Anode-Less (Anode-Free) Batteries: From Fundamental Principles to Practical Pathways Toward Solid-State Implementation
Highlights
- A comprehensive review on anode-less battery design for metal-ion systems.
- Detailed analysis of electrolyte design (liquid and solid) and interface engineering.
- Critical assessment of advanced operando characterization techniques for anode-less cells.
- Identifies plating/stripping efficiency and dendrite growth as key challenges.
- Solid-state electrolytes offer a promising path for safer, high-energy anode-less cells.
- Provides a roadmap for developing next-generation, high-energy-density batteries.
- Highlights the critical importance of current collector and interlayer functionalization.
- Underlines the necessity for standardized performance evaluation protocols.
- Bridges fundamental material insights with industrial scale-up requirements.
- Positions the anode-less architecture as a viable strategy for sustainable batteries.
Abstract
1. Introduction
2. Anode-Less Cells
2.1. Concept of Anode-Less Cells
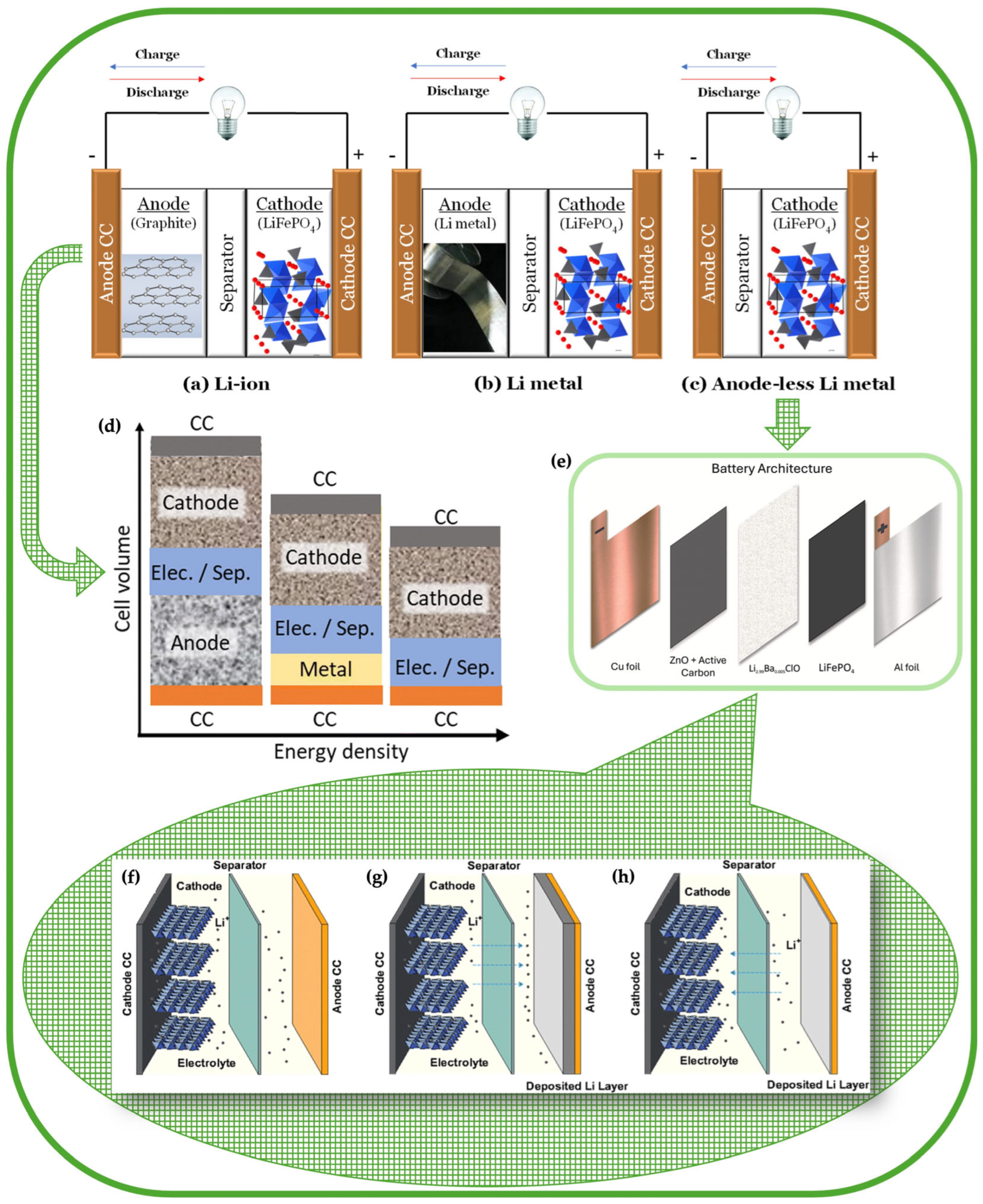
2.2. Advantages and Challenges
2.3. Characterization and Analysis Techniques
2.3.1. Electrochemical Analyses: Charge–Discharge Cycling, PEIS, CV, GITT
2.3.2. Interfacial Potential Mapping: SKP, KPFM
2.3.3. Structural and Chemical Analyses: XRD, XPS, ToF-SIMS, TEM, LIBS, Operando Techniques
2.3.4. Morphological Studies: SEM–EDX, AFM, in Operando Tomography
2.3.5. Thermal Analyses: In Operando Calorimetry, DSC, ARC
3. Anode-Less with Different Electrolytes
3.1. With Liquid Electrolytes
3.1.1. Current Collector and Interface Engineering
3.1.2. Electrolyte Design and Optimization
3.1.3. In Situ Strategies and Nucleation
3.1.4. Advanced Morphologies and Deposition Control
3.2. With Solid Electrolytes
3.2.1. Fundamentals of Anode-Less Operation in Solid-State Cells
3.2.2. Influence of Electrolyte Family and Chemistry
Inorganic Solid Electrolytes
Solid Polymer Electrolytes
Hybrid and Solid Composite Electrolytes
3.2.3. Interface and Interlayer Engineering
Fundamental Interfacial Instabilities and Design Principles
Metal-Based and Lithiophilic Interlayers
Composite and Artificial Interlayers
Collector Surface Modification and Morphology Control
Extension of Interfacial Design Principles Across Alkali-Metal Systems (Li, Na, K)
3.2.4. Characterization and in Operando and Postmortem Studies in Solid ALMB
3.2.5. Beyond Li: Na and K Solid-State ALMB Systems
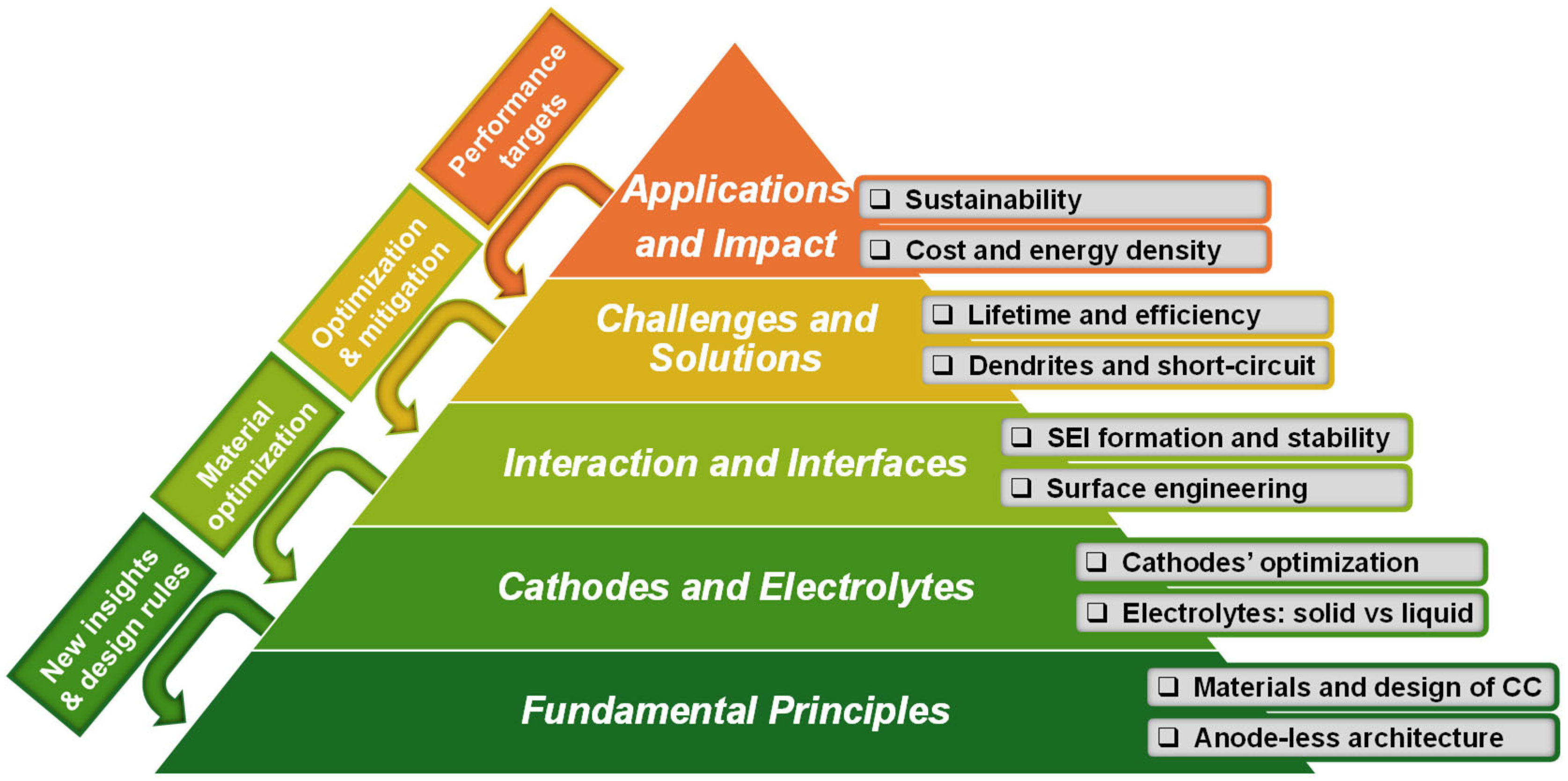
4. Outlook and Perspective: From Lab Bench to Industrial Reality
- Emerging pathway: The research focus has shifted from seeking pure ionic conductors to developing interface stabilizers. Promising strategies include designing compliant interlayers (e.g., soft polymers, lithiophilic alloys) between the solid electrolyte and current collector and creating hybrid or composite electrolytes that combine ceramic mechanical stability with polymer interfacial wettability. Furthermore, artificial SEI engineering via pretreatment or electrolyte additives aims to create a robust interface from the first cycle.
- Emerging pathway: Passive coatings are evolving into active, multifunctional 3D hosts. These structures are engineered with lithiophilic nucleation sites (e.g., Ag nanoparticles, N-doped carbon) to guide uniform plating, while being sodiophilic or lithiophobic in other regions to confine deposition and prevent top-growth, effectively lowering local current density.
- Emerging pathway: The “one-size-fits-all” constant current charge is being replaced by adaptive formation protocols. This involves using low initial currents to build a stable SEI, followed by multistep or pulse plating techniques to refine deposition morphology and maximize initial coulombic efficiency.
- Emerging pathway: For high energy density, high-nickel layered oxides (NMC) and sulfur cathodes are primary candidates, often requiring protective coatings (e.g., LiNbO3) for compatibility with solid electrolytes. For sustainability, manganese-rich layered oxides and iron-based polyanion compounds offer promising, earth-abundant alternatives.
- Emerging pathway: A major focus is reducing the external pressure requirement for solid-state cells by developing mechanically robust, self-standing electrolyte membranes. For temperature, research is directed at wide-temperature-range electrolytes and integrating efficient internal heating elements to ensure operation in subzero conditions.
- Emerging pathway: Advanced battery management system (BMS) algorithms are being researched to incorporate anode potential sensing or impedance tracking. This enables dynamic protocols that adjust based on the cell’s state of health, actively managing the metal inventory and mitigating side reactions over time.
- Emerging pathway: The development of in situ and operando techniques (e.g., ultrasonic imaging, pressure cell monitoring) is vital to observe degradation in real time. These data are essential for creating digital twins of the cell, which can predict failure and inform both cell redesign and BMS strategies.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AFM | atomic force microscopy |
| ALBs | anode-less batteries |
| ALMBs | anode-less lithium-metal batteries |
| ALSSBs | anode-less solid-state batteries |
| AM | additive manufacturing |
| ARC | accelerating rate calorimetry |
| BMS | battery management system |
| CC | current collector |
| CE | coulombic efficiency |
| CPD | contact potential difference |
| CV | cyclic voltammetry |
| DFAM | design for additive manufacturing |
| DFT | density functional theory |
| DMC | dimethyl carbonate |
| DME | dimethoxyethane |
| DSC | differential scanning calorimetry |
| EC | ethylene carbonate |
| EDX | energy-dispersive X-ray spectroscopy |
| FFF | fused filament fabrication |
| GITT | galvanostatic intermittent titration technique |
| HG | holey graphene |
| KPFM | Kelvin probe force microscopy |
| LFP | lithium iron phosphate (LiFePO4) |
| LIBS | laser-induced breakdown spectroscopy |
| LIPON | lithium phosphorus oxynitride |
| LLZO | lithium lanthanum zirconium oxide (Li7La3Zr2O12) |
| LPSCl | lithium phosphorus sulfur chloride (Li6PS5Cl) |
| MBs | metal batteries |
| ML | machine learning |
| NMC111 | nickel manganese cobalt oxide (LiNi0.33Mn0.33Co0.33O2) |
| NMC532 | nickel manganese cobalt oxide (LiNi0.5Mn0.3Co0.2O2) |
| NMC622 | nickel manganese cobalt oxide (LiNi0.6Mn0.2Co0.2O2) |
| NMC811 | nickel manganese cobalt oxide (LiNi0.8Mn0.1Co0.1O2) |
| PBAs | Prussian blue analogues |
| PDMS | polydimethylsiloxane |
| PEIS | potentiostatic electrochemical impedance spectroscopy |
| PEO | polyethylene oxide |
| PVDF | polyvinylidene fluoride |
| SEI | solid-electrolyte interphase |
| SEM | scanning electron microscopy |
| SHE | standard hydrogen electrode |
| SKP | scanning Kelvin probe |
| SS | stainless steel |
| SSE | solid-state electrolyte |
| SUS | sustained discharge |
| TEM | transmission electron microscopy |
| TMSP | trimethylsilyl phosphate |
| ToF-SIMS | time-of-flight secondary ion mass spectrometry |
| XPS | X-ray photoelectron spectroscopy |
| XRD | X-ray diffraction |
References
- Li, P.; Kim, H.; Ming, J.; Jung, H.-G.; Belharouak, I.; Sun, Y.-K. Quasi-Compensatory Effect in Emerging Anode-Free Lithium Batteries. eScience 2021, 1, 3–12. [Google Scholar] [CrossRef]
- Tian, Y.; An, Y.; Wei, C.; Jiang, H.; Xiong, S.; Feng, J.; Qian, Y. Recently Advances and Perspectives of Anode-Free Rechargeable Batteries. Nano Energy 2020, 78, 105344. [Google Scholar] [CrossRef]
- Tang, Z.; Han, C.; Li, W. Anode-Free Solid-State Rechargeable Batteries: Mechanisms, Challenges, and Design Strategies. Batter. Supercaps 2025, 8, e202400585. [Google Scholar] [CrossRef]
- Wang, J.; Zhu, H. Sulfide-Based Anode-Free Solid-State Batteries: Key Challenges and Emerging Solutions. ACS Energy Lett. 2025, 10, 2377–2391. [Google Scholar] [CrossRef] [PubMed]
- Lal, M.S.; Albertus, P.; Noked, M. Anode-Less Sulfide-Based All-Solid-State Batteries: Interfacial Challenges, Material Strategies, and Future Prospects. Small 2025, 21, e10624. [Google Scholar] [CrossRef]
- Jeon, S.; Kim, U.; Kwag, S.H.; Min, J.; Kim, D.; Kim, H.; Lee, Y.; Jung, Y. All-Solid-State Batteries with Anodeless Electrodes: Research Trend and Future Perspective. Adv. Mater. Interfaces 2025, 12, 2400953. [Google Scholar] [CrossRef]
- Sharma, V.; Singh, K.; Krishnamurthy, N. A Review on Conventional to Bipolar Design of Anode Less All Solid-State Batteries. Energy Adv. 2024, 3, 1222–1237. [Google Scholar] [CrossRef]
- Molaiyan, P.; Abdollahifar, M.; Boz, B.; Beutl, A.; Krammer, M.; Zhang, N.; Tron, A.; Romio, M.; Ricci, M.; Adelung, R. Optimizing Current Collector Interfaces for Efficient “Anode-free” Lithium Metal Batteries. Adv. Funct. Mater. 2024, 34, 2311301. [Google Scholar] [CrossRef]
- Wang, C.; Kim, J.T.; Wang, C.; Sun, X. Progress and Prospects of Inorganic Solid-state Electrolyte-based All-solid-state Pouch Cells. Adv. Mater. 2023, 35, 2209074. [Google Scholar] [CrossRef]
- He, P.; Xavier, N.F., Jr.; Golomb, M.J.; Han, Y.; Du, Y.; Cai, Q.; Xu, Y. Toward Zero-Excess Alkali Metal Batteries: Bridging Experimental and Computational Insights. Adv. Mater. 2025, 37, 2502052. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Ganapathy, S.; Wagemaker, M. A Direct View on Li-Ion Transport and Li-Metal Plating in Inorganic and Hybrid Solid-State Electrolytes. Acc. Chem. Res. 2022, 55, 333–344. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Liu, L.; Wang, X.; Zheng, Q.; Han, C.; Li, W. Current Progress of Anode-free Rechargeable Sodium Metal Batteries: Origin, Challenges, Strategies, and Perspectives. Adv. Funct. Mater. 2024, 34, 2313823. [Google Scholar] [CrossRef]
- Skundin, A.M.; Kulova, T.L. All-Solid-State Anode-Free Sodium Batteries: Challenges and Prospects. Batteries 2025, 11, 292. [Google Scholar] [CrossRef]
- Yang, T.; Luo, D.; Liu, Y.; Yu, A.; Chen, Z. Anode-Free Sodium Metal Batteries as Rising Stars for Lithium-Ion Alternatives. iScience 2023, 26, 105982. [Google Scholar] [CrossRef]
- Dong, Y.; Xu, C.; Zhao, H.; Chen, L.; Shi, W.; Irvine, J.; Lei, Y. Interface Engineering Strategies for Realizing Anode-Free Sodium Batteries: A Review. Adv. Energy Mater. 2025, 16, 2500407. [Google Scholar] [CrossRef]
- Wang, J.; Zhou, Y.; Zhuo, Y.; Fang, K.; Wang, S.; Zhao, B.; Zhou, J.; Wang, H. The Challenges and Strategies towards High-Performance Anode-Free Post-Lithium Metal Batteries. Chem. Sci. 2025, 16, 552–574. [Google Scholar] [CrossRef]
- Petersen, D.; Gronenberg, M.; Lener, G.; Leiva, E.P.M.; Luque, G.L.; Rostami, S.; Paolella, A.; Hwang, B.J.; Adelung, R.; Abdollahifar, M. Anode-Free Post-Li Metal Batteries. Mater. Horiz. 2024, 11, 5914–5945. [Google Scholar] [CrossRef]
- Zhou, Y.; Wang, P.; Wang, K.; Fang, X.; Li, W.; Nai, J.; Liu, Y.; Wang, Y.; Zou, S.; Yuan, H. Developing High-Performance Anode-Free Lithium Batteries: Challenges, Strategies, and Opportunities. Adv. Funct. Mater. 2025, 35, 2424022. [Google Scholar] [CrossRef]
- Menkin, S.; Ayerbe, E.; Gunnarsdóttir, A.B.; del Olmo, D.; López-Aranguren, P.; Rafique, A.; Rikarte, J.; Chauque, S.; Shi, X.; Guillon, O.; et al. Anode-Free Cell Concepts: Critical Analysis and Development of Practical Batteries. Small 2026, e13633. [Google Scholar] [CrossRef] [PubMed]
- Nikodimos, Y.; Shitaw, K.N.; Hagos, T.M.; Yeh, T.-I.; Huang, Y.-C.; Hsieh, C.-L.; Su, H.-H.; Su, W.-N.; Hwang, B.J. Anode-Free Batteries: Pioneering Energy Storage Revolution. Chem. Rev. 2026, 126, 2391–2483. [Google Scholar] [CrossRef]
- Vishweswariah, K.; Ningappa, N.G.; Bouguern, M.D.; Kumar MR, A.; Armand, M.B.; Zaghib, K. Evaluation and Characterization of SEI Composition in Lithium Metal and Anode-Free Lithium Batteries. Adv. Energy Mater. 2025, 15, 2501883. [Google Scholar] [CrossRef]
- Duan, X.; Sun, J.; Shi, L.; Dong, S.; Cui, G. Exploring the Active Lithium Loss in Anode-Free Lithium Metal Batteries: Mechanisms, Challenges, and Strategies. Interdiscip. Mater. 2025, 4, 217–234. [Google Scholar] [CrossRef]
- Qian, J.; Adams, B.D.; Zheng, J.; Xu, W.; Henderson, W.A.; Wang, J.; Bowden, M.E.; Xu, S.; Hu, J.; Zhang, J.-G.; et al. Anode-Free Rechargeable Lithium Metal Batteries. Adv. Funct. Mater. 2016, 26, 7094–7102. [Google Scholar] [CrossRef]
- Braga, M.H.; Murchison, A.J.; Ferreira, J.A.; Singh, P.; Goodenough, J.B. Glass-Amorphous Alkali-Ion Solid Electrolytes and Their Performance in Symmetrical Cells. Energy Environ. Sci. 2016, 9, 948–954. [Google Scholar] [CrossRef]
- Naikwade, M.B.; Katkar, P.K.; Lee, S.-W. Understanding the Impact of Porosity on Li-Ion Diffusion Enhancement in Micro-Sized Silicon Particles for Advanced Batteries. Ceram. Int. 2024, 50, 54778–54790. [Google Scholar] [CrossRef]
- Baptista, M.C.; Gomes, B.M.; Capela, D.; Ferreira, M.F.S.; Guimarães, D.; Silva, N.A.; Jorge, P.A.S.; Silva, J.J.; Braga, M.H. Conditioning Solid-State Anode-Less Cells for the Next Generation of Batteries. Batteries 2023, 9, 402. [Google Scholar] [CrossRef]
- Zhang, J.G. Anode-Less. Nat. Energy 2019, 4, 637–638. [Google Scholar] [CrossRef]
- Chavan, V.D.; Patil, P.D.; Patil, C.S.; Patil, S.R.; Katkar, P.K.; Sheikh, Z.A.; Ustad, R.E.; Kim, H.; Kadam, K.D.; Patil, H.S.; et al. Advanced Functional Materials and Their Coordinated Composites for Next-Generation Li-S Batteries: A Brief Review. J. Energy Storage 2024, 88, 111572. [Google Scholar] [CrossRef]
- Danzi, F.; Valente, M.; Terlicka, S.; Braga, M.H. Sodium and Potassium Ion Rich Ferroelectric Solid Electrolytes for Traditional and Electrode-Less Structural Batteries. APL Mater. 2022, 10, 031111. [Google Scholar] [CrossRef]
- Tang, K.; Tian, L.; Zhang, Y.; Xu, Z.J. Anode-Free Lithium Metal Batteries: A Promising Flexible Energy Storage System. J. Mater. Chem. A Mater. 2024, 12, 16268–16292. [Google Scholar] [CrossRef]
- Capela, D.; Baptista, M.C.; Gomes, B.M.; Jorge, P.A.S.; Silva, N.A.; Braga, M.H.; Guimarães, D. Laser-Induced Breakdown Spectroscopy for Surface Analysis of Solid-State Anode-Less Battery. J. Power Sources 2025, 647, 237328. [Google Scholar] [CrossRef]
- Wang, D.; Qiu, J.; Inui, N.; Hagiwara, R.; Hwang, J.; Matsumoto, K. Between Promise and Practice: A Comparative Look at the Energy Density of Li Metal-Free Batteries and Li Metal Batteries. ACS Energy Lett. 2023, 8, 5248–5252. [Google Scholar] [CrossRef]
- Finkelstein, S.H.; Ricci, M.; Bötticher, T.; Schmidt-Rohr, K. How Lithium-Ion Batteries Work Conceptually: Thermodynamics of Li Bonding in Idealized Electrodes. Phys. Chem. Chem. Phys. 2024, 26, 24157–24171. [Google Scholar] [CrossRef]
- Braga, M.H.; Grundish, N.S.; Murchison, A.J.; Goodenough, J.B. Thermodynamic Considerations of Same-Metal Electrodes in an Asymmetric Cell. Mater. Theory 2019, 3, 1. [Google Scholar] [CrossRef]
- Gomes, B.M.; Moutinho, J.F.R.; Braga, M.H. A Perspective on the Building Blocks of a Solid-State Battery: From Solid Electrolytes to Quantum Power Harvesting and Storage. J. Mater. Chem. A Mater. 2024, 12, 690–722. [Google Scholar] [CrossRef]
- Aydinol, M.K.; Ceder, G. First-principles Prediction of Insertion Potentials in Li-Mn Oxides for Secondary Li Batteries. J. Electrochem. Soc. 1997, 144, 3832. [Google Scholar] [CrossRef]
- Kim, D.K.; Muralidharan, P.; Lee, H.-W.; Ruffo, R.; Yang, Y.; Chan, C.K.; Peng, H.; Huggins, R.A.; Cui, Y. Spinel LiMn2O4 Nanorods as Lithium Ion Battery Cathodes. Nano Lett. 2008, 8, 3948–3952. [Google Scholar] [CrossRef] [PubMed]
- Baptista, M.C.; Gomes, B.M.; Vale, A.B.; Braga, M.H. In-Series All-Solid-State Anode-Less Cells. J. Energy Storage 2024, 102, 113983. [Google Scholar] [CrossRef]
- Oh, J.; Sohn, Y.; Choi, J.W. High-Performance Anode-Less All-Solid-State Batteries Enabled by Multisite Nucleation and an Elastic Network. EES Batter. 2025, 1, 566–575. [Google Scholar] [CrossRef]
- Sohn, Y.; Oh, J.; Lee, J.; Kim, H.; Hwang, I.; Noh, G.; Lee, T.; Kim, J.Y.; Bae, K.Y.; Lee, T. Dual-Seed Strategy for High-Performance Anode-Less All-Solid-State Batteries. Adv. Mater. 2024, 36, 2407443. [Google Scholar] [CrossRef]
- Tao, J.; Zhang, C.; Li, X.; Chen, X.; Ji, C.; Wan, W.; Wang, C. Advancing Anode-Less Lithium Metal Batteries: ZnF2 Modification and in Situ Structural Regulation for Enhanced Performance. J. Mater. Chem. A Mater. 2024, 12, 18127–18136. [Google Scholar] [CrossRef]
- Yang, X.; Hao, J.; Zhao, Y.; Chen, J.; Guo, J.; Chi, C. Synthesis 3D Copper Foam Current Collector with Lithiophilicity Cu2O and LiF-Rich Interface Layer for Anode-Free Lithium Metal Batteries. J. Energy Storage 2025, 135, 118367. [Google Scholar] [CrossRef]
- Salvatierra, R.V.; Chen, W.; Tour, J.M. What Can Be Expected from “Anode-Free” Lithium Metal Batteries? Adv. Energy Sustain. Res. 2021, 2, 2000110. [Google Scholar] [CrossRef]
- Zhang, J.; Khan, A.; Liu, X.; Lei, Y.; Du, S.; Lv, L.; Zhao, H.; Luo, D. Research Progress of Anode-Free Lithium Metal Batteries. Crystals 2022, 12, 1241. [Google Scholar] [CrossRef]
- Li, S.; Wu, F.; Chen, T.; Kang, K.; Guo, R.; Liu, C.; Niu, Y.; Gao, A.; Zhao, R.; Wang, X. Progress and Challenges for Energy-Dense and Cost-Effective Anode-Free Lithium Metal Batteries. Energy Mater. Adv. 2025, 6, 0168. [Google Scholar] [CrossRef]
- Tong, Z.; Bazri, B.; Hu, S.-F.; Liu, R.-S. Interfacial Chemistry in Anode-Free Batteries: Challenges and Strategies. J. Mater. Chem. A Mater. 2021, 9, 7396. [Google Scholar] [CrossRef]
- Acebedo, B.; Cid, R.; Villaverde, A.; Armand, M.; Ruiz De Larramendi, I.; Gonzalo, E.; Devaraj, S. Enabling Anode-Less Lithium Batteries via Sacrificial Salts: An In-Depth Feasibility Analysis. ACS Appl. Mater. Interfaces 2025, 17, 49573–49583. [Google Scholar] [CrossRef]
- Cheng, L.; Liu, J.; Wang, H.; Guo, Y.; Shao, A.; Li, Y.; Wang, Z.; Zhang, Y.; Tang, J.; Li, C. A Lightweight, Li Supplementary and Lithiophilic Interface Enables Anode-Less Lithium Metal Battery Prototyping. EES Batter. 2025, 1, 555–565. [Google Scholar] [CrossRef]
- Oh, J.; Choi, S.H.; Kim, J.Y.; Lee, J.; Lee, T.; Lee, N.; Lee, T.; Sohn, Y.; Chung, W.J.; Bae, K.Y. Anode-less All-solid-state Batteries Operating at Room Temperature and Low Pressure. Adv. Energy Mater. 2023, 13, 2301508. [Google Scholar] [CrossRef]
- Zhan, J.; Deng, L.; Liu, Y.; Hao, M.; Wang, Z.; Dong, L.; Yang, Y.; Song, K.; Qi, D.; Wang, J. Self-Selective (220) Directional Grown Copper Current Collector Design for Cycling-Stable Anode-Less Lithium Metal Batteries. Adv. Mater. 2025, 37, 2413420. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Dong, H.; Katyal, N.; Hao, H.; Liu, P.; Celio, H.; Henkelman, G.; Watt, J.; Mitlin, D. A Sodium–Antimony–Telluride Intermetallic Allows Sodium-metal Cycling at 100% Depth of Discharge and as an Anode-free Metal Battery. Adv. Mater. 2022, 34, 2106005. [Google Scholar] [CrossRef]
- Kim, J.; Lee, S.; Kim, S.; Park, C.-Y.; Kim, J.; Lee, K.B.; Oh, J.; Lee, J. Anode-Less Hybrid Na–CO2 Battery with Sodium Harvesting from Seawater for Both Electricity Storage and Various Chemical Production. ACS Energy Lett. 2023, 8, 5079–5087. [Google Scholar] [CrossRef]
- Menkin, S.; O’Keefe, C.A.; Gunnarsdóttir, A.B.; Dey, S.; Pesci, F.M.; Shen, Z.; Aguadero, A.; Grey, C.P. Toward an Understanding of SEI Formation and Lithium Plating on Copper in Anode-Free Batteries. J. Phys. Chem. C 2021, 125, 16719–16732. [Google Scholar] [CrossRef]
- Jung, H.; Jung, H.-T. Highly Ordered and High-Aspect-Ratio Au Nanopatterns Directly Fabricated on Cu for Efficient Anode-Less Li Metal Batteries. J. Mater. Chem. A Mater. 2025, 13, 18766–18775. [Google Scholar] [CrossRef]
- Oh, J.; Kim, J.; Bae, S.; Kim, H.; Lee, T.; Choi, S.H.; Park, G.; Ji, S.; Kim, K.J.; Choi, J.W. Comprehensive Analysis of Anode-Less Batteries with Lithiophilic Seeds in Liquid and Solid-State Electrolytes. Adv. Energy Mater. 2025, 15, e02711. [Google Scholar] [CrossRef]
- Zhou, H.; Wan, L.; Han, J.; Wu, J.-C.; Deng, Y.; Gu, J.; Wang, H.; Gao, H.; Su, Z. Dense Integration of Chlorocatechols Crosslinked Polyphenylene Sulfide Solid-State Separator for Li Metal-Free Batteries. Chem. Eng. J. Adv. 2025, 21, 100694. [Google Scholar] [CrossRef]
- Oh, J.; Choi, S.H.; Kim, H.; Kim, J.Y.; Lee, G.-J.; Bae, K.Y.; Lee, T.; Lee, N.; Sohn, Y.; Chung, W.J. Lithio-Amphiphilic Nanobilayer for High Energy Density Anode-Less All-Solid-State Batteries Operating under Low Stack Pressure. Energy Environ. Sci. 2024, 17, 7932–7943. [Google Scholar] [CrossRef]
- Oh, J.; Lee, T.; Lee, N.; Sohn, Y.; Kim, J.Y.; Bae, K.Y.; Choi, S.H.; Choi, J.W. Kinetically Engineered Lithiophilic Dual-Metal Layers for Dendrite-Free, High-Energy Anode-Less All-Solid-State Batteries. Battery Energy 2025, 4, e70038. [Google Scholar] [CrossRef]
- Nwaogu, E.C.; Bakenov, Z.; Nurpeissova, A. Three-Dimensional Copper Current Collector as an Anode for Rechargeable Anode-Less Lithium-Ion Batteries. Int. J. Electrochem. Sci. 2024, 19, 100463. [Google Scholar] [CrossRef]
- Bai, M.; Zhong, M.; Shen, W.; Zhang, J.; Guo, S. Free-Standing Graphene Films Decorated with Lithiophilic Particles as Host Electrodes for Lithium Plating/Stripping in Anode-Less Lithium Metal Batteries. J. Mater. Chem. A Mater. 2025, 13, 8865–8875. [Google Scholar] [CrossRef]
- Ping Zheng, Y.; Hu, X.J.; Xu, X.M.; Wang, H.F.; Sun, Y.L.; Liu, T.; Bin Liu, X.; Wu, Z.P.; Xia, B.Y. Lithiated Graphene Current Collector for Stable Anode-Free and Anode-Less Lithium Metal Batteries. Batter. Supercaps 2024, 7, e202400144. [Google Scholar] [CrossRef]
- Zhuang, R.; Zhang, X.; Qu, C.; Xu, X.; Yang, J.; Ye, Q.; Liu, Z.; Kaskel, S.; Xu, F.; Wang, H. Fluorinated Porous Frameworks Enable Robust Anode-Less Sodium Metal Batteries. Sci. Adv. 2023, 9, eadh8060. [Google Scholar] [CrossRef]
- Kang, S.; Geng, F.; Wu, X.; Fan, Y.; Li, C.; Shen, M.; Lou, X.; Hu, B. Bilayer-Separator Strategy Facilitates Uniform and Dense Na Deposition for Stable Anode-Less Sodium Metal Batteries Revealed by EPR Imaging. ACS Energy Lett. 2025, 10, 3876–3886. [Google Scholar] [CrossRef]
- Sandoval, S.E.; Haslam, C.G.; Vishnugopi, B.S.; Liao, D.W.; Yoon, J.S.; Park, S.H.; Wang, Y.; Mitlin, D.; Hatzell, K.B.; Siegel, D.J. Electro-Chemo-Mechanics of Anode-Free Solid-State Batteries. Nat. Mater. 2025, 24, 673–681. [Google Scholar] [CrossRef] [PubMed]
- Sreedeep, S.; Lee, Y.-S.; Aravindan, V. In Situ Transformation of a Liquid Electrolyte into a Solid Polymer Electrolyte: Influence of TMSP in a Layered LiNi0.82Mn0.12Al0.06O2 Cathode. J. Mater. Chem. A Mater. 2025, 13, 13262–13275. [Google Scholar] [CrossRef]
- Freitas, Â.; Baptista, M.C.; Braga, M.H. Sustainable Solid-State Sodium-Ion Batteries Featuring Ferroelectric Electrolytes. Int. J. Mol. Sci. 2024, 25, 12694. [Google Scholar] [CrossRef]
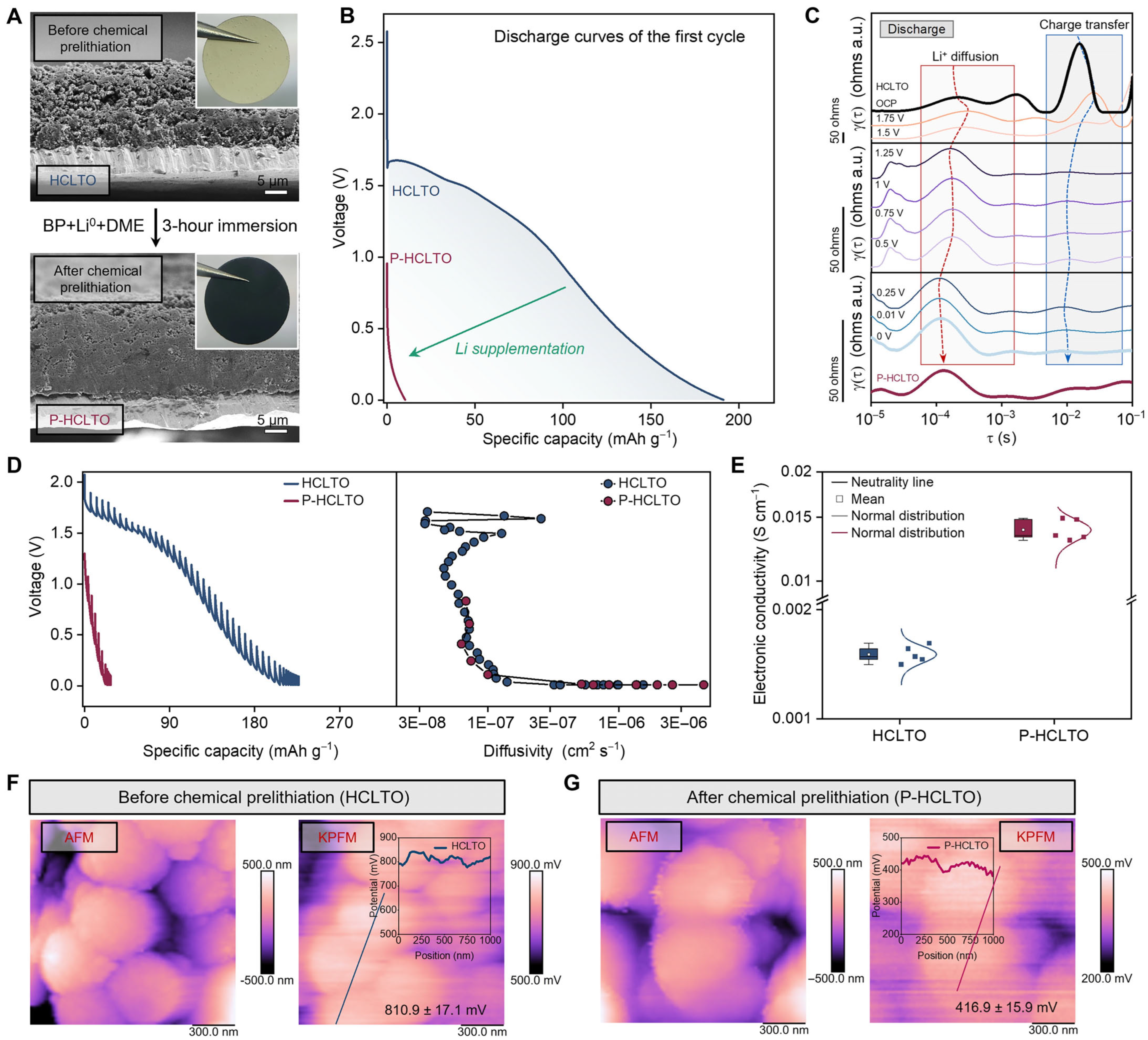
- Shao, A.; Wang, H.; Zhang, M.; Liu, J.; Cheng, L.; Li, Y.; Guo, Y.; Wang, Z.; Jia, Q.; Wang, X.; et al. Multiscale interfacial stabilization via prelithiation separator engineering for Ah-level anode-free lithium batteries. Nat. Commun. 2025, 16, 4145. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Tang, Y.; Wei, Y.; Li, H.; Yan, J.; Feng, Z.; Du, W.; Yang, Q.; Ye, M.; Zhang, Y. Hydrophobic Ion Barrier-Enabled Ultradurable Zn (002) Plane Orientation towards Long-Life Anode-Less Zn Batteries. Angew. Chem. Int. Ed. 2024, 63, e202407639. [Google Scholar] [CrossRef]
- Peng, L.; Wang, X.; Liu, X.; Xi, Z.; Li, Y.; Zhang, J.; Ning, Y.; Zhao, Q. Manipulating Metal Growth in Hollow Ionic-Electronic Conductor for Anode-Free Lithium Metal Batteries. Sci. Adv. 2026, 12, eadt2630. [Google Scholar] [CrossRef] [PubMed]
- Guerreiro, A.N.; Baptista, M.C.; Maia, B.A.; Braga, M.H. Interfacial Chemistry with ZnO: In Operando Work Functions in Hetero Cells. ACS Appl. Energy Mater. 2022, 5, 9811–9822. [Google Scholar] [CrossRef]
- Guerreiro, A.N.; Maia, B.A.; Khalifa, H.; Baptista, M.C.; Braga, M.H. What Differentiates Dielectric Oxides and Solid Electrolytes on the Pathway toward More Efficient Energy Storage? Batteries 2022, 8, 232. [Google Scholar] [CrossRef]
- Park, S.K.; Kim, S.; He, R.; Sanders, K.; Hwang, U.; An, Z.; Hamidinejad, M.; Kim, J.; De Volder, M. Synergistic Lithium Alloying and Plating in 3D Cu/CNT/Sn Electrodes for Stable Lithium Metal Batteries. Small 2025, 21, 2501292. [Google Scholar] [CrossRef]
- To-A-Ran, W.; Mastoi, N.R.; Ha, C.Y.; Song, Y.J.; Kim, Y.-J. Kelvin Probe Force Microscopy and Electrochemical Atomic Force Microscopy Investigations of Lithium Nucleation and Growth: Influence of the Electrode Surface Potential. J. Phys. Chem. Lett. 2024, 15, 7265–7271. [Google Scholar] [CrossRef] [PubMed]
- Masuda, H.; Matsushita, K.; Ito, D.; Fujita, D.; Ishida, N. Dynamically Visualizing Battery Reactions by Operando Kelvin Probe Force Microscopy. Commun. Chem. 2019, 2, 140. [Google Scholar] [CrossRef]
- Masuda, H.; Ishida, N.; Ogata, Y.; Ito, D.; Fujita, D. Internal Potential Mapping of Charged Solid-State-Lithium Ion Batteries Using in Situ Kelvin Probe Force Microscopy. Nanoscale 2017, 9, 893–898. [Google Scholar] [CrossRef]
- Zhu, C.; Fuchs, T.; Weber, S.A.L.; Richter, F.H.; Glasser, G.; Weber, F.; Butt, H.-J.; Janek, J.; Berger, R. Understanding the Evolution of Lithium Dendrites at Li6.25Al0.25La3Zr2O12 Grain Boundaries via Operando Microscopy Techniques. Nat. Commun. 2023, 14, 1300. [Google Scholar] [CrossRef] [PubMed]
- Prior, T.; Figueira, J.; Freitas, Â.; Carvalho, D.; Gomes, B.M.; Baptista, M.C.; Lebre, H.; Martins, R.; Pereira, L.; Pinto, J.V.; et al. Surface Morphology and Electrochemical Behavior of Microstructured Cu Electrodes in All-Solid-State Sodium Batteries. Molecules 2025, 30, 3493. [Google Scholar] [CrossRef]
- Wu, X.; Song, B.; Chien, P.; Everett, S.M.; Zhao, K.; Liu, J.; Du, Z. Structural Evolution and Transition Dynamics in Lithium Ion Battery under Fast Charging: An Operando Neutron Diffraction Investigation. Adv. Sci. 2021, 8, 2102318. [Google Scholar] [CrossRef]
- Lee, J.-H.; Heo, J.Y.; Kim, J.Y.; Bae, K.Y.; Son, S.; Lee, J.H. Lithium–Silver Alloys in Anode-Less Batteries: Comparison in Liquid-and Solid-Electrolytes. Chem. Commun. 2024, 60, 8268–8271. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Liu, Y.; Nguyen, M.; Cho, J.; Katyal, N.; Vishnugopi, B.S.; Hao, H.; Fang, R.; Wu, N.; Liu, P. Stable Anode-free All-solid-state Lithium Battery through Tuned Metal Wetting on the Copper Current Collector. Adv. Mater. 2023, 35, 2206762. [Google Scholar] [CrossRef]
- Ge, J.; Ma, C.; Zhang, Y.; Ma, P.; Zhang, J.; Xie, Z.; Wen, L.; Tang, G.; Wang, Q.; Li, W. Edge Electron Effect Induced High-Entropy SEI for Durable Anode-Free Sodium Batteries. Adv. Mater. 2024, 37, 2413253. [Google Scholar] [CrossRef]
- Lee, J.-A.; Kang, H.; Kim, S.; Lee, K.; Byun, J.H.; Kwon, E.; Seo, S.; Kwak, K.; Ryu, K.H.; Choi, N.-S. Unveiling Degradation Mechanisms of Anode-Free Li-Metal Batteries. Energy Storage Mater. 2024, 73, 103826. [Google Scholar] [CrossRef]
- Xie, C.; Zhao, C.; Jeong, H.; Liu, Q.; Li, T.; Xu, W.; Cheng, L.; Xu, G.; Amine, K.; Chen, G. Regulating Li Nucleation and Growth Heterogeneities via Near-Surface Lithium-Ion Irrigation for Stable Anode-Less Lithium Metal Batteries. Small 2024, 20, 2306868. [Google Scholar] [CrossRef]
- Xia, L.; Lin, L.; Li, J.; Zhang, Y.; Zheng, H.; Chang, X.; Liang, J.; Sa, B.; Wang, L.; Lin, J. Lithium-Containing Hybrid SEI Layer Enabling High Mass Loading and Anode-Less Sodium Metal Batteries. Angew. Chem. Int. Ed. 2025, 64, e202423090. [Google Scholar] [CrossRef]
- Dai, J.; Wang, H.; Zhang, R.; Wang, J.; Wang, P.; Qiu, T.; Wang, H. Co-Sputtering Strategy to Construct Robust Sodiophilic Interfaces for Anode-Less Sodium Metal Batteries. Chem. Commun. 2024, 60, 11120–11123. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Liu, Q.; Wang, D.; Shi, J.; Zhai, D.; Zhang, B. Unraveling Electrode Surface Chemistry in Determining Interphase Stability and Deposition Homogeneity for Anode-Free Potassium Metal Batteries. Angew. Chem. Int. Ed. 2025, 64, e202502091. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Yang, Z.; Zhang, X.; Zhao, Y.; Wang, Y.; Chen, J.; Wang, S.; Zhao, Q.; Hou, Z. High Modulus Na2SiO3-Rich Solid Electrolyte Interphase Enable Long-Cycle and Energy-Dense Sodium Metal Battery. Adv. Funct. Mater. 2024, 34, 2407783. [Google Scholar] [CrossRef]
- Ko, D.-S.; Kim, S.; Lee, S.; Yoon, G.; Kim, D.; Shin, C.; Kim, D.; Lee, J.; Sul, S.; Yun, D.-J. Mechanism of Stable Lithium Plating and Stripping in a Metal-Interlayer-Inserted Anode-Less Solid-State Lithium Metal Battery. Nat. Commun. 2025, 16, 1066. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Pei, Z.; Luan, D.; Lou, X.W. Anchoring Sn Nanoparticles in Necklace-Like B, N, F-Doped Carbon Fibers Enables Anode-Less 5V-Class Li-Metal Batteries. Angew. Chem. 2025, 137, e202423454. [Google Scholar] [CrossRef]
- Kwon, H.; Lee, J.-H.; Roh, Y.; Baek, J.; Shin, D.J.; Yoon, J.K.; Ha, H.J.; Kim, J.Y.; Kim, H.-T. An Electron-Deficient Carbon Current Collector for Anode-Free Li-Metal Batteries. Nat. Commun. 2021, 12, 5537. [Google Scholar] [CrossRef]
- Qian, L.; Zheng, Y.; Or, T.; Park, H.W.; Gao, R.; Park, M.; Ma, Q.; Luo, D.; Yu, A.; Chen, Z. Advanced Material Engineering to Tailor Nucleation and Growth towards Uniform Deposition for Anode-Less Lithium Metal Batteries. Small 2022, 18, 2205233. [Google Scholar] [CrossRef]
- Lee, J.; Choi, S.H.; Im, G.; Lee, K.; Lee, T.; Oh, J.; Lee, N.; Kim, H.; Kim, Y.; Lee, S. Room-temperature Anode-less All-solid-state Batteries via the Conversion Reaction of Metal Fluorides. Adv. Mater. 2022, 34, 2203580. [Google Scholar] [CrossRef]
- Li, H.; Zhang, H.; Wu, F.; Zarrabeitia, M.; Geiger, D.; Kaiser, U.; Varzi, A.; Passerini, S. Sodiophilic Current Collectors Based on MOF-derived Nanocomposites for Anode-less Na-metal Batteries. Adv. Energy Mater. 2022, 12, 2202293. [Google Scholar] [CrossRef]
- Koul, S.; Morita, Y.; Fujisaki, F.; Ogasa, H.; Fujiwara, Y.; Kushima, A. Effect of Liquid Metal Coating on Improved Cycle Performance of Anode-Free Lithium Metal Battery. J. Electrochem. Soc. 2022, 169, 020542. [Google Scholar] [CrossRef]
- Lee, N.; Oh, J.; Choi, J.W. Anode-Less All-Solid-State Batteries: Recent Advances and Future Outlook. Mater. Futures 2023, 2, 013502. [Google Scholar] [CrossRef]
- Santagata, A.; Pace, M.L.; Trucchi, D.M.; Curcio, M.; De Bonis, A.; Teghil, R.; Carboni, N.; Gentile, A.; Sette, A.; Mesina, L. Towards Anode-Less Lithium Metal Negative Electrodes for Secondary Aprotic Batteries. Future Batter. 2025, 7, 100095. [Google Scholar] [CrossRef]
- Wang, H.; Yuan, Y.; Jia, Q.; Shao, A.; Zhang, M.; Wang, Z.; Cheng, L.; Tang, X.; Li, S.; Ma, Y. A Thin-Layer, Li+ Compensation, Moisture-Tolerant Interfacial Modification of Cu Substrate Toward the Anode-Less, Energy/Power Dense Li Metallic Batteries. Adv. Funct. Mater. 2024, 34, 2314186. [Google Scholar] [CrossRef]
- Li, S.; Zhu, H.; Liu, Y.; Wu, Q.; Cheng, S.; Xie, J. Space-Confined Guest Synthesis to Fabricate Sn-monodispersed N-doped Mesoporous Host toward Anode-Free Na Batteries. Adv. Mater. 2023, 35, 2301967. [Google Scholar] [CrossRef] [PubMed]
- Ponraj, R.; Dai, X.; Kim, D.G.; Kim, D.K.; Kim, D.J. Enhancing the Cycle Life of Lithium-Anode-Free Batteries through Polydopamine-Coated Substrates. Adv. Energy Sustain. Res. 2023, 4, 2300051. [Google Scholar] [CrossRef]
- Kim, Y.C.; Kulkarni, P.; Kim, S.; Kim, H.; Jung, H.Y. Hybrid Interfacial Modulation for Stabilizing Anode-Less Lithium-Sulfur Batteries: Guided Lithium Nucleation and Polysulfide Regulation. Small Methods 2025, 9, e01183. [Google Scholar] [CrossRef] [PubMed]
- Metzler, M.; Doerrer, C.; Sun, Y.; Matthews, G.; Liotti, E.; Grant, P.S. Effect of Silver Particle Distribution in a Carbon Nanocomposite Interlayer on Lithium Plating in Anode-Free All-Solid-State Batteries. ACS Appl. Mater. Interfaces 2025, 17, 39089–39096. [Google Scholar] [CrossRef]
- Pahari, P.; Ramakrishnan, S. Interfacial Passivation in Anodeless Solid-State Batteries Enabled by a Porous, Li2O-Rich Interphase Generated from a Corrosion-Conversion Reaction. ACS Electrochem. 2025, 1, 2364–2375. [Google Scholar] [CrossRef]
- Kim, Y.; Künzel, M.; Steinle, D.; Dong, X.; Kim, G.-T.; Varzi, A.; Passerini, S. Anode-Less Seawater Batteries with a Na-Ion Conducting Solid-Polymer Electrolyte for Power to Metal and Metal to Power Energy Storage. Energy Environ. Sci. 2022, 15, 2610–2618. [Google Scholar] [CrossRef]
- Garcia-Calvo, O.; Gutiérrez-Pardo, A.; Combarro, I.; Orue, A.; Lopez-Aranguren, P.; Urdampilleta, I.; Kvasha, A. Selection and Surface Modifications of Current Collectors for Anode-Free Polymer-Based Solid-State Batteries. Front. Chem. 2022, 10, 934365. [Google Scholar] [CrossRef]
- Jin, D.; Roh, Y.; Jo, T.; Ryou, M.; Lee, H.; Lee, Y.M. Robust Cycling of Ultrathin Li Metal Enabled by Nitrate-preplanted Li Powder Composite. Adv. Energy Mater. 2021, 11, 2003769. [Google Scholar] [CrossRef]
- Lin, J.; Ma, C.; Li, C.; Zou, X.; Chen, W.; Zhou, J.; Lian, P. In Situ Forming a Black Phosphorene Mixed Ion/Electron Conductor Layer by External Pressure without Binder for Anode-Free Li Metal Batteries. ACS Appl. Mater. Interfaces 2025, 17, 33191–33198. [Google Scholar] [CrossRef]
- Zhou, X.; Zhang, Q.; Hao, Z.; Ma, Y.; Drozhzhin, O.A.; Li, F. Unlocking the Allometric Growth and Dissolution of Zn Anodes at Initial Nucleation and an Early Stage with Atomic Force Microscopy. ACS Appl. Mater. Interfaces 2021, 13, 53227–53234. [Google Scholar] [CrossRef]
- You, X.; Feng, Y.; Ning, D.; Yao, H.; Wang, M.; Wang, J.; Chen, B.; Zhong, G.-H.; Yang, C.; Wu, W. Phosphorized 3D Current Collector for High-Energy Anode-Free Lithium Metal Batteries. Nano Lett. 2024, 24, 11367–11375. [Google Scholar] [CrossRef]
- Srivastava, P.; Hung, Y.-T.; Cheng, C.-Y.; Huang, S.-M.; Wu, Y.-T.; Liu, R.-S. Toward Anode-Less Lithium Metal Batteries Enabled by Garnet-Based Composite Polymer Electrolytes. ACS Mater. Lett. 2025, 7, 3283–3290. [Google Scholar] [CrossRef]
- Shi, J.; Wang, D.; Liu, Q.; Yu, Z.; Huang, J.-Q.; Zhang, B. Intermetallic Layers with Tuned Na Nucleation and Transport for Anode-Free Sodium Metal Batteries. Nano Lett. 2025, 25, 1800–1807. [Google Scholar] [CrossRef]
- Li, N.; Kang, G.; Huang, S.; Biao, J.; Liu, Y.; Kang, F.; Cao, Y. Protonated Polyaniline-Modified Copper Current Collector for Anode-Free Lithium Metal Battery. ACS Appl. Mater. Interfaces 2025, 17, 36627–36638. [Google Scholar] [CrossRef]
- Rafique, A.; Fallarino, L.; Accardo, G.; Pesce, A.; Bonilla, F.; Zhang, Y.; Lanceros-Mendez, S.; Casas-Cabanas, M.; López-Aranguren, P. Interfacial Analysis of In-Situ Anode Formation in Solid-State Batteries with Nanometric Current Collector. Chem. Eng. J. 2025, 509, 160956. [Google Scholar] [CrossRef]
- Xu, L.; Le Houx, J.; Kachkanov, V.; Zhang, J.; Wullich, R.N.; Fankhauser, M.; Löffel, K.; Schmidt, T.J.; El Kazzi, M. Operando X-Ray Computed Tomography Reveals the Role of Interfacial Nucleation Nanolayers in Suppressing Mechanical Failure in Zero-Excess Lithium All-Solid-State Batteries. Small 2026, 22, e12284. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Perlmutter, D.; Fei-Huei Su, A.; Quenum, J.; Shevchenko, P.; Parkinson, D.Y.; Zenyuk, I.V.; Ushizima, D. Detecting Lithium Plating Dynamics in a Solid-State Battery with Operando X-Ray Computed Tomography Using Machine Learning. NPJ Comput. Mater. 2023, 9, 93. [Google Scholar] [CrossRef]
- Vanpeene, V.; Stamati, O.; Guilloud, C.; Tucoulou, R.; Holliger, B.; Chandesris, M.; Lyonnard, S.; Villanova, J. Comparative Study of the Quantitative Analysis of Battery Materials with X-Ray Nano-Tomography: From Ex Situ toward Operando Measurements. ACS Nano 2025, 19, 9994–10012. [Google Scholar] [CrossRef]
- Liu, H.; Kazemiabnavi, S.; Grenier, A.; Vaughan, G.; Di Michiel, M.; Polzin, B.J.; Thornton, K.; Chapman, K.W.; Chupas, P.J. Quantifying Reaction and Rate Heterogeneity in Battery Electrodes in 3D through Operando X-Ray Diffraction Computed Tomography. ACS Appl. Mater. Interfaces 2019, 11, 18386–18394. [Google Scholar] [CrossRef]
- Tonin, G.; Vaughan, G.; Bouchet, R.; Alloin, F.; Di Michiel, M.; Boutafa, L.; Colin, J.-F.; Barchasz, C. Multiscale Characterization of a Lithium/Sulfur Battery by Coupling Operando X-Ray Tomography and Spatially-Resolved Diffraction. Sci. Rep. 2017, 7, 2755. [Google Scholar] [CrossRef]
- Sottmann, J.; Di Michiel, M.; Fjellvåg, H.; Malavasi, L.; Margadonna, S.; Vajeeston, P.; Vaughan, G.B.M.; Wragg, D.S. Chemical Structures of Specific Sodium Ion Battery Components Determined by Operando Pair Distribution Function and X-ray Diffraction Computed Tomography. Angew. Chem. Int. Ed. 2017, 56, 11385–11389. [Google Scholar] [CrossRef]
- Barchasz, C.; Boutafa, L.; Mayousse, E.; Chavillon, B. Diagnostic of Failure Mechanisms in Li/S Rechargeable Batteries Using Thermal Micro-Calorimetry Technique Applied to Pouch and Cylindrical Type Cells. Electrochim. Acta 2018, 292, 974–981. [Google Scholar] [CrossRef]
- Ming, F.; Zhu, Y.; Huang, G.; Emwas, A.-H.; Liang, H.; Cui, Y.; Alshareef, H.N. Co-Solvent Electrolyte Engineering for Stable Anode-Free Zinc Metal Batteries. J. Am. Chem. Soc. 2022, 144, 7160–7170. [Google Scholar] [CrossRef]
- Heo, J.; Shin, K.; Kim, H. A Zinc–Bromine Battery with Deep Eutectic Electrolytes. Adv. Sci. 2022, 9, 2204908. [Google Scholar] [CrossRef]
- Sabato, A.G.; Eroles, M.N.; Anelli, S.; Sierra, C.D.; Gonzalez-Rosillo, J.C.; Torrell, M.; Pesce, A.; Accardo, G.; Casas-Cabanas, M.; López-Aranguren, P. 3D Printing of Self-Supported Solid Electrolytes Made of Glass-Derived Li1.5Al0.5Ge1.5P3O12 for All-Solid-State Lithium-Metal Batteries. J. Mater. Chem. A Mater. 2023, 11, 13677–13686. [Google Scholar] [CrossRef]
- Charbonnel, J.; Bengaouer, A.; Thivel, P.-X.; Vincent, R. Can the Shock Wave Generated by the Thermal Runaway of a Lithium-Metal All-Solid-State Cell Be Predicted by Material-Scale Analysis. Chem. Eng. J. 2024, 494, 153234. [Google Scholar] [CrossRef]
- Chen, D.; Liao, J.; Trdinich, Z.J.; Wang, C.; Shi, F. Impact of Electrolyte and SEI Passivity on Thermal Runaway: A Comparative DSC and ARC Analysis of Anode-Free Lithium Metal Batteries. J. Power Sources 2026, 667, 239261. [Google Scholar] [CrossRef]
- Ma, P.; Zhang, Y.; Li, W.; Luo, J.; Wen, L.; Tang, G.; Gai, J.; Wang, Q.; Zhao, L.; Ge, J. Tailoring Alloy-Reaction-Induced Semi-Coherent Interface to Guide Sodium Nucleation and Growth for Long-Term Anode-Less Sodium-Metal Batteries. Sci. China Mater. 2024, 67, 3648–3657. [Google Scholar] [CrossRef]
- Cheng, X.; Li, D.; Peng, S.; Shi, P.; Yu, H.; Jiang, Y.; Li, S. In-Situ Alloy-Modified Sodiophilic Current Collectors for Anode-Less Sodium Metal Batteries. Batteries 2023, 9, 408. [Google Scholar] [CrossRef]
- Wang, C.; Zheng, Y.; Chen, Z.; Zhang, R.; He, W.; Li, K.; Yan, S.; Cui, J.; Fang, X.; Yan, J. Robust Anode-Free Sodium Metal Batteries Enabled by Artificial Sodium Formate Interface. Adv. Energy Mater. 2023, 13, 2204125. [Google Scholar] [CrossRef]
- Sagar, R.U.R.; Nelson, A.; Fazal, M.W.; Khan, M.W.; Mahmood, N.; Rahman, M.M.; Chen, Y.I. An Ultralight Porous Carbon Scaffold for Anode-Free Lithium Metal Batteries. J. Mater. Chem. A Mater. 2025, 13, 5081–5090. [Google Scholar] [CrossRef]
- Jang, E.; Park, S.; Lee, J.; Kim, P.J.; Yoo, J. Revolutionizing Anode-Free Batteries with Holey Graphene Interlayers: Achieving Uniform Lithium Deposition and Long-Term Stability. J. Energy Storage 2025, 115, 115974. [Google Scholar] [CrossRef]
- Huang, F.; Hu, C.; Luo, Z.; Qin, M.; Li, W.; Lai, G.; Liu, S.; Lei, Y.; Liang, S.; Fang, G. Sodiophilic Host with 85% Depth-of-Discharge Reversibility towards Robust Anode-Free Na Metal Batteries. Nano Energy 2025, 137, 110780. [Google Scholar] [CrossRef]
- Guo, W.; Liu, X.; Mu, Y.; Yue, G.; Liu, J.; Zhu, K.; Cui, Z.; Wang, N.; Chen, Z.; Zhao, Y. Outside-in Directional Sodium Deposition through Self-Supporting Gradient Fluorinated Magnesium Alloy Framework toward High-Rate Anode-Free Na Batteries. Energy Storage Mater. 2024, 73, 103840. [Google Scholar] [CrossRef]
- Hu, Z.; Liu, L.; Wang, X.; Lu, H.; Zheng, Q.; Gao, Y.; Wang, J.; Qi, Y.; Han, C.; Li, W. In Situ Integration of Rapid Ion-Diffusion Interlayers on Cu Current Collectors toward Ultrafast Anode-Free Sodium Metal Batteries. ACS Nano 2025, 19, 23193–23208. [Google Scholar] [CrossRef]
- Chen, J.; Li, Q.; Pollard, T.P.; Fan, X.; Borodin, O.; Wang, C. Electrolyte Design for Li Metal-Free Li Batteries. Mater. Today 2020, 39, 118–126. [Google Scholar] [CrossRef]
- Zhou, C.; Zheng, L.; He, T.; Garakani, M.A.; Abouali, S.; Shen, Y.; Chen, L.; Thangadurai, V. Rational Design of a Carbonate-Glyme Hybrid Electrolyte for Practical Anode-Free Lithium Metal Batteries. Energy Storage Mater. 2021, 42, 295–306. [Google Scholar] [CrossRef]
- Weber, R.; Genovese, M.; Louli, A.J.; Hames, S.; Martin, C.; Hill, I.G.; Dahn, J.R. Long Cycle Life and Dendrite-Free Lithium Morphology in Anode-Free Lithium Pouch Cells Enabled by a Dual-Salt Liquid Electrolyte. Nat. Energy 2019, 4, 683–689. [Google Scholar] [CrossRef]
- Beyene, T.T.; Bezabh, H.K.; Weret, M.A.; Hagos, T.M.; Huang, C.-J.; Wang, C.-H.; Su, W.-N.; Dai, H.; Hwang, B.-J. Concentrated Dual-Salt Electrolyte to Stabilize Li Metal and Increase Cycle Life of Anode Free Li-Metal Batteries. J. Electrochem. Soc. 2019, 166, A1501–A1509. [Google Scholar] [CrossRef]
- Kang, D.W.; Moon, J.; Choi, H.Y.; Shin, H.C.; Kim, B.G. Stable Cycling and Uniform Lithium Deposition in Anode-Free Lithium-Metal Batteries Enabled by a High-Concentration Dual-Salt Electrolyte with High LiNO3 Content. J. Power Sources 2021, 490, 229504. [Google Scholar] [CrossRef]
- Eldesoky, A.; Louli, A.J.; Benson, A.; Dahn, J.R. Cycling Performance of NMC811 Anode-Free Pouch Cells with 65 Different Electrolyte Formulations. J. Electrochem. Soc. 2021, 168, 120508. [Google Scholar] [CrossRef]
- Pathirana, T.; Kerr, R.; Forsyth, M.; Howlett, P.C. Application of Super-Concentrated Phosphonium Based Ionic Liquid Electrolyte for Anode-Free Lithium Metal Batteries. Sustain. Energy Fuels 2021, 5, 4141–4152. [Google Scholar] [CrossRef]
- Lu, Z.; Yang, H.; Yang, Q.-H.; He, P.; Zhou, H.; Lu, Z.; Yang, H.; Zhou, H.; Yang, Q.; He, P. Building a Beyond Concentrated Electrolyte for High-Voltage Anode-Free Rechargeable Sodium Batteries. Angew. Chem. 2022, 134, e202200410. [Google Scholar] [CrossRef]
- Wu, S.; Nishigaki, Y.; Hagiwara, R.; Hwang, J.; Matsumoto, K. Entropy-Driven 60 Mol% Li Electrolyte for Li Metal-Free Batteries. Small 2024, 20, 2405007. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Lan, H.; Wang, S.; Liu, X.; Zhu, Q.; Zhang, X.; Tang, M.; Dong, S.; Yang, J.; Kurbanov, M. Realizing an Energy-Dense Potassium Metal Battery At− 40° C via an Integrated Anode-Free and Dual-Ion Strategy. J. Am. Chem. Soc. 2025, 147, 2393–2402. [Google Scholar] [CrossRef] [PubMed]
- Louli, A.J.; Eldesoky, A.; Weber, R.; Genovese, M.; Coon, M.; DeGooyer, J.; Deng, Z.; White, R.T.; Lee, J.; Rodgers, T.; et al. Diagnosing and Correcting Anode-Free Cell Failure via Electrolyte and Morphological Analysis. Nat. Energy 2020, 5, 693–702. [Google Scholar] [CrossRef]
- Cohn, A.P.; Muralidharan, N.; Carter, R.; Share, K.; Pint, C.L. Anode-Free Sodium Battery through in Situ Plating of Sodium Metal. Nano Lett. 2017, 17, 1296–1301. [Google Scholar] [CrossRef]
- Tang, X.; Bai, M.; Shao, A.; Wang, Z.; Wang, H.; Zhang, M.; Ma, Y. Lithium Carbide Prelithiation Agent Enhanced the Energy Density and Lifespan of Anode-Free Battery. Small 2025, 21, 2501041. [Google Scholar] [CrossRef]
- Kim, S.; Didwal, P.N.; Fiates, J.; Dawson, J.A.; Weatherup, R.S.; De Volder, M. Effect of the Formation Rate on the Stability of Anode-Free Lithium Metal Batteries. ACS Energy Lett. 2024, 9, 4753–4760. [Google Scholar] [CrossRef]
- Lin, X.; Shen, Y.; Yu, Y.; Huang, Y. In Situ NMR Verification for Stacking Pressure-Induced Lithium Deposition and Dead Lithium in Anode-Free Lithium Metal Batteries. Adv. Energy Mater. 2024, 14, 2303918. [Google Scholar] [CrossRef]
- Tang, M.; Dong, S.; Wang, J.; Cheng, L.; Zhu, Q.; Li, Y.; Yang, X.; Guo, L.; Wang, H. Low-Temperature Anode-Free Potassium Metal Batteries. Nat. Commun. 2023, 14, 6006. [Google Scholar] [CrossRef]
- Su, L.; Charalambous, H.; Cui, Z.; Manthiram, A. High-Efficiency, Anode-Free Lithium–Metal Batteries with a Close-Packed Homogeneous Lithium Morphology. Energy Environ. Sci. 2022, 15, 843–854. [Google Scholar] [CrossRef]
- Lee, K.; Lee, Y.J.; Lee, M.J.; Han, J.; Lim, J.; Ryu, K.; Yoon, H.; Kim, B.; Kim, B.J.; Lee, S.W. A 3D Hierarchical Host with Enhanced Sodiophilicity Enabling Anode-free Sodium-metal Batteries. Adv. Mater. 2022, 34, 2109767. [Google Scholar] [CrossRef]
- Sreedeep, S.; Athira, K.M.; Lee, Y.; Aravindan, V. Crucial Role of Solvent and Additive During the Transformation of Liquid to Solid Polymer Electrolyte for Stabilizing 4.8 V Class LiCo0.9Fe0.1PO4 Cathode in Anode and Anodeless Assemblies. Small 2025, 21, 2503310. [Google Scholar] [CrossRef]
- Nguyen, M.H.; Kim, D.; Kim, B.; Park, S. Harnessing Liquid Metals with In Situ Polymerized Electrolyte for Anode-Free Lithium Metal Batteries. Adv. Funct. Mater. 2024, 34, 2407179. [Google Scholar] [CrossRef]
- Sreedeep, S.; Lee, Y.; Aravindan, V. Tailoring the Salt Concentration in a Gel-Solid Polymer Electrolyte to Stabilize High-Voltage LiCoMnO4 Cathode. Small 2025, 21, 2503594. [Google Scholar] [CrossRef] [PubMed]
- Rajendran, S.; Sekar, A.; Li, J. Deciphering the Effect of Nucleation/Growth Kinetics on the Morphology of Li Plating/Stripping on Vertically Aligned Carbon Nanofibers-A Low-Tortuosity 3-D Nanostructured Carbon Host. Chem. Eng. J. 2024, 484, 149515. [Google Scholar] [CrossRef]
- Thorpe, M.A.; Zhang, M.; Liao, D.W.; Sandoval, S.E.; Kim, Y.; McDowell, M.T.; Thouless, M.D.; Dasgupta, N.P. Controlling Stack Pressure Inhomogeneity in Anode-Free Solid-State Batteries Using Elastomeric Interlayers. Matter 2025, 8, 101955. [Google Scholar] [CrossRef]
- Neudecker, B.J.; Dudney, N.J.; Bates, J.B. “Lithium-Free” Thin-Film Battery with In Situ Plated Li Anode. J. Electrochem. Soc. 2000, 147, 517. [Google Scholar] [CrossRef]
- Baptista, M.C.; Vale, A.B.; Costa, J.M.; Braga, M.H. Robust All-Solid-State Batteries with Sodium Ion Electrolyte, Aluminum and Additive Manufacturing Inconel 625 Electrodes. Molecules 2025, 30, 4465. [Google Scholar] [CrossRef]
- Kazyak, E.; Wang, M.J.; Lee, K.; Yadavalli, S.; Sanchez, A.J.; Thouless, M.D.; Sakamoto, J.; Dasgupta, N.P. Understanding the Electro-Chemo-Mechanics of Li Plating in Anode-Free Solid-State Batteries with Operando 3D Microscopy. Matter 2022, 5, 3912–3934. [Google Scholar] [CrossRef]
- Shozib, I.A.; Cúñez, F.D.; Ganter, M.; Leggiero, A.; Liu, R.; Tu, Q.H. Interplay of Interfacial Adhesion and Mechanical Degradation in Anode-Free Solid-State Batteries. J. Electrochem. Soc. 2024, 171, 090524. [Google Scholar] [CrossRef]
- Jeong, M.-G.; Hatzell, K.B.; Banerjee, S.; Vishnugopi, B.S.; Mukherjee, P.P. Temperature Impact on Lithium Metal Morphology in Lithium Reservoir-Free Solid-State Batteries. PRX Energy 2024, 3, 023003. [Google Scholar] [CrossRef]
- Park, S.H.; Ayyaswamy, A.; Gjerde, J.; Andrews, W.B.; Vishnugopi, B.S.; Drakopoulos, M.; Vo, N.T.; Zhong, Z.; Thornton, K.; Mukherjee, P.P. Filament-Induced Failure in Lithium-Reservoir-Free Solid-State Batteries. ACS Energy Lett. 2025, 10, 1174–1182. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.L.; Kazyak, E.; Liao, D.W.; Wood, K.N.; Dasgupta, N.P. Operando Analysis of Interphase Dynamics in Anode-Free Solid-State Batteries with Sulfide Electrolytes. J. Electrochem. Soc. 2021, 168, 070557. [Google Scholar] [CrossRef]
- Yin, Q.; Li, T.; Zhang, H.; Zhong, G.; Yang, X.; Li, X. SEI/Dead Li-Turning Capacity Loss for High-Performance Anode-Free Solid-State Lithium Batteries. J. Energy Chem. 2024, 96, 145–152. [Google Scholar] [CrossRef]
- Tron, A.; Nosenko, A.; Paolella, A. Dendrite-Free Formation of Anode-Less Lithium Metal Through a Solid Glassy Electrolyte Film for Lithium Metal Batteries. Phys. Status Solidi A 2025, 223, 2500285. [Google Scholar] [CrossRef]
- Zegeye, T.A.; Su, W.-N.; Fenta, W.; Zeleke, S.; Jiang, S.-K.; Hwang, B.J. Ultrathin Li6.75La3Zr1.75Ta0.25O12-Based Composite Solid Electrolytes Laminated on Anode and Cathode Surfaces for Anode-Free Lithium Metal Batteries. ACS Publ. 2020, 3, 11713–11723. [Google Scholar] [CrossRef]
- Bert, M.; Colonna, J.-P.; Cele’, J.; Casiez, L.; Oukassi, S.; Franger, S. Influence of Charge Current Density on the Lithium Metal Plating at Ti/LiPON Interface of Anode-Less Solid-State Batteries Observed by SEM. In Proceedings of the Electrochemical Society Meeting Abstracts 247, Montreal, QC, Canada, 18--22 May 2025; The Electrochemical Society, Inc.: Pennington, NJ, USA, 2025; p. 668. [Google Scholar]
- Westover, A.S.; Dudney, N.J.; Sacci, R.L.; Kalnaus, S. Deposition and Confinement of Li Metal along an Artificial Lipon-Lipon Interface. ACS Energy Lett. 2019, 4, 651–655. [Google Scholar] [CrossRef]
- Lee, S.H.; Liu, P.; Tracy, C.E. Lithium Thin-Film Battery with a Reversed Structural Configuration SS/Li/Lipon/LixV2O5/Cu. Electrochem. Solid-State Lett. 2003, 6, A275. [Google Scholar] [CrossRef]
- Choi, S.H.; Baek, C.H.; Oh, J.; Lee, G.-J.; Kim, M.; Lee, H.; Yoo, D.-J.; Jung, Y.S.; Kim, K.; Yu, J.-S. Silver Exsolution from Li-Argyrodite Electrolytes for Initially Anode-Free All-Solid-State Batteries. Nat. Commun. 2025, 16, 5871. [Google Scholar] [CrossRef]
- Lee, J.H.; Oh, S.-H.; Yim, H.; Lee, H.-J.; Kwon, E.; Yu, S.; Kim, J.S.; Song, J.; Koo, J.; Cho, J. Interfacial Stabilization Strategy via In-Doped Ag Metal Coating Enables a High Cycle Life of Anode-Free Solid-State Li Batteries. Energy Storage Mater. 2024, 69, 103398. [Google Scholar] [CrossRef]
- Kang, J.; Kim, J.; Kim, R.; Lim, Y.J.; Lee, J.-W. Mechanistic Insight into Calendar Aging of Anode-Less All-Solid-State Batteries. Energy Storage Mater. 2025, 76, 104164. [Google Scholar] [CrossRef]
- Sahore, R.; Mayer, A.; Steinle, D.; Counihan, M.J.; Chen, X.C.; Tepavcevic, S.; Bresser, D.; Westover, A.S. Electroanalytical Exploration of Li Loss at the Solid Electrolyte-Anode Interface in Anode-Free Batteries with Polymer Electrolytes. J. Electrochem. Soc. 2024, 171, 050505. [Google Scholar] [CrossRef]
- Guan, X.; Brandell, D.; Mindemark, J. Exploring the Efficiency of Lithium Stripping/Plating in Solid Polymer Anode-Free Lithium Cells. In Proceedings of the Electrochemical Society Meeting Abstracts 244, Gothenburg, Sweden, 8--12 October 2023; The Electrochemical Society, Inc.: Pennington, NJ, USA, 2023; p. 3031. [Google Scholar]
- Zhao, J.; Huang, S.; Zhao, Y.; Cui, C.; Zhang, Y.; Lin, H.; Zhao, C.; Dai, W.; Liu, Z.; Song, X. Ultra-Stable Solid-State Lithium Metal Batteries with Ferroelectric Oxide-Enhanced PVDF-Based Hybrid Solid Electrolytes. J. Mater. Chem. A Mater. 2025, 13, 9347–9356. [Google Scholar] [CrossRef]
- Xie, F.; Diallo, M.S.; Kim, H.; Tu, Q.H.; Ceder, G. The Microscopic Mechanism of Lithiation and Delithiation in the Ag/C Buffer Layer for Anode-free Solid-state Batteries. Adv. Energy Mater. 2024, 14, 2302960. [Google Scholar] [CrossRef]
- Wang, M.J.; Carmona, E.; Gupta, A.; Albertus, P.; Sakamoto, J. Enabling “Lithium-Free” Manufacturing of Pure Lithium Metal Solid-State Batteries through in Situ Plating. Nat. Commun. 2020, 11, 5201. [Google Scholar] [CrossRef] [PubMed]
- Seo, D.-B.; Kim, D.; Kim, M.-R.; Kwon, J.; Kook, H.J.; Kang, S.; Yim, S.; Lee, S.S.; Shin, D.O.; An, K.-S. Tailoring Artificial Solid Electrolyte Interphase via MoS2 Sacrificial Thin Film for Li-Free All-Solid-State Batteries. Nanomicro Lett. 2025, 17, 224. [Google Scholar] [CrossRef] [PubMed]
- Romio, M.; Kahr, J.; Miele, E.; Krammer, M.; Surace, Y.; Boz, B.; Molaiyan, P.; Dimopoulos, T.; Armand, M.; Paolella, A. Operando Optical Microscopy of Dead Lithium Growth in Anode-Less Configuration. Adv. Mater. Technol. 2024, 9, 2301902. [Google Scholar] [CrossRef]
- Fang, C.; Li, J.; Zhang, M.; Zhang, Y.; Yang, F.; Lee, J.Z.; Lee, M.-H.; Alvarado, J.; Schroeder, M.A.; Yang, Y. Quantifying Inactive Lithium in Lithium Metal Batteries. Nature 2019, 572, 511–515. [Google Scholar] [CrossRef]
- Gunnarsdóttir, A.B.; Amanchukwu, C.V.; Menkin, S.; Grey, C.P. Noninvasive in Situ NMR Study of “Dead Lithium” Formation and Lithium Corrosion in Full-Cell Lithium Metal Batteries. J. Am. Chem. Soc. 2020, 142, 20814–20827. [Google Scholar] [CrossRef]
- Hsieh, Y.-C.; Leißing, M.; Nowak, S.; Hwang, B.-J.; Winter, M.; Brunklaus, G. Quantification of Dead Lithium via In Situ Nuclear Magnetic Resonance Spectroscopy. Cell Rep. Phys. Sci. 2020, 1, 100139. [Google Scholar] [CrossRef]





| Ref. | Focus | System/Chemistry Scope | Key Emphasis | How the Present Review Differs from Cited |
|---|---|---|---|---|
| [18] | General review of anode-less lithium batteries | Li anode-less batteries | Challenges, strategies, failure mechanisms, and characterization | Broader fundamentals-to-application framework |
| [19] | Critical analysis of anode-less cell concepts and practical battery development | Anode-less batteries in liquid and solid electrolyte systems, including Li and Na plating contexts | Practical cell performance, plating/stripping mechanisms at the negative current collector, degradation processes, influence of other cell components, and gaps in data accessibility, reporting standards, and metrics | Broader fundamentals to future directions framework with stronger emphasis on conceptual organization and development roadmap |
| [16] | Review of post-lithium anode-less metal batteries | Post-lithium systems, including Na, K, and Zn | Challenges, reversibility, and strategies beyond Li | Architecture-centered rather than chemistry-centered |
| [15] | Interface engineering in anode-less sodium batteries | Sodium anode-less batteries | Interfacial chemistry, Na deposition/stripping, and stabilization strategies | Broader than interface-focused sodium scope |
| [20] | Broad overview of anode-less batteries | Broad anode-less battery landscape | Mechanisms, challenges, opportunities, energy density, and safety | Structured around a conceptual pyramid and development roadmap |
| [21] | SEI-focused review with characterization emphasis | Lithium-metal and anode-less lithium batteries | SEI formation, composition, and in situ/operando characterization | Broader than SEI characterization alone |
| [22] | Mechanistic review of active lithium loss in anode-less batteries | Li anode-less lithium-metal batteries | Active lithium loss, degradation mechanisms, and mitigation strategies | Broader than single-mechanism degradation analysis |
| [4] | Focused review on sulfide-based of anode-less batteries | Sulfide-based anode-less solid-state batteries | Sulfide interfaces, non-uniform Li growth, voids, and solid-state solutions | Broader than sulfide-based solid-state scope |
| Category | Technique | Main Information Provided | Mechanistic Relevance | Example Applications | References |
|---|---|---|---|---|---|
| Electrochemical analyses | Charge–discharge cycling | Capacity, coulombic efficiency, reversibility, stability | Overall cycling instability, active lithium loss, and reversibility limitations | Distinguishes stable Li nucleation vs. interfacial degradation; validates separators and frameworks; conditioning effects | [26,39,40,55,56,57,58,59,60,61,62,63,64] |
PEIS | Internal resistance, charge transfer, interfacial properties | Interfacial degradation, contact loss, and resistance buildup during cycling | Nucleation engineering; polymer separator integration; in situ polymerization; in-series solid-state cells | [26,38,65] | |
CV | Redox processes, interfacial reactions, stability | Nucleation behavior, plating/stripping reversibility, and reaction kinetics | Preconditioning; polymerization peaks; redox stability of SSEs; ferroelectric systems | [26,38,65,66] | |
GITT | Ionic diffusivity, kinetic limitations | Diffusion/transport limitations and polarization-related kinetic bottlenecks | Diffusion coefficients in elastic networks; dual-seed strategies; Ah-level separators; Zn2+ transport stabilization | [39,40,67,68] | |
| Interfacial potential mapping | SKP | Macroscopic surface potential/work function mapping | Surface chemical-potential gradients and interfacial stability evolution | Surface potentials shift in heterocells; dielectric oxides vs. SSEs; Li alloying/plating in 3D collectors | [70,71,72] |
KPFM | Nanoscale CPD mapping | Local interfacial heterogeneity, nucleation-active regions, and surface instability | Li nucleation pathways; in operando redistribution; suppressed gradients; dendrite nucleation at grain boundaries; combined with AFM, ToF-SIMS | [73,74,75,76] | |
| Structural and chemical analyses | XRD | Crystal structure, phase transitions | Structural transformation pathways and reaction-product formation | Cathode lattice variations; Li–Ag alloy phases; reversible alloy phases; lithiophilic interlayers | [40,48,78,79] |
XPS | Surface chemistry, SEI/interphase composition | Interphase formation, surface decomposition pathways, and chemical instability | Wetting layers on Cu; SEI composition (organic/inorganic); conditioning effects; high-entropy SEIs (Na); degradation pathways | [21,26,80,81,82] | |
ToF-SIMS | Nanoscale chemical mapping | Interphase nonuniformity and depth-dependent chemical degradation | Lithiophilic seeds, dual-metal interlayers, near-surface Li-ion irrigation; lithio-amphiphilic bilayers; Na/K SEIs | [55,57,58,83,84,85,86,87] | |
TEM | Interfacial structures, deposition morphology, phase transformations | Nanoscale interfacial degradation, filament formation, and local structural failure | Metallic interlayers; Sn-decorated fibers; electron-deficient collectors; engineered coatings; conversion-type anodes; Na collectors; Cu substrates | [48,50,88,89,90,91,92,93,94] | |
LIBS | Surface/interfacial elemental analysis, depth profiling | Spatially heterogeneous degradation and nonuniform elemental redistribution | Interfacial chemistry in SSEs; cycling-induced compositional changes; postmortem analysis; Li detection | [26,31,95,96] | |
In operando techniques (XRD, TEM, XPS, Raman) | Real-time structural/chemical/morphological changes | Real-time interfacial evolution, reaction pathways, and failure development during cycling | Phase transitions; Li nucleation and dendrites; SEI chemistry; polydopamine coatings; Li–S polysulfides; Na SEI; oriented Cu | [50,81,88,94,97,98,99,100] | |
| Morphological studies | SEM–EDX | Surface morphology, elemental distribution | Surface degradation, deposit morphology, and compositional heterogeneity | Li plating/stripping (not detected directly), interphase degradation, Ag distribution, conditioning cells, Na deposition, surface modifications | [26,101,102,103,104,105,106] |
AFM | Nanoscale morphology, nucleation sites | Surface roughness evolution, local mechanical heterogeneity, and early deposition instability | ZnF2 coatings; Li–S systems; in operando Zn nucleation; engineered collectors and electrolytes | [41,68,100,107,108,109,110,111] | |
In operando tomography | 3D real-time morphological evolution | Void formation, filament growth, and 3D morphological evolution during cycling | Zero-excess Li failure suppression; machine learning-enhanced tomography; multiscale tomography/diffraction coupling | [113,114,115,116,117,118] | |
| Thermal analyses | In operando calorimetry | Heat generation during cycling, parasitic reactions | Heat generation pathways, parasitic reactions, and thermal signatures of interfacial instability during cycling | Failure diagnosis in Li–S (potential for anode-less systems) | [119] |
DSC | Thermal stability, phase transitions | Thermal reactivity, decomposition behavior, and safety-related phase or interfacial instability | Seawater Na batteries; Zn co-solvent electrolytes; deep eutectic solvents; glass-derived SSEs | [103,120,121,122] | |
ARC | Thermal runaway and abuse testing | Thermal runaway propensity, self-heating behavior, and abuse-response safety limits | Safety margins in Li-metal SSEs; ultralight integrated anodes; porous carbons for heat dissipation | [123,124] |
| Year | Cell Type | Cell Configuration | Electrolyte | Interlayer/Surface Modification | Key Results/Performance | Ref. |
|---|---|---|---|---|---|---|
| 2016 | Coin | Cu||NMC622 | LiPF6 in EC/DMC | Cu current collector, focus on fundamentals of anode-less Li metal plating | >100 cycles, CE > 95%, stable Li plating/stripping with minimal dendrite formation | [23] |
| 2017 | Coin | Cu||carbon coated NVP composite | NaPF6 in EC/DE | In situ plating of sodium metal on a bare Cu current collector | >150 cycles, CE > 95%, stable Na plating/stripping, minimized dead sodium | [144] |
| 2019 | Coin | Cu||NMC811 | Concentrated dual salt electrolyte: LiFSI and LiPF6 in EC/EMC | Electrolyte induced stable SEI formation on lithium metal | >250 cycles, CE > 97%, improved cycling stability and lithium-metal protection | [136] |
| 2019 | Pouch | Cu||NMC532 | Dual-salt liquid electrolyte: LiPF6 and LiTFSI in EC/EMC | Formation of stable SEI promoted by dual-salt electrolyte | >300 cycles, CE > 98%, dendrite-free lithium morphology, enhanced cycling stability | [135] |
| 2020 | Coin | Cu||NMC532 | Optimized electrolyte: baseline is a conventional carbonate | Morphological control via electrolyte tuning to mitigate failure mechanisms | >200 cycles, CE > 96%, reduced dendrite formation, improved cycle life | [143] |
| 2020 | Coin | Cu||LFP | LiFSI in DME/BTFE | Electrolyte chemistry optimizing SEI formation and lithium plating behavior | >150 cycles, CE > 95%, improved stability without lithium-metal anode | [133] |
| 2021 | Coin | Cu||LFP | LiFSI in P1114FSI ionic liquid | Electrolyte enabled stable SEI and improved lithium plating | >200 cycles, CE > 98%, enhanced cycling stability and dendrite suppression | [139] |
| 2021 | Coin | Cu||NMC622 | LiFSI, LiTFSI and LiNO3 in DME/DOL | Electrolyte induced uniform lithium deposition and stable SEI formation | >300 cycles, CE > 99%, enhanced cycling stability, dendrite suppression | [137] |
| 2021 | Coin | Cu||NMC811 | LiPF6 in EC/EMC/Triglyme | None or intrinsic interface formation promoted by electrolyte design | >200 cycles, CE > 98%, improved stability and dendrite suppression | [134] |
| 2021 | Pouch | Cu||NMC811 | 65 different electrolyte formulations: based on carbonate solvents (EC: EMC) with varied salts (LiPF6) and a wide range of additives | None or electrolyte-driven SEI modifications studied | >300 cycles, CE > 99%, electrolyte dependent cycling stability | [138] |
| 2022 | Coin | Cu||NMC811 | LiFSI in a mixture of DME and TTE | Interface engineering for homogeneous lithium plating | >300 cycles, CE > 99%, uniform Li morphology, suppressed dendrites | [149] |
| 2022 | Coin | 3D Cu-based hierarchical host||NVP | NaPF6 in EC: DEC | 3D hierarchical sodiophilic host structure to promote uniform Na plating | >300 cycles, CE > 99%, enhanced Na utilization, suppressed dendrites and dead sodium formation | [150] |
| 2022 | Coin | Cu||NaMnO2 | Beyond concentrated sodium salt electrolyte tailored for high voltage | Electrolyte engineered interface for stable sodium plating | 80% capacity retention after 350 cycles, CE > 97%, high voltage stability, suppressed dendrites | [140] |
| 2023 | Coin | Cu||NVP | NaPF6 in EC: DEC with FEC | HCOONa artificial layer formed by HCOOH vapor pretreatment | 80% capacity retention after 350 cycles, average CE 99.69%, stable Na plating, suppressed dendrites | [127] |
| 2023 | Coin | Cu||NVP | NaPF6 in EC: PC: EMC with FEC | In situ formed Sn-Cu alloy layer from SnO2 coating | 80% capacity retention after 400 cycles, average CE ~99.5% | [126] |
| 2023 | Coin and pouch | Cu||KPTCDA | KPF6 in DME and PDMS | In situ formed interfacial layer via polydimethylsiloxane (PDMS) additive, creating a potassiophilic interface and robust organic-inorganic hybrid SEI | 99.80% average CE at −40 °C, 82% capacity retention after 50 cycles | [148] |
| 2024 | Coin | Cu||LFP | LiTFSI in DOL/DME and LiNO3 | No surface modification or interlayer was applied | Achieves the lowest dead lithium content (2.7 µAh), a high average Coulombic efficiency of ~96%, and the longest cycle life | [147] |
| 2024 | Coin | Cu||NVP | NaPF6 in EC: DEC with FEC | Semi-coherent Cu2Sb alloy interface | 80.5% capacity retention after 500 cycles, average CE ~99.8% | [125] |
| 2024 | Coin | Cu||LFP | Entropy-driven 60 mol% LiF-LiI halide solid electrolyte | Electrolyte—driven interface enabling stable lithium plating without metal anode | Stable cycling for >400 cycles, average CE ~99.4% | [141] |
| 2024 | Pouch | MgF2@NCHNFs||NVP | NaTFSI in FEC:P | 3D host of nitrogen-doped carbon hollow nanofibers with MgF2 that, during initial plating, forms an in situ gradient fluorinated alloy architecture with a surface NaF layer and subsurface Mg site | Dendrite free Na deposition at high rates (10 mA·cm−2) and capacities (10 mAh.cm−2), achieving > 500 h stability in symmetric cells and 96% capacity retention after 400 cycles in pouch cells | [131] |
| 2024 | Pouch | Cu||NMC811 | LiPF6 in EC/DEC; dual-salt electrolyte (DSE) and localized high-concentration electrolyte (LHCE) | None: the study focused on optimizing the formation protocol (initial charging rate), with no current collector surface modification | ~20% capacity retention after 20 cycles, optimal formation at max non-dendritic rate (C/2 for standard electrolyte) | [146] |
| 2024 | Coin | Cu||NVP | NaPF6 in EC: PC with FEC | High-entropy SEI formed by edge electron effect to enhance durability on N-doped carbon nanotube modified Cu | 86.5% capacity retention after 500 cycles, average CE ~99.6% | [81] |
| 2025 | Coin | Al||PTPAn | KPF6/KTFSI-DME/diglyme | Anion derived SEI formed in situ via KTFSI salt additive, creating a robust, inorganic-rich interface | 99.98% average CE at −40 °C, 407 Wh·kg−1 energy density, 80% capacity retention after 183 cycles | [142] |
| 2025 | Coin | Cu||LFP | LiFSI in DME | Holey graphene (HG) or graphene (GR) coated as an interlayer on the Cu | HG-based achieved 75% capacity retention after 200 cycles, CE > 99.6% | [129] |
| 2025 | Coin | Cu||NMC811 | LiPF6 in EC: EMC with VC | Lithium carbide prelithiation agent to improve initial lithium inventory and cycling stability | 80% capacity retention after 420 cycles, CE > 99.1% | [145] |
| 2025 | Coin | Cu||LFP | LiPF6 in EC: DEC with FEC | Protonated polyaniline coating on Cu to improve lithium nucleation | Stable cycling for 200 cycles, average CE ~99.2%, uniform Li deposition | [111] |
| 2025 | Coin | Cu||NMC111 | LiPF6 in EC: EMC with LiDFOB sacrificial salt | Sacrificial lithium salts favoring stable SEI and CEI formation | Feasibility analysis for anode-less cells, CE ~99.1% in Cu | [47] |
| 2025 | Coin | SnSb/C-Cu foam||NVP | NaPF6 in EC: PC with FEC | 3D sodiophilic SnSb/C-coated Cu foam host | 85% DOD reversibility, stable for 400 cycles, average CE ~99.5%, dendrite suppression | [130] |
| 2025 | Coin | PBA@Cu||NVP PBA@Cu||LFP | NaPF6 in G2; LiTFSI in DME/DOL and LiNO3 | In situ integrated Prussian blue analogue (PBA) interlayer on Cu | 75.3% capacity retention after 300 cycles at 5C and an average CE of ~99.7% | [132] |
| 2025 | Coin | Cu2O on Cu||LFP | LiPF6 in EC: EMC with FEC | 3D Cu foam current collector with lithiophilic Cu2O coating | 92.8% capacity retention after 200 cycles, average CE ~99.2% | [42] |
| Aspect | Liquid-Electrolyte Anode-Less Systems | Solid-State Anode-Less Systems | Main Implication |
|---|---|---|---|
| Interfacial contact | Assisted by electrolyte wetting and dynamic interfacial accommodation | Limited by solid–solid contact, surface conformity, and contact retention during cycling | Solid-state systems are more sensitive to local interfacial defects and contact loss |
| Current-density tolerance | Can often be improved through electrolyte formulation, additives, and current collector design, but remains sensitive to nonuniform plating and parasitic reactions | Frequently constrained by local current constriction, interfacial resistance, and chemomechanical instability | Stable high-rate operation is generally more challenging in solid-state systems |
| Pressure requirements | Usually low to moderate | Often significant to maintain interfacial contact and suppress void formation during stripping/plating | Pressure is a major design variable in solid state |
| Interfacial resistance evolution | Strongly influenced by SEI composition, electrolyte depletion, and continuous side reactions | Strongly influenced by contact loss, void formation, and interphase growth at solid–solid interfaces | The dominant origins of resistance growth differ substantially between the two systems |
| Practical energy-density considerations | Potentially high, but strongly affected by electrolyte excess, inactive components, and lithium inventory loss | High theoretical promise, but stack pressure hardware, SSE thickness, and packaging constraints may reduce practical gains | Stack-level metrics must be critically interpreted in both cases |
| Year | Cell Type | Cell Configuration | Electrolyte | Interlayer/Surface Modification | Key Results/Performance | Ref. |
|---|---|---|---|---|---|---|
| 2016 | Symmetric (hermetically sealed in epoxy resin) | Cu||Cu Li||Li | Li2.99Ba0.005OCl1-x(OH)x and Na2.99Ba0.005OCl1-x(OH)x | No surface modification or interlayer reported; the glassy electrolyte is applied directly onto the copper or lithium current collector | Demonstrated high ionic conductivity (~best liquid), low activation energy (~0.06 eV), stable dendrite-free Li/Na plating/stripping for 19 days, and anomalous self-charging behavior | [24] |
| 2000 | Thin film | Cu||LiCoO2 | LiPON | A dense overlayer (LiPON or Parylene C) was deposited on the copper current collector; this layer was crucial to confine the plated lithium and prevent detrimental side reactions, enabling stable long-term cycling | Achieved > 1000 cycles via in situ-plated Li anode enabled by a protective overlayer | [156] |
| 2003 | Thin film | SS||LixV2O5 | LiPON | No interlayer: lithium anode formed in situ during first charge between SS and LiPON | Li plating during first charge; stable cycling over 100 cycles; discharge capacity of 110 mAh g−1 at 5 μA cm−2 | [168] |
| 2019 | Thin film | LiPON–Cu–LiPON||LCO/Al2O3 | LiPON | Artificial LiPON–LiPON interface created by depositing a second LiPON layer over Cu current collector | Li metal deposits and propagates along the artificial LiPON–LiPON interface but does not penetrate through bulk LiPON, confirming its ability to suppress Li dendrites; planar Li growth observed over ~2 mm distance | [167] |
| 2020 | Custom research | Cu||PEO-NCA | LLZO | No interlayer or surface modification reported for the anode interface; the cathode interface uses PEO–LiTFSI as a contact/composite layer | Demonstrated “Li-free” solid-state battery in situ plated Li anodes (up to 5 mAh.cm−2) achieving ~100% CE over 50 cycles without LLZO degradation | [176] |
| 2020 | Coin | Cu||NMC111 | LLZTO–PEO composite solid electrolyte (PEO–LiTFSI with 10 wt% LLZTO filler) | Ultrathin (7–10 μm) LLZTO–PEO–CPE layer spin-coated on both Cu current collector and NMC cathode surfaces | Achieved high ionic conductivity, 4.76 × 10−4 S·cm−1 at RT; dendrite free Li, and 98.8% average CE with 41.2% capacity retention after 65 cycles at 0.2 mA·cm−2 | [165] |
| 2021 | Coin | Cu||LTO | LPSCI and LGPS | A Mo-sputtered current collector and an LPSCI interlayer were used for operando analysis and to isolate interface effects, with no surface modification applied to the copper | LPSCI electrolyte achieved efficient lithium plating with stable SEI formation after initial decomposition, while LGPS showed continuous electrolyte degradation without lithium deposition due to conductive SEI formation | [162] |
| 2022 | Coin | Cu||NMC622 | LPSCl | Unmodified Cu and carbon-coated Cu | <50 cycles, low CE, heterogeneous Li plating and void formation quantified as root cause of rapid capacity fade | [158] |
| 2022 | Pouch | Cu with Ag-C||LFP | PEO–LiTFSI with Al2O3 nanoparticles | Ag–C nanocomposite coating on Cu foil | Doubled initial discharge capacity (93 vs. 46 mAh·g−1) vs. bare Cu; CE > 99% after 50 cycles; 54% capacity retention after 50 cycles | [104] |
| 2023 | Pouch | Cu and Cu with ZnO||LFP | Hybrid electrolyte: Li2.99Ba0.005ClO glass ceramic ferroelectric matrix impregnated in cellulose with PVAc binder | ZnO interlayer used to promote uniform lithium nucleation; electrochemical conditioning with controlled load resistors to optimize interface and cycling stability | Confirmed uniform lithium plating (~2.9 µm thick); cells with ZnO layer deliver higher discharge power and longer retention; cycling efficiency over 99%; stable discharge plateau ~3.2 V indicating high lithium chemical potential | [26] |
| 2023 | Coin | Cu||LFP | PEO, PTMC, and PCL with LiTFSI | No surface modification or interlayer | Demonstrated the feasibility of anode-less cells with solid polymer electrolyte; CE limited by “dead lithium” formation; need to improve lithium deposition morphology | [173] |
| 2024 | Custom-pressurized | SS||Li | LPSCl | No surface modification or interlayer was mentioned; the current collector is plain SS | High temperature (80 °C) promoted uniform, film like Li morphology with 100% active area and dominant horizontal growth, whereas low temperature (25 °C) led to vertical growth, discontinuous islands, and reduced active area (63%), with the study focusing solely on the first deposition | [160] |
| 2024 | Coin | Ag–C buffer layer||NMC811 | LPSCl | Ag–C buffer layer facilitating lithiation/delithiation and enhancing interfacial stability | >250 cycles, CE > 96%, improved interface stability and lithium plating/stripping efficiency | [175] |
| 2024 | Coin | Cu||LFP | PDOL polymer electrolyte with LiPF6–LiFSI–LiFSI and CsClO4 | SEI modification via electrolyte composition | LiFSI + CsClO4 electrolyte achieved 89.4% initial efficiency and 98.5% stable efficiency by reducing dead lithium formation by 20× compared to conventional electrolyte | [163] |
| 2024 | Pouch | Cu with ZnO and Cu with Li2O||LFP | Hybrid electrolyte: Li2.99Ba0.005OCl glass ceramic ferroelectric matrix impregnated in cellulose with PVAc binder | ZnO or Li2O nucleation layer was doctor bladed onto the copper current collector to enhance lithium-metal plating | Single ZnO-based cells achieved a 76% CE and 0.82 mAh capacity, while series-connected cells powered LEDs for multiple cycles with a final voltage of 5.3 V | [38] |
| 2024 | Coin | Cu||NMC811 | In situ polymerized electrolyte with liquid metal additives (LM-PE) | Liquid metal (Ga–In–Sn alloy) nanoparticles as nucleation seeds on Cu current collector | Achieved 80% capacity retention after 150 cycles; high average CE of 99.2%; stable Li plating/stripping with dense; dendrite-free morphology | [152] |
| 2024 | Coin | Ag–In coated SUS||NMC811 | Li3PSeCl2Br0.5 | In-doped Ag metal coating on SUS current collector via CCS sputtering | 80% capacity retention after 250 cycles at 1C with 99.8% CE, enabled by stable Li–Ag–In alloy interlayer | [170] |
| 2024 | Coin | Cu/Ag–C||NMC | LPSCl | Ag–C nanocomposite interlayer (Ag nanoparticles and carbon black) coated on Cu | Optimal assembly pressure (500–530 MPa) achieved 198.1 mAh·g−1 initial capacity and 410 Wh·kg−1 energy density by enhancing interfacial adhesion, while higher pressures caused electrolyte fracture and rapid performance decay | [159] |
| 2024 | Coin | Cu||NMC811 | Polyester-based polymer electrolyte | Investigation of lithium loss mechanisms at the polymer electrolyte–anode interface | >200 cycles, CE > 95%, lithium loss quantified impacting cycling stability and interface durability | [172] |
| 2024 | Pouch | Cu||Zn | Hybrid electrolyte: Na2.99Ba0.005OCl glass ceramic ferroelectric matrix impregnated in cellulose with PVAc binder | The copper and zinc current collectors were sanded to create texture for improved sodium nucleation; a carbon felt interlayer was added adjacent to the positive copper current collector | Electrodeless sodium pouch cells demonstrated self-charging, with voltage increasing during discharge to 1.2 V and confirmed sodium metal plating on zinc | [66] |
| 2025 | Custom pressurized | SS||Li | LPSCl | No surface modification or interlayer was used; the current collector was plain SS | Low stack pressure (2 MPa) caused early failure due to irregular plating and Li filaments, while high pressure (20 MPa) induced electrolyte fracture, with optimal performance at 10 MPa achieving 4.56 mAh·cm−2 | [161] |
| 2025 | Pouch | Ag–C||NCA | LPSCl | Ag–C nanocomposite interlayer | 20 MPa pressure during storage reduces cathode degradation at high SOC, improving capacity retention to ~89% over 200 cycles | [171] |
| 2025 | Coin | Cu||Li | Nb–LLZO | Cu current collector (50–1200 nm) was deposited by sputtering directly onto polished LLZO (~100 nm roughness), without any additional interlayers or lithiophilic materials | Cells operated for 180 h (40 cycles) without external pressure, with 75–90% CE, until short circuit due to dendrites; Li morphology varied with Cu thickness, with preferential nucleation along polishing lines and interfacial resistance reduction from 3187 Ω to 82 Ω | [112] |
| 2025 | Pouch | Microstructured Cu||Zn | Hybrid electrolyte: Na2.99Ba0.005OCl glass ceramic ferroelectric matrix impregnated in cellulose with PVAc binder | The positive copper current collector was microstructured using colloidal lithography and O2 plasma reactive ion etching (0–15 min) to create a hexagonal pattern of micro-cones; this process increased the surface area by up to ~30% and modified the surface chemical potential | Cell performance was optimized with a 12-min etched copper collector, where its surface properties proved more critical than its increased area for boosting conductivity and capacity | [77] |
| 2025 | Coin | SS|MoS2||LiNbO3-coated NMC622 | LPSCl | MoS2 thin film on current collector forms artificial SEI (Li2S + Mo) | MoS2—15 m cell achieved 161.1 mAh·g−1, 58.9% retention (20 cycles), and 96.7% CE | [177] |
| 2025 | Coin | Stainless steel|Ag–C interlayer||NMC811 | LPSCl | Spray-printed Ag–C nanocomposite bilayer (Ag-rich at current collector and CB-rich at electrolyte) | Structured Ag–C bilayer improved initial discharge capacity to >190 mAh·g−1 and capacity retention vs. unstructured interlayer; >98% CE over 100 cycles; promoted uniform Li plating directly on CC | [101] |
| 2025 | Coin | Cu–Ag interlayer||NMC111 | LLZO | Ag interlayer (200 nm) on LLZO forms Li–Ag alloy for uniform Li plating | ~86% CE, >960 cycles in half-cell, operates up to 0.9 mA·cm−2 in full-cell, dendrite-free Li plating via Li–Ag alloy mechanism | [88] |
| 2025 | Coin | Cu||NMC811 | LPSCl | Lithiophilic Au–Sn dual-metal layer on the Cu | >400 cycles, CE > 99.5%, kinetically controlled Li plating via Au–Sn layer enabling uniform deposition and superior cyclability | [58] |
| 2025 | Coin | Cu||NMC811 | LPSCl | In situ silver exsolution from electrolyte at Cu interface | ~99.7% average CE, 100+ cycles stability at 1.2 mAh·cm−2 via in situ Ag interlayer in all-solid-state cell | [169] |
| 2025 | Pouch | Cu/ZnO + C||LFP | Li2.99Ba0.005OCl | ZnO and active carbon coating on Cu | Stable Li plating confirmed via LIBS mapping, achieving 0.16 µm Li thickness and 2.10 V discharge plateau over 219 h in anode-less cells | [31] |
| 2025 | Coin | Cu||LFP | Solid glassy electrolyte Li3PS4–P2O5 composite film | Solid glassy electrolyte film on current collector to enable dendrite-free lithium plating | >200 cycles, CE > 97%, dendrite suppression, enhanced interfacial stability | [164] |
| 2025 | Coin | Ti||LCO | LiPON | Unmodified Ti surface | Higher current densities (2.5 mA·cm−2) enable uniform Li deposition with dense columnar morphology and reduce interfacial resistance | [166] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Baptista, M.C.; Braga, M.H. Anode-Less (Anode-Free) Batteries: From Fundamental Principles to Practical Pathways Toward Solid-State Implementation. Materials 2026, 19, 1232. https://doi.org/10.3390/ma19061232
Baptista MC, Braga MH. Anode-Less (Anode-Free) Batteries: From Fundamental Principles to Practical Pathways Toward Solid-State Implementation. Materials. 2026; 19(6):1232. https://doi.org/10.3390/ma19061232
Chicago/Turabian StyleBaptista, Manuela Carvalho, and Maria Helena Braga. 2026. "Anode-Less (Anode-Free) Batteries: From Fundamental Principles to Practical Pathways Toward Solid-State Implementation" Materials 19, no. 6: 1232. https://doi.org/10.3390/ma19061232
APA StyleBaptista, M. C., & Braga, M. H. (2026). Anode-Less (Anode-Free) Batteries: From Fundamental Principles to Practical Pathways Toward Solid-State Implementation. Materials, 19(6), 1232. https://doi.org/10.3390/ma19061232







