1. Introduction
Recent advances in electronic devices and wireless communication technologies have raised increasing concerns regarding electromagnetic interference (EMI), attracting significant attention from both academia and industry. EMI is known to induce malfunctions and unexpected failures in electronic devices under service conditions [
1,
2,
3]. Consequently, effective EMI shielding is essential to ensure the reliable performance and durability of modern electronic systems.
The shielding effectiveness (SE), expressed in decibels (dB), is a key parameter for quantifying the EMI shielding capability of a material. As an electromagnetic wave interacts with a shielding medium, three primary mechanisms contribute to its attenuation: reflection, absorption, and multiple reflections. The total shielding effectiveness (
) is expressed as [
4]:
where
,
and
represent the contributions from reflection, absorption, and multiple reflection, respectively.
,
and
are defined as follows [
4].
and denote the frequency of electromagnetic radiation, relative magnetic permeability, magnetic permeability, relative electrical conductivity, electrical conductivity, and shield thickness, respectively. For conductive materials such as metals, the majority of incident electromagnetic waves are reflected at the surface, while only a small fraction penetrates into the material. As a result, attenuation by reflection () generally provides the dominant contribution to , with absorption and multiple reflection further attenuating the transmitted waves.
Highly conductive metals such as Cu and Ag exhibit excellent EMI shielding performance. However, their high density limits their applicability in fields such as transportation and telecommunications. Accordingly, the development of lightweight materials with superior EMI shielding capabilities has attracted growing research interest over the past decade.
Magnesium and its alloys have emerged as promising candidates due to their unique combination of properties, including the lowest density among structural metals, high specific strength, excellent damping capacity, and favorable electrical conductivity [
5,
6,
7]. The EMI shielding effectiveness of magnesium alloys has been shown to depend on alloying elements, crystallographic texture, and the formation of secondary phases [
8]. For example, Song et al. [
9] reported that strengthening of the basal-type texture enhances the SE of AZ31 alloy. Chen et al. [
10] demonstrated that the orientation of precipitates in Mg–3Sn, Mg-5Sn alloys significantly affects EMI shielding, with improved SE observed when precipitates align along the RD–TD plane, i.e., when the incident electromagnetic wave is perpendicular to the basal plane. These studies highlight the importance of microstructural features such as precipitate morphology, crystallographic texture, and grain size in governing the EMI shielding performance of Mg alloys.
In the present work, the microstructural evolution of Mg–Al–Zn alloy subjected to hot rolling and subsequent aging treatment was systematically characterized. Particular attention was given to the effects of deformation-induced microstructural features and secondary-phase precipitation on both the shielding effectiveness and the mechanical properties.
3. Results and Discussion
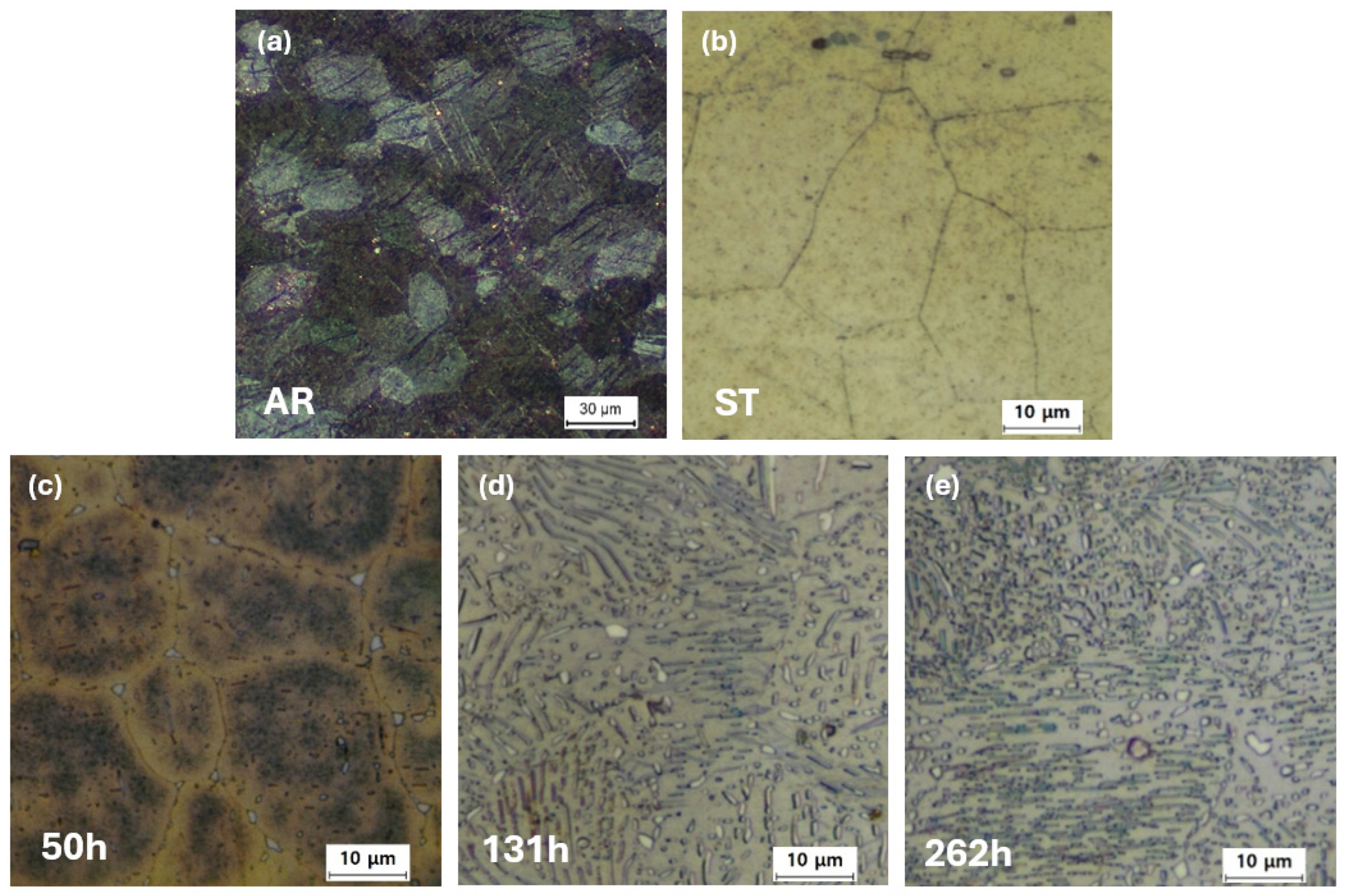
Optical micrographs of the samples subjected to various heat treatments are presented in
Figure 2. All observations were performed on the RD–ND plane of the sheet specimens. The as-rolled (AR) sample exhibits a high density of deformation twins with an average grain size of 20–40 µm (
Figure 2a). Following solution treatment (ST) at 400 °C for 18 h, the grain size remains essentially unchanged, confirming that grain coarsening is effectively suppressed during the treatment (
Figure 2b). This stability is attributed to the grain boundary pinning effect of fine precipitates in the AZ61 alloy [
12]. With subsequent aging treatments, the amount of precipitates increases progressively, as shown in
Figure 2c–e. Especially, lamellar precipitates were observed within the Mg matrix after 131 h of aging (
Figure 2d,e). The precipitate length ranged from <1 µm to over 20 µm on the observed surface.
The calculated phase diagram of the (2-9)Al–1Zn–0.2Mn–Mg alloy with Al contents ranging from 2 to 9 wt.% is shown in
Figure 3, obtained by using the thermodynamic simulation software Pandat-2025. A guideline is included to indicate the composition of the present AZ61 alloy (
Figure 3). The phase diagram predicts the formation of two types of precipitates in this alloy system: Al–Mn phases and Al–Mg phases. The Al–Mg phases are thermally unstable and dissolve during solution heat treatment at 400 °C, whereas the Al–Mn phases remain stable and do not dissolve at this temperature. Consequently, the precipitates observed in the solution-treated (ST) specimen are mainly the Al–Mn phases, which suppressed grain coarsening by exerting a grain boundary pinning effect. With subsequent aging, the number density and size of precipitates continuously increased, as shown in
Figure 2c–e. These precipitates were identified as Mg
17Al
12 phases, consistent with the phase diagram.
The chemical compositions of the precipitates were analyzed by SEM–EDX analysis,
Figure 4. All SEM micrographs were acquired at the RD–ND plane. SEM–EDX analysis of the ST specimen confirmed the persistence of Al–Mn phases,
Figure 4a. Upon subsequent aging heat treatment, Mg
17Al
12 precipitates is identified, as exemplified in the 50 h specimen,
Figure 4b, consistent with the phase diagram in
Figure 3. With prolonged aging, the volume fraction of Mg
17Al
12 increased,
Figure 4c. A comparison between
Figure 4c,d indicates that the 262 h specimen contained coarser Mg
17Al
12 precipitates than the 131 h specimen. The thickness of the Mg
17Al
12 precipitates in the 131 h specimen ranged from 210 to 761 nm, whereas those in the 262 h specimen ranged from 328 to 836 nm. The EDX mapping corresponding to the area of
Figure 4d,e indicated a high Al content within the precipitates, confirming that they are Mg
17Al
12 precipitates.
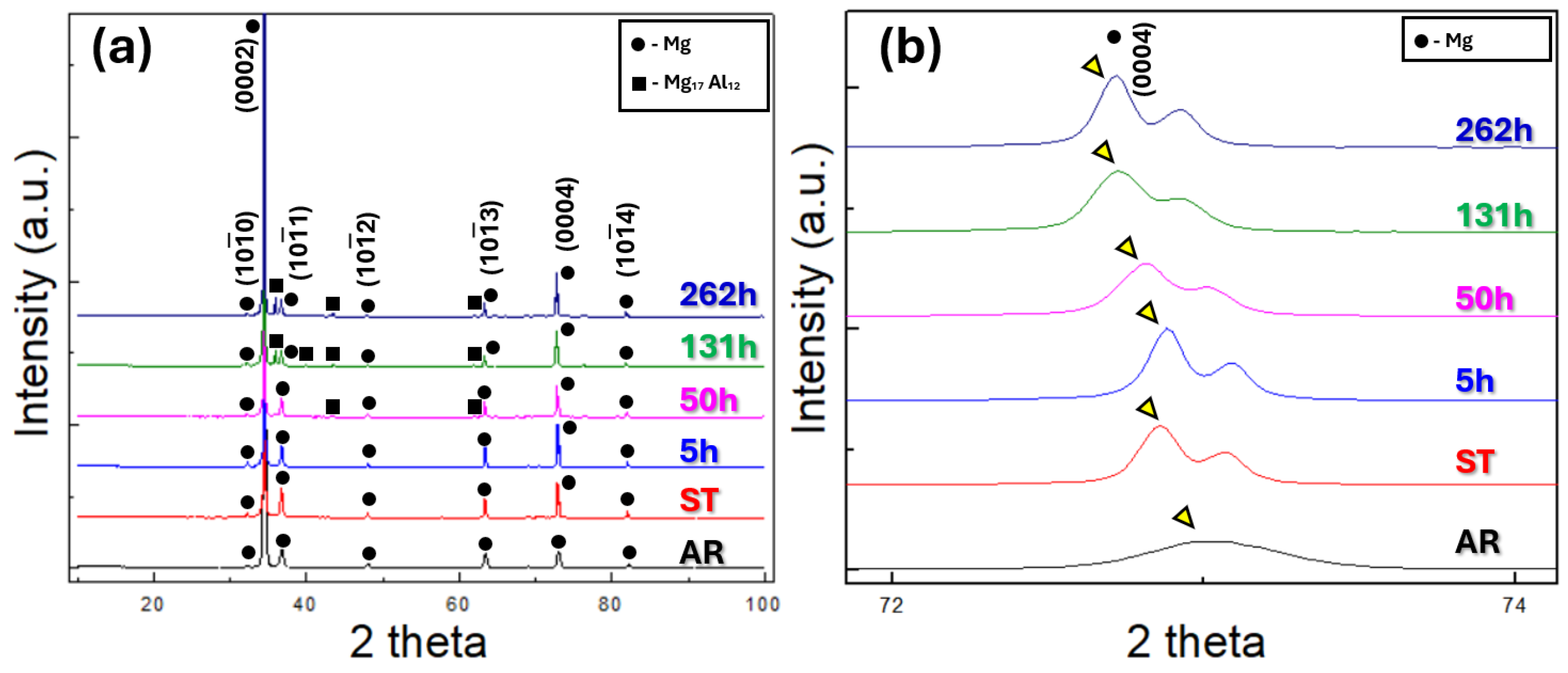
Figure 5 presents the XRD diffraction profiles of the examined specimens. All specimens exhibit a strong (0002) diffraction peak, which can be attributed to the crystallographic texture developed during the hot rolling process. The XRD patterns of the AR and ST specimens exhibit only Mg peaks, without the diffraction peaks corresponding to secondary phases. In the 5 h specimen, diffraction peaks corresponding to the Mg
17Al
12 phase appear as a result of the aging treatment. As the aging time increases from 5 h to 262 h, the intensity of the Mg
17Al
12 peaks progressively increases, confirming the continuous precipitation and growth of Mg
17Al
12 in the aged specimens.
Figure 5b presents an enlarged view of the XRD profiles at high 2θ range. A comparison of the (0004) peak positions, indicated by yellow triangles, reveals distinct differences among the specimens. The AR specimen exhibits its (0004) peak at the rightmost position compared to the others. This shift can be attributed to compressive residual stresses introduced during hot rolling. Such stresses reduce the lattice constant, and according to Bragg’s law (nλ = 2dsinθ), a decreased lattice constant results in a peak shift toward higher 2θ values.
Among the remaining specimens, the solution-treated and 5 h-aged samples also show their (0004) peaks located at the rightmost side. During solution treatment of AZ61 alloy, Al atoms dissolve into the Mg matrix to form a solid solution. Since the atomic radius of Al is smaller than that of Mg, the lattice constant of the Mg matrix decreases, leading to a peak shift to higher 2θ values in accordance with Bragg’s law. Upon subsequent aging, these Al solutes precipitate as Mg
17Al
12, allowing the Mg matrix to recover its intrinsic lattice parameter, which is larger than that of the Al-containing Mg solid solution. Consequently, the diffraction peak shifts back toward lower 2θ values. Nevertheless, after 5 h of aging, a considerable amount of Al solutes still remains in the Mg matrix, comparable to that of the solution-treated state. As a result, the (0004) peak of the 5 h aged specimen also remains at relatively high 2θ values, consistent with the observed XRD results in
Figure 5b. Thus, the precipitation of the Mg
17Al
12 phase can also be observed through the shift in the (0004) diffraction peak.
The EMI SE of the examined specimens is demonstrated in
Figure 6. As shown in
Figure 6a, the AR and 131 h specimens exhibit the highest EMI SE values among the examined AZ61 samples. The electromagnetic interference (EMI) shielding effectiveness (SE) values of the AZ61 specimens at different incident frequencies are summarized in
Table 2. Among the investigated samples, the 131 h-aged specimen exhibited the highest SE of 77.8 dB, which is approximately 32.5% greater than that of the 5 h-aged specimen. The superior EMI shielding performance of the AR and 131 h-aged specimens can be attributed to diverse mechanisms.
Table 3 presents the electrical conductivity of AZ61 alloy samples subjected to different processing conditions. The as-rolled (AR) sample exhibits the lowest electrical conductivity, which can be attributed to the high density of dislocations introduced during the rolling process. The solution-treated (ST) sample shows a slightly higher conductivity than the AR sample. Solution heat treatment reduces lattice defects such as dislocations, thereby improving electrical conductivity. Although solution heat treatment increases the concentration of solute atoms, particularly Al, in the Mg matrix—which generally leads to a reduction in electrical conductivity—this effect appears to be offset by the decrease in defect density during the solution treatment. Furthermore, the electrical conductivity increases with increasing aging time when comparing the aged samples with the ST condition. This improvement can be attributed to the precipitation of Mg
17Al
12 phases during aging, which reduces the concentration of solute atoms in the Mg matrix and consequently enhances electrical conductivity.
It is noteworthy that the as-rolled (AR) specimen exhibits the lowest electrical conductivity while demonstrating the highest EMI shielding effectiveness (EMI SE). As described in the introduction, a reduction in electrical conductivity generally decreases EMI SE by reducing the reflection component (), which is the dominant contributor to the shielding performance of metallic materials. Therefore, the observed high EMI SE cannot be explained solely by electrical conductivity. Instead, the results suggest that microstructural factors introduced during the hot-rolling process play a significant role in enhancing the shielding effectiveness of the AZ61 alloy.
Firstly, defects such as grain boundaries and secondary phases can attenuate incident electromagnetic waves by reflecting the incoming radiation [
8,
13]. In addition, the superior EMI SE of the AR specimen, which contains a high density of dislocations and deformation twins introduced during hot rolling, indicates that dislocations and twins also play a significant role in the attenuation of electromagnetic waves. When comparing the AR specimen with its high dislocation density and a lot of dislocation twins to the ST specimen which has relatively low dislocation density and dislocation twins, the former possesses a more complex atomic arrangement. Such structural complexity increases the probability of collisions between the penetrating electromagnetic waves and lattice atoms, thereby reducing the mean free path of the radiation. Conversely, in the matrix with lower deformation induced defects such as dislocation and twins, electromagnetic waves experience longer mean free paths, resulting in reduced attenuation. Consequently, the AR specimen exhibits excellent EMI SE, whereas the shielding performance of the ST specimen and the aged specimens up to 50 h is diminished.
To examine the microstructural characteristics of the 131 h-aged specimen, electron backscatter diffraction (EBSD) analysis was performed on slice samples sectioned along the RD–TD plane.
Figure 7a shows the EBSD inverse pole figure (IPF) map of the 131 h specimen. A pronounced basal texture was observed, which is consistent with the XRD results, where the (0002) diffraction peak exhibited strong intensity,
Figure 5a. To obtain detailed information on the orientation of the Mg
17Al
12 precipitates, two FIB slices were extracted from the RD–TD plane along the ND. The orientation of the FIB slices with respect to the sample geometry is schematically illustrated in
Figure 7b. One slice was prepared from a grain with its crystallographic c-axis aligned parallel to the ND (grain I in
Figure 7a), hereafter referred to as 131h-I. The other slice was taken from a grain with the c-axis tilted by approximately 52° from the ND (grain II in
Figure 7a), denoted as 131h-II.
The microstructural features including crystallographic orientations of 131h-I and 131h-II were analyzed using transmission Kikuchi diffraction (TKD). The 131 h-aged specimen exhibited a high density of precipitates, consistent with the microstructural features observed in
Figure 2d. The precipitates displayed a wide range of lengths, from less than 1 μm to more than 8.5 μm. In the case of 131h-I, the thickness of the Mg
17Al
12 precipitates was measured to be in the range of 154–210 nm (
Figure 8b).
It is to note that the Mg
17Al
12 precipitates are nearly perpendicular to the ND, clearly seen in
Figure 8. Because the 131h-I specimen is extracted from the grain having its c-axis almost parallel to the ND, this gives evidence that the Mg
17Al
12 precipitates are formed along the (0001) basal plane of the Mg matrix.
Figure 8c,d show the IPF map obtained from TKD analysis and the grain orientation, respectively. From the pole figures,
Figure 8d, it is clear that the long axis of the Mg
17Al
12 precipitates is aligned perpendicular to the c-axis of the matrix, i.e., parallel to the <10–10> and <11–20> axes. The long axis of the precipitate appears to rise from left to right in the SEM image, whereas it appears to be oriented downward in the TKD IPF map. The discrepancy between the SEM and TKD analyses arises from the observation of opposite sides of the FIB-prepared slice, resulting in a mirrored image of the microstructural orientation. The thickness of the Mg
17Al
12 precipitates in the 131h-II specimen is in the range of 206 nm to 653 nm, which is substantially thicker than the precipitates in
Figure 8b. That is, the thickness of the Mg
17Al
12 phases formed on the (0001) basal plane varies with changing the observation plane. For example, if they are observed on a pyramidal plane the precipitates look thicker than in the case of observation from the prismatic plane. From the above results the formation of plate-shaped Mg
17Al
12 phase on (0001) plane is evident.
A schematic representation indicating a strong basal-type texture of the 131 h specimen is shown in
Figure 9a. Given this strong basal-type texture with the c-axes aligned parallel to the ND and the fact that the precipitates are oriented parallel to the (0002) plane, the alignment of Mg
17Al
12 precipitates can be schematically depicted as shown in
Figure 9b. When incident electromagnetic waves penetrate the specimen perpendicular to the RD–TD plane, the plate-like Mg
17Al
12 phases act as secondary reflection sites, thereby enhancing the EMI SE [
10].
In conclusion, the superior EMI SE of the 131 h specimen can be attributed to the increased volume fraction of Mg
17Al
12 precipitates with favorable orientation with the Mg matrix. These secondary phases enhance shielding performance by inducing multiple reflections of incident electromagnetic waves at the interfaces between the Mg matrix and the precipitate phase [
8,
10,
14,
15]. Consequently, the formation of dense and well-aligned precipitates is highly beneficial for improving EMI SE.
As shown in
Figure 6 and
Table 2, the 262 h specimen showed a lower EMI SE compared to the 131 h specimen. This degradation in shielding effectiveness can be attributed to the coarsening of the Mg
17Al
12 precipitates, as observed in
Figure 4c,d. As already mentioned, 131 h specimen’s Mg
17Al
12 precipitate’s thickness ranged from 210 to 761 nm, whereas those in the 262 h specimen ranged from 328 to 836 nm. Such coarsening of the secondary phases accompanies the reduction in their length, which is also an effective way to reduce the interface energy between the Mg matrix and the Mg
17Al
12 phase. As a result, the multiple reflection of incident electromagnetic waves is diminished, leading to reduced EMI SE.
Figure 10 presents the Vickers hardness values of the AZ61 alloy at different sample conditions. The AR specimen has the highest dislocation density, resulting in a hardness of 72.2 HV. Since dislocation density is dramatically reduced by annealing, the ST specimen shows a decrease in hardness by 57.0 HV. The specimens aged for 5 h and 50 h exhibited hardness values comparable to that of the ST condition, indicating that age hardening in AZ61 is negligible up to 50 h.
After prolonged aging, the specimens aged for 131 h and 262 h exhibited increased hardness values of 71.2 HV and 73.1 HV, respectively, corresponding to increments of 12.3 HV and 14.2 HV compared with the 50 h specimen. Notably, this hardening response contrasts with the behavior of conventional age-hardenable alloys, where over-aging typically results in a decrease in hardness.
This anomalous hardening can be explained with reference to
Figure 2. As shown in
Figure 2c–e, the 131 h and 262 h specimens contain a markedly higher volume fraction of Mg
17Al
12 precipitates compared to the 50 h specimen. At these later stages of aging, the precipitates are densely distributed, and their interparticle spacing becomes smaller than the size of the Vickers indentation. As a result, the indenter inevitably presses the Mg
17Al
12 precipitates during hardness tests. Since Mg
17Al
12 precipitates is an intermetallic phase that is harder than the Mg matrix, this increment of the pressed Mg
17Al
12 phase during the operation of the Vickers hardness test contributes directly to the rise in measured hardness. Therefore, the rise in hardness observed from 50 h to 131 h and 262 h can be attributed to the substantially increased amount of Mg
17Al
12 phase during prolonged aging.