Transparent Conductive Copper-Doped Zinc Oxide (ZnO:Cu) Thin Films: PVco-D Fabrication and Applications in Perovskite Solar Cells
Highlights
- The vapor phase co-deposition technique was successfully implemented for ZnO:Cu thin films.
- An annealing process was performed to modify the optical and electrical properties.
- AFM imaging and qualitative and quantitative XRF composition analysis were performed.
- ZnO:Cu layers exhibit high transparency, uniformity and ohmic conductivity over a wide temperature range.
- A complete structure for a thin-film perovskite solar cell with an efficiency of 17% was proposed.
Abstract
1. Introduction
2. Materials and Methods
2.1. Physical Vapor Deposition
2.2. Annealing
3. Results and Discussion
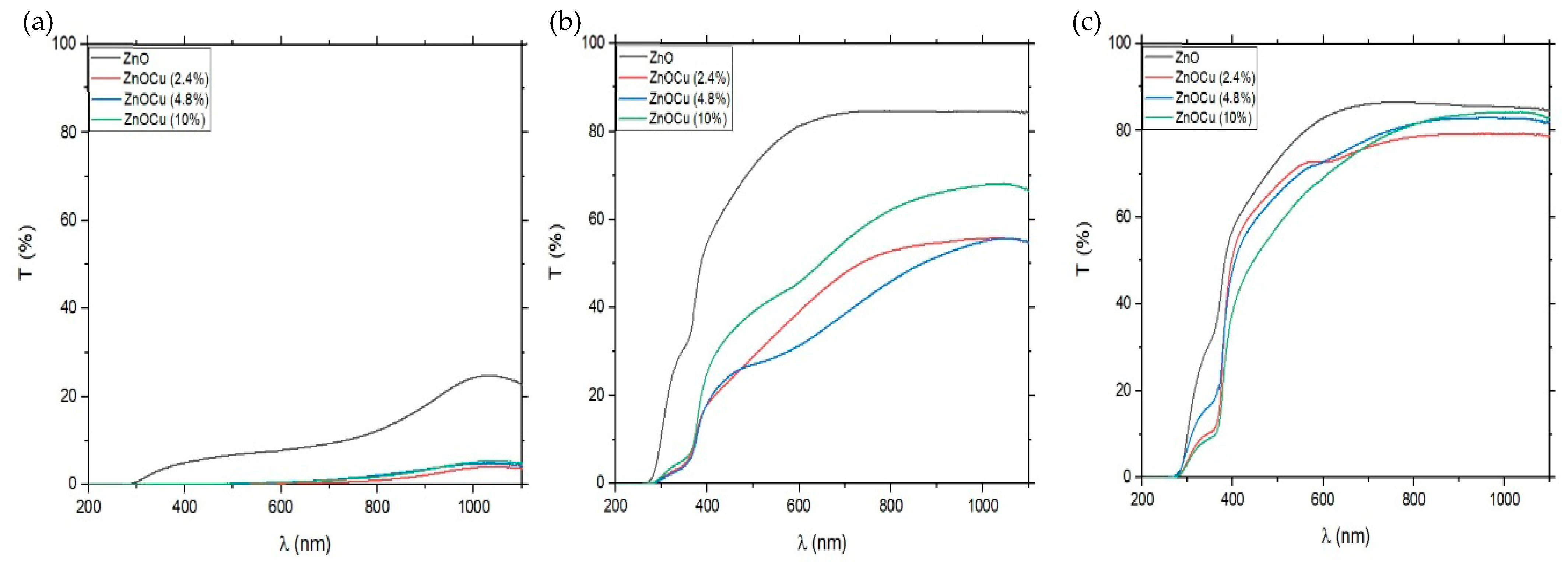
3.1. Transmission Measurements
3.2. Energy Bandgaps
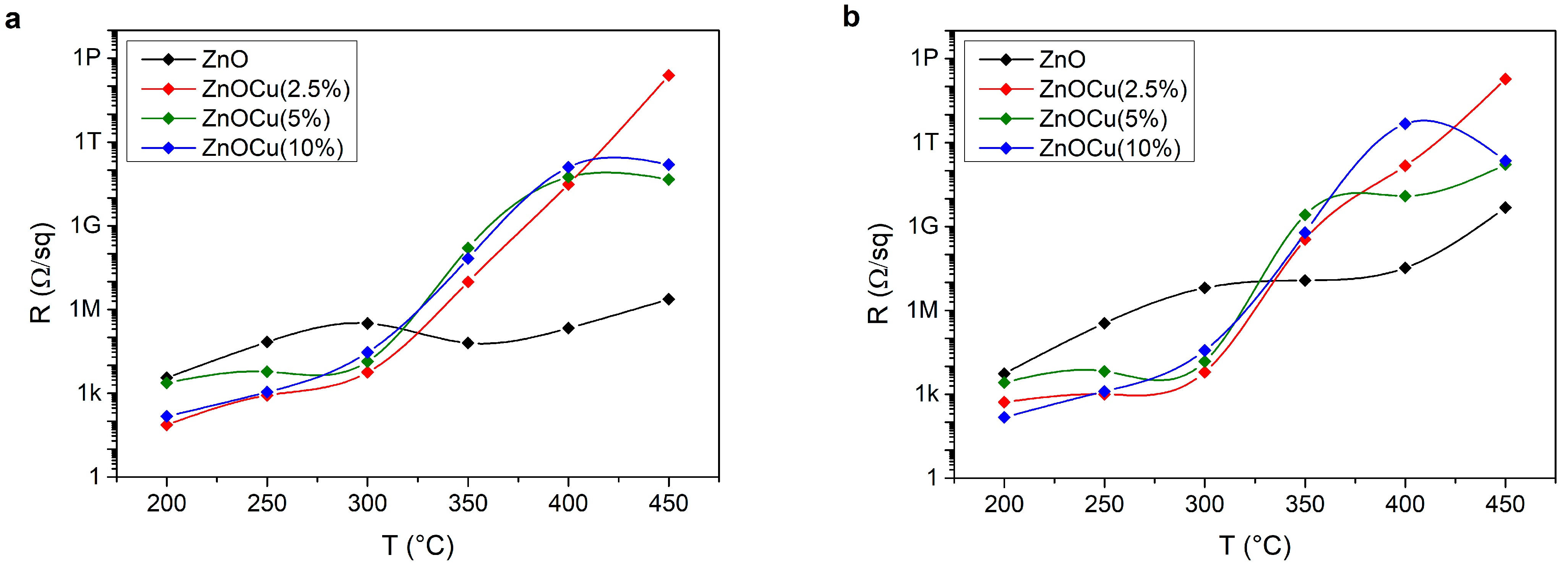
3.3. I(U)Characteristics and Sheet Resistance of ZnO:Cu
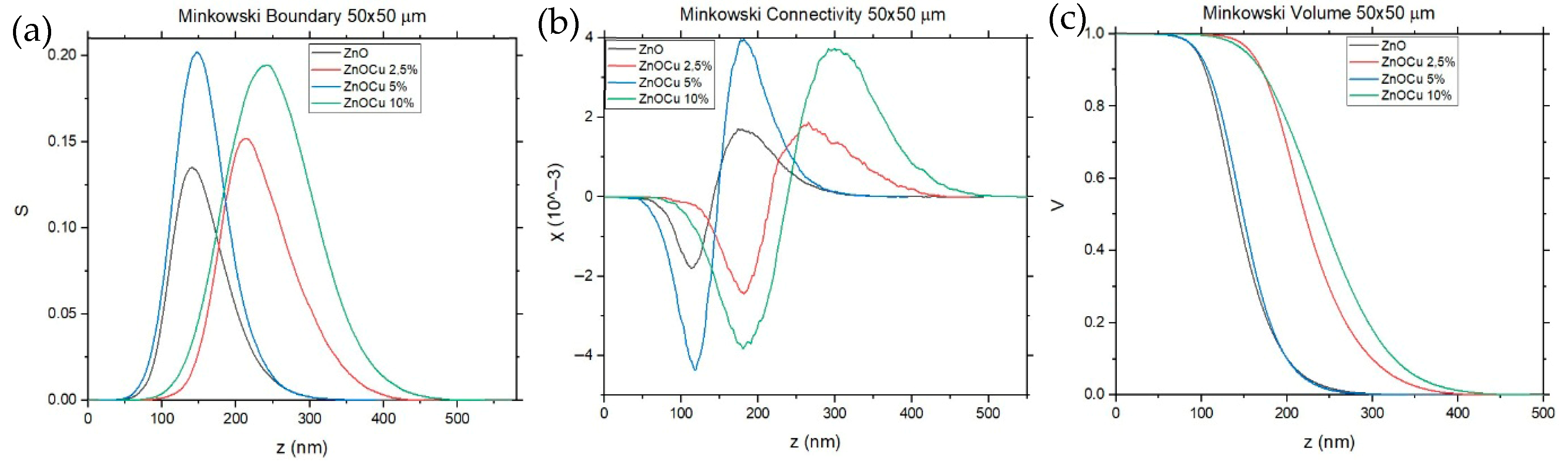
3.4. Atomic Force Microscopy
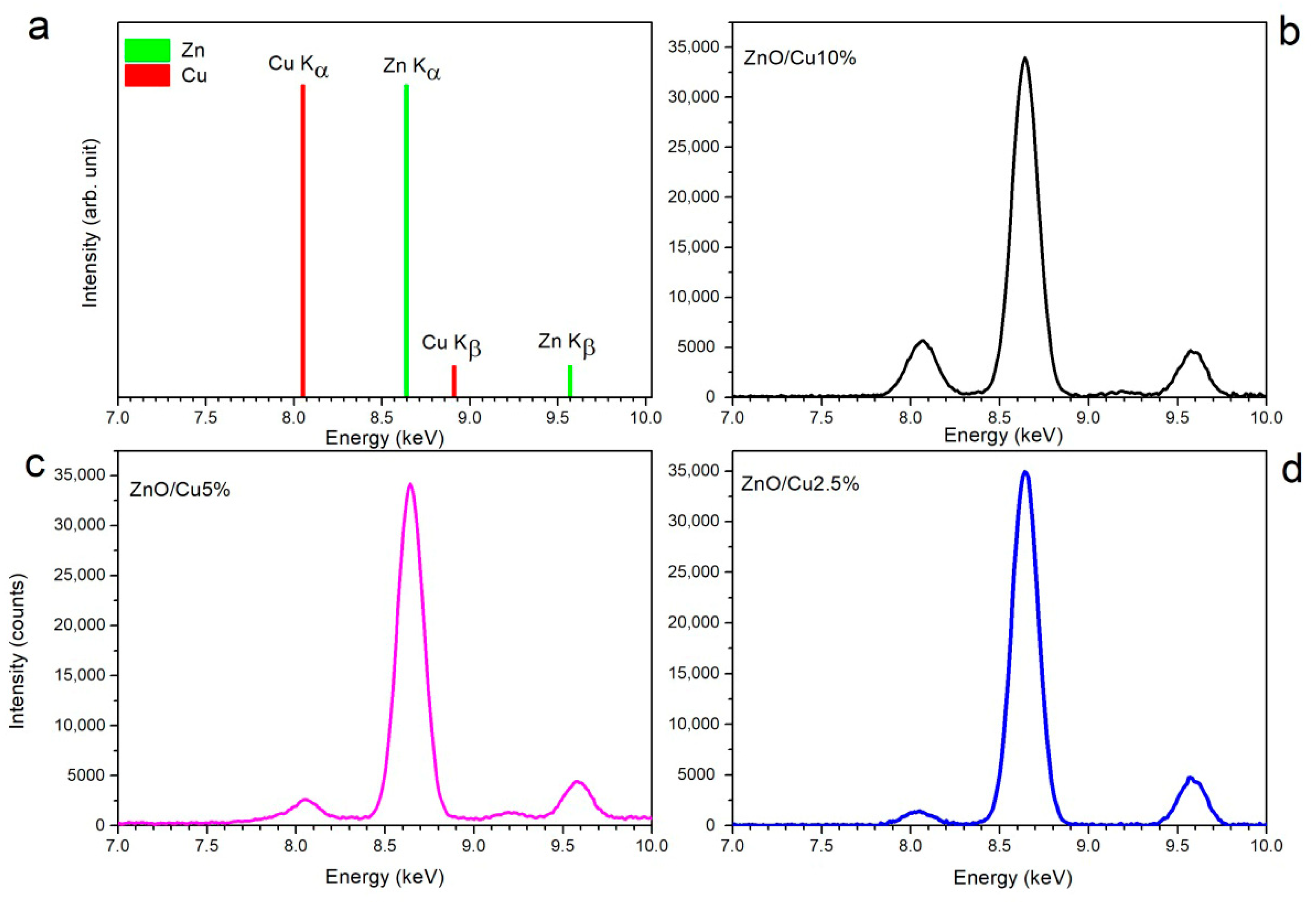
3.5. X-Ray Fluorescence
3.6. Thin-Film Perovskite Solar Cell Using ZnO:Cu as a Transparent Conductive Layer
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Intal, D.; Ebong, A.U. Thin-Film Solar Photovoltaics: Trends and Future Directions. Renew. Sustain. Energy Rev. 2025, 226, 116464. [Google Scholar] [CrossRef]
- Green, M.A.; Dunlop, E.D.; Yoshita, M.; Kopidakis, N.; Bothe, K.; Siefer, G.; Hao, X. Solar cell efficiency tables (Version 63). Prog. Photovolt. 2024, 32, 3–13. [Google Scholar] [CrossRef]
- Jelle, B.P.; Breivik, C.; Røkenes, H.D. Building integrated photovoltaic products: A state-of-the-art review and future research opportunities. Sol. Energy Mater. Sol. 2012, 100, 69–96. [Google Scholar] [CrossRef]
- Kidowaki, H.; Oku, T.; Akiyama, T. Fabrication and characterization of CuO/ZnOsolar cells. J. Phys. Conf. Ser. 2012, 352, 012022. [Google Scholar] [CrossRef]
- Fortunato, E.; Ginley, D.; Hosono, H.; Paine, D.C. Transparent Conducting Oxides for Photovoltaics. MRS Bull. 2007, 32, 242–247. [Google Scholar] [CrossRef]
- Minami, T. Transparent conducting oxide semiconductors for transparent electrodes. Semicond. Sci. Technol. 2005, 20, s35. [Google Scholar] [CrossRef]
- Ellmer, K. Past achievements and future challenges in the development of optically transparent electrodes. Nat. Photonics 2012, 6, 809–817. [Google Scholar] [CrossRef]
- Ginley, S.; Hosono, H.; Paine, D.C. Handbook of Transparent Conductors; Springer: Boston, MA, USA, 2011. [Google Scholar]
- Ozgür, Ü.; Alivov, Y.I.; Liu, C.; Teke, A.; Reshchikov, M.A.; Doğan, S.; Avrutin, V.; Cho, S.-J.; Morkoç, H. A comprehensive review of ZnO materials and devices. J. Appl. Phys. 2005, 98, 041301. [Google Scholar] [CrossRef]
- Boiko, B.; Yermakov, M.; Pshenychnyi, R.; Klymov, O.; Opanasyuk, A.; Dobrozhan, O.; Diachenko, O.; Muñoz-Sanjosé, V. Structural, optical, and electrical properties of copper-alloyed ZnO films deposited by the pulsed spray-pyrolysis with molecular solutions. Phys. B Condens. Matter 2025, 705, 417086. [Google Scholar] [CrossRef]
- Biswas, S.K.; Karim, M.A.; Rajib, A.; Rahman, M.A. Study the Optical and Structural Properties of Cu-doped ZnO Thin Films for Solar Energy Applications. In Proceedings of the 2019 International Conference on Computer, Communication, Chemical, Materials and Electronic Engineering (IC4ME2), Rajshahi, Bangladesh, 11–12 July 2019; pp. 1–4. [Google Scholar] [CrossRef]
- Zawadzka, A.; Marjanowska, A.; Laouid, A.; Wisniewski, K.; El Kouari, Y.; El Hani, Y.; Płóciennik, P. Low-temperature influence on the properties and efficiency of thin-film perovskite solar cells fabricated by the PVco-D technique. Sol. Energy Mater. Sol. Cells 2024, 274, 112993. [Google Scholar] [CrossRef]
- Płóciennik, P.; Guichaoua, D.; Korcala, A.; Zawadzka, A. Studies of aluminium oxide thin films deposited by laser ablation technique. Opt. Mater. 2016, 56, 49–57. [Google Scholar] [CrossRef]
- Laouid, A.; Belghiti, A.A.; Wisniewski, K.; Strzelecki, J.; Karakas, A.; Gozutok, A.; El Kouari, Y.; Bouich, A.; Tlemçani, M.; Plociennik, P.; et al. Optical and morphological properties of DCM thin films co-doped of Znq2 by PVD: Theoretical and experimental investigations. Vacuum 2024, 222, 112997. [Google Scholar] [CrossRef]
- Tauc, J.; Grigorovici, R.; Vancu, A. Optical properties and electronic structure of amorphous germanium. Phys. Status Solidi B 1966, 15, 627–637. [Google Scholar] [CrossRef]
- Yu, P.Y.; Cardona, M. Fundamentals of Semiconductors: Physics and Materials Properties, 4th ed.; Springer: Berlin/Heidelberg, Germany, 2010. [Google Scholar]
- Viezbicke, B.D.; Patel, S.; Davis, B.E.; Birnie, D.P., III. Evaluation of the Tauc method for optical absorption edge determination: ZnO thin films as a model system. Phys. Status Solidi B 2015, 252, 1700–1710. [Google Scholar] [CrossRef]
- Kumar, P.; Das, M.K. Temperature-dependent characterization of RF-Sputtered ZnO thin films for optoelectronic applications. Appl. Phys. A 2026, 132, 83. [Google Scholar] [CrossRef]
- Kumar, V.; Singh, S.K.; Sharma, H.; Kumar, S.; Banerjee, M.; Vij, A. Investigation of structural and optical properties of ZnO thin films deposited by pulsed laser deposition. Phys. B Condens. Matter 2019, 552, 221–226. [Google Scholar] [CrossRef]
- Gboglo, A.D.; Baneto, M.; Ako, O.; Gadedjisso-Tossou, K.S.; Grandidier, B.; Haris, M.; Senthilkumar, M.; N’konou, K. Structural, morphological and optical properties of ZnO thin films grown by time-dependent chemical bath deposition. Int. J. Renew. Energy Dev. 2026, 15, 66–75. [Google Scholar] [CrossRef]
- Chen, G.-J.; Jian, S.-R.; Juang, J.-Y. Surface Analysis and Optical Properties of Cu-Doped ZnO Thin Films Deposited by Radio Frequency Magnetron Sputtering. Coatings 2018, 8, 266. [Google Scholar] [CrossRef]
- Aadnan, I.; Zegaoui, O.; El Mragui, A.; Moussout, H.; da Silva, J.C.E. Structural, optical and photocatalytic properties under UV-A and visible lights of Co–, Ni- and Cu-doped ZnO nanomaterials. Comparative study. Arab. J. Chem. 2023, 17, 105336. [Google Scholar] [CrossRef]
- Saoud, T.; Benramache, S.; Diha, A. The Effect of Co and Cu Co-Doping ZnO Thin Films on Structural and Optical Properties. Chem. Didact. Ecol. Metrol. 2024, 28, 171–178. [Google Scholar] [CrossRef]
- Keithley Instruments, Inc. Keithley Model 4200-SCS Semiconductor Characterization System User Manual; Keithley Instruments: Cleveland, OH, USA, 2015. [Google Scholar]
- Janis Research Company. Janis Cryogenics Model 22 User Manual; Janis Cryogenics: Wilmington, MA, USA, 2012. [Google Scholar]
- Lake Shore Cryotronics, Inc. Lake Shore 335 Temperature Controller User Manual; Lake Shore Cryotronics: Westerville, OH, USA, 2016. [Google Scholar]
- Cisneros-Contreras, I.R.; Muñoz-Rosas, A.L.; Rodríguez-Gómez, A. Resolution improvement in Haacke’s figure of merit for transparent conductive films. Results Phys. 2019, 15, 102695. [Google Scholar] [CrossRef]
- McCluskey, M.D.; Jokela, S.J. Defects in ZnO. J. Appl. Phys. 2009, 106, 071101. [Google Scholar] [CrossRef]
- Nunes, P.; Fortunato, E.; Tonello, P.; Fernandes, F.B.; Vilarinho, P.; Martins, R. Effect of different dopant elements on the properties of ZnO thin films. Vacuum 2002, 64, 281–285. [Google Scholar] [CrossRef]
- Marjanowska, A.; El Karout, H.; Guichaoua, D.; Sahraoui, B.; Płóciennik, P.; Zawadzka, A. Topography and Nonlinear Optical Properties of Thin Films Containing Iodide-Based Hybrid Perovskites. Nanomaterials 2024, 14, 50. [Google Scholar] [CrossRef] [PubMed]
- Ţălu, Ş.; Bramowicz, M.; Kulesza, S.; Solaymani, S.; Ghaderi, A.; Dejam, L.; Elahi, S.M.; Boochani, A. Microstructure and micromorphology of ZnO thin films: Case study on Al doping and annealing effects. Superlattices Microstruct. 2016, 93, 109–121. [Google Scholar] [CrossRef]
- Harth, A. X-ray fluorescence (XRF) on painted heritage objects: A review using topic modelling. Herit. Sci. 2024, 12, 17. [Google Scholar] [CrossRef]
- Sung, N.-E.; Kang, S.-W.; Shin, H.-J.; Lee, H.-K.; Lee, I.-J. Cu doping effects on the electronic and optical properties of Cu-doped ZnO thin films fabricated by radio frequency sputtering. Thin Solid Films 2013, 547, 285–288. [Google Scholar] [CrossRef]
- Software: ESPRIT Family. Available online: https://i-mag.com.tr/tr/urun/eds-wds-ebsd-micro-edxrf/software-esprit-family (accessed on 17 February 2026).
- Burgelman, M.; Decock, K.; Niemegeers, A.; Verschraegen, J.; Degrave, S. University of Gent Department of Electronics and Information Systems (ELIS). Available online: https://scaps.elis.ugent.be/SCAPS%20manual%20most%20recent.pdf (accessed on 15 February 2026).
- Plóciennik, P.; Zawadzka, A.; Idrissi, I.E.; Sofiani, Z.; Wiśniewski, K.; Laouid, A.; Marjanowska, A.; Łukasiak, Z. ZnO-based thin-film transparent contacts for applications in perovskite solar cells. In Proceedings of the2024 24th International Conference on Transparent Optical Networks (ICTON), Bari, Italy, 14–18 July 2024; pp. 1–4. [Google Scholar] [CrossRef]
- Laouid, A.; Belghiti, A.A.; Wisniewski, K.; Abouais, A.; Tlemçani, M.; Płóciennik, P.; Hajjaji, A.; Zawadzka, A. Enhancing the efficiency and stability of Non_Toxic RbSn0.5Ge0.5I3_Based perovskite solar cells through optimization. Mater. Sci. Eng. B 2024, 310, 117672. [Google Scholar] [CrossRef]
- Tarbi, A.; Erguig, H.; Sellam, M.; Zawadzka, A.; Marjanowska, A.; Migalska-Zalas, A.; Goncharova, I.; Myronchuk, G.; Kityk, A.; Sahraoui, B. Diagnostics of the stability of CsPbX3 (X = I, Br, and Cl) inorganic halide perovskites for photovoltaic applications: An in-depth review with an original contribution on impedance characterization. Renew. Sustain. Energy Rev. 2026, 226, 116234. [Google Scholar] [CrossRef]













| Experimental Data | Other Scientific Works—Similar Methods | Other Scientific Works—Different Methods | |||
|---|---|---|---|---|---|
| Cu-Doping [%] | Energy Bandgap [eV] | Cu-Doping [%] | Energy Bandgap [eV] | Cu-Doping [%] | Energy Bandgap [eV] |
| 0 | 3.15 ± 0.05 | 0 [17,18,19,20] | 3.2–3.5 | 0 [22,23] | 3.09, 3.37 |
| - | - | 1 [20] | 3.41 | 1.5 [23] | 3.35 |
| 2.5 | 3.23 ± 0.07 | 2 [21] | 3.18 | 2 [23] | 3.50 |
| 5 | 3.22 ± 0.09 | 6 [21] | 3.02 | 3 [23] | 3.48 |
| 10 | 3.20 ± 0.06 | 10 [21] | 2.85 | 5 [23] | 3.31 |
| - | - | 25 [20] | 2.87 | 10 [22] | 3.14 |
| Sample | Annealing Temperature [°C] | Sheet Resistance [Ω/sq] | Transmittance at 750 nm | FOMH-HR, n = 10 [Ω/sq]1/10 |
|---|---|---|---|---|
| ZnO:Cu (2.5%) | 200 | 74.4 | 0.039 | 0.025 |
| 250 | 842.4 | 0.180 | 0.092 | |
| 300 | 5701.2 | 0.566 | 0.238 | |
| 350 | 9.9 × 106 | 0.863 | 0.172 | |
| 400 | 3.1 × 1010 | 0.951 | 0.085 | |
| 450 | 2.4 × 1014 | 0.943 | 0.034 | |
| ZnO:Cu (5%) | 200 | 2385.6 | 0.074 | 0.034 |
| 250 | 6016.7 | 0.152 | 0.064 | |
| 300 | 13,814.3 | 0.472 | 0.182 | |
| 350 | 1.6 × 108 | 0.890 | 0.135 | |
| 400 | 5.6 × 1010 | 0.912 | 0.077 | |
| 450 | 4.6 × 1010 | 0.890 | 0.076 | |
| ZnO:Cu (10%) | 200 | 150.3 | 0.057 | 0.034 |
| 250 | 1105.1 | 0.197 | 0.098 | |
| 300 | 29,372.3 | 0.657 | 0.235 | |
| 350 | 6.8 × 107 | 0.882 | 0.145 | |
| 400 | 1.3 × 1011 | 0.914 | 0.071 | |
| 450 | 1.6 × 1011 | 0.916 | 0.069 |
| TCO Layer Material | Parameter | |||||
|---|---|---|---|---|---|---|
| UOC (V) | JSC (mA/cm2) | FF (%) | UMPP (V) | IMPP (mA/cm2) | η (%) | |
| ZnO:Cu | 1.03 | 22.80 | 72.46 | 0.82 | 20.80 | 16.97 |
| ITO | 1.07 | 22.80 | 78.85 | 0.92 | 20.83 | 19.24 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Mientki, M.; Zawadzka, A.; Kowalska, M.; Zawadzki, M.; Tarbi, A.; Sahraoui, B.; Płóciennik, P. Transparent Conductive Copper-Doped Zinc Oxide (ZnO:Cu) Thin Films: PVco-D Fabrication and Applications in Perovskite Solar Cells. Materials 2026, 19, 1455. https://doi.org/10.3390/ma19071455
Mientki M, Zawadzka A, Kowalska M, Zawadzki M, Tarbi A, Sahraoui B, Płóciennik P. Transparent Conductive Copper-Doped Zinc Oxide (ZnO:Cu) Thin Films: PVco-D Fabrication and Applications in Perovskite Solar Cells. Materials. 2026; 19(7):1455. https://doi.org/10.3390/ma19071455
Chicago/Turabian StyleMientki, Mateusz, Anna Zawadzka, Magdalena Kowalska, Michał Zawadzki, Amal Tarbi, Bouchta Sahraoui, and Przemysław Płóciennik. 2026. "Transparent Conductive Copper-Doped Zinc Oxide (ZnO:Cu) Thin Films: PVco-D Fabrication and Applications in Perovskite Solar Cells" Materials 19, no. 7: 1455. https://doi.org/10.3390/ma19071455
APA StyleMientki, M., Zawadzka, A., Kowalska, M., Zawadzki, M., Tarbi, A., Sahraoui, B., & Płóciennik, P. (2026). Transparent Conductive Copper-Doped Zinc Oxide (ZnO:Cu) Thin Films: PVco-D Fabrication and Applications in Perovskite Solar Cells. Materials, 19(7), 1455. https://doi.org/10.3390/ma19071455











