; Zhou, H.; Wang, W.; Liu, Y. Solvothermal Synthesis of ZnO Nanoparticles for Photocatalytic Degradation of Methyl Orange and p-Nitrophenol. Water 2021, 13, 3224. [Google Scholar] [CrossRef]
- Patil, M.S.; Sarvalkar, P.D.; Mulani, S.R.; Sheikh, A.D.; Devan, R.S.; Sharma, K.K.K. Effect of solvothermal reaction time on adsorption and photocatalytic activity of spinel ZnFe2O4 nanoparticles. J. Photochem. Photobiol. A Chem. 2025, 459, 116001. [Google Scholar] [CrossRef]
- Khan, M.I.; Bhatti, K.A.; Qindeel, R.; Bousiakou, L.G.; Alonizan, N.; Fazal-e-Aleem. Investigations of the structural, morphological and electrical properties of multilayer ZnO/TiO2 thin films, deposited by sol–gel technique. Results Phys. 2016, 6, 156–160. [Google Scholar] [CrossRef]
- García-Molina, R.; Suárez-Velázquez, G.G.; Pech-Rodríguez, W.J.; Ordóñez, L.C.; Melendez-Gonzalez, P.C.; Sánchez-Padilla, N.M.; González-Quijano, D. Soft chemistry synthesis of size-controlled ZnO nanostructures as photoanode for dye-sensitized solar cell. Rev. Mex. Ing. Quím. 2024, 23, 12. [Google Scholar] [CrossRef]
- Zhan, F.; Qi, J.; Li, R.; Zhao, H.; Liu, Y.; La, P. Synthesis, photocatalytic applications and future prospects of noble metal-modified semiconductors fabricated via photodeposition. Phys. Chem. Chem. Phys. 2025, 27, 13770–13792. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Gu, X.; Zhang, N.; Wu, Z.; Du, Y. Solvothermal synthesis of PtPb nanoparticles with efficient alcohol oxidation performance. Nanoscale 2025, 17, 21756–21765. [Google Scholar] [CrossRef]
- Matias, M.L.; Reis-Machado, A.S.; Rodrigues, J.; Calmeiro, T.; Deuermeier, J.; Pimentel, A.; Fortunato, E.; Martins, R.; Nunes, D. Microwave Synthesis of Visible-Light-Activated g-C3N4/TiO2 Photocatalysts. Nanomaterials 2023, 13, 1090. [Google Scholar] [CrossRef] [PubMed]
- Ortega, P.P.; Amoresi, R.A.C.; Teodoro, M.D.; Longo, E.; Ponce, M.A.; Simões, A.Z. Relationship among morphology, photoluminescence emission, and photocatalytic activity of Eu-doped ceria nanostructures: A surface-type effect. Ceram. Int. 2023, 49, 21411–21421. [Google Scholar] [CrossRef]
- Al-Keisy, A.; Ahmed, D.S.; Abid, H.N. Impact of pH on the morphology and photocatalytic activity of Bi2WO6 nanoplates for the degradation of rhodamine and methylene orange. AIP Conf. Proc. 2025, 3169, 060005. [Google Scholar] [CrossRef]
- de Almeida, J.C.; Rodrigues, T.A.; da Silva, G.T.S.T.; Ribeiro, C.; de Mendonça, V.R. Unveiling the influence of alkaline modifiers in CuO synthesis on its photocatalytic activity for CO2 reduction. Mater. Adv. 2024, 5, 6479–6488. [Google Scholar] [CrossRef]
- Li, L.; Li, J.; Kim, B.H.; Huang, J. The effect of morphology and crystal structure on the photocatalytic and photoelectrochemical performances of WO3. RSC Adv. 2024, 14, 2080–2087. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Li, Y.; Hou, Q.; Gao, L.; Ding, Z. One-Step Solvothermal Synthesis of Fe3O4 Acicular Aggregates Induced by Reaction Medium and Urea for Photocatalytic Degradation of Azo Dyes. Nanomaterials 2025, 15, 341. [Google Scholar] [CrossRef]
- Shi, D.; Ma, Y.; Zhang, B. Solvothermal Synthesis of Magnetic CoFe2O4/Carbon Nanocomposites with Superior Visible Light Photocatalysis. Langmuir 2025, 41, 9548–9558. [Google Scholar] [CrossRef]
- Bikerchalen, S.; Mllaoiy, L.; Saddik, N.; Bakiz, B.; Villain, S.; Taoufyq, A.; Guinneton, F.; Valmalette, J.C.; Gavarri, J.R.; Benlhachemi, A. Optimization of solvothermal synthesis parameters for Bi24O31Cl10: Enhanced photocatalytic performance, degradation pathways, and mechanism of organic pollutants. J. Phys. Chem. Solids 2026, 209, 113267. [Google Scholar] [CrossRef]
- Ortega López, Y.; Medina Vázquez, H.; Salinas Gutiérrez, J.; Guzmán Velderrain, V.; López Ortiz, A.; Collins Martínez, V. Synthesis Method Effect of CoFe2O4 on Its Photocatalytic Properties for H2 Production from Water and Visible Light. J. Nanomater. 2015, 2015, 985872. [Google Scholar] [CrossRef]
- Nyankson, E.; Yeboah, N.; Jnr, S.O.; Onaja, S.; Mensah, T.; Efavi, J.K. The effect of synthesis route on the photocatalytic performance of Ag-TiO2 using rhodamine b dyes, pesticides, and pharmaceutical waste as model pollutants. Mater. Res. Express 2022, 9, 094001. [Google Scholar] [CrossRef]
- Estrada-Flores, S.; Martínez-Luévanos, A.; Perez-Berumen, C.M.; García-Cerda, L.A.; Flores-Guia, T.E. Relationship between morphology, porosity, and the photocatalytic activity of TiO2 obtained by sol–gel method assisted with ionic and nonionic surfactants. Bol. Soc. Esp. Cerám. Y Vidr. 2020, 59, 209–218. [Google Scholar] [CrossRef]
- Ebrahimi, A.; Haghighi, M.; Shabani, M. Design of novel solar-light-induced KBi6O9I/Ag–AgVO3 nanophotocatalyst with Ag-bridged Z-scheme charge carriers separation and boosted photo-elimination of hospital effluents. Environ. Pollut. 2024, 346, 123584. [Google Scholar] [CrossRef]
- Liu, X.; Li, H.; Xu, N.; Guo, Y.; Zhang, X.; Wang, X. Rational Design of Co(II)-Pyridine-Decorated Reduced Phosphomolybdate Photocatalysts for Efficient Aniline Oxidation under Mild Conditions. Chin. J. Chem. 2024, 42, 2970–2978. [Google Scholar] [CrossRef]
- Belik, Y.A.; Vergilessov, R.; Kovaleva, E.A.; Svetlichnyi, V.A.; Vodyankina, O.V. Design of heterostructure photocatalysts based on layered perovskite-like bismuth silicate. Appl. Surf. Sci. 2025, 682, 161733. [Google Scholar] [CrossRef]
- Zhong, R.; Liao, H.; Deng, Q.; Zou, X.; Wu, L. Preparation of a novel composite photocatalyst BiOBr/ZIF-67 for enhanced visible-light photocatalytic degradation of RhB. J. Mol. Struct. 2022, 1259, 132768. [Google Scholar] [CrossRef]
- Li, Y.-X.; Chen, X.; Jiang, Z.-Y.; Luan, J.; Guo, F. Rational Design and Synthesis of Fe-Doped Co-Based Coordination Polymer Composite Photocatalysts for the Degradation of Norfloxacin and Ciprofloxacin. Inorg. Chem. 2024, 63, 6514–6525. [Google Scholar] [CrossRef]
- Li, Y.; Gan, L.-H. NiS2/CdS photocatalysts with high specific surface area and excellent H2 evolution performance. Int. J. Hydrogen Energy 2024, 60, 1500–1508. [Google Scholar] [CrossRef]
- Lee, Y.; Kim, T.; Kim, B.; Choi, S.; Kim, K. Synthesis of TiO2/MoSx/Ag nanocomposites via photodeposition for enhanced photocatalysis and membrane fouling mitigation. J. Environ. Chem. Eng. 2023, 11, 109266. [Google Scholar] [CrossRef]
- Selvaraj, S.; Patrick, D.S.; Manikandan, V.S.; Vangari, G.A.; Mohan, M.K.; Navaneethan, M. Synergistic effects of La-doping on ZnO nanostructured photocatalysts for enhanced MB dye degradation. Surf. Interfaces 2024, 51, 104538. [Google Scholar] [CrossRef]
- Phan, C.M.; Nguyen, H.M. Role of Capping Agent in Wet Synthesis of Nanoparticles. J. Phys. Chem. A 2017, 121, 3213–3219. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Zhu, M.; Liu, T. Effects of chain length of surfactants and their adsorption on nanoparticles on stability of CO2-in-water emulsions. Colloids Surf. A Physicochem. Eng. Asp. 2022, 644, 128877. [Google Scholar] [CrossRef]
- Aulakh, M.K.; Dua, J.; Pal, B. Influence of capping agents on morphology and photocatalytic response of ZnS nanostructures towards crystal violet degradation under UV and sunlight. Sep. Purif. Technol. 2022, 281, 119869. [Google Scholar] [CrossRef]
- Xia, Y.; Xia, X.; Peng, H.-C. Shape-Controlled Synthesis of Colloidal Metal Nanocrystals: Thermodynamic versus Kinetic Products. J. Am. Chem. Soc. 2015, 137, 7947–7966. [Google Scholar] [CrossRef]
- Yuan, S.; Heng, X.; Zhang, Q.; Hu, J. pH-induced aggregation growth of large Au nanoparticles from zwitterionic ligand-modified small Au nanoparticles. Colloids Surf. A Physicochem. Eng. Asp. 2016, 506, 6–12. [Google Scholar] [CrossRef]
- Alarcon, R.; Walter, M.; Paez, M.; Azócar, M.I. Ostwald Ripening and Antibacterial Activity of Silver Nanoparticles Capped by Anti-Inflammatory Ligands. Nanomaterials 2023, 13, 428. [Google Scholar] [CrossRef] [PubMed]
- Wayman, T.M.R.; Lomonosov, V.; Ringe, E. Capping Agents Enable Well-Dispersed and Colloidally Stable Metallic Magnesium Nanoparticles. J. Phys. Chem. C 2024, 128, 4666–4676. [Google Scholar] [CrossRef]
- Perez, M. Gibbs–Thomson effects in phase transformations. Scr. Mater. 2005, 52, 709–712. [Google Scholar] [CrossRef]
- Behrens, M.A.; Franzén, A.; Carlert, S.; Skantze, U.; Lindfors, L.; Olsson, U. On the Ostwald ripening of crystalline and amorphous nanoparticles. Soft Matter 2025, 21, 2349–2354. [Google Scholar] [CrossRef]
- Alcorn, F.M.; Chattoraj, M.; van der Veen, R.M.; Jain, P.K. Watching Plasmon-Induced Nanoparticle Ostwald Ripening. J. Phys. Chem. C 2023, 127, 16538–16544. [Google Scholar] [CrossRef]
- Liu, L.; Nakouzi, E.; Sushko, M.L.; Schenter, G.K.; Mundy, C.J.; Chun, J.; De Yoreo, J.J. Connecting energetics to dynamics in particle growth by oriented attachment using real-time observations. Nat. Commun. 2020, 11, 1045. [Google Scholar] [CrossRef] [PubMed]
- Piwoński, I.; Spilarewicz-Stanek, K.; Kisielewska, A.; Kądzioła, K.; Cichomski, M.; Ginter, J. Examination of Ostwald ripening in the photocatalytic growth of silver nanoparticles on titanium dioxide coatings. Appl. Surf. Sci. 2016, 373, 38–44. [Google Scholar] [CrossRef]
- Salzmann, B.B.V.; van der Sluijs, M.M.; Soligno, G.; Vanmaekelbergh, D. Oriented Attachment: From Natural Crystal Growth to a Materials Engineering Tool. Acc. Chem. Res. 2021, 54, 787–797. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Wang, X.; Cao, K.; An, Y.; Song, X.; Liu, N.; Xu, F.; Gao, Z.; Jiang, K. ZnO Nanorods with Tunable Aspect Ratios Deriving from Oriented-attachment for Enhanced Performance in Quantum-dot Sensitized Solar Cells. Electrochim. Acta 2017, 231, 1–12. [Google Scholar] [CrossRef]
- Sarahan, M.C.; Osterloh, F.E.; Browning, N.D. Morphology Effects on Photocatalytic Activity of Niobate Materials. Microsc. Microanal. 2008, 14, 338–339. [Google Scholar] [CrossRef]
- Hu, K.H.; Hu, X.G.; Xu, Y.F.; Pan, X.Z. The effect of morphology and size on the photocatalytic properties of MoS2. React. Kinet. Mech. Catal. 2010, 100, 153–163. [Google Scholar] [CrossRef]
- Cen, W.; Xiong, T.; Tang, C.; Yuan, S.; Dong, F. Effects of Morphology and Crystallinity on the Photocatalytic Activity of (BiO)2CO3 Nano/microstructures. Ind. Eng. Chem. Res. 2014, 53, 15002–15011. [Google Scholar] [CrossRef]
- Li, L.; Jing, Y.; Zhang, J.; Guo, J. Photocatalytic degradation of NO by MnO2 catalyst: The decisive relationship between crystal phase, morphology and activity. J. Hazard. Mater. 2025, 487, 137228. [Google Scholar] [CrossRef]
- Xu, Y.; Wu, S.; Wan, P.; Sun, J.; Hood, Z.D. Introducing Ti3+ defects based on lattice distortion for enhanced visible light photoreactivity in TiO2 microspheres. RSC Adv. 2017, 7, 32461–32467. [Google Scholar] [CrossRef]
- Cheng, H.; Wang, J.; Zhao, Y.; Han, X. Effect of phase composition, morphology, and specific surface area on the photocatalytic activity of TiO2 nanomaterials. RSC Adv. 2014, 4, 47031–47038. [Google Scholar] [CrossRef]
- Roškarič, M.; Žerjav, G.; Zavašnik, J.; Finšgar, M.; Pintar, A. Effect of TiO2 Morphology on the Properties and Photocatalytic Activity of g-C3N4/TiO2 Nanocomposites Under Visible-Light Illumination. Molecules 2025, 30, 460. [Google Scholar] [CrossRef] [PubMed]
- Deng, Z.; Osuga, R.; Matsubara, M.; Kanie, K.; Muramatsu, A. Morphological effect of TiO2 nanoparticles in TiO2/g-C3N4 heterojunctions on photocatalytic dye degradation. Chem. Lett. 2024, 53, upae171. [Google Scholar] [CrossRef]
- Yin, X.; Li, X.; Gu, W.; Zou, W.; Liu, H.; Zhu, L.; Fu, Z.; Lu, Y. Morphology effect on photocatalytic activity in Bi3Fe0.5Nb1.5O9. Nanotechnology 2018, 29, 265706. [Google Scholar] [CrossRef] [PubMed]
- Cao, Q.; Feng, J.; Chang, K.T.; Liang, W.; Lu, H. Emerging Opportunities of Colloidal Quantum Dots for Photocatalytic Organic Transformations. Adv. Mater. 2025, 37, 2409096. [Google Scholar] [CrossRef]
- Gui, X.; Lu, Y.; Wang, Q.; Cai, M.; Sun, S. Application of Quantum Dots for Photocatalytic Hydrogen Evolution Reaction. Appl. Sci. 2024, 14, 5333. [Google Scholar] [CrossRef]
- Ghasemzadeh, R.; Akhbari, K. Band Gap Engineering of MOF-801 via Loading of γ-Fe2O3 Quantum Dots Inside It as a Visible Light-Responsive Photocatalyst for Degradation of Acid Orange 7. Cryst. Growth Des. 2023, 23, 6359–6368. [Google Scholar] [CrossRef]
- Ghasemzadeh, R.; Akhbari, K. Templated synthesis of ZnO quantum dots via double solvents method inside MOF-801 as emerging photocatalyst for photodegradation of Acid Blue 25 under UV light. J. Photochem. Photobiol. A Chem. 2024, 448, 115306. [Google Scholar] [CrossRef]
- Ishfaq, M.; Khan, S.A.; Nazir, M.A.; Ali, S.; Younas, M.; Mansha, M.; Shah, S.S.A.; Arshad, M.; ur Rehman, A. The in situ synthesis of sunlight-driven Chitosan/MnO2@MOF-801 nanocomposites for photocatalytic reduction of Rhodamine-B. J. Mol. Struct. 2024, 1301, 137384. [Google Scholar] [CrossRef]
- Lv, M.; Wang, S.; Shi, H. Carbon quantum dots/BiVO4 S-scheme piezo-photocatalysts improved carrier separation for efficient antibiotic removal. J. Mater. Sci. Technol. 2024, 201, 21–31. [Google Scholar] [CrossRef]
- Zhao, P.; Jin, B.; Zhang, Q.; Peng, R. Facile synthesis of quantum dots/TiO2 photocatalyst with superior photocatalytic activity: The effect of carbon nitride quantum dots and N-doped carbon dots. Res. Chem. Intermed. 2021, 47, 5229–5247. [Google Scholar] [CrossRef]
- Jiang, Z.; Feng, Q.; Hao, Y.; Xu, L.; Liu, C. Enhanced photocatalytic activity by loading Zn0.5Cd0.5S quantum dots onto flower-like BiOI under visible light. J. Ind. Eng. Chem. 2024, 129, 202–210. [Google Scholar] [CrossRef]
- Malitha, M.D.; Molla, M.T.H.; Bashar, M.A.; Chandra, D.; Ahsan, M.S. Fabrication of a reusable carbon quantum dots (CQDs) modified nanocomposite with enhanced visible light photocatalytic activity. Sci. Rep. 2024, 14, 17976. [Google Scholar] [CrossRef] [PubMed]
- Alshoaibi, A.; Islam, S.; Alamer, K. CdS Quantum Dot Encapsulated in Anatase/Silica Core–Shell Nanostructures: A Synergistic Approach for Efficient Photocatalytic Water Purification. Catalysts 2025, 15, 182. [Google Scholar] [CrossRef]
- Jawale, N.; Umarji, G.; Damkale, S.; Arbuj, S. Enhanced photocatalytic performance of hydrothermally synthesized TiO2 nanowires for H2 production via water splitting. Next Energy 2025, 6, 100205. [Google Scholar] [CrossRef]
- Giuffrida, F.; Calcagno, L.; Leonardi, A.A.; Cantarella, M.; Zimbone, M.; Impellizzeri, G. Enhancing the photocatalytic properties of doped TiO2 nanowires grown by seed-assisted thermal oxidation. Thin Solid Films 2023, 771, 139783. [Google Scholar] [CrossRef]
- Yan, Z.; Sun, Z.; Liu, X.; Jia, H.; Du, P. Cadmium sulfide/graphitic carbon nitride heterostructure nanowire loading with a nickel hydroxide cocatalyst for highly efficient photocatalytic hydrogen production in water under visible light. Nanoscale 2016, 8, 4748–4756. [Google Scholar] [CrossRef]
- Ji, Z.; Wang, X.; Zhang, H.; Lin, S.; Meng, H.; Sun, B.; George, S.; Xia, T.; Nel, A.E.; Zink, J.I. Designed Synthesis of CeO2 Nanorods and Nanowires for Studying Toxicological Effects of High Aspect Ratio Nanomaterials. ACS Nano 2012, 6, 5366–5380. [Google Scholar] [CrossRef]
- Shehab, M.A.; Sharma, N.; Valsesia, A.; Karacs, G.; Kristály, F.; Koós, T.; Leskó, A.K.; Nánai, L.; Hernadi, K.; Németh, Z. Preparation and Photocatalytic Performance of TiO2 Nanowire-Based Self-Supported Hybrid Membranes. Molecules 2022, 27, 2951. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Yang, G.; Xiang, Z.; Wang, C.; Chu, K.; Guo, W.; Han, N. Design of two-dimensional porous photocatalysts and their applications in solar fuel and valuable chemical production. J. Environ. Chem. Eng. 2024, 12, 113483. [Google Scholar] [CrossRef]
- El Ouardi, M.; Madigou, V.; Chevallier, V.; Merlen, A.; BaQais, A.; Saadi, M.; Ait Ahsaine, H.; Arab, M. Synthesis of ZnV2O6 nanosheet photocatalysts for efficient photodegradation of Rhodamine B: Experimental and RSM modeling. J. Environ. Chem. Eng. 2024, 12, 113505. [Google Scholar] [CrossRef]
- Linh, P.H.; Do Chung, P.; Van Khien, N.; Oanh, L.T.M.; Thu, V.T.; Bach, T.N.; Hang, L.T.; Hung, N.M.; Lam, V.D. A simple approach for controlling the morphology of g-C3N4 nanosheets with enhanced photocatalytic properties. Diam. Relat. Mater. 2021, 111, 108214. [Google Scholar] [CrossRef]
- Luo, X.; Dong, Y.; Wang, D.; Duan, Y.; Lei, K.; Mao, L.; Li, Y.; Zhao, Q.; Sun, Y. Facile synthesis of g-C3N4 nanosheets for effective degradation of organic pollutants via ball milling. Rev. Adv. Mater. Sci. 2023, 62, 20230123. [Google Scholar] [CrossRef]
- Wang, M.; Wang, M.; Peng, F.; Sun, X.; Han, J. Fabrication of g-C3N4 Nanosheets Anchored With Controllable CdS Nanoparticles for Enhanced Visible-Light Photocatalytic Performance. Front. Chem. 2021, 9, 746031. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Sun, X.; Li, H.; Huang, H.; Ma, T. Rational design of dimensionally matched 2D/2D COF based photocatalysts for highly efficient noble-metal-free solar energy catalysis. J. Mater. Chem. C 2024, 12, 15224–15231. [Google Scholar] [CrossRef]
- Otaif, K.D.; Mnefgui, S.; Elgazzar, E. Development of Prussian blue analogue nanosheets as highly efficient photocatalysts for the degradation of organic pollutants in water sources. Inorg. Chem. Commun. 2025, 177, 114421. [Google Scholar] [CrossRef]
- Luo, Z.; Leo, C.P.; Guo, F.; Zhao, X.-y. Transition of BiOCl nanosheet into cotton-like clusters for piezoelectric enhanced photocatalytic degradation of dye and photocatalytic hydrogen production. Chem. Eng. J. Adv. 2025, 21, 100698. [Google Scholar] [CrossRef]
- Chen, X.-L.; Ni, M.; Li, Y.-R.; Pan, Y.-A.; Chi, C.; Yang, Y.; Li, S.-M.; Biswas, S.; Sun, Y.-B.; Xia, X.-H. Design of 2D/2D Pt SA-MoS2/ZnIn2S4 photocatalysts for boosting hydrogen production. Chem. Eng. J. 2025, 520, 165713. [Google Scholar] [CrossRef]
- Kouser, S.; Hezam, A.; Namratha, K.; Byrappa, K.; Khanum, S.A. Morphology effect on photocatalytic activity of self-doped ZnO nanoflowers. J. Iran. Chem. Soc. 2023, 20, 2561–2568. [Google Scholar] [CrossRef]
- Wang, H.; Lian, Y. A mechanistic study of oleic acid-mediated solvothermal shape controllable preparation of zinc oxide nanostructures. J. Alloys Compd. 2014, 594, 141–147. [Google Scholar] [CrossRef]
- Wang, Z.; Byun, J.; Lee, S.; Seo, J.; Park, B.; Kim, J.C.; Jeong, H.Y.; Bang, J.; Lee, J.; Oh, S.H. Vacancy driven surface disorder catalyzes anisotropic evaporation of ZnO (0001) polar surface. Nat. Commun. 2022, 13, 5616. [Google Scholar] [CrossRef]
- Zare, M.; Namratha, K.; Byrappa, K.; Surendra, D.M.; Yallappa, S.; Hungund, B. Surfactant assisted solvothermal synthesis of ZnO nanoparticles and study of their antimicrobial and antioxidant properties. J. Mater. Sci. Technol. 2018, 34, 1035–1043. [Google Scholar] [CrossRef]
- Lu, J.; Li, Z.; Wu, B.; Jiang, Z.; Pei, C. Nanosheet-Stacked g-C3N4 Tubes with Carbon Vacancies for Enhanced Photocatalytic H2 Evolution. ACS Appl. Nano Mater. 2025, 8, 6133–6143. [Google Scholar] [CrossRef]
- Li, R.; Zheng, M.; Zhou, X.; Zhang, D.; Shi, Y.; Li, C.; Yang, M. Carbon vacancies in porous g-C3N4 nanosheets induced robust H2O2 production for highly efficient photocatalysis-self-Fenton system for metronidazole degradation. Chem. Eng. J. 2023, 464, 142584. [Google Scholar] [CrossRef]
- Sun, Y.; Pei, X.; Wang, B.; Hau Ng, Y.; Zhu, R.; Zhang, Q.; Deng, J.; Liu, Y.; Jing, L.; Dai, H. Point defect engineering of elemental phosphorus for photocatalytic hydrogen evolution. Chem. Eng. J. 2023, 463, 142488. [Google Scholar] [CrossRef]
- He, H.; Zhai, W.; Liu, P.; Wang, J. Rational design of metal–organic framework based photocatalysts correlated with specific additives for driving gas–liquid-solid CO2 reduction. Mater. Today 2025, 83, 382–403. [Google Scholar] [CrossRef]
- Fabrizio, K.; Lazarou, K.A.; Payne, L.I.; Twight, L.P.; Golledge, S.; Hendon, C.H.; Brozek, C.K. Tunable Band Gaps in MUV-10(M): A Family of Photoredox-Active MOFs with Earth-Abundant Open Metal Sites. J. Am. Chem. Soc. 2021, 143, 12609–12621. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Li, Y.; Tan, Y.; Xu, B.; Cai, J.; Zhang, Y.; Wang, Q.; Wu, Q.; Yang, B.; Huang, J. Recent Advances in Metal-Organic Framework (MOF)-Based Photocatalysts: Design Strategies and Applications in Heavy Metal Control. Molecules 2023, 28, 6681. [Google Scholar] [CrossRef]
- Yuan, L.; Du, P.; Zhang, C.; Xi, Y.; Zou, Y.; Li, J.; Bi, Y.; Bao, T.; Liu, C.; Yu, C. Facet-dependent spatial separation of dual cocatalysts on MOF photocatalysts for H2O2 production coupling biomass oxidation with enhanced performance. Appl. Catal. B Environ. Energy 2025, 364, 124855. [Google Scholar] [CrossRef]
- Luo, Y.; Ni, C.; Zhao, J.; Cui, J.; Zhang, J.; Zhu, Q.; Fan, F.; Li, C. Intrafacet Charge Separation toward Efficient Overall Water Splitting on SrTiO3 Single Crystal Photocatalysts. J. Phys. Chem. Lett. 2025, 16, 2981–2986. [Google Scholar] [CrossRef]
- Mahto, B.; Ali, H.; Barhoi, A.; Hussain, S. Morphology engineering of inorganic nanocrystals with deep eutectic solvents (DESs): Current developments and future prospects. Coord. Chem. Rev. 2025, 527, 216406. [Google Scholar] [CrossRef]
- Wojnicki, M.; Luty-Błocho, M.; Kwolek, P.; Gajewska, M.; Socha, R.P.; Pędzich, Z.; Csapó, E.; Hessel, V. The influence of dielectric permittivity of water on the shape of PtNPs synthesized in high-pressure high-temperature microwave reactor. Sci. Rep. 2021, 11, 4851. [Google Scholar] [CrossRef]
- Liu, J.; Liang, C.; Zhu, X.; Lin, Y.; Zhang, H.; Wu, S. Understanding the Solvent Molecules Induced Spontaneous Growth of Uncapped Tellurium Nanoparticles. Sci. Rep. 2016, 6, 32631. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.Y.; Check, B.; Fang, X.; Blum, S.; Pham, H.T.B.; Tayman, K.; Park, J. Photocatalytic Hydrogen Peroxide Production through Functionalized Semiconductive Metal–Organic Frameworks. J. Am. Chem. Soc. 2024, 146, 11319–11327. [Google Scholar] [CrossRef]
- Hao, Y.-C.; Chen, L.-W.; Li, J.; Guo, Y.; Su, X.; Shu, M.; Zhang, Q.; Gao, W.-Y.; Li, S.; Yu, Z.-L.; et al. Metal-organic framework membranes with single-atomic centers for photocatalytic CO2 and O2 reduction. Nat. Commun. 2021, 12, 2682. [Google Scholar] [CrossRef]
- Du, P.; Yuan, L.; Bao, T.; Xi, Y.; Li, J.; Bi, Y.; Yin, L.; Wang, J.; Liu, C. Facet effect of metal-organic frameworks on supporting co-catalysts for photocatalytic hydrogen peroxide production. Chin. Chem. Lett. 2025, 36, 110472. [Google Scholar] [CrossRef]
- Lei, H.; Zhang, J.; Wu, Z.; Ye, Y.; Li, Z.; Zhao, Y.; Chen, L.; Liu, Y.; Hong, M. Crystal Facet-Dependent Photocatalytic Hydrogen Evolution from Ultra-Stable Cu-Zr/Hf Heterobimetallic Metal–Organic Frameworks. Angew. Chem. Int. Ed. 2025, 64, e202509572. [Google Scholar] [CrossRef]
- Qi, Y.; Zhang, J.; Kong, Y.; Zhao, Y.; Chen, S.; Li, D.; Liu, W.; Chen, Y.; Xie, T.; Cui, J.; et al. Unraveling of cocatalysts photodeposited selectively on facets of BiVO4 to boost solar water splitting. Nat. Commun. 2022, 13, 484. [Google Scholar] [CrossRef]
- Li, Q.; Zhao, C.; Jia, S.; Chen, Q.; Li, X.; She, M.; Liu, H.; Liu, P.; Wang, Y.; Li, J. Design and fabrication of CuI/CuII-MOF-incorporated hydrogel photocatalysts for synergy removal of Cr(VI) and congo red. Chin. Chem. Lett. 2025, 36, 109936. [Google Scholar] [CrossRef]
- Hemdan, M.; Ragab, A.H.; Gumaah, N.F.; Mubarak, M.F. Sodium alginate-encapsulated nano-iron oxide coupled with copper-based MOFs (Cu-BTC@Alg/Fe3O4): Versatile composites for eco-friendly and effective elimination of Rhodamine B dye in wastewater purification. Int. J. Biol. Macromol. 2024, 274, 133498. [Google Scholar] [CrossRef] [PubMed]
- Edirisooriya, E.M.N.T.; Senanayake, P.S.; Xu, P.; Wang, H. Recyclability and regeneration of Au/TiO2 nanocomposite and Pt/TiO2 atom-nano composite catalysts in photo-reforming plastics for hydrogen production. J. Environ. Chem. Eng. 2025, 13, 116467. [Google Scholar] [CrossRef]
- Bockenstedt, J.; Vidwans, N.A.; Gentry, T.; Vaddiraju, S. Catalyst Recovery, Regeneration and Reuse during Large-Scale Disinfection of Water Using Photocatalysis. Water 2021, 13, 2623. [Google Scholar] [CrossRef]
- Yuan, G.; Tan, L.; Wang, P.; Wang, Y.; Wang, C.; Yan, H.; Wang, Y.-Y. MOF-COF Composite Photocatalysts: Design, Synthesis, and Mechanism. Cryst. Growth Des. 2022, 22, 893–908. [Google Scholar] [CrossRef]
- Li, X.; Wang, Q.; Sun, Y.; Sun, S.; Ge, L. Photogenerated charge carriers’ regulation strategies: Structure design, mechanism, and characterization technology. Int. J. Hydrogen Energy 2024, 69, 1341–1365. [Google Scholar] [CrossRef]
- Qi, S.; Wu, S.; Zhang, K.; Guan, L.; Hu, X.; Li, H. Design, preparation and mechanism of Bi2MoO6-modified cobalt-doped CdS solid solution photocatalysts. J. Alloys Compd. 2024, 1003, 175565. [Google Scholar] [CrossRef]
- Raza, Q.; Iqbal, S.; Bibi, F.; Aldosari, E.; Kalsoom, A.; Jamshaid, M.; Iqbal, R.; Lim, S. A novel g-C3N4 based CeO2 loaded ternary CeO2/CoFe2O4@g-C3N4 hetero-junction catalyst for the efficient removal of enrofloxacin from the veterinary wastewater. J. Sol-Gel Sci. Technol. 2025, 114, 532–548. [Google Scholar] [CrossRef]
- Yuan, L.; Du, P.; Yin, L.; Yao, J.; Wang, J.; Liu, C. Metal–organic framework-based S-scheme heterojunction photocatalysts. Nanoscale 2024, 16, 5487–5503. [Google Scholar] [CrossRef]
- Tian, H.; Li, Y.; Dai, Q.; Wei, S.; Qiu, Z.; Wang, W. P-N heterojunction to boost photoelectrochemical H2 evolution of CdS/AgVO3 nanorod arrays photoanode. J. Power Sources 2025, 632, 236341. [Google Scholar] [CrossRef]
- Qiao, S.; Di, M.; Jiang, J.-X.; Han, B.-H. Conjugated porous polymers for photocatalysis: The road from catalytic mechanism, molecular structure to advanced applications. EnergyChem 2022, 4, 100094. [Google Scholar] [CrossRef]
- Wang, Y.; Du, K.; Xu, R.; Cui, D.; Shi, Y.; Hao, W.; Du, Y. Bismuth-based semiconductor heterostructures for photocatalytic pollution gases removal. Curr. Opin. Green Sustain. Chem. 2023, 41, 100824. [Google Scholar] [CrossRef]
- Fang, W.; Wang, L. S-Scheme Heterojunction Photocatalyst for Photocatalytic H2O2 Production: A Review. Catalysts 2023, 13, 1325. [Google Scholar] [CrossRef]
- Zhi, Y.; Tian, J.; Sun, J.; Guo, H.; Sun, L.; Ma, P.; Li, M. Recent advances on Bi2XO6-based (X = W, Mo) S-scheme heterojunction photocatalysts: Design, synthesis, and photocatalytic performance of the systems for the degradation of organic pollutants. J. Alloys Compd. 2023, 965, 171377. [Google Scholar] [CrossRef]
- Yan, C.; Xu, M.; Li, J.; Chang, B.; Chen, Q.; Cao, W.; Xiao, W.; Wang, H.; Huo, P. Rational Construction of CuO/CdS for Highly Selective CO2 to CO Conversion with S-Scheme Photocatalysts. Energy Technol. 2025, 13, 2401137. [Google Scholar] [CrossRef]
- Ahmad, I.; Alfaifi, M.Q.; Alassmy, Y.A.; Abduljawad, M.M.; Khasawneh, M.A.; Ali, M.D.; Karim, M.R.; Ashraf, I.M. Design of S-scheme CdS/La2O3 heterojunction for excellent photocatalytic H2 evolution performance under simulated sunlight irradiation. Int. J. Hydrogen Energy 2025, 98, 1052–1066. [Google Scholar] [CrossRef]
- Li, D.; Li, R.; Zhou, D.; Qin, X.; Yan, W. The formation of S-scheme anatase/rutile TiO2 heterojunction and the enhancement of solar-to-hydrogen efficiency driven by dual electric field synergy. Fuel 2025, 388, 134237. [Google Scholar] [CrossRef]
- Liu, J.; Wan, Y.; Xie, Y.; Dong, F.; Yang, K.; Li, X. Boosting photocatalytic overall water splitting by β-NiS/TiO2-x Ohmic junction. Appl. Catal. B Environ. Energy 2025, 379, 125690. [Google Scholar] [CrossRef]
- Huang, H.; Cai, Y.; Xu, Q.; Xiong, M.; Ding, L.; Wang, X.; Jiang, Q.; Li, Q.; Han, X.; Hu, J.; et al. S-Scheme CdS/Co3S4 Double-Shelled Hollow Nanoboxes for Enhanced Photocatalytic Hydrogen Evolution. Small 2025, 21, 2501710. [Google Scholar] [CrossRef]
- Lu, Y.; Wan, Y.; Liu, J.; Hu, B.; Xie, Y.; Li, X. Robust photocatalytic hydrogen evolution performance of 0D/1D NiO/CdS S-scheme heterojunction. Sep. Purif. Technol. 2025, 358, 130126. [Google Scholar] [CrossRef]
- Li, L.; Wu, C.-q.; Zhang, Z.-x.; Zhang, N.; Huang, T.; Wang, Y. Rational Design of CeO2/CdSe Heterojunction Photocatalysts toward Enhanced Charge Separation for Efficient Photoreduction of Cr(VI) and Photodegradation of Tetracycline Hydrochloride. Langmuir 2025, 41, 18184–18199. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.-E.; Mameda, N.; Reddy, K.P.; Abraham, B.M.; Jo, W.-K.; Tonda, S. Bifunctional S-scheme hybrid heterojunction comprising CdS nanorods and BiOIO3 nanosheets for efficient solar-induced antibiotic degradation and highly-selective CO2 reduction. J. Mater. Sci. Technol. 2023, 161, 74–87. [Google Scholar] [CrossRef]
- Zhao, F.; Ahmad, I.; AlFaify, S.; Alanezi, K.M.; Bayahia, H.; Aladsani, E.A.; Alshahrani, T. Synthesis of CdS/N-rGO S-scheme heterojunction photocatalyst for efficient H2 evolution. Mater. Sci. Semicond. Process. 2024, 182, 108716. [Google Scholar] [CrossRef]
- Ahmad, N.; Khan, A.R.; Siddiqui, M.R.; Khan, M.D.; Benamor, A.; Anwer, A.H.; Ahmad, N. Advanced porphyrin-integrated Cu-MOF/semiconductor hybrids for high-efficiency removal of micropollutant via photocatalytic and adsorptive pathways. J. Water Process Eng. 2025, 77, 108447. [Google Scholar] [CrossRef]
- Xi, Y.; Xiang, Y.; Bao, T.; Li, Z.; Zhang, C.; Yuan, L.; Li, J.; Bi, Y.; Yu, C.; Liu, C. Nanoarchitectonics of S-Scheme Heterojunction Photocatalysts: A Nanohouse Design Improves Photocatalytic Nitrate Reduction to Ammonia Performance. Angew. Chem. Int. Ed. 2024, 63, e202409163. [Google Scholar] [CrossRef]
- Kozyr, E.G.; Njoroge, P.N.; Chapek, S.V.; Shapovalov, V.V.; Skorynina, A.A.; Pnevskaya, A.Y.; Bulgakov, A.N.; Soldatov, A.V.; Pellegrino, F.; Groppo, E.; et al. Operando Laboratory X-ray Absorption Spectroscopy and UV–Vis Study of Pt/TiO2 Photocatalysts during Photodeposition and Hydrogen Evolution Reactions. Catalysts 2023, 13, 414. [Google Scholar] [CrossRef]
- Sakata, K.; Amemiya, K. Real-time and operando observation of intermediates on TiO2 photoelectrocatalysis by soft X-ray absorption spectroscopy. Electrochem. Commun. 2024, 165, 107771. [Google Scholar] [CrossRef]
- Abudukade, M.; Pinna, M.; Spanu, D.; De Amicis, G.; Minguzzi, A.; Vertova, A.; Recchia, S.; Ghigna, P.; Mul, G.; Altomare, M. In Situ X-ray Absorption Spectroscopy Study of the Deactivation Mechanism of a Ni-SrTiO3 Photocatalyst Slurry Active in Water Splitting. J. Phys. Chem. C 2024, 128, 16020–16031. [Google Scholar] [CrossRef] [PubMed]
- Mei, B.; Shen, D.; Wei, Y.; Ma, J.; Sun, F. In situ photothermal catalytic cell for X-ray absorption fine structure spectroscopy measurement. Mater. Today Catal. 2024, 7, 100071. [Google Scholar] [CrossRef]
- Vicente, R.A.; Neckel, I.T.; Sankaranarayanan, S.K.R.S.; Solla-Gullon, J.; Fernández, P.S. Bragg Coherent Diffraction Imaging for In Situ Studies in Electrocatalysis. ACS Nano 2021, 15, 6129–6146. [Google Scholar] [CrossRef]
- Park, S.H.; Kim, S.; Park, J.W.; Kim, S.; Cha, W.; Lee, J. In-situ and wavelength-dependent photocatalytic strain evolution of a single Au nanoparticle on a TiO2 film. Nat. Commun. 2024, 15, 5416. [Google Scholar] [CrossRef]
- Anderson, J.; Nazirkar, N.P.; Ndiaye, A.; Barringer, J.; Tran, V.; Bassène, P.; Cha, W.; Jiang, J.; Shi, J.; Harder, R.; et al. Real-Time Tracking of Nanoscale Morphology and Strain Evolution in Bi2WO6 via Operando Coherent X-Ray Imaging. Adv. Mater. 2025, 37, 2504445. [Google Scholar] [CrossRef]
- Pech-Rodríguez, W.J.; González-Quijano, D.; Vargas-Gutiérrez, G.; Morais, C.; Napporn, T.W.; Rodríguez-Varela, F.J. Electrochemical and in situ FTIR study of the ethanol oxidation reaction on PtMo/C nanomaterials in alkaline media. Appl. Catal. B Environ. 2017, 203, 654–662. [Google Scholar] [CrossRef]
- Elsheikh, A.; Torrero, J.; Rojas, S.; McGregor, J. In-situ FTIR spectroscopy investigation of carbon-supported PdAuNi electrocatalysts for ethanol oxidation. J. Electroanal. Chem. 2023, 928, 116985. [Google Scholar] [CrossRef]
- Palharim, P.H.; Wang, X.; Varesio, E.; Bürgi, T.; Teixeira, A.C.S.C. In Situ ATR-IR Spectroscopy for the Degradation of Acetaminophen on WO3–AgCl Photocatalysts. J. Phys. Chem. C 2024, 128, 20042–20052. [Google Scholar] [CrossRef]
- Illana, A.; Doña-Rodríguez, J.M.; Robles, A.; González-Díaz, Ó.M.; Pulido-Melián, E.; Azofra, L.M.; Araña, J. In-situ FT-IR study of alcohols degradation in the gas phase using different TiO2 composites. Catal. Today 2024, 432, 114603. [Google Scholar] [CrossRef]
- Yin, Z.; Liu, B.; Fan, S.; Wang, P.; Wang, X.; Long, D.; Zhang, L.; Yang, X.; Li, X. In situ FTIR spectra investigation of the photocatalytic degradation of gaseous toluene over a novel hedgehog-like CaFe2O4 hollow-structured materials. Catal. Commun. 2019, 130, 105754. [Google Scholar] [CrossRef]
- Ali, F.M.; Gouda, A.; Duchesne, P.N.; Hmadeh, M.; O’Brien, P.G.; Mohan, A.; Ghoussoub, M.; Tountas, A.A.; Ibrahim, H.; Perovic, D.D.; et al. In situ probes into the structural changes and active state evolution of a highly selective iron-based CO2 reduction photocatalyst. Chem Catal. 2024, 4, 100983. [Google Scholar] [CrossRef]
- Berdini, F.; Heffner, H.; Marchetti, J.M.; López-Corral, I.; Brigante, M. Theoretical-experimental design of TiO2 photocatalysts for removal of emerging pollutants from water: The effect of Ga doping on photodegradation of methyl orange. J. Water Process Eng. 2024, 68, 106426. [Google Scholar] [CrossRef]
- Alhasani, Z.A.A.; Ajeel, F.N. DFT investigations of structural and electronic properties of two-dimensional Y2C and Y2CF2 monolayers. Nano Trends 2025, 9, 100073. [Google Scholar] [CrossRef]
- Er, C.-C.; Putri, L.K.; Ang, Y.S.; Chai, S.-P. Unveiling fundamental first-principles insights into single-atom transition metal photocatalysts for carbon dioxide reduction. Fuel 2025, 382, 133746. [Google Scholar] [CrossRef]
- Ribeiro, R.A.P.; Oliveira, M.C.; Bomio, M.R.D.; de Lazaro, S.R.; Andrés, J.; Longo, E. Connecting the surface structure, morphology and photocatalytic activity of Ag2O: An in depth and unified theoretical investigation. Appl. Surf. Sci. 2020, 509, 145321. [Google Scholar] [CrossRef]
- Gouveia, A.F.; Lemos, S.C.S.; Leite, E.R.; Longo, E.; Andrés, J. Back to the Basics: Probing the Role of Surfaces in the Experimentally Observed Morphological Evolution of ZnO. Nanomaterials 2023, 13, 978. [Google Scholar] [CrossRef]
- Huang, B.; Liu, Y.; Zhang, Y.; Zhang, F.; Yang, Y.; Li, J. Effect of Vacancy Defects on the Electronic Structure and Optical Properties of Bi4O5Br2: First-Principles Calculations. Coatings 2024, 14, 1361. [Google Scholar] [CrossRef]
- Katai, M.; Edalati, P.; Hidalgo-Jimenez, J.; Shundo, Y.; Akbay, T.; Ishihara, T.; Arita, M.; Fuji, M.; Edalati, K. Black brookite rich in oxygen vacancies as an active photocatalyst for CO2 conversion: Experiments and first-principles calculations. J. Photochem. Photobiol. A Chem. 2024, 449, 115409. [Google Scholar] [CrossRef]
- Quirk, J.A.; Lazarov, V.K.; McKenna, K.P. First-Principles Modeling of Oxygen-Deficient Anatase TiO2 Nanoparticles. J. Phys. Chem. C 2020, 124, 23637–23647. [Google Scholar] [CrossRef]
- Saroar, S.; Sultana, S.; Nishat, S.S.; Hossain, Q.S.; Khan, M.N.I.; Islam, D.; Akhtar, U.S.; Shahriar Bashar, M.; Jahan, S.; Hossain, K.S.; et al. First-Principles Calculations on Electronic, Optical, and Phonon Properties of γ-Bi2MoO6. ACS Omega 2024, 9, 36314–36325. [Google Scholar] [CrossRef]
- Hussain, S.; Rehman, J.U.; Tahir, M.B.; Hussain, A. First-principles study of structural, mechanical, optical, and electronic properties of double perovskite RbBa2Ti3O10 material for photocatalytic applications. Int. J. Hydrogen Energy 2024, 78, 1123–1132. [Google Scholar] [CrossRef]
- Esbergenova, A.; Yusupov, M.; Ghasemitarei, M.; Jalolov, R.; Hojamberdiev, M.; Shaislamov, U. Understanding the effect of morphological change on photocatalytic activity of ZnO nanostructures and reaction mechanism by molecular dynamics. Colloids Surf. A Physicochem. Eng. Asp. 2023, 677, 132386. [Google Scholar] [CrossRef]
- Haounati, R.; Ighnih, H.; Malekshah, R.E.; Alahiane, S.; Alakhras, F.; Alabbad, E.; Alghamdi, H.; Ouachtak, H.; Addi, A.A.; Jada, A. Exploring ZnO/Montmorillonite photocatalysts for the removal of hazardous RhB Dye: A combined study using molecular dynamics simulations and experiments. Mater. Today Commun. 2023, 35, 105915. [Google Scholar] [CrossRef]
- Schossler, R.T.; Ojo, S.; Jiang, Z.; Hu, J.; Yu, X. A novel interpretable machine learning model approach for the prediction of TiO2 photocatalytic degradation of air contaminants. Sci. Rep. 2024, 14, 13070. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wang, J.; He, T.; Li, Z.; Xu, H.; Fan, Z.; Liao, F.; Liu, Y.; Kang, Z. Dynamic Machine Learning-Driven Optimization of Microwave-Synthesized Photocatalysts for Enhanced Hydrogen Peroxide Production. ChemCatChem 2025, 17, e00341. [Google Scholar] [CrossRef]
- Wu, M.; Song, Z.; Cui, Y.; Fu, Z.; Hong, K.; Li, Q.; Lyu, Z.; Liu, W.; Wang, J. Machine Learning-Assisted Design of Nitrogen-Rich Covalent Triazine Frameworks Photocatalysts. Adv. Funct. Mater. 2025, 35, 2413453. [Google Scholar] [CrossRef]
- Mai, H.; Li, X.; Le, T.C.; Russo, S.P.; Winkler, D.A.; Chen, D.; Caruso, R.A. Rapid Design of Efficient Mn3O4-Based Photocatalysts by Machine Learning and Density Functional Theory Calculations. Adv. Energy Sustain. Res. 2025, 6, 2400397. [Google Scholar] [CrossRef]
- Zhai, X.; Chen, M. A machine learning-based nano-photocatalyst module for accelerating the design of Bi2WO6/MIL-53(Al) nanocomposites with enhanced photocatalytic activity. Nanoscale Adv. 2023, 5, 4065–4073. [Google Scholar] [CrossRef]
- Kumar, R.; Singh, A.K. Chemical hardness-driven interpretable machine learning approach for rapid search of photocatalysts. npj Comput. Mater. 2021, 7, 197. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, X. Machine Learning Band Gaps of Doped-TiO2 Photocatalysts from Structural and Morphological Parameters. ACS Omega 2020, 5, 15344–15352. [Google Scholar] [CrossRef] [PubMed]
- Pellegrino, F.; Isopescu, R.; Pellutiè, L.; Sordello, F.; Rossi, A.M.; Ortel, E.; Martra, G.; Hodoroaba, V.-D.; Maurino, V. Machine learning approach for elucidating and predicting the role of synthesis parameters on the shape and size of TiO2 nanoparticles. Sci. Rep. 2020, 10, 18910. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Yang, Y.; Chen, X. A First-Principles and Machine Learning Study on Design of Graphitic Carbon Nitride-Based Single-Atom Photocatalysts. ACS Appl. Nano Mater. 2024, 7, 11862–11870. [Google Scholar] [CrossRef]
- Oh, S.-H.V.; Yoo, S.-H.; Jang, W. Small dataset machine-learning approach for efficient design space exploration: Engineering ZnTe-based high-entropy alloys for water splitting. npj Comput. Mater. 2024, 10, 166. [Google Scholar] [CrossRef]

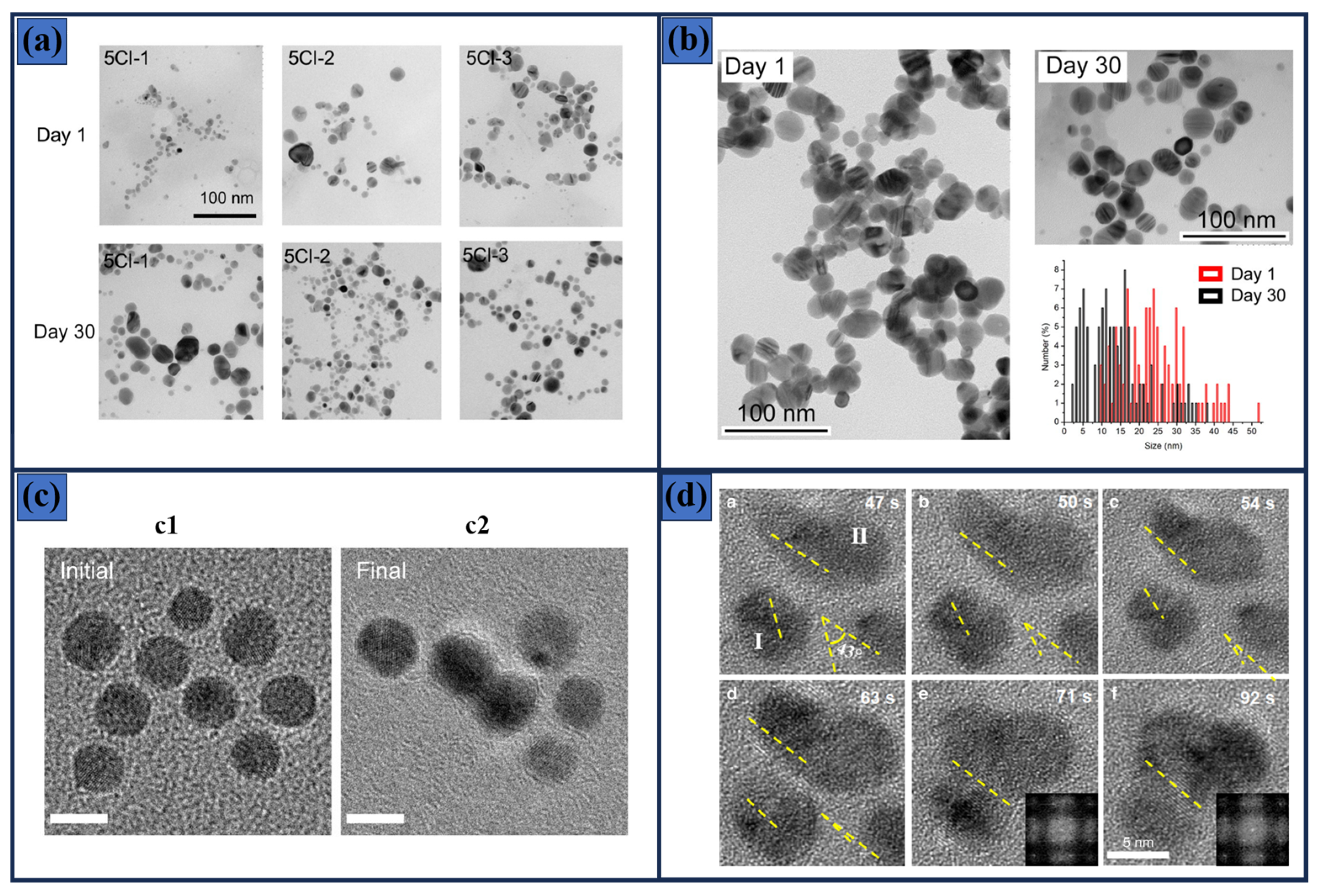






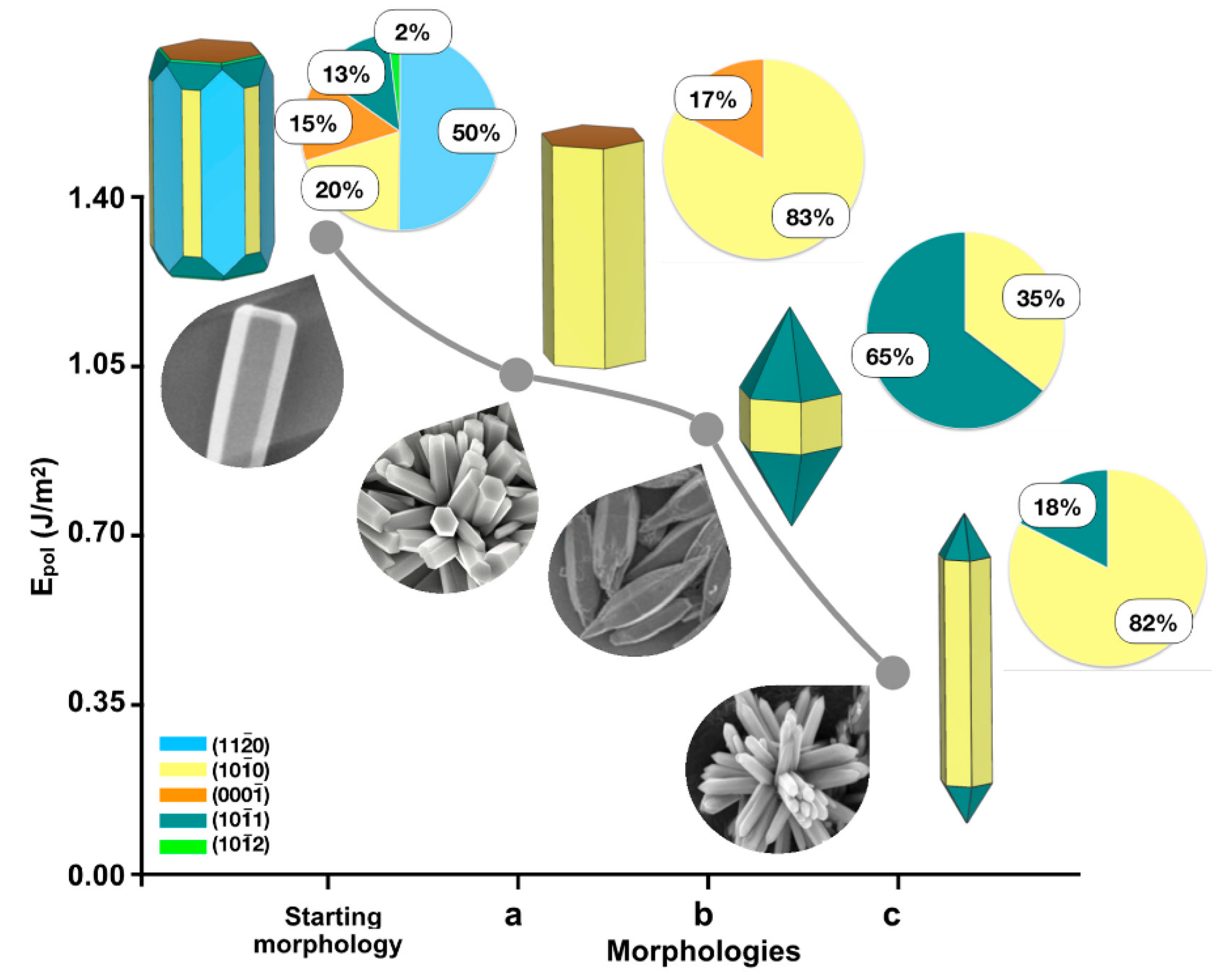


| Material | Synthesis Process | Conditions | Solvent and Caping Agent | Morphology | Ref. |
|---|---|---|---|---|---|
| Ce1-(3/4)xEuxO2 | Microwave-assisted hydrothermal | 8 min irradiation to reach 100, 140, 180, or 200 °C. | DI water, 2, 0.06, or 6 mol L−1, NaOH solution. | Nanorods and nanocubes. | [54] |
| TiO2 | Sol-gel | Stirring for 24 h at 25 °C and 60 °C. Calcined at 450 °C for 4 h, 1 °C min−1. | DI water and ethanol. SDS, CTAB, and PEG. | Spherical, semi-spherical and worm-like shapes. | [63] |
| Bi2WO6 | Hydrothermal | Hydrothermal for 15 h at 180 °C. | DI water and 20 mL of HNO3, NaOH solution. | Nanoplates. | [55] |
| WO3 | Solvothermal | Heated at 120 °C for 12 h. | Acetone: DI water, 2:5 v/v, oxalate or citric. | Nanospheres and nanocages. | [57] |
| KBi6O9I/Ag-AgVO3 | Combustion synthesis/sonochemical | At 80 °C, the gel was obtained, heated at 300 °C, sonicated for 45 min, and treated at 180 °C for 24 h. | DI water | Irregular polygon-like surface structure. | [64] |
| Co(II)-Pyridine-Decorated | Hydrothermal | Reagent dissolved in H2O and pH adjusted by H3PO4 and NaOH, heated in a reactor at 160 °C for 4 days. | DI water | Unevenly shaped blocks. | [65] |
| Bi2O3-Bi2SiO5 | Sol-gel | Reagents were mixed with the solvents, and then the gel was calcinated at 400 °C for 2 h or 450 °C for 4 h. | Ethylene glycol, tetraethoxysilane. | Nanoparticles | [66] |
| BiOBr | Solvothermal and one-pot method | The Bi-reagent was dissolved in ethylene glycol, and NaBr was added under stirring and heated at 170 °C for 6 h. | Ethylene glycol | Flower-like structure. | [67] |
| Fe@Co-CP-2 | Hydrothermal method and ion-exchange | Reagents were mixed with NaOH solution and heated at 130 °C for 4 days. Ion-exchange was achieved by mixing the obtained samples in a methanol solution. | H2O, methanol solution. | Irregular polyhedral. | [68] |
| NiS2/CdS | Hydrothermal synthesis + Ultrasonic-assisted composite synthesis | H2O2-CdS was dissolved in H2O2, DI water, and put in an autoclave at 180 °C for 10 h. For NiS2 the chemicals were dissolved in DI water and reacted at 80 °C for 12 h. | DI water, H2O2, and ethanol. | Cone shape to mesoporous particles. | [69] |
| TiO2/MoSx/Ag | Photodeposition | TiO2/a-MoSx: TiO2 nanoparticles in DI water, then (NH4)2MoS4 in ethanol, was added and exposed to a Xe lamp. Ag nanoparticles: AgNO3 in ethanol was added and exposed to the Xe lamp. | DI water, ethanol | Nanoparticles. | [70] |
| La-doping ZnO | Co-precipitation | Reagents dissolved in DI water, NaOH solution was added until the pH was 12. Then calcinated for 2 h at 500 °C. | DI H2O | Disrupted flower-like morphology. | [71] |
| Heterojunction | Band Alignment | Charge Transfer Mechanism | Advantages | Disadvantages | Ref. |
|---|---|---|---|---|---|
| Type I heterojunction (straddling gap). | CB and VB of one material straddle between the bands of the other. | Both electrons and holes migrate into the narrower bandgap semiconductor. | Strong recombination process, useful for light emission. | Poor charge separation, not ideal for photocatalysis. | [147] |
| Type II heterojunction (staggered gap). | CB of one is lower, VB of the other is higher. | Electrons and holes are spatially separate in different materials. | Efficient charge separation process. | Reduced redox potential due to charge loss. | [143] |
| p-n heterojunction. | Fermi levels align at the interface, forming a depletion region. | Built-in electric field drives the separation of electrons and holes. | Simple design, strong internal field, scalable. | Recombination at the interface is possible. | [148,149] |
| Z-scheme heterojunction. | Resembles natural photosynthesis. | Only high-energy electrons and holes are retained. | Strong redox ability mimics a natural process. | Requires mediator (redox pair, solid bridge). | [150] |
| S-scheme heterojunction. | Band bending leads to selective recombination at the interface. | Unfavorable electrons and holes recombine, leaving the most energetic ones. | High redox ability, strong charge separation, no mediator needed. | More complex design, less explored than other materials. | [151] |
| Material | Algorithm/Dataset | Remarks | Considerations | Ref. |
|---|---|---|---|---|
| Covalent triazine frameworks | DimeNet++/14,920 CTFs structures. | R2 > 0.98 and MAE < 0.008 eV or 0.0014 eV/atom. | Thermodynamic stability, Perdew-Burke-Ernzerhof band gap, and band edge positions. | [191] |
| Al0.5Mn2.5O4/35 wt. % Ag3PO4 | RF, EXT, GBR, KRR, and SVR/30 materials. | For EXT R2 = 1.00 | Effective mass, band gap, and degradation efficiency. | [192] |
| Bi2WO6/MIL-53(Al) | SVR/53 samples | R = 0.823 for the degradation rate of RhB dye. | Mole ratio, temperature, concentration of HNO3, surfactant, and nine other variables. | [193] |
| TiO2 | GPR model/60 samples | RME: 0.0012%; MAE 0.0010%. | Surface area and lattice parameter. | [195] |
| TiO2 | Artificial Neural Network and Genetic algorithm/20 samples. | Not defined | Triethanolamine, titanatrane (TeoaH3), initial pH, and operating temperature. | [196] |
| ZnTe-based alloys | Sure, independence screening, sparsifying operator, and the agreement approach/13 ternary component. | RMES | The lattice constant, the equilibrium temperature of the compounds, and the band edge positions. | [198] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Pech-Rodríguez, W.J.; Şahin, N.E.; Suarez-Velázquez, G.G. Engineering Design Strategies for Boosting Photocatalytic Activity: Theory-to-Data-Driven Perspective. Materials 2026, 19, 1472. https://doi.org/10.3390/ma19071472
Pech-Rodríguez WJ, Şahin NE, Suarez-Velázquez GG. Engineering Design Strategies for Boosting Photocatalytic Activity: Theory-to-Data-Driven Perspective. Materials. 2026; 19(7):1472. https://doi.org/10.3390/ma19071472
Chicago/Turabian StylePech-Rodríguez, Wilian Jesús, Nihat Ege Şahin, and Gladis Guadalupe Suarez-Velázquez. 2026. "Engineering Design Strategies for Boosting Photocatalytic Activity: Theory-to-Data-Driven Perspective" Materials 19, no. 7: 1472. https://doi.org/10.3390/ma19071472
APA StylePech-Rodríguez, W. J., Şahin, N. E., & Suarez-Velázquez, G. G. (2026). Engineering Design Strategies for Boosting Photocatalytic Activity: Theory-to-Data-Driven Perspective. Materials, 19(7), 1472. https://doi.org/10.3390/ma19071472








