Effect of Electrode Potential on Oxygen Adsorption and Electronic Structure on WC (0001) Surface: An Implicit Solvent DFT Study
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
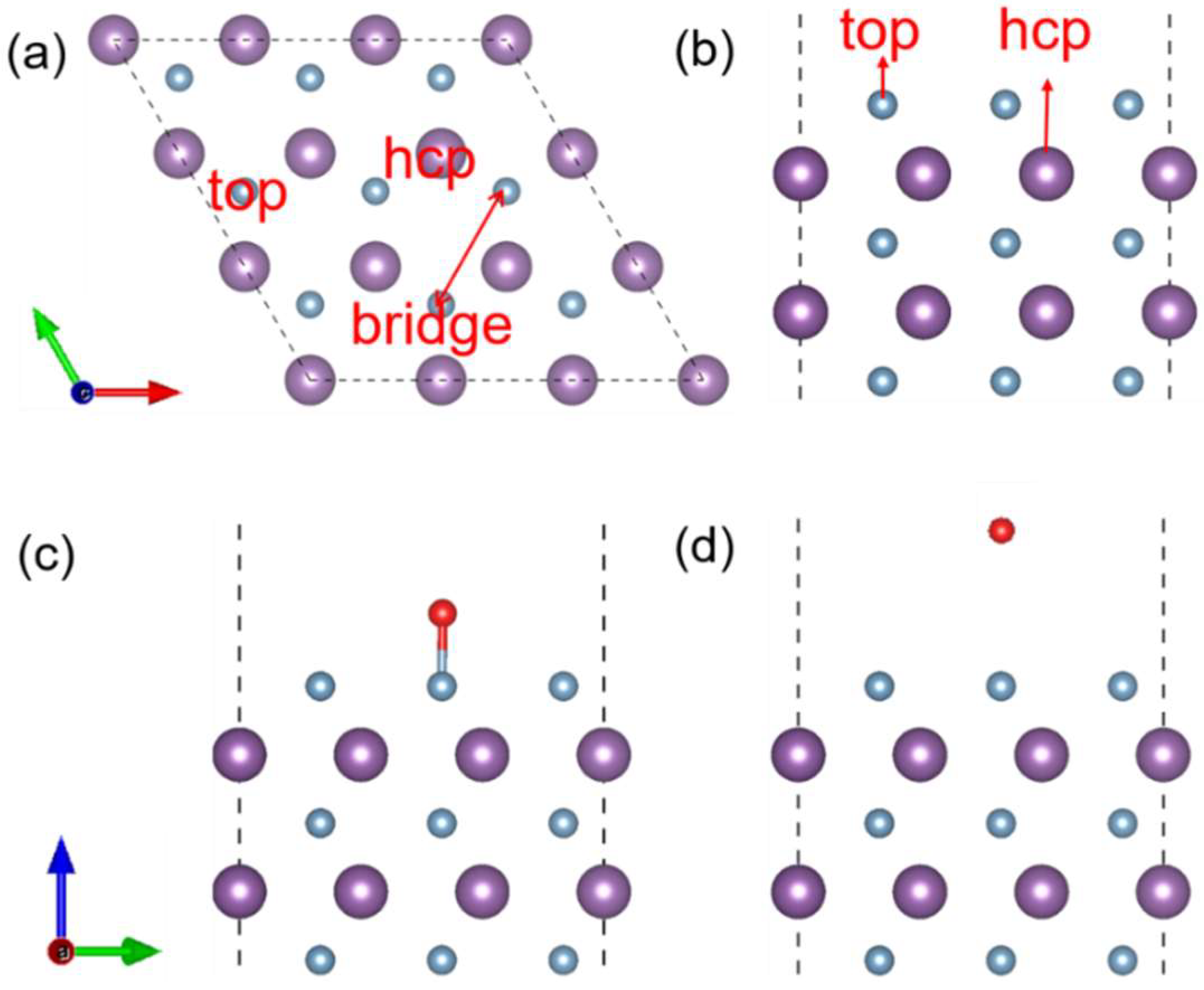
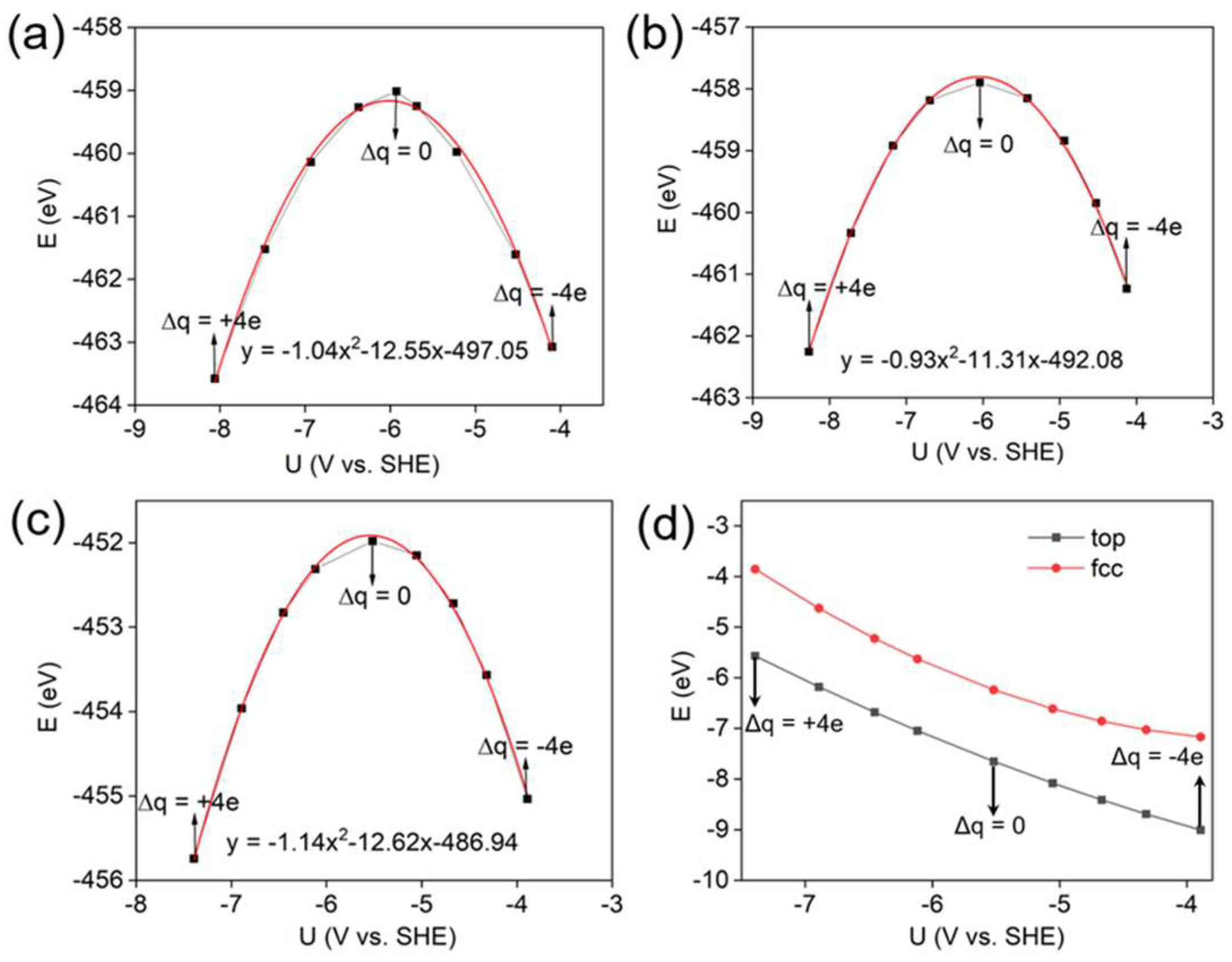
3.1. Adsorption Energy and Configuration
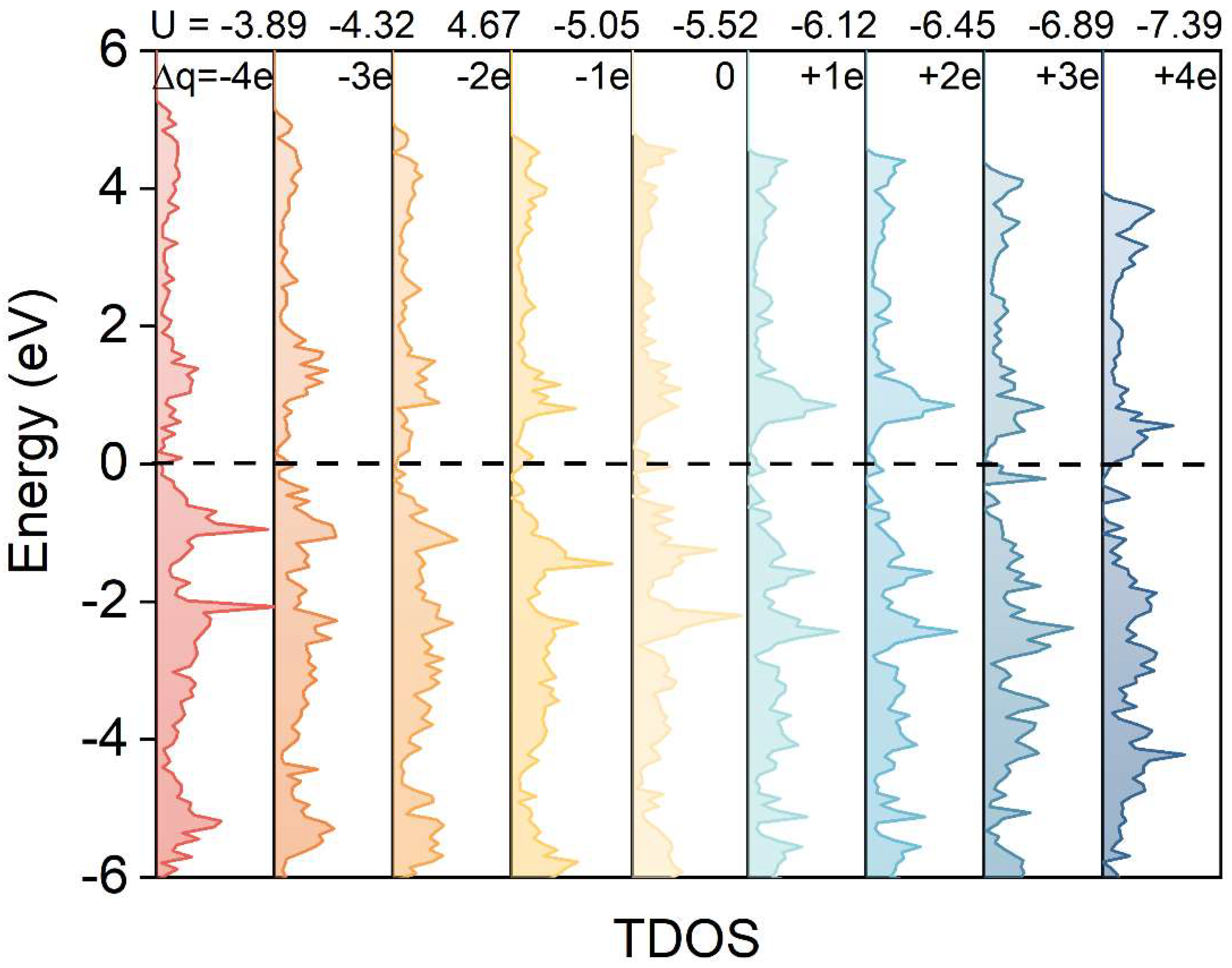
3.2. Electronic Structure of Oxygen Adsorption on the WC (0001) Surface
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Hoseini, S.; Seyedkanani, A.; Najafi, G.; Sasmito, A.; Akbarzadeh, A. Multiscale architected porous materials for renewable energy conversion and storage. Energy Storage Mater. 2023, 59, 102768. [Google Scholar] [CrossRef]
- Sun, Q.; Dai, L.; Luo, T.; Wang, L.; Liang, F.; Liu, S. Recent advances in solid-state metal–air batteries. Carbon Energy 2023, 5, e276. [Google Scholar] [CrossRef]
- Zhang, L.; Xia, Z. Mechanisms of Oxygen Reduction Reaction on Nitrogen-Doped Graphene for Fuel Cells. J. Phys. Chem. C. 2011, 115, 11170–11176. [Google Scholar] [CrossRef]
- Chen, F.; Wu, Z.; Adler, Z.; Wang, H. Stability challenges of electrocatalytic oxygen evolution reaction: From mechanistic understanding to reactor design. Joule 2021, 5, 1704–1731. [Google Scholar] [CrossRef]
- Peng, L.; Wei, Z. Catalyst Engineering for Electrochemical Energy Conversion from Water to Water: Water Electrolysis and the Hydrogen Fuel Cell. Engineering 2020, 6, 653–679. [Google Scholar] [CrossRef]
- Wang, J.; Pan, F.; Chen, W.; Li, B.; Yang, D.; Ming, P.; Wei, X.; Zhang, C. Pt-Based Intermetallic Compound Catalysts for the Oxygen Reduction Reaction: Structural Control at the Atomic Scale to Achieve a Win–Win Situation Between Catalytic Activity and Stability. Electrochem. Energy Rev. 2023, 6, 6. [Google Scholar] [CrossRef]
- Huang, L.; Zaman, S.; Tian, X.; Wang, Z.; Fang, W.; Xia, B. Advanced Platinum-Based Oxygen Reduction Electrocatalysts for Fuel Cells. Acc. Chem. Res. 2021, 54, 311–322. [Google Scholar] [CrossRef]
- Chen, C.; Rasal, A.; Chang, J.; Yu, W. Unlocking the catalytic potential of tungsten carbide coordinated with 3d transition metals in oxygen electrochemistry. Chem. Eng. J. 2024, 497, 154145. [Google Scholar] [CrossRef]
- Rafique, M.; Fu, Q.; Han, J.; Wang, R.; Yao, T.; Wang, X.; Song, B. Tungsten Carbide-Based Materials for Electrocatalytic Water Splitting: A Review. Chem. Electro. Chem 2024, 11, e202300722. [Google Scholar] [CrossRef]
- Sohail, U.; Pervaiz, E.; Ali, M.; Khosa, R.; Shakoor, A.; Abdullah, U. Role of tungsten carbide (WC) and its hybrids in electrochemical water splitting application—A comprehensive review. FlatChem 2022, 35, 100404. [Google Scholar] [CrossRef]
- Yan, D.; Xia, C.; Zhang, W.; Hu, Q.; He, C.; Xia, B.; Wang, S. Cation Defect Engineering of Transition Metal Electrocatalysts for Oxygen Evolution Reaction. Adv. Energy Mater. 2022, 12, 2202317. [Google Scholar] [CrossRef]
- Ma, Y.; Wang, J.; Yu, J.; Zhou, J.; Zhou, X.; Li, H.; He, Z.; Long, H.; Wang, Y.; Lu, P.; et al. Surface modification of metal materials for high-performance electrocatalytic carbon dioxide reduction. Matter 2021, 4, 888–926. [Google Scholar] [CrossRef]
- Yeh, K.; Janik, M.J. Density functional theory-based electrochemical models for the oxygen reduction reaction: Comparison of modeling approaches for electric field and solvent effects. J. Comput. Chem. 2011, 32, 3399–3408. [Google Scholar] [CrossRef] [PubMed]
- Kopač Lautar, A.; Hagopian, A.; Filhol, J.-S. Modeling interfacial electrochemistry: Concepts and tools. Phys. Chem. Chem. Phys. 2020, 22, 10569–10580. [Google Scholar] [CrossRef]
- Bramley, G.A.; Nguyen, M.-T.; Glezakou, V.-A.; Rousseau, R.; Skylaris, C.-K. Understanding Adsorption of Organics on Pt(111) in the Aqueous Phase: Insights from DFT Based Implicit Solvent and Statistical Thermodynamics Models. J. Chem. Theory Comput. 2022, 18, 1849–1861. [Google Scholar] [CrossRef]
- Liu, J.-C.; Luo, F.; Li, J. Electrochemical Potential-Driven Shift of Frontier Orbitals in M–N–C Single-Atom Catalysts Leading to Inverted Adsorption Energies. J. Am. Chem. Soc. 2023, 145, 25264–25273. [Google Scholar] [CrossRef]
- Zhang, J.; Yang, H.B.; Zhou, D.; Liu, B. Adsorption Energy in Oxygen Electrocatalysis. Chem. Rev. 2022, 122, 17028–17072. [Google Scholar] [CrossRef]
- Li, K.; Zeng, J.; Wang, Y.; Zhang, J.; Zhou, Y. Adsorption and migration behavior of lithium and potassium ions on Ti3C2O2 MXene by constant-potential implicit solvation. Phys. Rev. Appl. 2025, 23, 014011. [Google Scholar] [CrossRef]
- Li, K.; Ou, Y.; Wang, Y.; Zhang, J.; Zhou, Y. Single-atom lead ion adsorption behavior on Ti2CO2 MXene under different electrode potentials. Phys. Chem. Chem. Phys. 2025, 27, 3083–3088. [Google Scholar] [CrossRef]
- Mathew, K.; Sundararaman, R.; Letchworth-Weaver, K.; Arias, T.A.; Hennig, R.G. Implicit solvation model for density-functional study of nanocrystal surfaces and reaction pathways. J. Chem. Phys. 2014, 14, 084106. [Google Scholar] [CrossRef]
- Hagopian, A.; Doublet, M.-L.; Filhol, J.-S.; Binninger, T. Advancement of the Homogeneous Background Method for the Computational Simulation of Electrochemical Interfaces. J. Chem. Theory Comput. 2022, 18, 1883–1893. [Google Scholar] [CrossRef]
- Wang, V.; Xu, N.; Liu, J.-C.; Tang, G.; Geng, W.-T. VASPKIT: A User-Friendly Interface Facilitating High-Throughput Computing and Analysis Using VASP Code. Comput. Phys. Commun. 2021, 267, 108033. [Google Scholar] [CrossRef]
- Momma, K.; Izumi, F. VESTA 3 for Three-Dimensional Visualization of Crystal, Volumetric and Morphology Data. J. Appl. Crystallogr. 2011, 44, 1272–1276. [Google Scholar] [CrossRef]
- Deringer, V.L.; Tchougréeff, A.L.; Dronskowski, R. Crystal Orbital Hamilton Population (COHP) Analysis As Projected from Plane-Wave Basis Sets. J. Phys. Chem. A 2011, 115, 5461–5466. [Google Scholar] [CrossRef] [PubMed]
- Maintz, S.; Deringer, V.L.; Tchougréeff, A.L.; Dronskowski, R. LOBSTER: A Tool to Extract Chemical Bonding from Plane-Wave Based DFT. J. Comput. Chem. 2016, 37, 1030–1035. [Google Scholar] [CrossRef]
- Li, D.; Zhao, L.; Wang, J.; Yang, C. Tailoring the d-Band Center over Isomorphism Pyrite Catalyst for Optimized Intrinsic Affinity to Intermediates in Lithium–Oxygen Batteries. Adv. Energy Mater. 2023, 13, 2204057. [Google Scholar] [CrossRef]
- Kim, D.; Shi, J.; Liu, Y. Substantial impact of charge on electrochemical reactions of two-dimensional materials. J. Am. Chem. Soc. 2018, 140, 9127–9131. [Google Scholar] [CrossRef] [PubMed]
- Kundu, S.; Bhattacharjee, S.; Lee, S.-C.; Jain, M. Population analysis with Wannier orbitals. J. Chem. Phys. 2021, 154, 104111. [Google Scholar] [CrossRef]
- George, J.; Petretto, G.; Naik, A.; Esters, M.; Jackson, A.J.; Nelson, R.; Dronskowski, R.; Rignanese, G.M.; Hautier, G. Automated bonding analysis with crystal orbital Hamilton populations. ChemPlusChem 2022, 87, e202200123. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Wang, L.; Wei, J.; Yin, C.; Liu, Y.; Bai, F.; Dong, B. Effect of Electrode Potential on Oxygen Adsorption and Electronic Structure on WC (0001) Surface: An Implicit Solvent DFT Study. Materials 2026, 19, 1129. https://doi.org/10.3390/ma19061129
Wang L, Wei J, Yin C, Liu Y, Bai F, Dong B. Effect of Electrode Potential on Oxygen Adsorption and Electronic Structure on WC (0001) Surface: An Implicit Solvent DFT Study. Materials. 2026; 19(6):1129. https://doi.org/10.3390/ma19061129
Chicago/Turabian StyleWang, Li, Jiawei Wei, Chaofan Yin, Ying Liu, Fan Bai, and Binbin Dong. 2026. "Effect of Electrode Potential on Oxygen Adsorption and Electronic Structure on WC (0001) Surface: An Implicit Solvent DFT Study" Materials 19, no. 6: 1129. https://doi.org/10.3390/ma19061129
APA StyleWang, L., Wei, J., Yin, C., Liu, Y., Bai, F., & Dong, B. (2026). Effect of Electrode Potential on Oxygen Adsorption and Electronic Structure on WC (0001) Surface: An Implicit Solvent DFT Study. Materials, 19(6), 1129. https://doi.org/10.3390/ma19061129




