CQD-Modified SrTiO3 for Enhanced Photocatalytic CO2 Reduction to Methane
Abstract
1. Introduction
2. Experimental and Theoretical Section
2.1. Chemicals
2.2. Synthesis of SrTiO3/CQD Nanomaterials
2.3. Characterization
2.4. CO2 Photodissociation Test
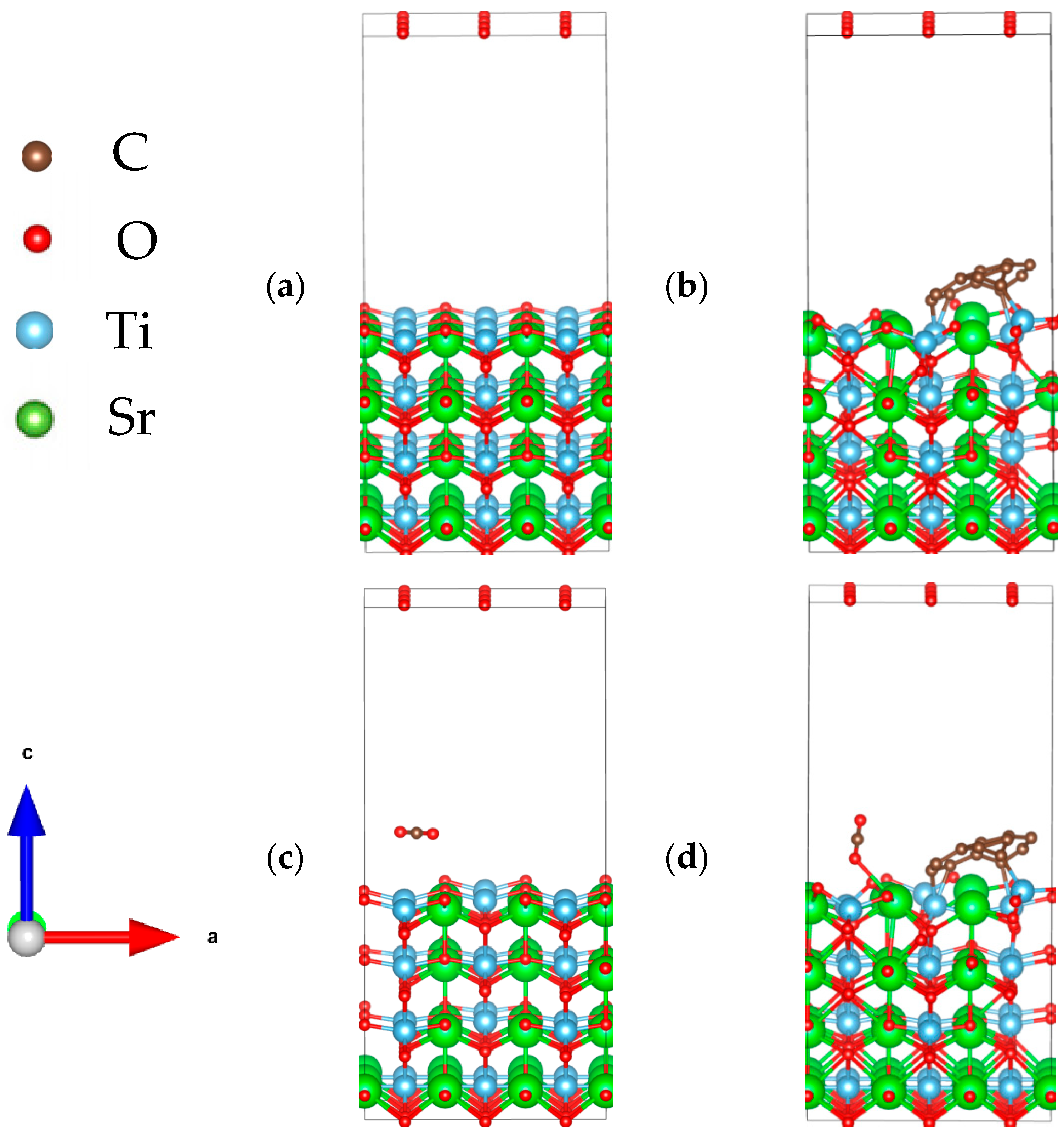
2.5. Computational Method and Details
3. Results and Discussion
3.1. Phase Structure of SrTiO3/CQD Nanomaterials
3.2. Morphology and Composition of SrTiO3/CQD Nanomaterials
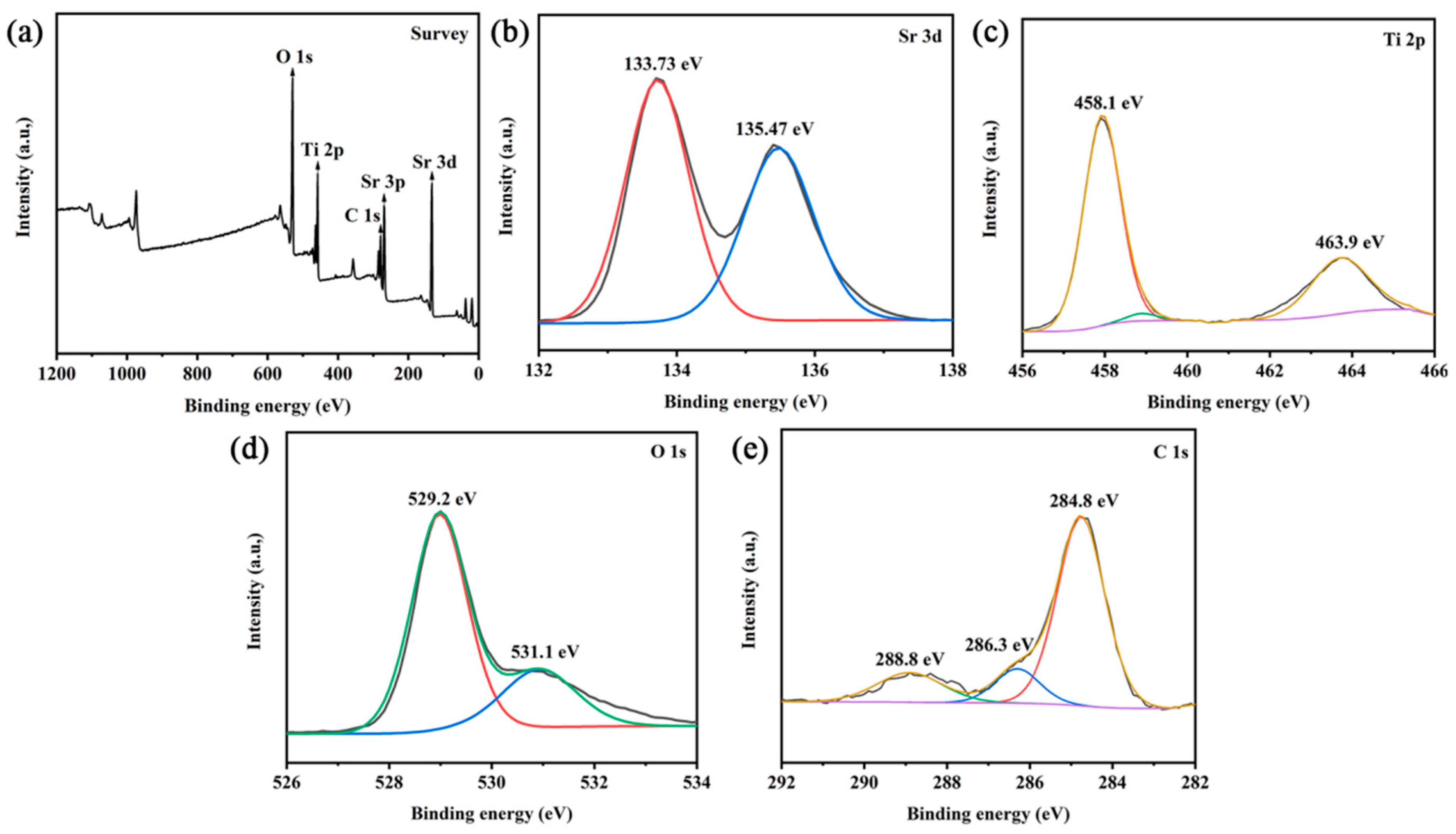
3.3. Chemical Valence State of SrTiO3/CQD Composite
3.4. Performance Evaluation of Photocatalyst
3.5. Optical Properties and Electrochemical Analysis
3.6. Adsorption Properties of CO2 on the CQD-Modified SrTiO3 Surface
3.7. Photocatalytic Schematic Illustration of SrTiO3/CQDs
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Aresta, M.; Dibenedetto, A.; Angelini, A. Catalysis for the valorization of exhaust carbon: From CO2 to chemicals, materials, and fuels. Technological use of CO2. Chem. Rev. 2014, 114, 1709–1742. [Google Scholar] [CrossRef]
- Hussain, I.; Jalil, A.A.; Hassan, N.S.; Hamid, M. Recent advances in catalytic systems for CO2 conversion to substitute natural gas (SNG): Perspective and challenges. J. Energy Chem. 2021, 62, 377–407. [Google Scholar] [CrossRef]
- Jiang, H.; Geng, Y.; Tian, X.; Zhang, X.; Chen, W.; Gao, Z. Uncovering CO2 emission drivers under regional industrial transfer in China’s Yangtze River Economic Belt: A multi-layer LMDI decomposition analysis. Front. Energy 2021, 15, 292–307. [Google Scholar] [CrossRef]
- Kiatphuengporn, S.; Chareonpanich, M.; Limtrakul, J. Effect of unimodal and bimodal MCM-41 mesoporous silica supports on activity of Fe–Cu catalysts for CO2 hydrogenation. Chem. Eng. J. 2014, 240, 527–533. [Google Scholar] [CrossRef]
- Akshay, V.R.; Arun, B.; Mandal, G.; Mutta, G.R.; Chanda, A.; Vasundhara, M. Observation of optical band-gap narrowing and enhanced magnetic moment in Co-doped Sol–Gel-derived anatase TiO2 nanocrystals. J. Phys. Chem. C 2018, 122, 26592–26604. [Google Scholar] [CrossRef]
- Abdullah, H.; Khan, M.M.R.; Ong, H.R.; Yaakob, Z. Modified TiO2 photocatalyst for CO2 photocatalytic reduction: An overview. J. CO2 Util. 2017, 22, 15–32. [Google Scholar] [CrossRef]
- Xiong, Z.; Kuang, C.C.; Lin, K.Y.; Lei, Z.; Chen, X.; Gong, B.; Yang, J.; Zhao, Y.; Zhang, J.; Xia, B.; et al. Enhanced CO2 photocatalytic reduction through simultaneously accelerated H2 evolution and CO2 hydrogenation in a twin photoreactor. J. CO2 Util. 2018, 24, 500–508. [Google Scholar] [CrossRef]
- Iwashina, K.; Kudo, A. Rh-doped SrTiO3 photocatalyst electrode showing cathodic photocurrent for water splitting under visible-light irradiation. J. Am. Chem. Soc. 2011, 133, 13272–13275. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.; Ueno, K.; Mori, Y.; Shi, X.; Oshikiri, T.; Murakoshi, K.; Inoue, H.; Misawa, H. Plasmon-assisted water splitting using two sides of the same SrTiO3 single-crystal substrate: Conversion of visible light to chemical energy. Angew. Chem. Int. Ed. 2014, 53, 10350–10354. [Google Scholar] [CrossRef]
- Bi, Y.; Ehsan, M.F.; Huang, Y.; Jin, J.; He, T. Synthesis of Cr-doped SrTiO3 photocatalyst and its application in visible-light-driven transformation of CO2 into CH4. J. CO2 Util. 2015, 12, 43–48. [Google Scholar] [CrossRef]
- Wu, X.; Wang, C.; Wei, Y.; Xiong, J.; Zhao, Y.; Zhao, Z.; Liu, J.; Li, J. Multifunctional photocatalysts of Pt-decorated 3DOM perovskite-type SrTiO3 with enhanced CO2 adsorption and photoelectron enrichment for selective CO2 reduction with H2O to CH4. J. Catal. 2019, 377, 309–321. [Google Scholar] [CrossRef]
- Almaev, A.V.; Karipbayev, Z.T.; Kakimov, A.B.; Yakovlev, N.N.; Kukenov, O.I.; Korchemagin, A.O.; Akmetova-Abdik, G.A.; Kumarbekov, K.K.; Zhunusbekov, A.M.; Mochalov, L.A.; et al. High-Temperature Methane Sensors Based on ZnGa2O4:Er Ceramics for Combustion Monitoring. Technologies 2025, 13, 286. [Google Scholar] [CrossRef]
- Jiao, M.-Z.; Chen, X.-Y.; Hu, K.-X.; Qian, D.-Y.; Zhao, X.-H.; Ding, E.-J. Recent developments of nanomaterials-based conductive type methane sensors. Rare Met. 2021, 40, 1515–1527. [Google Scholar] [CrossRef]
- Ateia, E.E.; Rabie, O.; Mohamed, A.T. Assessment of the correlation between optical properties and CQD preparation approaches. Eur. Phys. J. Plus 2024, 139, 24. [Google Scholar] [CrossRef]
- Subramani, M.C.; Budiman, I.; Subyakto, S.; Chitraningrum, N.; Widyaningrum, B.A.; Saputra, N.A.; Fauzi, A.A.B.; Hata, T.; Jebiti, H. A review on one-pot multicomponent organic reactions using carbon quantum dots as versatile heterogeneous catalyst. Top. Catal. 2025, 68, 2296–2331. [Google Scholar] [CrossRef]
- Ye, M.; Meng, L.F. Preparation and sensing study of Houttuynia cordata-based carbon quantum dots. Molecules 2025, 30, 3668. [Google Scholar] [CrossRef] [PubMed]
- Gawal, P.M.; Golder, A.K. Vegetal route for synthesis of CQDs/CdS nanocomposites for photocatalytic reduction of CO2 to methanol under visible light. Colloids Surf. A Physicochem. Eng. Asp. 2024, 683, 133068. [Google Scholar] [CrossRef]
- Brik, M.G.; Srivastava, A.M.; Popov, A. A few common misconceptions in the interpretation of experimental spectroscopic data. Opt. Mater. 2022, 127, 112276. [Google Scholar] [CrossRef]
- IEC 60736:1982; Testing Equipment for Electrical Energy Meters. International Electrotechnical Commission: Geneva, Switzerland, 1982.
- Kresse, G.; Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 1996, 54, 11169. [Google Scholar] [CrossRef]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized Gradient Approximation Made Simple. Phy. Rev. Lett. 1996, 77, 3865. [Google Scholar] [CrossRef] [PubMed]
- Ireta, J.; Neugebauer, J.; Scheffler, M. On the accuracy of DFT for describing hydrogen bonds: Dependence on the bond directionality. J. Phys. Chem. A 2004, 108, 5692–5698. [Google Scholar] [CrossRef]
- Okazaki, A.; Kawaminami, M. Lattice constant of strontium titanate at low temperatures. Mater. Res. Bull. 1973, 8, 545–550. [Google Scholar] [CrossRef]
- Politi, J.R.S.; Martins, J.B.L.; Cabral, B.J.C. A first principles approach to the interactions of alkali metal atoms with carbon quantum dots. Comput. Mater. Sci. 2021, 197, 110614. [Google Scholar] [CrossRef]
- Liu, S.; Liu, Z.; Li, Q.; Xia, H.; Yang, W.; Wang, R.; Li, Y.; Zhao, H.; Tian, B. Facile synthesis of carbon dots from wheat straw for colorimetric and fluorescent detection of fluoride and cellular imaging. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2021, 246, 118964. [Google Scholar] [CrossRef]
- Huang, K.; Lin, L.; Zhang, L.; Zhao, M.; Dai, X.; Jiang, Y.; Yang, R.; Liao, J.; Zhang, Y.; Wang, Y. Rapid synthesis of ultra-bright blue and cyan CQDs fluorescent powders based on chemical dispersion and concentration effects. Ceram. Int. 2024, 50, 4046–4052. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhong, L.; Duan, D. Single-step hydrothermal synthesis of strontium titanate nanoparticles from crystalline anatase titanium dioxide. Ceram. Int. 2015, 41, 13516–13524. [Google Scholar] [CrossRef]
- Xu, N.; Gao, S.; Xu, C.; Fang, Y.; Xu, L.; Zhang, W. Carbon quantum dots derived from waste acorn cups and its application as an ultraviolet absorbent for polyvinyl alcohol film. Appl. Surf. Sci. 2021, 556, 149774. [Google Scholar] [CrossRef]
- Zhou, M.; Chen, J.; Zhang, Y.; Jiang, M.; Xu, S.; Liang, Q.; Li, Z. Shape-controlled synthesis of golf-like, star-like, urchin-like and flower-like SrTiO3 for highly efficient photocatalytic degradation and H2 production. J. Alloys Compd. 2020, 817, 152796. [Google Scholar] [CrossRef]
- Chen, X.; Mao, S.S. Titanium dioxide nanomaterials: Synthesis, properties, modifications, and applications. Chem. Rev. 2007, 107, 2891–2959. [Google Scholar] [CrossRef]
- Jiang, X.; Zhang, Y.; Jiang, J.; Rong, Y.; Wang, Y.; Wu, Y.; Pan, C. Characterization of oxygen vacancy associates within hydrogenated TiO2: A positron annihilation study. J. Phys. Chem. C 2012, 116, 22619–22624. [Google Scholar] [CrossRef]
- Lin, Y.; Wu, S.; Li, X.; Wu, X.; Yang, C.; Zeng, G.; Peng, Y.; Zhou, Q.; Lu, L. Microstructure and performance of Z-scheme photocatalyst of silver phosphate modified by MWCNTs and Cr-doped SrTiO3 for malachite green degradation. Appl. Catal. B Environ. 2018, 227, 557–570. [Google Scholar] [CrossRef]
- Miot, C.; Proust, C.; Husson, E.; Blondiaux, G.; Coutures, J. Ageing influence on residual carbon content in different grain-sized BaTiO3 ceramics analyzed by 12C(d,p)13C nuclear method. J. Eur. Ceram. Soc. 1997, 17, 1335–1340. [Google Scholar] [CrossRef]
- Chen, F.; Liu, L.-L.; Zhang, Y.-J.; Wu, J.-H.; Huang, G.-X.; Yang, Q.; Chen, J.-J.; Yu, H.-Q. Enhanced full solar spectrum photocatalysis by nitrogen-doped graphene quantum dots decorated BiO2−x nanosheets: Ultrafast charge transfer and molecular oxygen activation. Appl. Catal. B Environ. 2020, 277, 119218. [Google Scholar] [CrossRef]
- Li, M.; Zhang, Y.; Li, X.; Wang, Y.; Dong, F.; Ye, L.; Yu, S.; Huang, H. Nature-derived approach to oxygen and chlorine dual-vacancies for efficient photocatalysis and photoelectrochemistry. ACS Sustain. Chem. Eng. 2018, 6, 2395–2406. [Google Scholar] [CrossRef]
- Bajiri, M.A.M.; Khan, N.; Diaz, J.C.C.A.; Avansi, W., Jr.; Gouvêa, D.; Gonçalves, R.V. Engineering Stable Cu-Doped SrTiO3 Perovskites for Enhanced Photocatalytic CO2 Reduction. Inorg. Chem. 2025, 64, 15370–15380. [Google Scholar] [CrossRef] [PubMed]
- Boga, B.; Moustakas, N.G.; Han, Y.; Jiao, H.; Kreyenschulte, C.; Naliwajko, P.; Duong, T.T.H.; Ding, S.; Ngo, A.B.; Hezam, A.; et al. Design of SrTiO3-based catalysts for photocatalytic CO2 reduction. Catal. Sci. Technol. 2024, 14, 3459–3472. [Google Scholar] [CrossRef]
- Fang, S.; Xia, Y.; Lv, K.; Li, Q.; Sun, J.; Li, M. Effect of carbon-dots modification on the structure and photocatalytic activity of g-C3N4. Appl. Catal. B Environ. 2016, 185, 225–232. [Google Scholar] [CrossRef]
- Huang, X.; Zhang, H.; Zhao, J.; Jiang, D.; Zhan, Q. Carbon quantum dot (CQD)-modified Bi3O4Br nanosheets possessing excellent photocatalytic activity under simulated sunlight. Mater. Sci. Semicond. Process. 2021, 122, 105489. [Google Scholar] [CrossRef]
- Liu, Z.; Liu, C.; Zheng, L.; Chen, F.; Qian, J.; Qiu, Y.; Meng, X.; Chen, Z. Progress on Synthesis of CQDs/g-C3N4 Composites and Their Photocatalytic Performance. Mater. Mech. Eng. 2024, 48, 1–10. [Google Scholar]
- Ji, M.; Liu, L.Y.; Di, J.; Chen, R.; Chen, Z.; Xia, J.; Li, H. N-CQDs accelerating surface charge transfer of Bi4O5I2 hollow nanotubes with broad spectrum photocatalytic activity. Appl. Catal. B Environ. 2018, 237, 1033–1043. [Google Scholar] [CrossRef]
- Zhao, Y.; Yang, D.; Yu, C.; Yan, H. A review on photocatalytic CO2 reduction of g-C3N4 and g-C3N4-based photocatalysts modified by CQDs. J. Environ. Chem. Eng. 2025, 13, 115348. [Google Scholar] [CrossRef]
- Li, Q.; Wang, S.; Sun, Z.; Tang, Q.; Liu, Y.; Wang, L.; Wang, H.; Wu, Z. Enhanced CH4 selectivity in CO2 photocatalytic reduction over carbon quantum dots decorated and oxygen doping g-C3N4. Nano Res. 2019, 12, 2749–2759. [Google Scholar] [CrossRef]
- Indrakanti, V.P.; Kubicki, J.D.; Schobert, H.H. Photoinduced activation of CO2 on Ti-based heterogeneous catalysts: Current state, chemical physics-based insights and outlook. Energy Environ. Sci. 2009, 2, 745–758. [Google Scholar] [CrossRef]
- Wang, R.; Lu, K.-Q.; Tang, Z.-R.; Xu, Y.-J. Recent progress in carbon quantum dots: Synthesis, properties and applications in photocatalysis. J. Mater. Chem. A 2017, 5, 3717–3734. [Google Scholar] [CrossRef]
- Tan, L.-L.; Ong, W.-J.; Chai, S.-P.; Goh, B.T.; Mohamed, A.R. Visible-light-active oxygen-rich TiO2 decorated 2D graphene oxide with enhanced photocatalytic activity toward carbon dioxide reduction. Appl. Catal. B Environ. 2015, 179, 160–170. [Google Scholar] [CrossRef]
- Kulandaivalu, T.; Rashid, S.A.; Sabli, N.; Tan, T.L. Visible light assisted photocatalytic reduction of CO2 to ethane using CQDs/Cu2O nanocomposite photocatalyst. Diam. Relat. Mater. 2019, 91, 64–73. [Google Scholar] [CrossRef]







Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Sun, S.; Liu, Y.; Hu, C.; Zhang, Y.; Zhong, Y.; Wang, D. CQD-Modified SrTiO3 for Enhanced Photocatalytic CO2 Reduction to Methane. Materials 2026, 19, 1075. https://doi.org/10.3390/ma19061075
Sun S, Liu Y, Hu C, Zhang Y, Zhong Y, Wang D. CQD-Modified SrTiO3 for Enhanced Photocatalytic CO2 Reduction to Methane. Materials. 2026; 19(6):1075. https://doi.org/10.3390/ma19061075
Chicago/Turabian StyleSun, Shaohang, Yize Liu, Chaohao Hu, Yanli Zhang, Yan Zhong, and Dianhui Wang. 2026. "CQD-Modified SrTiO3 for Enhanced Photocatalytic CO2 Reduction to Methane" Materials 19, no. 6: 1075. https://doi.org/10.3390/ma19061075
APA StyleSun, S., Liu, Y., Hu, C., Zhang, Y., Zhong, Y., & Wang, D. (2026). CQD-Modified SrTiO3 for Enhanced Photocatalytic CO2 Reduction to Methane. Materials, 19(6), 1075. https://doi.org/10.3390/ma19061075





