Research on the Mechanism of Hydrogen Plasma Heating and Reduction of Acidic Pellets
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Materials
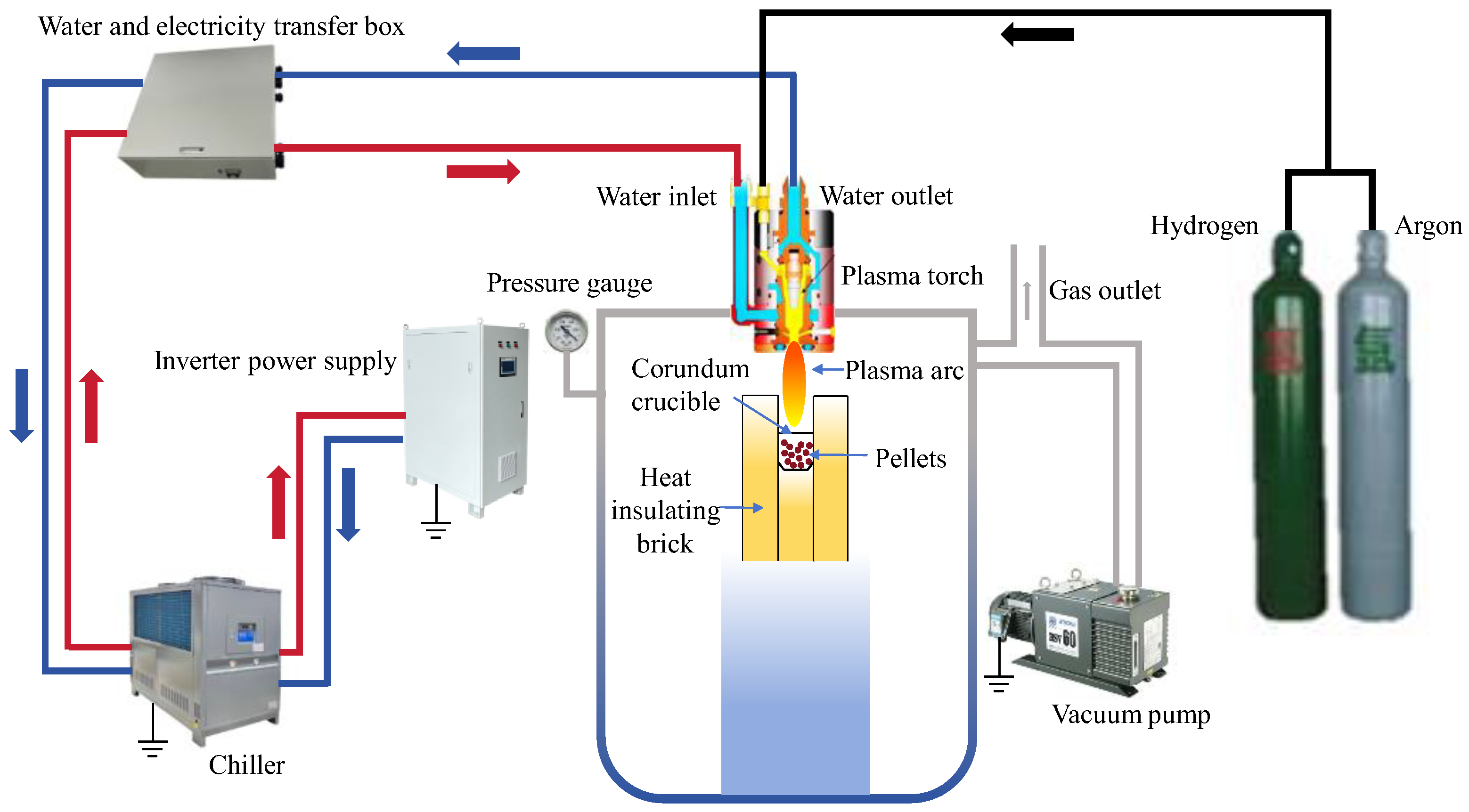
2.2. Experimental Apparatus and Research Methods
3. Experimental Results and Discussion
3.1. Macroscopic Analysis of Acidic Pellets Reduced by Hydrogen Plasma
3.2. Phase Composition Analysis of Acidic Pellets Reduced by Hydrogen Plasma
3.3. Microscopic Morphology and Composition Analyses of Acidic Pellets Reduced by Hydrogen Plasma
3.4. Analysis of Conversion and Conversion Rate of Acidic Pellets by Hydrogen Plasma
3.5. Analysis of Reaction Kinetics Model for Hydrogen Plasma Reduction of Acidic Pellets
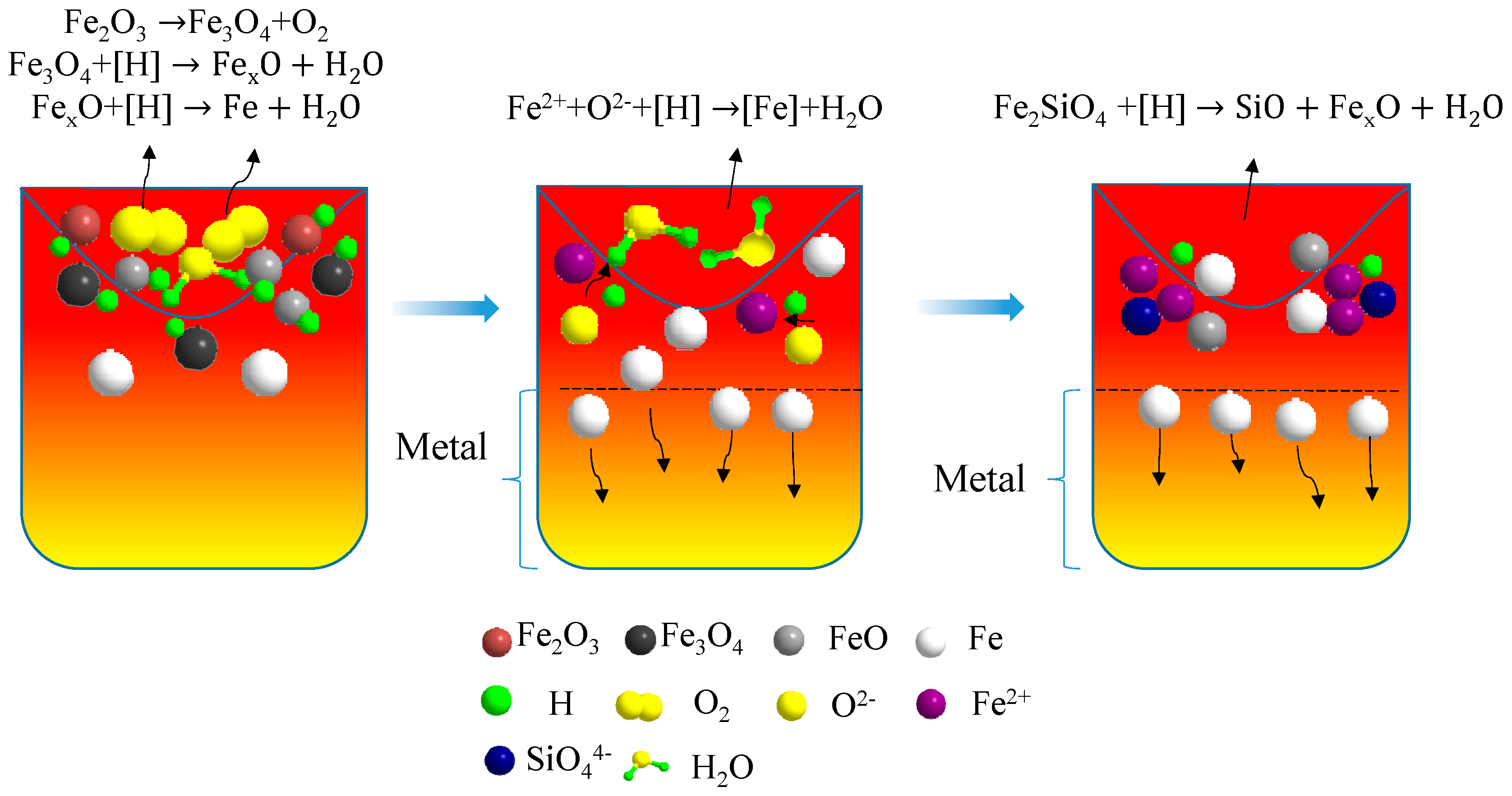
3.6. Phase Transformation Rules of Acidic Pellets Reduced by Hydrogen Plasma
- (1)
- First stage: High-temperature melting of Fe2O3 and surface reduction by hydrogen plasma (t ≤ 10 min)
- (2)
- Second stage: Rapid reduction of hydrogen plasma and effective deposition of metallic iron (10 min ≤ t ≤ 30 min)
- (3)
- Third stage: Slow reaction stage of FexO and Fe2SiO4 (t ≥ 30 min)
4. Conclusions
- (1)
- The hydrogen plasma reduction of acidic pellets generally exhibited a stepped trend. At a hydrogen concentration of 15%, the gas flow rate of 4.5 L min−1 achieved a conversion rate of 99.89% for acidic pellets in 40 min, while the gas flow rate of 3 L min−1 achieved a maximum of 82.5% reduction in 40 min. The deoxygenation rate per area increased from 0.87 g (cm2 min)−1 at 3 L min−1 to 1.11 g (cm2 min)−1 at 4.5 L min−1. This suggested that it is possible to achieve a high reduction rate of acidic pellets with lower hydrogen concentrations and higher gas flow rates.
- (2)
- The hydrogen plasma reduction of acidic pellets followed a three-dimensional diffusion-controlled reaction model, with the controlling step primarily being the diffusion of the reactants Fe2+, [H], and O2− particles toward the reaction interface. The mechanism function was determined to be 2/3(1 + α)3/2[(1 + α)1/3 − 1]−1. Under different gas flow rates (3 and 4.5 L min−1), the corresponding reduction reaction rate constants k(T) were 12.6 × 10−2 and 12.8 × 10−2 s−1, respectively. The apparent activation energy was 21.9 kJ mol−1, and the empirical equation for the reaction reduction rate was ln r = −2637.5/T − 0.407.
- (3)
- After the acidic pellets underwent HPSR, the contents of impurity elements such as Si and Al could be effectively reduced. After 40 min of reduction, the metallic iron achieved a purity of 99.90%, meeting the purity requirements for industrial 3N-grade high-purity iron. The findings of this study can accelerate the application of hydrogen plasma in the metallurgical industry and provide a theoretical basis for developing new technologies in the steel industry.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Wang, L.X.; Zeng, H.; Luan, J.Y.; Chen, W.F.; Wang, C.Z. Research on the Advancement of Steel Process Flow under the Background of “Double Carbon”. Shandong Metall. 2024, 46, 49–53. [Google Scholar]
- Meng, X.; Li, C.X.; Zhao, P.; Feng, Z.K.; Ji, Z.H.; Sun, H.K. Technological progress of CO2 resource utilization in whole process of iron and steel production. Sinter. Pelletizing 2024, 49, 21–34. [Google Scholar]
- Zhang, J.; Li, K.; Liu, Z.; Yang, T. Primary Exploration of Hydrogen Metallurgy; Springer Nature: New York, NY, USA, 2024. [Google Scholar]
- Zhang, J.L.; Zong, Y.B.; Li, K.J.; Liang, Z.; Yang, T.J. Progress and future perspective of low-carbon ironmaking process in the world. Iron Steel 2024, 59, 45–55+155. [Google Scholar]
- Haki, B.; Akbar Rhamdhan, M.; Hidayat, T.; Hendrawan, Y.; Zulhan, Z. Fast Iron Production from Limonite Iron Ore in a Lab-Scale Hydrogen Plasma Smelting Reduction (HPSR) Reactor. J. Sustain. Metall. 2025, 11, 4437–4450. [Google Scholar] [CrossRef]
- Song, X.R.; Liu, Z.J.; Zhang, J.L.; Zong, Y.B.; Wang, Y.Z. Research progress of plasma hydrogen reduction of iron oxides. China Metall. 2022, 32, 7–14. [Google Scholar]
- Ding, C.Y.; Xue, S.; Chang, R.D.; Jiang, F.; Long, H.M.; Yu, Z.W. Application of plasma reduction technology in metallurgical processes. J. Iron Steel Res. 2024, 36, 568–579. [Google Scholar]
- Rajput, P.; Bhoi, B.; Sahoo, S.; Paramguru, R.K.; Mishra, B.K. Preliminary investigation into direct reduction of iron in low temperature hydrogen plasma. Ironmak. Steelmak. 2013, 40, 61–68. [Google Scholar] [CrossRef]
- Nagasaka, T.; Hino, M.; Ban-Ya, S. Interfacial kinetics of hydrogen with liquid slag containing iron oxide. Metall. Mater. Trans. B 2000, 31, 945–955. [Google Scholar] [CrossRef]
- Hiebler, H.; Plaul, J.F. Hydrogen plasma smelting reduction-an option for steelmaking in the future. Metalurgija 2004, 43, 155–162. [Google Scholar]
- Stokes, C.S. Chemistry in high temperature plasma jets. Adv. Chem. 1969, 80, 390–405. [Google Scholar]
- Behera, P.R.; Bhoi, B.; Paramguru, R.K.; Mukherjee, P.S.; Mishra, B.K. Hydrogen plasma smelting reduction of Fe2O3. Metall. Mater. Trans. B 2019, 50, 262–270. [Google Scholar] [CrossRef]
- Nakamura, Y.; Ito, M.; Ishikawa, H. Reduction and dephosphorization of molten iron oxide with hydrogen-argon plasma. Plasma Chem. Plasma Process. 1981, 1, 149–160. [Google Scholar] [CrossRef]
- Naseri, S.M.; Schenk, J.; Zarl, M.A. Reduction of haematite using hydrogen thermal plasma. Materials 2019, 12, 1608. [Google Scholar] [CrossRef]
- Wang, X.R.; Chen, D.Y.; Sun, D.H.; Yang, P.Y.; Lee, F.S.C. Research progress on the application of thermal plasma technology for pollutant treatment. Chemistry 1999, 4, 1–6. [Google Scholar]
- Zhang, Y.W.; Ding, W.Z.; Lu, X.; Guo, S.; Xu, K.D. Reduction of TiO2 with hydrogen cold plasma in DC pulsed glow discharge. Trans. Nonferrous Met. Soc. China 2005, 15, 594–599. [Google Scholar]
- Anand, R.A.; Pande, M.M.; Kumar, D.; Viswanathan, N.N. Thermal Decomposition of Hematite Ore Fines in Air. Steel Res. Int. 2025, 96, 2400200. [Google Scholar] [CrossRef]
- Satritama, B.; Cooper, C.; Fellicia, D.; Pownceby, M.I.; Palanisamy, S.; Ang, A.; Mukhlis, R.Z.; Pye, J.; Rahbari, A.; Brooks, G.A.; et al. Hydrogen Plasma for Low-Carbon Extractive Metallurgy: Oxides Reduction, Metals Refining, and Wastes Processing. J. Sustain. Metall. 2024, 10, 1845–1894. [Google Scholar] [CrossRef]
- Naseri, S.M.; Schenk, J. Thermodynamic of liquid iron ore reduction by hydrogen thermal plasma. Metals 2018, 8, 1051. [Google Scholar] [CrossRef]
- Zhang, Y.W.; Ding, W.Z.; Guo, S.Q.; Hu, X.K. Reduction of metal oxide by non-equilibrium hydrogen plasma. Shanghai Met. 2004, 26, 17–20. [Google Scholar] [CrossRef]
- Cooper, C.; Brooks, G.; Rhamdhani, M.A.; Pye, J.; Rahbari, A. Technoeconomic analysis of low-emission steelmaking using hydrogen thermal plasma. J. Clean. Prod. 2025, 495, 144896. [Google Scholar] [CrossRef]
- Li, Y. Study on Phase Evolution and Reduction Kinetics of Molten Iron Oxides Reduced by Hydrogen. Ph.D. Thesis, University of Science and Technology Beijing, Beijing, China, 2022. [Google Scholar]
- Sabat, K.C.; Rajput, P.; Paramguru, R.K.; Bhoi, B.; Mishra, B.K. Reduction of oxide minerals by hydrogen plasma: An overview. Plasma Chem. Plasma Process. 2014, 34, 1–23. [Google Scholar] [CrossRef]
- Gilles, H.L.; Clump, C.W. Reduction of iron ore with hydrogen in a direct current plasma jet. Ind. Eng. Chem. Process Des. Dev. 1970, 9, 194–207. [Google Scholar] [CrossRef]
- Souza, F.I.R.; Ma, Y.; Kulse, M.; Ponge, D.; Gault, B.; Springer, H.; Raabe, D. Sustainable steel through hydrogen plasma reduction of iron ore: Process, kinetics, microstructure, Chemistry. Acta Mater. 2021, 213, 116971. [Google Scholar] [CrossRef]
- Naseri, S.M.; Schenk, J.; Spreitzer, D.; Andreas Zarl, M. Slag Formation during Reduction of Iron Oxide Using Hydrogen Plasma Smelting Reduction. Materials 2020, 13, 935. [Google Scholar] [CrossRef]
- Kamiya, K.; Kitahara, N.; Morinaka, I.; Sakuraya, K.; Ozawa, M.; Tanaka, M. Reduction of molten iron oxide and FeO bearing slags by H2-Ar plasma. Trans. Iron Steel Inst. Jpn. 1984, 24, 7–16. [Google Scholar] [CrossRef]
- Ban-Ya, S.; Iguchi, Y.; Nagasaka, T. Rate of Reduction of Liquid Iron Oxide with Hydrogen. Trans. Iron Steel Inst. Jpn. 1982, 23, b-197. [Google Scholar]
- Kaneko, K.; Sano, N.; Matsushita, Y. Decarburization and Denitrogenization of Iron and Iron-Chromium Alloys by Plasma Jet of Hydrogen-Argon Gas Mixture. Tetsu Hagane 1976, 62, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Lemperle, M.; Weigel, A. On the smelting reduction of iron ores with hydrogen-argon plasma. Steel Res. 1985, 56, 465–469. [Google Scholar] [CrossRef]
- Hayashi, S.; Iguchi, Y. Hydrogen reduction of liquid iron oxide fines in gas-conveyed systems. ISIJ Int. 1994, 34, 555–561. [Google Scholar] [CrossRef]












| Fetot | Si | Al | Ti | Mg | V | Mn | Ca | Ni | P | S | Zn | O |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 69.35 | 0.84 | 0.41 | 0.34 | 0.27 | 0.13 | 0.12 | 0.12 | 0.02 | 0.02 | 0.02 | 0.01 | 28.35 |
| Group | Gas Composition | Ar Flow Rate (L min−1) | H2 Flow Rate (L min−1) | Reduction Time (min) |
|---|---|---|---|---|
| 1 | Ar + 15%H2 | 17 | 3 | 10, 20, 30, 40 |
| 2 | Ar + 15%H2 | 25.5 | 4.5 | 10, 20, 30, 40 |
| Point | wt/% | |||||
|---|---|---|---|---|---|---|
| Fe | O | Si | Al | Mg | Ca | |
| Point 1 | 51.16 | 34.50 | 6.15 | 4.13 | 1.00 | 3.06 |
| Point 2 | 81.18 | 17.97 | 0.12 | 0.61 | 0.10 | 0.01 |
| Point 3 | 99.62 | 0.32 | 0.04 | 0.01 | 0.00 | 0.01 |
| Point 4 | 99.85 | 0.12 | 0.03 | 0.00 | 0.00 | 0.00 |
| Point | wt/% | |||||
|---|---|---|---|---|---|---|
| Fe | O | Si | Al | Mg | Ca | |
| Point 1 | 54.79 | 22.16 | 12.65 | 8.37 | 1.09 | 0.84 |
| Point 2 | 85.54 | 13.71 | 0.36 | 0.40 | 0.16 | 0.03 |
| Point 3 | 99.87 | 0.36 | 0.07 | 0.00 | 0.00 | 0.00 |
| Gas Flow Rate (L min−1) | Maximum Conversion Rate (%) | Deoxygenation Rate per Area (g (cm2 min)−1) |
|---|---|---|
| 17Ar + 3H2 | 82.67% | 0.87 |
| 25.5Ar + 4.5H2 | 99.89% | 1.11 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Fan, Z.; Zhang, X.; Geng, C.; Jin, X.; Li, L.; Zhao, P.; Wen, B.; Yang, J. Research on the Mechanism of Hydrogen Plasma Heating and Reduction of Acidic Pellets. Materials 2026, 19, 1269. https://doi.org/10.3390/ma19061269
Fan Z, Zhang X, Geng C, Jin X, Li L, Zhao P, Wen B, Yang J. Research on the Mechanism of Hydrogen Plasma Heating and Reduction of Acidic Pellets. Materials. 2026; 19(6):1269. https://doi.org/10.3390/ma19061269
Chicago/Turabian StyleFan, Zihao, Xiaoping Zhang, Chuanwen Geng, Xingyue Jin, Lin Li, Peng Zhao, Baoliang Wen, and Jialong Yang. 2026. "Research on the Mechanism of Hydrogen Plasma Heating and Reduction of Acidic Pellets" Materials 19, no. 6: 1269. https://doi.org/10.3390/ma19061269
APA StyleFan, Z., Zhang, X., Geng, C., Jin, X., Li, L., Zhao, P., Wen, B., & Yang, J. (2026). Research on the Mechanism of Hydrogen Plasma Heating and Reduction of Acidic Pellets. Materials, 19(6), 1269. https://doi.org/10.3390/ma19061269





