Retarding Effect and Hydration Mechanism of Sodium Polyacrylate on Magnesium Potassium Phosphate Cement
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Mix Design
2.3. Test Methods
3. Results
3.1. Macro Performance Testing
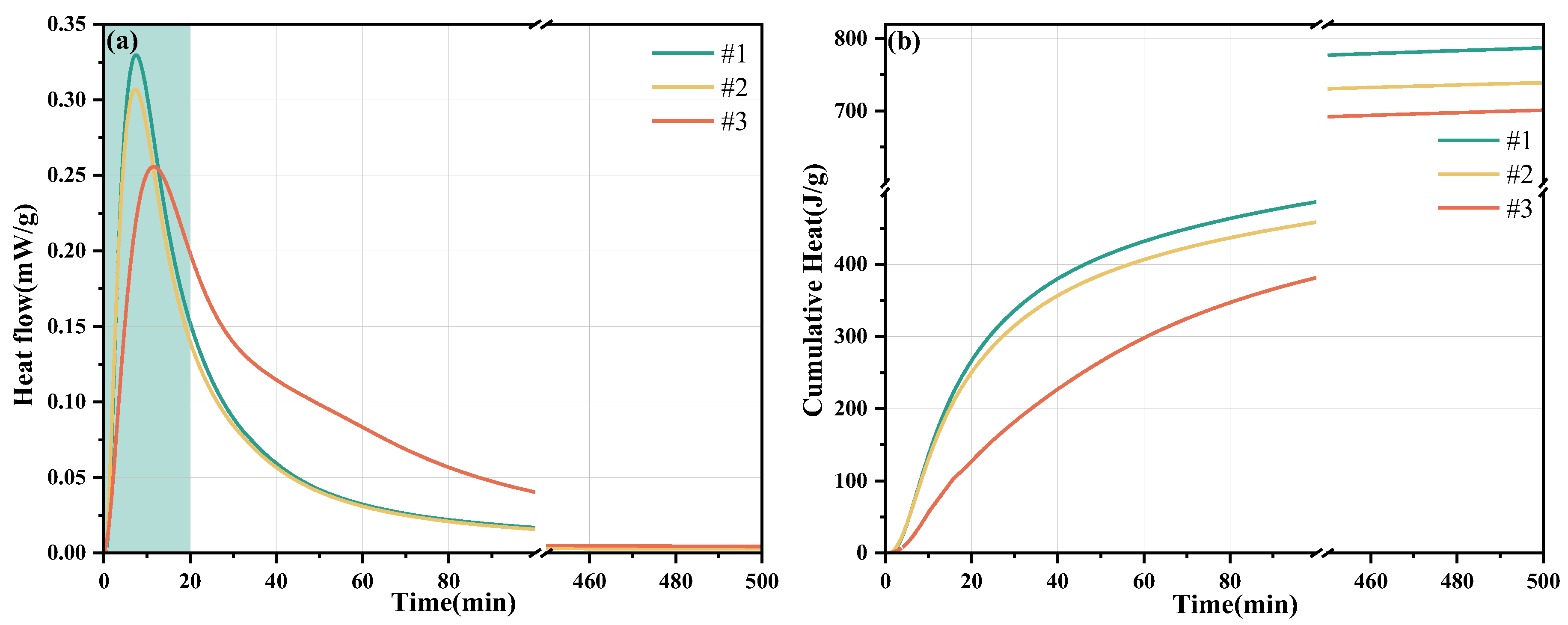
3.2. Hydration Heat Test
3.3. pH and Conductivity
3.4. XRD
3.5. Evolution of Ion Concentrations
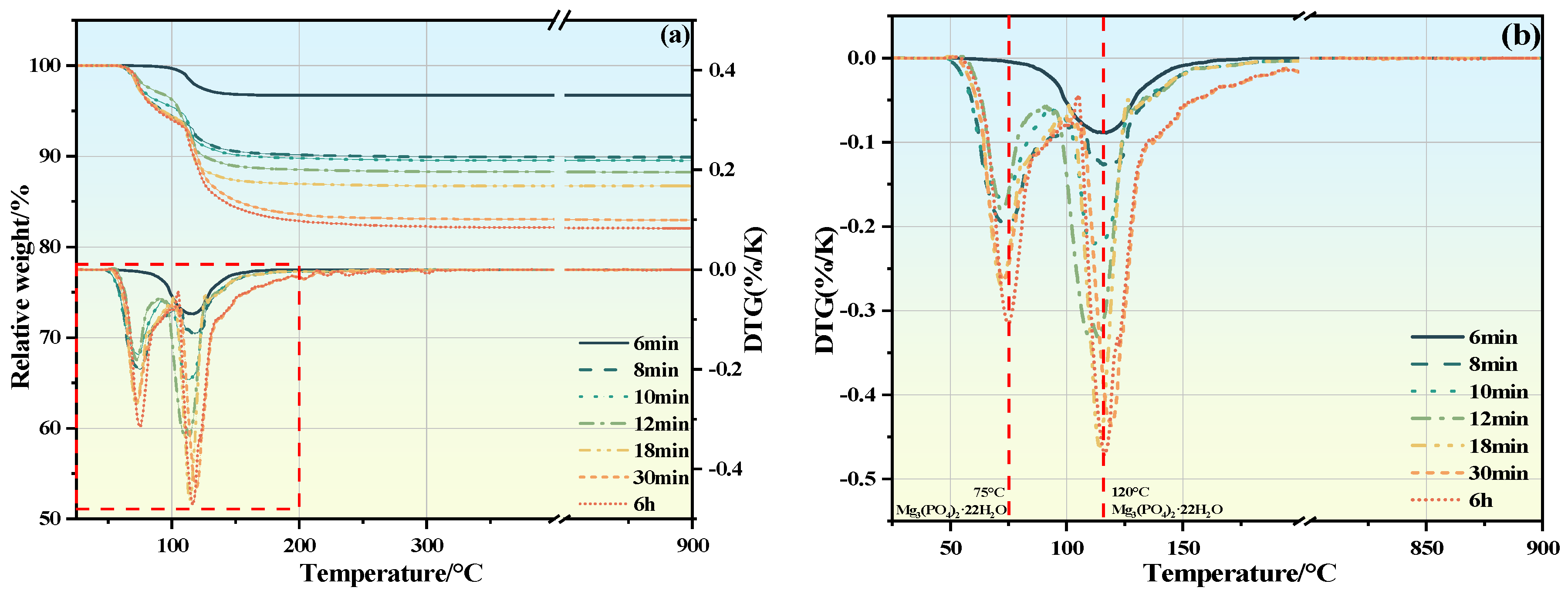
3.6. TG/DTG
3.7. FTIR
3.8. Microstructure
4. Discussions
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Xu, B.; Ma, H.; Shao, H.; Li, Z.; Lothenbach, B. Influence of fly ash on compressive strength and micro-characteristics of magnesium potassium phosphate cement mortars. Cem. Concr. Res. 2017, 99, 86–94. [Google Scholar] [CrossRef]
- Xu, B.; Winnefeld, F.; Kaufmann, J.; Lothenbach, B. Influence of magnesium-to-phosphate ratio and water-to-cement ratio on hydration and properties of magnesium potassium phosphate cements. Cem. Concr. Res. 2019, 123, 105781. [Google Scholar] [CrossRef]
- Qiao, F.; Chau, C.K.; Li, Z. Property evaluation of magnesium phosphate cement mortar as patch repair material. Constr. Build. Mater. 2010, 24, 695–700. [Google Scholar] [CrossRef]
- Qiao, F.; Chau, C.K.; Li, Z. Setting and strength development of magnesium phosphate cement paste. Adv. Cem. Res. 2009, 21, 175–180. [Google Scholar] [CrossRef]
- Qin, J.; Qian, J.; You, C.; Fan, Y.; Li, Z.; Wang, H. Bond behavior and interfacial micro-characteristics of magnesium phosphate cement onto old concrete substrate. Constr. Build. Mater. 2018, 167, 166–176. [Google Scholar] [CrossRef]
- Ma, S.; Cao, Z.; Wei, C.; Shao, Y.; Wu, P.; Zhang, Z.; Liu, X. Red mud-modified magnesium potassium phosphate cement used as rapid repair materials: Durability, bonding property, volume stability and environment performance optimization. Constr. Build. Mater. 2024, 415, 135144. [Google Scholar] [CrossRef]
- Chen, X.; Zhou, X.; Fan, Z.; Peng, Z.; Lu, Q. Competitive encapsulation of multiple heavy metals by magnesium potassium phosphate cement: Hydration characteristics and leaching toxicity properties. Waste Manag. 2024, 177, 115–124. [Google Scholar] [CrossRef]
- Buj, I.; Torras, J.; Casellas, D.; Rovira, M.; De Pablo, J. Effect of heavy metals and water content on the strength of magnesium phosphate cements. J. Hazard. Mater. 2009, 170, 345–350. [Google Scholar] [CrossRef]
- Wagh, A.S.; Jeong, S.Y. Chemically Bonded Phosphate Ceramics: I, A Dissolution Model of Formation. J. Am. Ceram. Soc. 2003, 86, 1838–1844. [Google Scholar] [CrossRef]
- Walling, S.A.; Provis, J.L. Magnesia-Based Cements: A Journey of 150 Years, and Cements for the Future? Chem. Rev. 2016, 116, 4170–4204. [Google Scholar] [CrossRef]
- Chau, C.K.; Qiao, F.; Li, Z. Microstructure of magnesium potassium phosphate cement. Constr. Build. Mater. 2011, 25, 2911–2917. [Google Scholar] [CrossRef]
- Ding, Z.; Li, Z. Effect of aggregates and water contents on the properties of magnesium phospho-silicate cement. Cem. Concr. Compos. 2005, 27, 11–18. [Google Scholar] [CrossRef]
- Lee, K.-H.; Yoon, H.-S.; Yang, K.-H. Tests on magnesium potassium phosphate composite mortars with different water-to-binder ratios and molar ratios of magnesium-to-phosphate. Constr. Build. Mater. 2017, 146, 303–311. [Google Scholar] [CrossRef]
- Li, Y.; Sun, J.; Chen, B. Experimental study of magnesia and M/P ratio influencing properties of magnesium phosphate cement. Constr. Build. Mater. 2014, 65, 177–183. [Google Scholar] [CrossRef]
- Ma, H.; Xu, B.; Li, Z. Magnesium potassium phosphate cement paste: Degree of reaction, porosity and pore structure. Cem. Concr. Res. 2014, 65, 96–104. [Google Scholar] [CrossRef]
- Tan, Y.; Yu, H.; Li, Y.; Wu, C.; Dong, J.; Wen, J. Magnesium potassium phosphate cement prepared by the byproduct of magnesium oxide after producing Li2CO3 from salt lakes. Ceram. Int. 2014, 40, 13543–13551. [Google Scholar] [CrossRef]
- Xu, B.; Ma, H.; Li, Z. Influence of magnesia-to-phosphate molar ratio on microstructures, mechanical properties and thermal conductivity of magnesium potassium phosphate cement paste with large water-to-solid ratio. Cem. Concr. Res. 2015, 68, 1–9. [Google Scholar] [CrossRef]
- Coumes, C.C.D.; Lambertin, D.; Lahalle, H.; Antonucci, P.; Cannes, C.; Delpech, S. Selection of a mineral binder with potentialities for the stabilization/solidification of aluminum metal. J. Nucl. Mater. 2014, 453, 31–40. [Google Scholar] [CrossRef]
- Gardner, L.J.; Bernal, S.A.; Walling, S.A.; Corkhill, C.L.; Provis, J.L.; Hyatt, N.C. Characterisation of magnesium potassium phosphate cements blended with fly ash and ground granulated blast furnace slag. Cem. Concr. Res. 2015, 74, 78–87. [Google Scholar] [CrossRef]
- Gardner, L.J.; Corkhill, C.L.; Walling, S.A.; Vigor, J.E.; Murray, C.A.; Tang, C.C.; Provis, J.L.; Hyatt, N.C. Early age hydration and application of blended magnesium potassium phosphate cements for reduced corrosion of reactive metals. Cem. Concr. Res. 2021, 143, 106375. [Google Scholar] [CrossRef]
- Lahalle, H.; Coumes, C.C.D.; Mercier, C.; Lambertin, D.; Cannes, C.; Delpech, S.; Gauffinet, S. Influence of the w/c ratio on the hydration process of a magnesium phosphate cement and on its retardation by boric acid. Cem. Concr. Res. 2018, 109, 159–174. [Google Scholar] [CrossRef]
- Klammert, U.; Vorndran, E.; Reuther, T.; Müller, F.A.; Zorn, K.; Gbureck, U. Low temperature fabrication of magnesium phosphate cement scaffolds by 3D powder printing. J. Mater. Sci. Mater. Med. 2010, 21, 2947–2953. [Google Scholar] [CrossRef]
- Wang, A.; Zhang, J.; Li, J.; Ma, A.; Liu, L. Effect of liquid-to-solid ratios on the properties of magnesium phosphate chemically bonded ceramics. Mater. Sci. Eng. C 2013, 33, 2508–2512. [Google Scholar] [CrossRef] [PubMed]
- Sugama, T.; Kukacka, L.E. Characteristics of magnesium polyphosphate cements derived from ammonium polyphosphate solutions. Cem. Concr. Res. 1983, 13, 499–506. [Google Scholar] [CrossRef]
- Lahalle, H.; Coumes, C.C.D.; Mesbah, A.; Lambertin, D.; Cannes, C.; Delpech, S.; Gauffinet, S. Investigation of magnesium phosphate cement hydration in diluted suspension and its retardation by boric acid. Cem. Concr. Res. 2016, 87, 77–86. [Google Scholar] [CrossRef]
- Pei, H.; Zhang, S.; Bai, L.; Hou, D.; Yang, Q.; Borana, L. Early-age shrinkage strain measurements of the graphene oxide modified magnesium potassium phosphate cement. Measurement 2019, 139, 293–300. [Google Scholar] [CrossRef]
- Deng, Q.; Lai, Z.; Xiao, R.; Wu, J.; Liu, M.; Lu, Z.; Lv, S. Effect of Waste Glass on the Properties and Microstructure of Magnesium Potassium Phosphate Cement. Materials 2021, 14, 2073. [Google Scholar] [CrossRef]
- Tao, Y.; Zhenyu, L.; Zhichao, H.; Chunrong, R.; Yuanyuan, W.; Xin, H.; Jie, W.; Mengliang, L.; Qiubai, D.; Khan, K.; et al. Mechanical and microstructure of magnesium potassium phosphate cement with a high concentration of Ni(II) and its leaching toxicity. Constr. Build. Mater. 2020, 245, 118425. [Google Scholar] [CrossRef]
- Le Rouzic, M.; Chaussadent, T.; Platret, G.; Stefan, L. Mechanisms of k-struvite formation in magnesium phosphate cements. Cem. Concr. Res. 2017, 91, 117–122. [Google Scholar] [CrossRef]
- Xu, B.; Lothenbach, B.; Leemann, A.; Winnefeld, F. Reaction mechanism of magnesium potassium phosphate cement with high magnesium-to-phosphate ratio. Cem. Concr. Res. 2018, 108, 140–151. [Google Scholar] [CrossRef]
- De Campos, M.; Davy, C.A.; Djelal, N.; Rivenet, M.; Garcia, J. Development of a stoichiometric magnesium potassium phosphate cement (MKPC) for the immobilization of powdered minerals. Cem. Concr. Res. 2021, 142, 106346. [Google Scholar] [CrossRef]
- Briki, Y.; Avet, F.; Zajac, M.; Bowen, P.; Haha, M.B.; Scrivener, K. Understanding of the factors slowing down metakaolin reaction in limestone calcined clay cement (LC3) at late ages. Cem. Concr. Res. 2021, 146, 106477. [Google Scholar] [CrossRef]
- Wei, X.; Lin, L.; Zhang, X.; Zhao, G.; Wang, J.; Jiang, C.; Feng, S. Effect of boric acid and triethanolamine compound on early hydration properties of magnesium potassium phosphate cement. Constr. Build. Mater. 2024, 449, 138364. [Google Scholar] [CrossRef]
- Coumes, C.C.D.; Rousselet, A.; Xu, B.; Mercier, C.A.; Gauffinet, S. Investigation of aluminum nitrate as a set retarder of magnesium potassium phosphate cement: Mechanisms involved in diluted suspension. Cem. Concr. Res. 2021, 150, 106608. [Google Scholar] [CrossRef]
- Gelli, R.; Tonelli, M.; Martini, F.; Calucci, L.; Borsacchi, S.; Ridi, F. Effect of borax on the hydration and setting of magnesium phosphate cements. Constr. Build. Mater. 2022, 348, 128686. [Google Scholar] [CrossRef]
- Yu, S.; Liu, L.; Xu, C.; Dai, H. Magnesium phosphate based cement with improved setting, strength and cytocompatibility properties by adding Ca(H2PO4)2·H2O and citric acid. J. Mech. Behav. Biomed. Mater. 2019, 91, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Haque, M.A.; Chen, B. Research progresses on magnesium phosphate cement: A review. Constr. Build. Mater. 2019, 211, 885–898. [Google Scholar] [CrossRef]
- Chen, X.; Wang, S.; Zhou, Y.; Cheeseman, C.; Bi, W.; Zhang, T. Improved low-carbon magnesium oxysulfate cement pastes containing boric acid and citric acid. Cem. Concr. Compos. 2022, 134, 104813. [Google Scholar] [CrossRef]
- Li, Y.; Wang, N.; Lin, H.; Li, Y.; Li, H.; Liu, X. Optimization of mechanical property and microstructure for magnesium phosphate cement-based canvas using borax. Constr. Build. Mater. 2024, 438, 137136. [Google Scholar] [CrossRef]
- Yu, Y.; Peng, R.; Yang, C.; Tang, Y. Eco-friendly and cost-effective superabsorbent sodium polyacrylate composites for environmental remediation. J. Mater. Sci. 2015, 50, 5799–5808. [Google Scholar] [CrossRef]
- Mechtcherine, V.; Wyrzykowski, M.; Schröfl, C.; Snoeck, D.; Lura, P.; De Belie, N.; Mignon, A.; Van Vlierberghe, S.; Klemm, A.J.; Almeida, F.C.R.; et al. Application of super absorbent polymers (SAP) in concrete construction—update of RILEM state-of-the-art report. Mater. Struct. 2021, 54, 80. [Google Scholar] [CrossRef]
- Craeye, B.; Geirnaert, M.; Schutter, G.D. Super absorbing polymers as an internal curing agent for mitigation of early-age cracking of high-performance concrete bridge decks. Constr. Build. Mater. 2011, 25, 1–13. [Google Scholar] [CrossRef]
- Chau, C.K.; Li, Z. Accelerated Reactivity Assessment of Light Burnt Magnesium Oxide. J. Am. Ceram. Soc. 2008, 91, 1640–1645. [Google Scholar] [CrossRef]
- JC-T 2537-2019; Magnesium Phosphate Repair Mortar. Ministry of Industry and Information Technology of the People’s Republic of China: Beijing, China, 2019.
- Frontera, C.; Rodrı, J. FullProf as a new tool for flipping ratio analysis. Phys. B Condens. Matter 2003, 335, 219–222. [Google Scholar] [CrossRef]
- McNeill, I.C.; Sadeghi, S.M.T. Thermal stability and degradation mechanisms of poly(acrylic acid) and its salts: Part 1—Poly(acrylic acid). Polym. Degrad. Stab. 1990, 29, 233–246. [Google Scholar] [CrossRef]
- McNeill, I.C.; Sadeghi, S.M.T. Thermal stability and degradation mechanisms of poly(acrylic acid) and its salts: Part 2—Sodium and potassium salts. Polym. Degrad. Stab. 1990, 30, 213–230. [Google Scholar] [CrossRef]
- Lothenbach, B.; Xu, B.; Winnefeld, F. Thermodynamic data for magnesium (potassium) phosphates. Appl. Geochem. 2019, 111, 104450. [Google Scholar] [CrossRef]











| Oxide | MgO | CaO | SiO2 | Other |
|---|---|---|---|---|
| Content/% | ≥90.42 | ≤1.51 | ≤3.16 | ≤0.79 |
| NO. | MgO (g) | KH2PO4 (g) | H2O (g) | PAAS (g) | PAAS/(M+P) (%) |
|---|---|---|---|---|---|
| M-0 | 35.07 | 14.93 | 9 | 0 | 0 |
| M-4 | 35.07 | 14.93 | 9 | 2 | 4 |
| M-8 | 35.07 | 14.93 | 9 | 4 | 8 |
| M-12 | 35.07 | 14.93 | 9 | 6 | 12 |
| M-16 | 35.07 | 14.93 | 9 | 8 | 16 |
| NO. | MgO (g) | KH2PO4 (g) | H2O (g) | PAAS (g) | PAAS/H2O (%) |
|---|---|---|---|---|---|
| #1 | 35.07 | 14.93 | 500 | 0 | 0 |
| #2 | 35.07 | 14.93 | 500 | 20 | 4% |
| #3 | 35.07 | 14.93 | 500 | 80 | 16% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Cui, Y.; Liu, R.; Yang, Y.; Pang, B.; Wang, Y. Retarding Effect and Hydration Mechanism of Sodium Polyacrylate on Magnesium Potassium Phosphate Cement. Materials 2026, 19, 1349. https://doi.org/10.3390/ma19071349
Cui Y, Liu R, Yang Y, Pang B, Wang Y. Retarding Effect and Hydration Mechanism of Sodium Polyacrylate on Magnesium Potassium Phosphate Cement. Materials. 2026; 19(7):1349. https://doi.org/10.3390/ma19071349
Chicago/Turabian StyleCui, Yunpeng, Runqing Liu, Yuanquan Yang, Bo Pang, and Yihe Wang. 2026. "Retarding Effect and Hydration Mechanism of Sodium Polyacrylate on Magnesium Potassium Phosphate Cement" Materials 19, no. 7: 1349. https://doi.org/10.3390/ma19071349
APA StyleCui, Y., Liu, R., Yang, Y., Pang, B., & Wang, Y. (2026). Retarding Effect and Hydration Mechanism of Sodium Polyacrylate on Magnesium Potassium Phosphate Cement. Materials, 19(7), 1349. https://doi.org/10.3390/ma19071349





