Magnetic Nanostructures for the Removal of Emerging Organic and Inorganic Pollutants: An Overview of Applications in Contaminated Water
Abstract
1. Introduction
2. Magnetic Properties of Nanomaterials
3. Environmental Applications of Magnetic Nanomaterials
4. Synthesis and Processing of Magnetic Nanoparticles
4.1. Physical Methods
4.1.1. Ball Milling Method
4.1.2. Laser Ablation Method
4.1.3. Electrical Wire Explosion Method
4.1.4. Electron Beam Lithography Method
4.2. Chemical Methods
4.2.1. Precipitation and Coprecipitation Method
4.2.2. Sol–Gel Method
4.2.3. Hydrothermal Method
4.2.4. Solvothermal Method
4.2.5. Microemulsion Method
4.2.6. Polyol Method
4.2.7. Thermal Decomposition Method
4.2.8. Spray Pyrolysis Method
4.2.9. Pechini Method
4.2.10. Self-Assembly Method
4.2.11. Laser Pyrolysis Method
4.2.12. Oxidation-Reduction Method
4.2.13. Microwave Method
4.2.14. Arc Discharge Method
4.2.15. Sonochemical Method
4.2.16. Combustion Method
4.2.17. Chemical Vapor Deposition
5. Mechanisms of Contaminant Removal by Magnetic Nanomaterials
6. Strategies for Removing Contaminants
7. Classification of Emerging Pollutants
7.1. Organic Pollutants
7.2. Inorganic Contaminants
8. Polymeric Nanofibers with Magnetic Nanoparticles
8.1. Electrospinning Technique
8.2. Self-Assembly
8.3. Phase Separation
8.4. Template Synthesis
8.5. Drawing
8.6. Centrifugal Spinning
9. Isotherms for Equilibrium Sorption Measurements
9.1. Freundlich Model
9.2. Langmuir Model
9.3. Dubinin-Radushkevich Model
9.4. Temkin Model
9.5. Sips Model
10. Adsorption Kinetics Models
10.1. Pseudo-First-Order (PFO) Model
10.2. Pseudo-Second-Order (PSO) Model
10.3. Intra-Particle Diffusion Model
11. Future Perspectives
12. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MNMs | Magnetic nanomaterials |
| NMs | Nanomaterials |
| MNPs | Magnetic nanoparticles |
| NPs | Nanoparticles |
| PFOA | Perfluorooctanoic Acid |
| (e−-h+) | electron–hole pairs |
| NOM | Natural Organic Matter |
| F | Freundlich model |
| L | Langmuir model |
| D-R | Dubinin-Radushkevich model |
| T | Temkin model |
| S | Sips model |
| R-P | Redlich-Peterson |
| Amount of pollutant adsorbed per unit mass of adsorbent at equilibrium | |
| Equilibrium concentration of the pollutant in solution | |
| PFO | Pseudo-First-Order Model |
| PSO | Pseudo-Second-Order |
References
- Israilova, E.; Voronina, A.; Shatila, K. Impact of water scarcity on socio-economic development. E3S Web Conf. 2023, 458, 08027. [Google Scholar] [CrossRef]
- Vallinayagam, S.; Rajendran, K.; Lakkaboyana, S.K.; Remya, S.K.; Sharma, V.K.; Kumar, V.; Venkateswarlu, K.; Koduru, J.R. Recent developments in magnetic nanoparticles and nano-composites for wastewater treatment. J. Environ. Chem. Eng. 2021, 9, 106553. [Google Scholar] [CrossRef]
- Khan, I.; Saeed, K.; Khan, I. Nanoparticles: Properties, applications and toxicities. Arab. J. Chem. 2019, 12, 908–931. [Google Scholar] [CrossRef]
- Sung, H.; Rudowicz, C. Physics behind the magnetic hysteresis loop—A survey of misconceptions in magnetism literature. J. Magn. Magn. Mater. 2003, 260, 250–260. [Google Scholar] [CrossRef]
- Martínez-Pedrero, F. Static and dynamic behavior of magnetic particles at fluid interfaces. Adv. Colloid Interface Sci. 2020, 284, 102233. [Google Scholar] [CrossRef]
- Kotnala, R.K.; Shah, J. Ferrite Materials. In Handbook of Magnetic Materials; Elsevier: Amsterdam, The Netherlands, 2015; pp. 291–379. [Google Scholar] [CrossRef]
- Cullity, B.D.; Graham, C.D. Introduction to Magnetic Materials, 2nd ed.; Wiley-Blackwell: Chichester, UK, 2007. [Google Scholar]
- Yamato, M.; Kimura, T. Magnetic Processing of Diamagnetic Materials. Polymers 2020, 12, 1491. [Google Scholar] [CrossRef]
- Amorim, C.O. A Compendium of Magnetic Nanoparticle Essentials: A Comprehensive Guide for Beginners and Experts. Pharmaceutics 2025, 17, 137. [Google Scholar] [CrossRef]
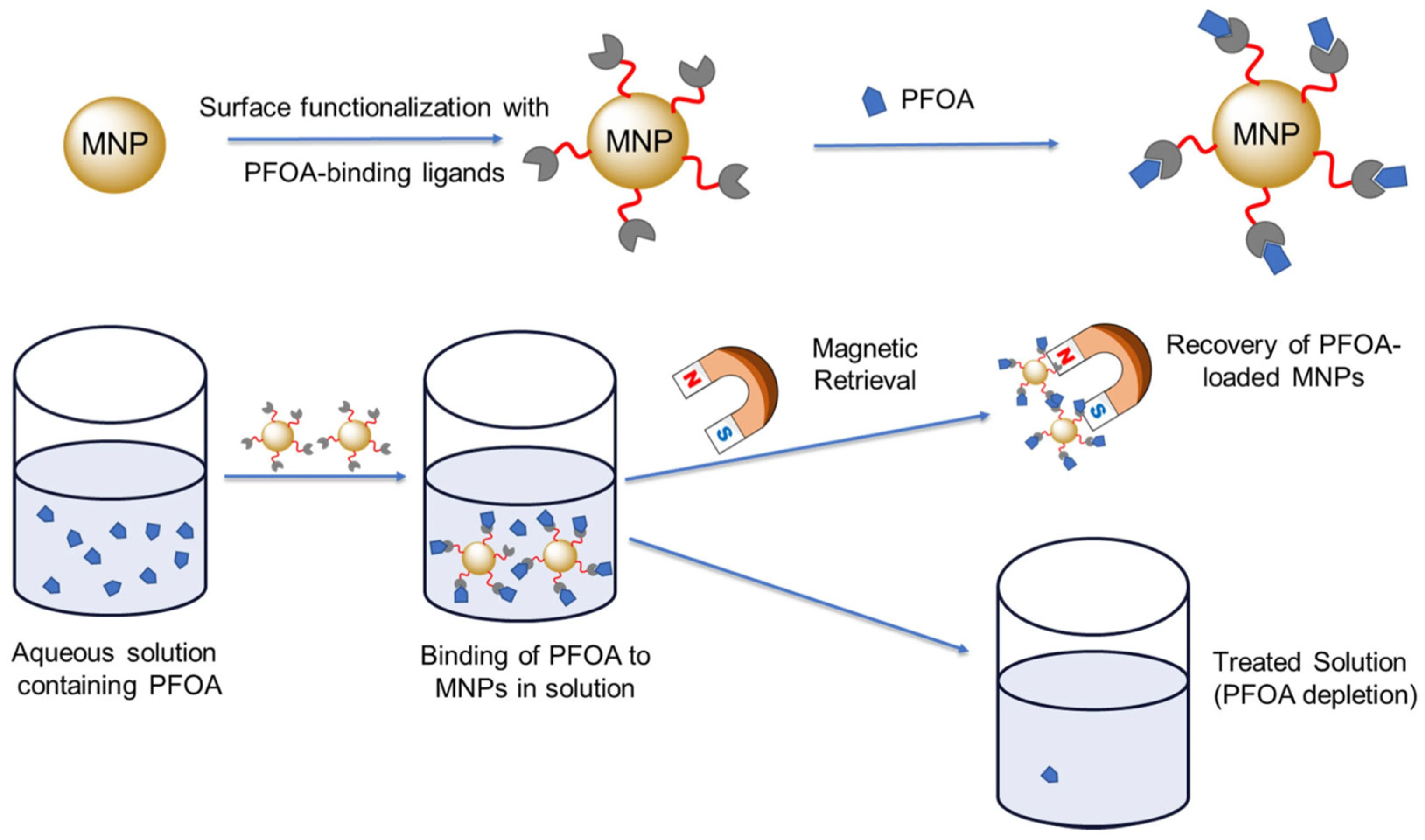
- Zhang, Y.; Ortiz, J.; He, S.; Li, X.; Kaur, B.; Cao, B.; Seiden, Z.; Wu, S.; Wei, H. Magnetically Retrievable Nanoparticles with Tailored Surface Ligands for Investigating the Interaction and Removal of Water-Soluble PFASs in Natural Water Matrices. Sensors 2025, 25, 4353. [Google Scholar] [CrossRef]
- Reddy, L.H.; Arias, J.L.; Nicolas, J.; Couvreur, P. Magnetic nanoparticles: Design and characterization, toxicity and biocompatibility, pharmaceutical and biomedical applications. Chem. Rev. 2012, 112, 5818–5878. [Google Scholar] [CrossRef]
- Patankar, K.K.; Jadhav, P.; Gayakvad, K. Introduction and applications of magnetic nanoparticles. In Fundamentals and Industrial Applications of Magnetic Nanoparticles; Elsevier: Amsterdam, The Netherlands, 2022; pp. 3–39. [Google Scholar] [CrossRef]
- Velásquez, A.; Urquijo, J. Synthesis and characterization of magnetite-maghemite nanoparticles in presence of polyethylene glycol obtained by mechanical milling. Mater. Sci. Eng. B 2021, 263, 114873. [Google Scholar] [CrossRef]
- Bedoya, P.A.C.; Botta, P.M.; Bercoff, P.G.; Fanovich, M.A. Influence of the milling materials on the mechanochemical synthesis of magnetic iron oxide nanoparticles. J. Alloys Compd. 2023, 939, 168720. [Google Scholar] [CrossRef]
- Zhang, D.; Gökce, B. Perspective of laser-prototyping nanoparticle-polymer composites. Appl. Surf. Sci. 2017, 392, 991–1003. [Google Scholar] [CrossRef]
- Piotto, V.; Litti, L.; Omelyanchik, A.; Martucci, A.; Riello, P.; Peddis, D.; Meneghetti, M. Synthesis of magnetic nanoparticles by laser ablation of strontium ferrite under water and their characterization by optically detected magnetophoresis supported by BEM calculations. J. Mater. Chem. C Mater. Opt. Electron. Devices 2022, 10, 3819–3825. [Google Scholar] [CrossRef]
- Kotov, Y.A. Electric Explosion of Wires as a Method for Preparation of Nanopowders. J. Nanoparticle Res. 2003, 5, 539–550. [Google Scholar] [CrossRef]
- Song, K.; Kim, W.; Suh, C.-Y.; Shin, D.; Ko, K.-S.; Ha, K. Magnetic iron oxide nanoparticles prepared by electrical wire explosion for arsenic removal. Powder Technol. 2013, 246, 572–574. [Google Scholar] [CrossRef]
- Tseng, A.A.; Chen, K.; Chen, C.D.; Ma, K.J. Electron beam lithography in nanoscale fabrication: Recent development. IEEE Trans. Electron. Packag. Manuf. 2003, 26, 141–149. [Google Scholar] [CrossRef]
- Jia, C.-J.; Sun, L.-D.; Luo, F.; Han, X.-D.; Heyderman, L.J.; Yan, Z.-G.; Yan, C.-H.; Zheng, K.; Zhang, Z.; Takano, M.; et al. Large-Scale Synthesis of Single-Crystalline Iron Oxide Magnetic Nanorings. J. Am. Chem. Soc. 2008, 130, 16968–16977. [Google Scholar] [CrossRef]
- Tan, P.T.; Hien, L.T.T.; Anh, N.N.; Minh, P.N.; Van Trinh, P.; Van Hao, N. Graphene oxide–carbon nanotube-magnetite nanocomposites for efficient arsenic removal from aqueous solutions. RSC Adv. 2025, 15, 20792–20809. [Google Scholar] [CrossRef]
- Sajjad, A.; Hussain, S.; Jaffari, G.H.; Hanif, S.; Qureshi, M.N.; Zia, M. Fabrication of Hematite (α-Fe2O3) nanoparticles under different spectral lights transforms physio chemical, biological, and nanozymatic properties. Nano Trends 2023, 2, 10001. [Google Scholar] [CrossRef]
- Parashar, M.; Shukla, V.K.; Singh, R. Metal oxides nanoparticles via sol–gel method: A review on synthesis, characterization and applications. J. Mater. Sci. Mater. Electron. 2020, 31, 3729–3749. [Google Scholar] [CrossRef]
- Tahir, M.; Fakhar-E-Alam, M.; Atif, M.; Mustafa, G.; Ali, Z. Investigation of optical, electrical and magnetic properties of hematite α-Fe2O3 nanoparticles via sol-gel and co-precipitation method. J. King Saud Univ. Sci. 2023, 35, 102695. [Google Scholar] [CrossRef]
- Rabenau, A. The Role of Hydrothermal Synthesis in Preparative Chemistry. Angew. Chem. Int. Ed. Engl. 1985, 24, 1026–1040. [Google Scholar] [CrossRef]
- da Silva, A.O.; Campos, A.F.C.; Rodrigues, M.O.; Sousa, M.H. Tuning magnetic and luminescent properties of iron oxide@C nanoparticles from hydrothermal synthesis: Influence of precursor reagents. Surf. Interfaces 2023, 36, 102624. [Google Scholar] [CrossRef]
- Li, C.; Wei, Y.; Liivat, A.; Zhu, Y.; Zhu, J. Microwave-solvothermal synthesis of Fe3O4 magnetic nanoparticles. Mater. Lett. 2013, 107, 23–26. [Google Scholar] [CrossRef]
- Sahadevan, J.; Sojiya, R.; Padmanathan, N.; Kulathuraan, K.; Shalini, M.; Sivaprakash, P.; Muthu, S.E. Magnetic property of Fe2O3 and Fe3O4 nanoparticle prepared by solvothermal process. Mater. Today Proc. 2022, 58, 895–897. [Google Scholar] [CrossRef]
- Mosayebi, J.; Kiyasatfar, M.; Laurent, S. Synthesis, functionalization, and design of magnetic nanoparticles for theranostic applications. Adv. Health Mater. 2017, 6, 1700306. [Google Scholar] [CrossRef]
- Bozkurt, G. Synthesis and Characterization of α-Fe2O3 nanoparticles by microemulsion method. Erzincan Üniversitesi Fen Bilim. Enstitüsü Derg. 2020, 13, 890–897. [Google Scholar] [CrossRef]
- Koventhan, C.; Kumar, N.K.R.; Chen, S.-M.; Pandi, K.; Sangili, A. Polyol mediated synthesis of hexagonal manganese cobaltate nanoparticles for voltammetric determination of thioridazine. Colloids Surf. A Physicochem. Eng. Asp. 2021, 621, 126625. [Google Scholar] [CrossRef]
- Anit, J.; Praveena, M.; Thoufeeq, S.; Al-Omari, I.; Anantharaman, M. A simple polyol one-shot synthesis of Maghemite and Hematite from inexpensive precursors. Inorg. Chem. Commun. 2023, 151, 110590. [Google Scholar] [CrossRef]
- Sun, S.; Zeng, H. Size-Controlled Synthesis of Magnetite Nanoparticles. J. Am. Chem. Soc. 2002, 124, 8204–8205. [Google Scholar] [CrossRef]
- Agarwal, P.; Preethi, J.; Bora, D.K. Direct thermal decomposition of FeCl3.6H2O in oleic acid forms hematite cube and nano octahedron structure with quasicrystalline and supercell symmetries for enhanced photoelectrochemical functionality. Mater. Chem. Phys. 2021, 273, 124977. [Google Scholar] [CrossRef]
- Tahir, M.B.; Rafique, M.; Rafique, M.S.; Nawaz, T.; Rizwan, M.; Tanveer, M. Photocatalytic nanomaterials for degradation of organic pollutants and heavy metals. In Nanotechnology and Photocatalysis for Environmental Applications; Elsevier: Amsterdam, The Netherlands, 2020; pp. 119–138. [Google Scholar] [CrossRef]
- Bomatí-Miguel, O.; Mazeina, L.; Navrotsky, A.; Veintemillas-Verdaguer, S. Calorimetric study of maghemite nanoparticles synthesized by laser-induced pyrolysis. Chem. Mater. 2008, 20, 591–598. [Google Scholar] [CrossRef]
- Pechini, M.P. Method of Preparing Lead and Alkaline Earth Titanates and Niobates and Coating Method Using the Same to Form a Capacitor. U.S. Patent 3330697A, 26 August 1963. Available online: https://patents.google.com/patent/US3330697A/en (accessed on 5 January 2026).
- Murillo-Ortíz, R.; Mirabal-García, M.; Cruz-Rivera, J.; Valdez-Pérez, D.; Martínez, J.; Pérez-Moreno, F.; Lobo-Guerrero, A. Properties and arsenic removal evaluation of polyvinyl alcohol nanofibers with embedded strontium hexaferrite nanoparticles. Mater. Chem. Phys. 2019, 234, 151–157. [Google Scholar] [CrossRef]
- Ateia, E.E.; Elsayed, K.; Ramadan, R. Tuning the properties of Ba-M hexaferrite BaFe11.5Co0.5O19: A road towards diverse applications. J. Inorg. Organomet. Polym. Mater. 2022, 32, 2502–2512. [Google Scholar] [CrossRef]
- Wang, L.; Gao, L. Self-assembly behavior of hematite nanoparticles with controllable anisotropic morphology. J. Colloid Interface Sci. 2010, 349, 519–526. [Google Scholar] [CrossRef]
- Bomati, O.; Morales, M.; Serna, C.; Veintemillas, S. Magnetic nanoparticles prepared by laser-induced pyrolysis. In IEEE International Digest of Technical Papers on Magnetics Conference; IEEE: Piscataway, NJ, USA, 2003. [Google Scholar] [CrossRef]
- Criveanu, A.; Dumitrache, F.; Fleaca, C.; Gavrila-Florescu, L.; Lungu, I.; Morjan, I.P.; Socoliuc, V.; Prodan, G. Chitosan-coated iron oxide nanoparticles obtained by laser pyrolysis. Appl. Surf. Sci. Adv. 2023, 15, 100405. [Google Scholar] [CrossRef]
- Khan, S.; Sayed, M.; Sohail, M.; Shah, L.A.; Raja, M.A. Chapter 6—Advanced Oxidation and Reduction Processes. In Advances in Water Purification Techniques; Satinder, A., Ed.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 135–164. [Google Scholar] [CrossRef]
- Ozkaya, T.; Baykal, A.; Koseoğlu, Y.; Kavas, H. Synthesis of Co3O4 nanoparticles by oxidation-reduction method and its magnetic characterization. Open Chem. 2009, 7, 410–414. [Google Scholar] [CrossRef]
- Desai, P.P.; Radha, M.; Savitha, G.; Boregowda, R. Versatile strategies for multifaceted nanoparticle synthesis—An overview. In Nanotechnology and In Silico Tools; Elsevier: Amsterdam, The Netherlands, 2024; pp. 155–168. [Google Scholar] [CrossRef]
- Nashaat, A.M.; Abu El-Fadl, A.; Kassem, M.A.; Nakamura, H. Optimizing a microwave-combustion synthesis and particle-size dependent magnetic properties of M-type Sr ferrite. Mater. Chem. Phys. 2023, 305, 128008. [Google Scholar] [CrossRef]
- Fernández-Pacheco, R.; Arruebo, M.; Marquina, C.; Ibarra, R.; Arbiol, J.; Santamaría, J. Highly magnetic silica-coated iron nanoparticles prepared by the arc-discharge method. Nanotechnology 2006, 17, 1188–1192. [Google Scholar] [CrossRef]
- Karami, H.; Goli, F.; Ordoukhanian, J. Synthesis of magnetite/hematite/iron nanocomposites by the low voltage arc discharge in water in the presence of external magnetic field. Int. J. Electrochem. Sci. 2016, 11, 3074–3085. [Google Scholar] [CrossRef]
- Suslick, K. Sonochemistry. In Comprehensive Coordination Chemistry II; Elsevier: Amsterdam, The Netherlands, 2003; pp. 731–739. [Google Scholar] [CrossRef]
- Yadav, V.K.; Ali, D.; Khan, S.H.; Gnanamoorthy, G.; Choudhary, N.; Yadav, K.K.; Thai, V.N.; Hussain, S.A.; Manhrdas, S. Synthesis and characterization of amorphous iron oxide nanoparticles by the sonochemical method and their application for the remediation of heavy metals from wastewater. Nanomaterials 2020, 10, 1551. [Google Scholar] [CrossRef] [PubMed]
- Aruna, S.T.; Mukasyan, A.S. Combustion synthesis and nanomaterials. Curr. Opin. Solid State Mater. Sci. 2008, 12, 44–50. [Google Scholar] [CrossRef]
- Căpraru, A.; Moacă, E.-A.; Păcurariu, C.; Ianoş, R.; Lazău, R.; Barbu-Tudoran, L. Development and characterization of magnetic iron oxide nanoparticles using microwave for the combustion reaction ignition, as possible candidates for biomedical applications. Powder Technol. 2021, 394, 1026–1038. [Google Scholar] [CrossRef]
- Bube, R. Cadmium Sulfide and Telluride. In Encyclopedia of Materials: Science and Technology; Elsevier: Amsterdam, The Netherlands, 2001; pp. 873–879. [Google Scholar] [CrossRef]
- Levish, A.; Joshi, S.; Winterer, M. Chemical vapor synthesis of nanocrystalline iron oxides. Appl. Energy Combust. Sci. 2023, 15, 100177. [Google Scholar] [CrossRef]
- Li, Z.; Xu, S.; Xiao, G.; Qian, L.; Song, Y. Removal of hexavalent chromium from groundwater using sodium alginate dispersed nano zero-valent iron. J. Environ. Manag. 2019, 244, 33–39. [Google Scholar] [CrossRef]
- Hübner, U.; Spahr, S.; Lutze, H.; Wieland, A.; Rüting, S.; Gernjak, W.; Wenk, J. Advanced oxidation processes for water and wastewater treatment—Guidance for systematic future research. Heliyon 2024, 10, e30402. [Google Scholar] [CrossRef]
- Sandhu, S.; Zumeit, A.; Tian, Z.; Vinciguerra, V.; Dahiya, R. Semiconductor manufacturing wastewater challenges and the potential solutions via printed electronics. iScience 2025, 28, 113576. [Google Scholar] [CrossRef]
- Fecht, H.J.; Hellstern, E.; Fu, Z.; Johnson, W.L. Nanocrystalline metals prepared by high-energy ball milling. Met. Trans. A 1990, 21, 2333–2337. [Google Scholar] [CrossRef]
- Amendola, V.; Barcikowski, S. A quarter-century of nanoparticle generation by lasers in liquids: Where are we now, and what’s next? J. Colloid Interface Sci. 2017, 489, 1–2. [Google Scholar] [CrossRef]
- Kurlyandskaya, G.; Madinabeitia, I.; Beketov, I.; Medvedev, A.; Larrañaga, A.; Safronov, A.; Bhagat, S. Structure, magnetic and microwave properties of FeNi nanoparticles obtained by electric explosion of wire. J. Alloys Compd. 2014, 615, S231–S235. [Google Scholar] [CrossRef]
- Chen, Y.F. Nanofabrication by electron beam lithography and its applications: A review. Microelectron. Eng. 2015, 135, 57–72. [Google Scholar] [CrossRef]
- Marciello, M.; Luengo, Y.; Morales, M.P. Iron oxide nanoparticles for cancer diagnosis and therapy. In Nanoarchitectonics for Smart Delivery and Drug Targeting; Elsevier: Amsterdam, The Netherlands, 2016; pp. 667–694. [Google Scholar]
- Khan, I.; Morishita, S.; Higashinaka, R.; Matsuda, T.D.; Aoki, Y.; Kuzmann, E.; Homonnay, Z.; Katalin, S.; Pavić, L.; Kubuki, S. Synthesis, characterization and magnetic properties of ε-Fe2O3 nanoparticles prepared by sol-gel method. J. Magn. Magn. Mater. 2021, 538, 168264. [Google Scholar] [CrossRef]
- Meierhofer, F.; Mädler, L.; Fritsching, U. Nanoparticle evolution in flame spray pyrolysis—Process design via experimental and computational analysis. AIChE J. 2020, 66, e16885. [Google Scholar] [CrossRef]
- Cubas, P.d.J.; Semkiw, A.W.; Monteiro, F.C.; Weinert, P.L.; Monteiro, J.F.H.L.; Fujiwara, S.T. Synthesis of CuCr2O4 by self-combustion method and photocatalytic activity in the degradation of Azo Dye with visible light. J. Photochem. Photobiol. A Chem. 2020, 401, 112797. [Google Scholar] [CrossRef]
- Glasgow, W.; Fellows, B.; Qi, B.; Darroudi, T.; Kitchens, C.; Ye, L.; Crawford, T.M.; Mefford, O.T. Continuous synthesis of iron oxide (Fe3O4) nanoparticles via thermal decomposition. Particuology 2016, 26, 47–53. [Google Scholar] [CrossRef]
- Shang, Z.; Xu, P.; Feng, T.; Sun, Y.; He, K.; Li, G.; Li, X. Probe into a novel surfactant-free microemulsion system of ethylene glycol monobutyl ether + water + diesel for crude oil removal and recovery from oily sludge. Sci. Total Environ. 2024, 945, 174037. [Google Scholar] [CrossRef] [PubMed]
- Tazim, T.Q.; Kawsar, M.; Hossain, M.S.; Bahadur, N.M.; Ahmed, S. Hydrothermal synthesis of nano-metal oxides for structural modification: A review. Next Nanotechnol. 2025, 7, 100167. [Google Scholar] [CrossRef]
- Wei, J.; Gao, Z.; Song, Y.; Yang, W.; Wang, J.; Li, Z.; Mann, T.; Zhang, M.; Liu, L. Solvothermal synthesis of Li–Al layered double hydroxides and their electrochemical performance. Mater. Chem. Phys. 2013, 139, 395–402. [Google Scholar] [CrossRef]
- Loghman-Estarki, M.; Torkian, S.; Rastabi, R.A.; Ghasemi, A. Effect of annealing temperature and copper mole ratio on the morphology, structure and magnetic properties of Mg0.5−xCuxZn0.5Fe2O4 nanoparticles prepared by the modified Pechini method. J. Magn. Magn. Mater. 2017, 442, 163–175. [Google Scholar] [CrossRef]
- Fievet, F.; Lagier, J.; Figlarz, M. Preparing monodisperse metal powders in micrometer and submicrometer sizes by the polyol process. MRS Bull. 1989, 14, 29–34. [Google Scholar] [CrossRef]
- Borah, R.; Ag, K.R.; Minja, A.C.; Verbruggen, S.W. A review on self-assembly of colloidal nanoparticles into clusters, patterns, and films: Emerging synthesis techniques and applications. Small Methods 2023, 7, e2201536. [Google Scholar] [CrossRef]
- Lungu, I.I.; Andronescu, E.; Dumitrache, F.; Gavrila-Florescu, L.; Banici, A.M.; Morjan, I.; Criveanu, A.; Prodan, G. Laser pyrolysis of iron oxide nanoparticles and the influence of laser power. Molecules 2023, 28, 7284. [Google Scholar] [CrossRef] [PubMed]
- Saxena, V.; Chandra, U. Microwave synthesis: A physical concept. In Microwave Heating; Chandra, U., Ed.; InTech: London, UK, 2011. [Google Scholar] [CrossRef][Green Version]
- Retnosari, I.; Hayati, I.N.; Amalia, A.; Hastuti, S.; Saraswati, T.E. The chemical characteristics of iron oxide/carbon synthesized by the arc discharge method in liquid media with the addition of ammonia. J. Kim. Sains Dan Apl. 2018, 21, 166–170. [Google Scholar] [CrossRef]
- Khan, M.M. Sonochemical synthesis method. In Photocatalysts: Synthesis and Characterization Methods; Elsevier: Amsterdam, The Netherlands, 2025; pp. 67–77. [Google Scholar] [CrossRef]
- Iida, H.; Nakanishi, T.; Takada, H.; Osaka, T. Preparation of Magnetic Iron-Oxide Nanoparticles by Successive Reduction–Oxidation in Reverse Micelles: Effects of Reducing Agent and Atmosphere. Electrochim. Acta 2006, 52, 292–296. [Google Scholar] [CrossRef]
- Besmann, T.; Stinton, D.; Lowden, R. Chemical vapor deposition techniques. MRS Bull. 1988, 13, 45–51. [Google Scholar] [CrossRef]
- Kumar, A.; Cross, A.; Manukyan, K.; Bhosale, R.; Broeke, L.v.D.; Miller, J.; Mukasyan, A.; Wolf, E. Combustion synthesis of copper–nickel catalysts for hydrogen production from ethanol. Chem. Eng. J. 2015, 278, 46–54. [Google Scholar] [CrossRef]
- Li, X.; Shen, X.; Jiang, W.; Xi, Y.; Li, S. Comprehensive review of emerging contaminants: Detection technologies, environmental impact, and management strategies. Ecotoxicol. Environ. Saf. 2024, 278, 116420. [Google Scholar] [CrossRef]
- Mukhopadhyay, A.; Duttagupta, S.; Mukherjee, A. Emerging organic contaminants in global community drinking water sources and supply: A review of occurrence, processes and remediation. J. Environ. Chem. Eng. 2022, 10, 107560. [Google Scholar] [CrossRef]
- Sharma, M.; Kalita, P.; Senapati, K.K.; Garg, A. Study on magnetic materials for removal of water pollutants. In Emerging Pollutants—Some Strategies for the Quality Preservation of Our Environment; InTech: London, UK, 2018. [Google Scholar] [CrossRef]
- Al-Salihi, S.; Bayati, M.; Jasim, A.M.; Fidalgo, M.M.; Xing, Y. Magnetic mesoporous TiO2/Fe3O4 nanocomposite adsorbent for removal of sulfamethazine from water. Environ. Adv. 2022, 9, 100283. [Google Scholar] [CrossRef]
- Muduli, S.P.; Veeralingam, S.; Badhulika, S. Free-standing, non-toxic and reusable 0.67BiFeO3–0.33BaTiO3 based polymeric piezo-catalyst for organic dye wastewater treatment. J. Water Process. Eng. 2022, 48, 102934. [Google Scholar] [CrossRef]
- Sanad, M.M.; Gaber, S.E.; El-Aswar, E.I.; Farahat, M.M. Graphene-magnetite functionalized diatomite for efficient removal of organochlorine pesticides from aquatic environment. J. Environ. Manag. 2023, 330, 117145. [Google Scholar] [CrossRef]
- Shi, B.; Wang, Y.; Ahmed, I.; Zhang, B. Catalytic degradation of refractory phenol sulfonic acid by facile, calcination-free cobalt ferrite nanoparticles. J. Environ. Chem. Eng. 2022, 10, 107616. [Google Scholar] [CrossRef]
- Harja, M.; Lupu, N.; Chiriac, H.; Herea, D.-D.; Buema, G. Studies on the removal of congo red dye by an adsorbent based on Fly-Ash@Fe3O4 mixture. Magnetochemistry 2022, 8, 125. [Google Scholar] [CrossRef]
- Bameri, I.; Saffari, J.; Baniyaghoob, S.; Ekrami-Kakhki, M.-S. Synthesis of magnetic nano-NiFe2O4 with the assistance of ultrasound and its application for photocatalytic degradation of Titan Yellow: Kinetic and isotherm studies. Colloid Interface Sci. Commun. 2022, 48, 100610. [Google Scholar] [CrossRef]
- AbouSeada, N.; Ahmed, M.; Elmahgary, M.G. Synthesis and characterization of novel magnetic nanoparticles for photocatalytic degradation of indigo carmine dye. Mater. Sci. Energy Technol. 2022, 5, 116–124. [Google Scholar] [CrossRef]
- Ezzat, A.O.; Tawfeek, A.M.; Rajabathar, J.R.; Al-Lohedan, H.A. Synthesis of new hybrid structured magnetite crosslinked poly ionic liquid for efficient removal of Coomassie Brilliant Blue R-250 dye in aqueous medium. Molecules 2022, 27, 441. [Google Scholar] [CrossRef]
- Rezania, S.; Kadi, A.; Kamyab, H.; Ghfar, A.A.; Nodeh, H.R.; Ibrahim, W.N.W. Lanthanum doped magnetic polyaniline for removal of phosphate ions from water. Chemosphere 2022, 307, 135809. [Google Scholar] [CrossRef]
- Zhao, J.; Huang, P.; Wang, X.; Yang, J.; Zhou, Z.; Du, X.; Lu, X. Efficient adsorption removal of organic nitrogen pesticides: Insight into a new hollow NiO/Co@C magnetic nanocomposites derived from metal-organic framework. Sep. Purif. Technol. 2022, 287, 120608. [Google Scholar] [CrossRef]
- Manojkumar, M.; Jeyajothi, K.; Jagadeesan, A.; Jeevanantham, V. Magnetic separation of green synthesized Fe3O4 nanoparticles on photocatalytic activity of methyl orange dye removal. J. Indian Chem. Soc. 2022, 99, 100559. [Google Scholar] [CrossRef]
- Wei, F.; Wang, K.; Li, W.; Ren, Q.; Qin, L.; Yu, M.; Liang, Z.; Nie, M.; Wang, S. Preparation of Fe/Ni-MOFs for the adsorption of ciprofloxacin from wastewater. Molecules 2023, 28, 4411. [Google Scholar] [CrossRef]
- Roostaee, M.; Sheikhshoaie, I. Synthesis of CoFe2O4 @ZnMOF/Graphene Nanoflake for Photocatalytic Degradation of Diazinon Under Visible Light Irradiation: Optimization and Modeling Using a Fractional Factorial Method; Research Square: Durham, NC, USA, 2022. [Google Scholar] [CrossRef]
- Muñoz-Sandoval, M.J.; Caravaca, M.; López-García, I.; Hernández-Córdoba, M.; Vicente-Martínez, Y. Complete and simultaneous removal of ionic silver and silver nanoparticles by using an ionic liquid supported on a magnetic nanoparticle core. Environ. Res. 2022, 214, 113943. [Google Scholar] [CrossRef] [PubMed]
- Rayaroth, M.P.; Oh, D.; Lee, C.-S.; Chang, Y.-S. Simultaneous removal of heavy metals and dyes in water using a MgO-coated Fe3O4 nanocomposite: Role of micro-mixing effect induced by bubble generation. Chemosphere 2022, 294, 133788. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Sun, Y.; Gu, Y. Catalysis and adsorption of Zr-doped Fe3O4 nanoparticles provide a new strategy for diazinon removal and phosphorus recovery from aqueous solution. J. Environ. Chem. Eng. 2022, 10, 107153. [Google Scholar] [CrossRef]
- Han, J.-H.; Jia, W.-H.; Liu, Y.; Wang, W.-D.; Zhang, L.-K.; Li, Y.-M.; Sun, P.; Fan, J.; Hu, S.-T. α-Fe2O3/Cu2O composites as catalysts for photoelectrocatalytic degradation of benzotriazoles. Water Sci. Eng. 2022, 15, 200–209. [Google Scholar] [CrossRef]
- Vidovix, T.B.; Januário, E.F.D.; Bergamasco, R.; Vieira, A.M.S. Evaluation of agro-industrial residue functionalized with iron oxide magnetic nanoparticles for chloroquine removal from contaminated water. Mater. Lett. 2022, 326, 132915. [Google Scholar] [CrossRef]
- Bassim, S.; Mageed, A.K.; AbdulRazak, A.A.; Majdi, H.S. Green synthesis of Fe3O4 nanoparticles and its applications in wastewater treatment. Inorganics 2022, 10, 260. [Google Scholar] [CrossRef]
- Shankara, A.H.; Prabagar, J.S.; Tenzin, T.; Yadav, S.; Kumar, K.M.A.; Shivaraju, H.P. Facile synthesis of NdFeO3 perovskite for photocatalytic degradation of organic dye and antibiotic. Mater. Today: Proc. 2023, 75, 15–23. [Google Scholar] [CrossRef]
- Khairudin, K.; Abu Bakar, N.F.; Osman, M.S. Magnetically recyclable flake-like BiOI-Fe3O4 microswimmers for fast and efficient degradation of microplastics. J. Environ. Chem. Eng. 2022, 10, 108275. [Google Scholar] [CrossRef]
- Zhang, J.; Li, X.; Xu, H.; Zhang, W.; Feng, X.; Yao, Y.; Ma, Y.; Su, L.; Ren, S.; Li, S. Coating magnetic nanoparticles with artificial humic acid derived from rice straw for effective removal of tetracycline antibiotics. Ind. Crop. Prod. 2023, 200, 116781. [Google Scholar] [CrossRef]
- Heidarinejad, F.; Kamani, H.; Khtibi, A. Magnetic Fe-doped TiO2@Fe3O4 for metronidazole degradation in aqueous solutions: Characteristics and efficacy assessment. Heliyon 2023, 9, e21414. [Google Scholar] [CrossRef]
- Kubiak, A.; Kubacka, M.; Gabała, E.; Dobrowolska, A.; Synoradzki, K.; Siwińska-Ciesielczyk, K.; Czaczyk, K.; Jesionowski, T. Hydrothermally assisted fabrication of TiO2-Fe3O4 composite materials and their antibacterial activity. Materials 2020, 13, 4715. [Google Scholar] [CrossRef]
- López, J.; Rey, A.; Viñuelas-Zahinos, E.; Álvarez, P.M. Preparation of a new green magnetic Fe3O4 @TiO2-P25 photocatalyst for solar advanced oxidation processes in water. J. Environ. Chem. Eng. 2023, 11, 109999. [Google Scholar] [CrossRef]
- Nassar, A.E.; El-Aswar, E.I.; Rizk, S.A.; Gaber, S.E.-S.; Jahin, H.S. Microwave-assisted hydrothermal preparation of magnetic hydrochar for the removal of organophosphorus insecticides from aqueous solutions. Sep. Purif. Technol. 2023, 306, 122569. [Google Scholar] [CrossRef]
- Yin, J.; Fang, K.; Li, J.; Du, N.; Hu, D.; Cao, D.; Tian, R.; Deng, L.; Li, K. Competitive adsorption mechanisms of pigments in sugarcane juice on starch-based magnetic nanocomposites. Int. J. Biol. Macromol. 2023, 231, 123134. [Google Scholar] [CrossRef]
- Bibi, F.; Iqbal, S.; Kalsoom, A.; Jamshaid, M.; Ahmed, A.; Mirza, M.; Qureshi, W.A. Magnetically separable Nd and Mn co-doped SrFe12O19 hexa-ferrites nanostructures for the evaluation of structural, magnetic and photo-catalytic studies under solar irradiation for the crystal violet dye removal from the industrial wastes. Ceram. Int. 2023, 49, 15990–16001. [Google Scholar] [CrossRef]
- Fu, X.; Sarker, S.; Ma, W.; Zhao, W.; Rong, Y.; Liu, Q. Novel phenylalanine-modified magnetic ferroferric oxide nanoparticles for ciprofloxacin removal from aqueous solution. J. Colloid Interface Sci. 2023, 632, 345–356. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Rasool, R.T.; Ashraf, G.A.; Guo, H. The stimulation of peroxymonosulfate via novel Co0.5Cu0.5Fe2O4 heterogeneous photocatalyst in aqueous solution for organic contaminants removal. Mater. Sci. Semicond. Process. 2023, 157, 107321. [Google Scholar] [CrossRef]
- Liu, Z.; Zhao, H.; Wang, J.; Wang, Z.; Di, S.; Xu, H.; Wang, Q.; Wang, X.; Qi, P. Green synthesis of Fe3O4@SiO2@Salg particles for organophosphorus pesticides removal: Mechanisms, biosafety, and application. Chemosphere 2023, 332, 138751. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, Q.; Zhang, X.; Dong, L.; Yuan, Y.; Peng, C.; Zhang, M.; Rao, P.; Pervez, N.; Gao, N. CoFe2O4/WS2 as a highly active heterogeneous catalyst for the efficient degradation of sulfathiazole by activation of peroxymonosulfate. J. Water Process. Eng. 2023, 57, 104714. [Google Scholar] [CrossRef]
- Xia, Y.; Guo, M.; Zhao, Y.; Ren, Z.; Deng, J. Preparation of Mn2O3-Co3O4 and its xylene removal by oxidation. Chem. Phys. Impact 2023, 6, 100160. [Google Scholar] [CrossRef]
- Santana, R.M.d.R.; Napoleão, D.C.; Rodriguez-Diaz, J.M.; Gomes, R.K.d.M.; Silva, M.G.; de Lima, V.M.E.; Neto, A.A.d.M.; Vinhas, G.M.; Duarte, M.M.M.B. Efficient microbial cellulose/Fe3O4 nanocomposite for photocatalytic degradation by advanced oxidation process of textile dyes. Chemosphere 2023, 326, 138453. [Google Scholar] [CrossRef]
- Saleh, A.K.; Salama, A.; Badawy, A.S.; Diab, M.A.; El-Gendi, H. Paper sludge saccharification for batch and fed-batch production of bacterial cellulose decorated with magnetite for dye decolorization by experimental design. Cellulose 2023, 30, 10841–10866. [Google Scholar] [CrossRef]
- Mohammadi, M.; Sabbaghi, S.; Binazadeh, M.; Ghaedi, S.; Rajabi, H. Type-1 α-Fe2O3/TiO2 photocatalytic degradation of tetracycline from wastewater using CCD-based RSM optimization. Chemosphere 2023, 336, 139311. [Google Scholar] [CrossRef] [PubMed]
- Wahyuni, E.T.; Lestari, N.D.; Cinjana, I.R.; Annur, S.; Natsir, T.A.; Mudasir, M. Doping TiO2 with Fe from iron rusty waste for enhancing its activity under visible light in the Congo red dye photodegradation. J. Eng. Appl. Sci. 2023, 70, 9. [Google Scholar] [CrossRef]
- Adeleke, B.K.; Olalekan, O.A.; Adewuyi, A.; Lau, W.J.; Adeyemi, O.G. Purification of metronidazole and penicillin-G contaminated water by MOF-5 imprinted cobalt ferrite. Results Chem. 2023, 6, 101158. [Google Scholar] [CrossRef]
- Adewuyi, A.; Ogunkunle, O.A.; Oderinde, R.A. Synthesis of tin ferrite-dopped zeolitic imidazolate framework and its application in the photocatalytic degradation of sulfamethoxazole, ciprofloxacin, erythromycin and ampicillin in water. Catal. Commun. 2023, 183, 106769. [Google Scholar] [CrossRef]
- Rafie, S.F.; Sayahi, H.; Abdollahi, H.; Abu-Zahra, N. Hydrothermal synthesis of Fe3O4 nanoparticles at different pHs and its effect on discoloration of methylene blue: Evaluation of alternatives by TOPSIS method. Mater. Today Commun. 2023, 37, 107589. [Google Scholar] [CrossRef]
- Ma, Z.; Liu, C.; Srinivasakannan, C.; Li, L.; Wang, Y. Synthesis of magnetic Fe3O4-HKUST-1 nanocomposites for azo dye adsorption. Arab. J. Chem. 2023, 16, 104767. [Google Scholar] [CrossRef]
- Kaveh, R.; Bagherzadeh, M. Simultaneous removal of mercury ions and cationic and anionic dyes from aqueous solution using epichlorohydrin cross-linked chitosan @ magnetic Fe3O4/activated carbon nanocomposite as an adsorbent. Diam. Relat. Mater. 2022, 124, 108923. [Google Scholar] [CrossRef]
- Bibi, S.; Ahmad, A.; Shah, S.S.; Siddiq, M.; Habila, M.A.; Choi, D. Synthesis of Cit-Fe3O4@TiO2 photocatalyst for the degradation of eosin-Y and methylene blue dye. Ceram. Int. 2024, 50, 37533–37540. [Google Scholar] [CrossRef]
- Chergui, F.; Mokhtar, A.; Abdelkrim, S.; Sardi, A.; Hachemaoui, M.; Boukoussa, B.; Djelad, A.; Sassi, M.; Viscusi, G.; Abboud, M. Optimizing catalytic performance: Reduction of organic dyes using synthesized Fe3O4@AC magnetic nano-catalyst. Mater. Chem. Phys. 2024, 329, 130094. [Google Scholar] [CrossRef]
- Adewuyi, A.; Oderinde, R.A. Photocatalytic removal of some selected antibiotics in polluted water system by graphitic carbon nitride-enhanced vanadium ferrite (VFe2O4@g-C3N4). Chemosphere 2024, 362, 142733. [Google Scholar] [CrossRef]
- Deivayanai, V.; Karishma, S.; Thamarai, P.; Saravanan, A.; Yaashikaa, P. Efficient red azo dye removal from wastewater using magnetic nanoparticle impregnated Prosopis juliflora biomass: ANN modeling approach. Desalination Water Treat. 2024, 320, 100746. [Google Scholar] [CrossRef]
- Ali, N.S.; Khader, E.H.; Khudhur, R.H.; Abdulrahman, M.A.; Salih, I.K.; Albayati, T.M. Removal of anionic azo dye from wastewater using Fe3O4 magnetic nanoparticles adsorbents in a batch system. Desalination Water Treat. 2024, 317, 100033. [Google Scholar] [CrossRef]
- Ariaeenejad, S.; Barani, M.; Roostaee, M.; Lohrasbi-Nejad, A.; Mohammadi-Nejad, G.; Salekdeh, G.H. Enhanced pollutant degradation via green-synthesized core-shell mesoporous Si@Fe magnetic nanoparticles immobilized with metagenomic laccase. Int. J. Biol. Macromol. 2024, 278, 134813. [Google Scholar] [CrossRef] [PubMed]
- Harki, D.A.H.; Naghipour, A.; Fardood, S.T. Eco-friendly synthesis and characterization of Mg0.5Co0.5Fe2O4 magnetic nanoparticles for photocatalytic degradation of congo red dye. Case Stud. Chem. Environ. Eng. 2024, 10, 101016. [Google Scholar] [CrossRef]
- Fardood, S.T.; Moradnia, F.; Aminabhavi, T.M. Green synthesis of novel Zn0.5Ni0.5FeCrO4 spinel magnetic nanoparticles: Photodegradation of 4-nitrophenol and aniline under visible light irradiation. Environ. Pollut. 2024, 358, 124534. [Google Scholar] [CrossRef]
- Muhaymin, A.; Mohamed, H.E.A.; Hkiri, K.; Safdar, A.; Kotsedi, L.; Maaza, M. Green synthesis of NiFe2O4 nanoparticles using Hyphaene thebaica: A facile route towards magnetic and photocatalytic application. Mater. Today Chem. 2024, 40, 102286. [Google Scholar] [CrossRef]
- Hussain, E.; Shahadat, M.; Ahtesham, A.; Ibrahim, M.N.M. Synthesis, characterization, and applications of ambi-functional PANI/GO/MOF-Fe3O4 magnetic nanocomposite for removing industrial dye and emerging contaminant. Sep. Purif. Technol. 2024, 351, 128052. [Google Scholar] [CrossRef]
- Thabet, M.; El-Monaem, E.M.A.; Alharbi, W.R.; Mohamoud, M.; Abdel-Aty, A.-H.; Ibrahim, I.; Abdel-Lateef, M.A.; Goda, A.E.; Elnasr, T.A.S.; Wang, R.; et al. Adsorption and photocatalytic degradation activities of a hybrid magnetic mesoporous composite of α-Fe2O3 nanoparticles embedded with sheets-like MgO. J. Water Process. Eng. 2024, 60, 105192. [Google Scholar] [CrossRef]
- Rezagholizade-Shirvan, A.; Ghasemi, A.; Mazaheri, Y.; Shokri, S.; Fallahizadeh, S.; Sani, M.A.; Mohtashami, M.; Mahmoudzadeh, M.; Sarafraz, M.; Darroudi, M.; et al. Removal of aflatoxin M1 in milk using magnetic laccase/MoS2/chitosan nanocomposite as an efficient sorbent. Chemosphere 2024, 365, 143334. [Google Scholar] [CrossRef]
- Ferfera-Harrar, H.; Sadi, A.; Benhalima, T. Magnetic recyclable carboxymethyl cellulose/gelatin/citrate@Fe3O4 photo-nanocomposite beads for ciprofloxacin removal via hybrid adsorption/photocatalysis process under solar light as a renewable energy source. Int. J. Biol. Macromol. 2024, 282, 136854. [Google Scholar] [CrossRef] [PubMed]
- Zou, J.; Yu, Q.; Cao, D.; Wang, Q.; Ma, N.; Dai, W. NiFe2O4 magnetic nanoparticles supported on MIL-101(Fe) as bimetallic adsorbent for boosted capture ability toward levofloxacin. Mater. Today Chem. 2024, 41, 102310. [Google Scholar] [CrossRef]
- Du, J.-T.; Wu, H.; Jie, Y.; Xia, Y.; Yang, Z.; Yan, H.; Wang, Q.; Wang, J.-X.; Chen, J.-F. Magnetically recyclable CoFe2O4 nanocatalysts for efficient glycolysis of polyethylene terephthalate. Chem. Eng. Sci. 2025, 304, 121042. [Google Scholar] [CrossRef]
- Liu, L.; Cui, Z.; Feng, B.; Sui, M.; Huang, H.; Wu, Z. Synthesis of Fe2O3/TiO2 photocatalytic composites for methylene blue degradation as a novel strategy for high-value utilisation of iron scales. Materials 2024, 17, 4546. [Google Scholar] [CrossRef]
- Kartikowati, C.W.; Syadana, D.; Ramadikadipura, M.M.; Puspitasari, D.A.; Poerwadi, B.; Fauziyah, M.; Arutanti, O. Synthesis of TiO2/Fe2O3 nanocomposites as photocatalyst for methyl orange degradation. E3S Web Conf. 2024, 481, 03008. [Google Scholar] [CrossRef]
- Indhur, R.; Kumar, A.; Bux, F.; Kumari, S. Efficient microplastic removal from wastewater using Fe3O4 functionalized g-C3N4 and BNNS: A comprehensive study. J. Environ. Chem. Eng. 2025, 13, 117145. [Google Scholar] [CrossRef]
- Deng, Z.; Wang, N.; Wang, Y.; Li, X.; Li, H.; Zou, D.; Liu, Y. Persulfate advanced oxidation based on a novel CeO2-Fe3O4 heterojunction catalyst for enhanced degradation of soil PAHs. Chem. Eng. Sci. 2025, 314, 121797. [Google Scholar] [CrossRef]
- Paulista, A.P.F.; Barbosa, F.F.; Júnior, M.A.D.N.; Cavalcanti, W.E.C.; Soares, J.d.O.; Morales, M.; Pergher, S.B.; Braga, T.P. Photocatalytic degradation of the remazol red ultra RGB dye using SrFe12O19-Fe3O4 magnetic oxides dispersed in silica: Effect of reduction temperature. Desalination Water Treat. 2024, 320, 100760. [Google Scholar] [CrossRef]
- Poormand, F.; Farhadi, S.; Zabardasti, A.; Maleki, M.; Mahmoudi, F. Silver (Ag) nanoparticles decorated on magnetic CoFe2O4/h-BN nanocomposites for efficient catalytic removal of toxic nitrophenols. Mater. Today Commun. 2025, 42, 111364. [Google Scholar] [CrossRef]
- Vinayagam, R.; Batra, S.; Murugesan, G.; Goveas, L.C.; Varadavenkatesan, T.; Menezes, A.; Selvaraj, R. Emerging contaminant removal using eco-friendly zinc ferrite nanoparticles: Sunlight-driven degradation of tetracycline. Emerg. Contam. 2025, 11, 100469. [Google Scholar] [CrossRef]
- Ahmed, H.M.; Kebede, W.L.; Yaebyo, A.B.; Digisu, A.W.; Tadesse, S.F. Adsorptive removal of methylene blue dye by a ternary magnetic rGO/AK/Fe3O4 nanocomposite: Studying adsorption isotherms, kinetics, and thermodynamics. Chem. Phys. Impact 2025, 10, 100800. [Google Scholar] [CrossRef]
- Joga, S.B.; Korabandi, D.; Lakkaboyana, S.K.; Kumar, V. Synthesis of iron nanoparticles on lemon peel carbon dots (LP-CDs@ Fe3O4) applied in Photo-Catalysis, Antioxidant, Antidiabetic, and Hemolytic activity. Inorg. Chem. Commun. 2025, 174, 113960. [Google Scholar] [CrossRef]
- Almeida-Naranjo, C.E.; Santana-Romo, F.; Gallegos-Castro, E.; Villamar-Ayala, C.A.; Debut, A. Triclosan removal from synthetic solution using corn cobs and their magnetic composites: Insights from batch adsorption and fixed-bed column studies. Ind. Crop. Prod. 2025, 228, 120917. [Google Scholar] [CrossRef]
- Nazeer, Z.A.M.; Praveen, M.; Harikrishna, R.; Kumar, M.; Nagarajaiah, S.; Nagabhushana, B.M. Photocatalytic degradation of the azo dye “Congo-Red” by ZnFe2O4/ZnO nanocomposite. J. Mines Met. Fuels 2024, 71, 192–199. [Google Scholar] [CrossRef]
- Ke, S.; Naghizadeh, M.; Sun, L.; Jin, H.; Dong, S.; Huang, T. Highly reactive ZnFe2O4/TiO2 p-n heterojunction photocatalyst accelerates interfacial charge transfer for boosted photodegradation of ammonia nitrogen. Chem. Eng. Sci. 2025, 307, 121361. [Google Scholar] [CrossRef]
- Farrer, B.T.; Pecoraro, V.L. Bioinorganic Chemistry. In Encyclopedia of Physical Science and Technology; Elsevier: Amsterdam, The Netherlands, 2003; pp. 117–139. [Google Scholar] [CrossRef]
- Raj, S.; Sinha, U.; Singh, H.; Bhattacharya, J. Novel GO/Fe–Mn hybrid for the adsorptive removal of Pb(II) ions from aqueous solution and the spent adsorbent disposability in cement mix: Compressive properties and leachability study for circular economy benefits. Environ. Sci. Pollut. Res. 2022, 29, 63898–63916. [Google Scholar] [CrossRef]
- Adisu, N.; Balakrishnan, S.; Tibebe, H. Synthesis and characterization of Fe3O4 -bentonite nanocomposite adsorbent for Cr(VI) removal from water solution. Int. J. Chem. Eng. 2022, 2022, 4441718. [Google Scholar] [CrossRef]
- Herab, A.A.; Salari, D.; Ostadrahimi, A.; Olad, A. Preparation of magnetic inulin nanocomposite and its application in the removal of methylene blue and heavy metals from aqueous solution. Mater. Chem. Phys. 2022, 291, 126580. [Google Scholar] [CrossRef]
- Kalimuthu, P.; Kim, Y.; Subbaiah, M.P.; Jeon, B.-H.; Jung, J. Novel magnetic Fe@NSC nanohybrid material for arsenic removal from aqueous media. Chemosphere 2022, 308, 136450. [Google Scholar] [CrossRef]
- Gheitasi, F.; Ghammamy, S.; Zendehdel, M.; Semiromi, F.B. Removal of mercury (II) from aqueous solution by powdered activated carbon nanoparticles prepared from beer barley husk modified with Thiol/Fe3O4. J. Mol. Struct. 2022, 1267, 133555. [Google Scholar] [CrossRef]
- Hu, X.; Dai, L.; Ma, Q.; Xu, J.; Ma, J.; Liu, X. One-pot synthesis of iron oxides decorated bamboo hydrochar for lead and copper flash removal. Ind. Crop. Prod. 2022, 187, 115396. [Google Scholar] [CrossRef]
- Anbalagan, K.; Kumar, M.M.; Sudarsan, J.; Nithiyanantham, S. Removal of Heavy Metal Ions from Industrial Wastewater Using Magnetic Nanoparticles. J. Eng. Res. 2022, 10, 59–71. [Google Scholar] [CrossRef]
- Borges, G.C.d.C.; Verdi, I.R.; Fidelis, M.Z.; Junior, H.E.Z.; Lenzi, G.G.; de Souza, É.C.F.; Alves, O.C.; Brackmann, R. Magnetic heterostructures of NiFe2O4 and TiO2: Pechini synthesis, characterization and photocatalytic performance in arsenite oxidation. J. Water Process. Eng. 2023, 56, 104352. [Google Scholar] [CrossRef]
- Jadhao, J.S.; Rathod, N.V.; Rao, A.; Ghugare, C.D.; Chavan, S.M.; Kubade, A.V.; Kalyani, V.S.; Patil, A.B. Efficient removal of toxic Cd(II) ions from waste streams by a novel modified biodegradable magnetic sorbent. Chem. Inorg. Mater. 2023, 1, 100016. [Google Scholar] [CrossRef]
- Li, Y.; Yan, X.; Yu, S.; Hou, G.; Yang, J.; Bi, W.; Bie, H.; Yang, C.; Mi, Q. Construction of magnetic COF composites for lead removal with fast dynamics and superior capacity. J. Taiwan Inst. Chem. Eng. 2023, 143, 104705. [Google Scholar] [CrossRef]
- Kothavale, V.; Sharma, A.; Dhavale, R.; Chavan, V.; Shingte, S.; Selyshchev, O.; Dongale, T.; Park, H.; Zahn, D.; Salvan, G.; et al. Carboxyl and thiol-functionalized magnetic nanoadsorbents for efficient and simultaneous removal of Pb(II), Cd(II), and Ni(II) heavy metal ions from aqueous solutions: Studies of adsorption, kinetics, and isotherms. J. Phys. Chem. Solids 2023, 172, 111089. [Google Scholar] [CrossRef]
- Şahi̇n, M.; Atasoy, M.; Arslan, Y.; Yildiz, D. Removal of Ni(II), Cu(II), Pb(II), and Cd(II) from aqueous phases by silver nanoparticles and magnetic nanoparticles/nanocomposites. ACS Omega 2023, 8, 34834–34843. [Google Scholar] [CrossRef]
- Zhang, K.; Guo, F.; Graham, N.; Yu, W. Engineering of 3d graphene oxide hydrogel-supported Mno2-feooh nanoparticles with synergistic effect of oxidation and absorption toward highly efficient removal of arsenic. SSRN Electron. J. 2022, 317, 120735. [Google Scholar] [CrossRef]
- Guo, Q.; Li, Y.; Zheng, L.-W.; Wei, X.-Y.; Xu, Y.; Shen, Y.-W.; Zhang, K.-G.; Yuan, C.-G. Facile fabrication of Fe/Zr binary MOFs for arsenic removal in water: High capacity, fast kinetics and good reusability. J. Environ. Sci. 2023, 128, 213–223. [Google Scholar] [CrossRef]
- Huang, S.; Zhang, Y.; Mei, B.; Tian, X.; Zhu, W.; Liao, J.; Sun, N. Construction of mesoporous Si-Fe-GO composite for the highly efficient removal of uranium. Sep. Purif. Technol. 2023, 317, 123878. [Google Scholar] [CrossRef]
- Yu, C.; Liao, Y. Removal of Cr(VI) ions from wastewater by Fe3O4-loaded porous sludge biochar. Water Sci. Technol. 2023, 88, 947–960. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Hua, J.; Li, R.; Zhang, Y.; Jin, H.; Wang, S.; Chen, G. Application of magnetic nanocomposites in water treatment: Core–Shell Fe3O4 material for efficient adsorption of Cr(VI). Water 2023, 15, 2827. [Google Scholar] [CrossRef]
- Singh, S.; N, P.; Naik, T.; Uppara, B.; Thamaraiselvan, C.; Behera, S.; Kour, R.; Dwivedi, P.; Subramanian, S.; Khan, N.A.; et al. Removal of Pb ions using green Co3O4 nanoparticles: Simulation, modeling, adsorption, and biological studies. Environ. Res. 2023, 222, 115335. [Google Scholar] [CrossRef]
- Loulic, F.B.; Shirazi, R.H.S.M.; Miralinaghi, M.; Panahi, H.A.; Moniri, E. Highly efficient removal of toxic As(V), Cd (II), and Pb(II) ions from water samples using MnFe2O4@SBA-15-(CH2)3-adenine as a recyclable bio-nanoadsorbent. Microporous Mesoporous Mater. 2023, 356, 112567. [Google Scholar] [CrossRef]
- Li, S.; Yang, L.; Wu, J.; Yao, L.; Han, D.; Liang, Y.; Yin, Y.; Hu, L.; Shi, J.; Jiang, G. Efficient and selective removal of Hg(II) from water using recyclable hierarchical MoS2/Fe3O4 nanocomposites. Water Res. 2023, 235, 119896. [Google Scholar] [CrossRef]
- Al-Kadhi, N.S.; Basha, M.T. Enhanced removal of Cd(II) Ions from aqueous media via adsorption on facilely synthesized copper ferrite nanoparticles. Molecules 2024, 29, 3711. [Google Scholar] [CrossRef]
- Nguyen, T.H.; Nguyen, V.D.; Vu, A.-T. Synthesis of CS-Fe3O4/GO nanocomposite for adsorption of heavy metal in aqueous environment. Nanotechnology 2024, 35, 345705. [Google Scholar] [CrossRef]
- Rind, I.K.; Memon, N.; Sarı, A.; Khuhawar, M.Y.; Tuzen, M.; Hasan, S.N.U.; Memon, A.A.; Soomro, W.A.; Brohi, R.O.Z.; Saleh, T.A. Magnetic nanoparticles loaded hydrochar for effective Cr(VI) removal from water: Batch and column studies. Mater. Chem. Phys. 2024, 318, 129077. [Google Scholar] [CrossRef]
- Mirza, M.; Bodaghifard, M.A.; Darvish, F. Synthesis of a nitrogen-rich dendrimer grafted on magnetic nanoparticles for efficient removal of Pb(ii) and Cd(ii) ions. RSC Adv. 2024, 14, 32559–32572. [Google Scholar] [CrossRef]
- Motawea, E.A.; El-Sabban, H.A.; Kang, J.-H.; Ko, Y.G. Efficient multifunctional PPy-NTs/PEI@alginate@NiFe2O4 magnetic beads for heavy metals removal: Experimental design and optimization interpretations. Int. J. Biol. Macromol. 2024, 282, 137437. [Google Scholar] [CrossRef] [PubMed]
- Hinsene, H.; Bhawawet, N.; Imyim, A. Rice husk biochar doped with deep eutectic solvent and Fe3O4/ZnO nanoparticles for heavy metal and diclofenac removal from water. Sep. Purif. Technol. 2024, 339, 126638. [Google Scholar] [CrossRef]
- Yao, L.; Hong, C.; Qi, Y.; Wu, L. Effective Co2+ removal from aqueous solution and industrial wastewater using ZIF-7 and MnFe2O4-modified walnut shell biochar composite. Sep. Purif. Technol. 2025, 353, 128574. [Google Scholar] [CrossRef]
- Jiang, Y.; Tian, Q.; Zhang, H.; Yue, X.; Xue, S.; Qiu, F.; Zhang, T. One-step removal of anionic/cationic heavy metal ions from wastewater by magnetic amphoteric adsorbent. J. Water Process. Eng. 2024, 65, 105847. [Google Scholar] [CrossRef]
- Korkmaz, Ş.; Özkan, E.H.; Uzun, D.; Yetim, N.K.; Özcan, C. Magnetic solid phase extraction of lead (II) and cadmium (II) from water samples using ZnO@Fe3O4 nanoparticles combined with flame atomic absorption spectrometry determination. J. Sep. Sci. 2025, 48, e70115. [Google Scholar] [CrossRef]
- Memon, S.A.; Duan, J.; Li, W.; Wei, Y. Fabrication of core-shell structured Fe3O4 @amide humic acid magnetic nanoparticles and their highly efficient adsorption for La (III). Sep. Sci. Technol. 2025, 60, 840–855. [Google Scholar] [CrossRef]
- Manzoor, Q.; Farrukh, M.A.; Qamar, M.T.; Sajid, A.; Aldossari, S.A.; Manikandan, A.; Iqbal, M. Polymer-assisted synthesis of ternary magnetic graphene oxide nanocomposite for the adsorptive removal of Cr(VI) and Pb(II) ions. Heliyon 2024, 10, e35204. [Google Scholar] [CrossRef]
- El-Aryan, Y. Facile hydrothermal synthesis and characterization of magnetic Cr-cobalt ferrite nanocomposite as a novel adsorbent for lanthanide ions. Inorg. Chem. Commun. 2025, 180, 114967. [Google Scholar] [CrossRef]
- Arabkhani, P.; Asfaram, A. A novel biowaste-derived magnetic adsorbent for efficient removal of cadmium, cobalt and strontium ions from industrial wastewater. Inorg. Chem. Commun. 2025, 174, 113956. [Google Scholar] [CrossRef]
- Alhameedawi, F.A.H.; Maybodi, A.S.; Khathi, M.T. Synthesis and characterization of AC@Fe3O4 nanocomposite and its application as an efficient adsorbent for removal of some heavy metal ions from aqueous solutions. Iran. J. Chem. Chem. Eng. 2025, 44, 2234–2246. [Google Scholar] [CrossRef]
- Alyasi, H.; Wahib, S.; Tong, Y.; Gomez, T.; Mahmoud, K.A. Magnetic MXene chitosan-lignosulfonate composite (Fe3O4@ MCLS) for the reductive removal of Cr(VI) and other heavy metals from water. J. Hazard. Mater. Adv. 2025, 17, 100536. [Google Scholar] [CrossRef]
- Behroozi, A.H.; Al-Shaeli, M.; Vatanpour, V. Fabrication and modification of nanofiltration membranes by solution electrospinning technique: A review of influential factors and applications in water treatment. Desalination 2023, 558, 116638. [Google Scholar] [CrossRef]
- Rogina, A. Electrospinning process: Versatile preparation method for biodegradable and natural polymers and biocomposite systems applied in tissue engineering and drug delivery. Appl. Surf. Sci. 2014, 296, 221–230. [Google Scholar] [CrossRef]
- Xu, D.; Samways, D.S.; Dong, H. Fabrication of self-assembling nanofibers with optimal cell uptake and therapeutic delivery efficacy. Bioact. Mater. 2017, 2, 260–268. [Google Scholar] [CrossRef]
- Zhao, J.; Han, W.; Chen, H.; Tu, M.; Zeng, R.; Shi, Y.; Cha, Z.; Zhou, C. Preparation, structure and crystallinity of chitosan nano-fibers by a solid–liquid phase separation technique. Carbohydr. Polym. 2011, 83, 1541–1546. [Google Scholar] [CrossRef]
- Ugo, P.; Moretto, L.M. Moretto, Template deposition of metals. In Handbook of Electrochemistry; Elsevier: Amsterdam, The Netherlands, 2007; pp. 678–709. [Google Scholar] [CrossRef]
- Saleem, H.; Trabzon, L.; Kilic, A.; Zaidi, S.J. Recent advances in nanofibrous membranes: Production and applications in water treatment and desalination. Desalination 2020, 478, 114178. [Google Scholar] [CrossRef]
- Chen, C.; Dirican, M.; Zhang, X. Centrifugal spinning—High rate production of nanofibers. In Electrospinning: Nanofabrication and Applications; Elsevier: Amsterdam, The Netherlands, 2019; pp. 321–338. [Google Scholar] [CrossRef]
- Khan, W.S.; Asmatulu, R.; Ceylan, M.; Jabbarnia, A. Recent progress on conventional and non-conventional electrospinning processes. Fibers Polym. 2013, 14, 1235–1247. [Google Scholar] [CrossRef]
- Gao, Y.; Shi, J.; Yuan, D.; Xu, B. Imaging enzyme-triggered self-assembly of small molecules inside live cells. Nat. Commun. 2012, 3, 1033. [Google Scholar] [CrossRef]
- Zhang, H.; Ji, X.; Li, P.; Liu, C.; Lou, J.; Wang, Z.; Wen, W.; Xiao, Y.; Zhang, M.; Zhu, X. Liquid-liquid phase separation in biology: Mechanisms, physiological functions and human diseases. Sci. China Life Sci. 2020, 63, 953–985. [Google Scholar] [CrossRef]
- Garg, T.; Rath, G.; Goyal, A.K. Biomaterials-based nanofiber scaffold: Targeted and controlled carrier for cell and drug delivery. J. Drug Target. 2015, 23, 202–221. [Google Scholar] [CrossRef]
- Xu, T.-C.; Han, D.-H.; Zhu, Y.-M.; Duan, G.-G.; Liu, K.-M.; Hou, H.-Q. High strength electrospun single copolyacrylonitrile (coPAN) nanofibers with improved molecular orientation by drawing. Chin. J. Polym. Sci. 2021, 39, 174–180. [Google Scholar] [CrossRef]
- Marjuban, S.M.H.; Rahman, M.; Duza, S.S.; Ahmed, M.B.; Patel, D.K.; Rahman, S.; Lozano, K. Recent advances in centrifugal spinning and their applications in tissue engineering. Polymers 2023, 15, 1253. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; He, D.; Zhao, H.; Wang, C.; Wang, K. Study on high efficiency and fast photodegradation of Bi2WO6/BiOBr/PAN nanofibrous film. J. Mater. Res. Technol. 2022, 17, 2818–2830. [Google Scholar] [CrossRef]
- Sanchez, L.M.; Espinosa, E.; Zélis, P.M.; Martín, R.M.; Niza, J.d.H.; Rodríguez, A. Cellulose nanofibers/PVA blend polymeric beads containing in-situ prepared magnetic nanorods as dye pollutants adsorbents. Int. J. Biol. Macromol. 2022, 209, 1211–1221. [Google Scholar] [CrossRef]
- Van-Pham, D.-T.; Nhi, P.T.Y.; Long, T.V.B.; Nguyen, C.-N.; Nhan, L.M.; Quyen, T.T.B.; Tuyen, L.T.C.; Mai, N.T.N.; Thien, D.V.H. Electrospun Fe-doped TiO2/chitosan/PVA nanofibers: Preparation and study on photocatalytic and adsorption properties. Mater. Lett. 2022, 326, 132930. [Google Scholar] [CrossRef]
- Long, J.; Ren, T.; Han, J.; Li, N.; Chen, D.; Xu, Q.; Li, H.; Lu, J. Heterostructured BiFeO3@CdS nanofibers with enhanced piezoelectric response for efficient piezocatalytic degradation of organic pollutants. Sep. Purif. Technol. 2022, 290, 120861. [Google Scholar] [CrossRef]
- Kim, C.-Y.; Kim, S.H.; An, H.-R.; Park, J.-I.; Jang, Y.; Seo, J.; Kim, H.; Son, B.; Jeong, Y.; Jeong, B.; et al. Iron oxide incorporated carbide nanofiber composites for removal of organic pollutants and heavy metals from water. Ceram. Int. 2023, 49, 17984–17992. [Google Scholar] [CrossRef]
- Liu, J.; Wang, H.; Chang, M.-J.; Sun, M.; He, Z.-W.; Zhang, C.-M.; Zhu, W.-Y.; Chen, J.-L.; Du, H.-L.; Peng, L.-G.; et al. Magnetically separatable CoFe2O4/BiOCl: Controllable synthesis, superior photocatalytic performance and mechanism towards decomposing RhB, NOR and Cr(VI) under visible light. Colloids Surf. A Physicochem. Eng. Asp. 2022, 648, 129299. [Google Scholar] [CrossRef]
- Kumar, M.; Tiwari, A.; Randhawa, J.K. Electrospun nanofibers of α-hematite/polyacrylonitrile/calcium carbonate/cellulose triacetate as a multifunctional platform in, wastewater treatment and remineralisation. Desalination 2022, 541, 116030. [Google Scholar] [CrossRef]
- Chaudhuri, S.; Wu, C.-M.; Motora, K.G. Highly efficient solar-light-driven self-floatable WO2.72@Fe3O4 immobilized cellulose nanofiber aerogel/polypropylene Janus membrane for interfacial photocatalysis. J. Photochem. Photobiol. A Chem. 2023, 438, 114525. [Google Scholar] [CrossRef]
- Gao, Y.; Zhao, Z.; Song, L.; Cao, D.; Zhou, S.; Gao, T.; Shang, J.; Cheng, X. Eggshell supported Cu doped FeOx magnetic nanoparticles as peroxymonosulfate activator for carbamazepine degradation. Chem. Eng. J. 2023, 454, 140282. [Google Scholar] [CrossRef]
- Mahadadalkar, M.A.; Park, N.; Yusuf, M.; Nagappan, S.; Nallal, M.; Park, K.H. Electrospun Fe doped TiO2 fiber photocatalyst for efficient wastewater treatment. Chemosphere 2023, 330, 138599. [Google Scholar] [CrossRef]
- Teng, Y.; Li, W.; Wang, J.; Jia, S.; Zhang, H.; Yang, T.; Li, X.; Li, L.; Wang, C. A green hydrothermal synthesis of polyacrylonitrile@carbon/MIL-101(Fe) composite nanofiber membrane for efficient selective removal of tetracycline. Sep. Purif. Technol. 2023, 315, 123610. [Google Scholar] [CrossRef]
- Yang, H.; Dai, H.; Chen, Y.; Wan, X.; Li, F.; Xu, Q. Efficient and simple simultaneous adsorption removal of multiple mycotoxins from environmental water. Sep. Purif. Technol. 2023, 317, 123888. [Google Scholar] [CrossRef]
- Vatanpour, V.; Paziresh, S.; Behroozi, A.H.; Karimi, H.; Esmaeili, M.S.; Parvaz, S.; Ghazanlou, S.I.; Maleki, A. Fe3O4@Gum Arabic modified polyvinyl chloride membranes to improve antifouling performance and separation efficiency of organic pollutants. Chemosphere 2023, 328, 138586. [Google Scholar] [CrossRef] [PubMed]
- Aboelfetoh, E.F.; Aboubaraka, A.E.; Ebeid, E.-Z.M. Synergistic Effect of iron and copper oxides in the removal of organic dyes through thermal induced catalytic degradation process. J. Clust. Sci. 2023, 34, 2521–2535. [Google Scholar] [CrossRef]
- Mao, Y.; Lin, L.; Chen, Y.; Yang, M.; Zhang, L.; Dai, X.; He, Q.; Jiang, Y.; Chen, H.; Liao, J.; et al. Preparation of site-specific Z-scheme g-C3N4/PAN/PANI@LaFeO3 cable nanofiber membranes by coaxial electrospinning: Enhancing filtration and photocatalysis performance. Chemosphere 2023, 328, 138553. [Google Scholar] [CrossRef]
- Fuentes-García, J.; Sanz, B.; Mallada, R.; Ibarra, M.; Goya, G. Magnetic nanofibers for remotely triggered catalytic activity applied to the degradation of organic pollutants. Mater. Des. 2023, 226, 111615. [Google Scholar] [CrossRef]
- Moatmed, S.M.; Khedr, M.; El-Dek, S.; El-Deen, A.G. Multifunctional nanofiber membrane for high removal efficiency of biological/organic contaminations and oil-in-water emulsion under gravity-driven separation. J. Mol. Liq. 2024, 394, 123748. [Google Scholar] [CrossRef]
- Huang, Z.; Zhang, X.; Zhu, Z.; Guo, Z.; Wang, X.; Zhu, L.; Zhang, G.; Liu, B.; Xu, D. Enhanced peroxymonosulfate activation by highly magnetic FeCo-CoFe2O4 biphasic fibers for norfloxacin degradation. Chem. Eng. J. 2024, 480, 147883. [Google Scholar] [CrossRef]
- Bi, F.; Zhou, B.; Li, R.; Du, R.; Zheng, Z.; Fu, X.; Zhao, L.; Xiao, S.; Wang, L.; Dong, X. An efficient, green, and magnetic recycling wastewater treatment technique enabled by ZnO/NiFe2O4/BiOBr 3D nanofibers photo-fenton process. Mater. Sci. Semicond. Process. 2025, 185, 108936. [Google Scholar] [CrossRef]
- Rong, J.; Haipeng, B.; Ijaz, M.F.; Shahzad, K.; Mushtaq, S.; Iqbal, Y. Multifunctional magnetic SiO2/Fe3O4@MXene nanofiber membranes for reusable removal of doxorubicin and organic dyes from industrial wastewater. Inorg. Chem. Commun. 2025, 182, 115678. [Google Scholar] [CrossRef]
- Amini, F.; Delavari, H.H.; Ghovvati, S.; Poursalehi, R. Electrospinning optimization for enhanced hydrophilicity and surface functionality of Poly(ε-caprolactone)/Polyethyleneimine nanofibers with embedded Fe3O4 nanoparticles. J. Polym. Res. 2025, 32, 366. [Google Scholar] [CrossRef]
- Lakshmi, C.N.; Irfan, M.; Singh, N. Doping effects of Sn in 1-D α-Fe2O3 electrospun nanofibers for improved photocatalytic degradation of ciprofloxacin: Kinetics, intermediates identification and mechanisms. Inorg. Chem. Commun. 2025, 176, 114243. [Google Scholar] [CrossRef]
- Wang, W.; Li, M.; Si, J.; Wang, Q.; Qiu, S.; Xu, Y.; Liu, X.; Cui, Z. Preparation and characterization of PA6/PANI/α-Fe2O3-x composite nanofiber membranes for the removal of tetracycline from wastewater. J. Water Process. Eng. 2025, 77, 108383. [Google Scholar] [CrossRef]
- Jian, S.; Shi, F.; Hu, R.; Liu, Y.; Chen, Y.; Jiang, W.; Yuan, X.; Hu, J.; Zhang, K.; Jiang, S.; et al. Electrospun magnetic La2O3–CeO2–Fe3O4 composite nanofibers for removal of fluoride from aqueous solution. Compos. Commun. 2022, 33, 101194. [Google Scholar] [CrossRef]
- Huang, Y.; Zhou, J.; Zhang, Y.; Yan, L.; Bao, S.; Yin, Y.; Lu, J. Encapsulating hollow Fe3O4 in intertwined N-doped carbon nanofibers for high-performance supercapacitors and sodium-ion batteries. J. Alloys Compd. 2022, 918, 165672. [Google Scholar] [CrossRef]
- Poudel, M.B.; Shin, M.; Kim, H.J. Interface engineering of MIL-88 derived MnFe-LDH and MnFe2O3 on three-dimensional carbon nanofibers for the efficient adsorption of Cr(VI), Pb(II), and As(III) ions. Sep. Purif. Technol. 2022, 287, 120463. [Google Scholar] [CrossRef]
- Neskoromnaya, E.A.; Khamizov, R.K.; Melezhyk, A.V.; Memetova, A.E.; Mkrtchan, E.S.; Babkin, A.V. Adsorption of lead ions (Pb2+) from wastewater using effective nanocomposite GO/CMC/FeNPs: Kinetic, isotherm, and desorption studies. Colloids Surfaces A Physicochem. Eng. Asp. 2022, 655, 130224. [Google Scholar] [CrossRef]
- Torasso, N.; Vergara-Rubio, A.; Pereira, R.; Martinez-Sabando, J.; Baudrit, J.R.V.; Cerveny, S.; Goyanes, S. An in situ approach to entrap ultra-small iron oxide nanoparticles inside hydrophilic electrospun nanofibers with high arsenic adsorption. Chem. Eng. J. 2022, 454, 140168. [Google Scholar] [CrossRef]
- Tripathy, M.; Hota, G. Maghemite and graphene oxide embedded polyacrylonitrile electrospun nanofiber matrix for remediation of arsenate ions. ACS Appl. Polym. Mater. 2020, 2, 604–617. [Google Scholar] [CrossRef]
- Ye, D.; Gao, Q.; Li, T.; Wu, X.; Wu, Y. Photo-Fenton catalytic anti-fouling membranes for efficient elimination of radionuclides and organic contaminants. Desalination 2023, 553, 116461. [Google Scholar] [CrossRef]
- Feng, S.; Gao, J.; Li, S.; Cao, X.; Ni, J.; Yue, X.; Zheng, W.; Li, Y.; Hu, X.; Zhang, Y.; et al. Amino modified nanofibers anchored to Prussian blue nanoparticles selectively remove Cs+ from water. J. Environ. Sci. 2024, 146, 39–54. [Google Scholar] [CrossRef] [PubMed]
- Halder, S.; Chakraborty, C. Fe(II)-based metallo-supramolecular polymer nanostructures: An efficient electrode material for highly sensitive electrochemical detection and abatement of hexavalent chromium. J. Environ. Chem. Eng. 2023, 11, 110183. [Google Scholar] [CrossRef]
- Musa, M.A.; Shao, H.; Xu, D.; Sun, F.; Dong, X.; Azis, R.S.; Ugya, A.Y.; Ari, H.A. Enhanced visible light photocatalytic reduction of Cr (VI) by Bi2WO6 nanosheet/CuFe2O4 nanofiber heterojunctions. J. Photochem. Photobiol. 2023, 14, 100166. [Google Scholar] [CrossRef]
- Rind, I.K.; Tuzen, M.; Sarı, A.; Lanjwani, M.F.; Memon, N.; Saleh, T.A. Synthesis of TiO2 nanoparticles loaded on magnetite nanoparticles modified kaolinite clay (KC) and their efficiency for As(III) adsorption. Chem. Eng. Res. Des. 2023, 191, 523–536. [Google Scholar] [CrossRef]
- Luo, Y.; Hu, Z.; Lei, X.; Wang, Y.; Guo, X. Fluorescent magnetic chitosan-based hydrogel incorporating Amino-Functionalized Fe3O4 and cellulose nanofibers modified with carbon dots for adsorption and detection of Cr (VI). Colloids Surf. A Physicochem. Eng. Asp. 2023, 658, 130673. [Google Scholar] [CrossRef]
- Yang, F.; Zhu, F.; Shi, H.; Dong, X.; Sheng, J.; Zhou, J. Magnetic nanofibers in heavy metal arsenic(V) pollution control research. Langmuir 2025, 41, 9392–9405. [Google Scholar] [CrossRef]
- Khalith, M.; Karuppannan, S.B.; Ramalingam, R.; Raiyan, D.; Vijayalakshmi, S.; Arunachalam, K.D. Fabrication and characterization of electrospun nanofibers infused with hematite nanoparticles for the remediation of heavy metals from aqueous medium. Nano-Struct. Nano-Objects 2025, 42, 101452. [Google Scholar] [CrossRef]
- Hassan, A.; El-Aziz, A.A.; Elwi, M.; Klingner, A. Fabrication of Iron oxide nanoparticle loaded recycled PET nanofibers for water purification applications. Discov. Mater. 2025, 5, 153. [Google Scholar] [CrossRef]
- Al-Fiydh, M.N.; Najm, H.F.; Karam, F.F.; Baqir, S.J. Thermodynamics, kinetic study and equilibrium isotherm analysis of cationic dye adsorption by ternary composite. Results Chem. 2024, 10, 101680. [Google Scholar] [CrossRef]
- Mashile, G.P.; Dimpe, K.M.; Nomngongo, P.N. A Biodegradable Magnetic Nanocomposite as a Superabsorbent for the Simultaneous Removal of Selected Fluoroquinolones from Environmental Water Matrices: Isotherm, Kinetics, Thermodynamic Studies and Cost Analysis. Polymers 2020, 12, 1102. [Google Scholar] [CrossRef] [PubMed]
- Melhi, S. Recyclable Magnetic Nanocomposites For Efficient Removal Of Cadmium Ions From Water: Performance, Mechanism And Isotherm Studies. Environ. Pollut. Bioavailab. 2023, 35, 2163922. [Google Scholar] [CrossRef]
- Cui, B.; Chen, Z.; Wang, F.; Zhang, Z.; Dai, Y.; Guo, D.; Liang, W.; Liu, Y. Facile Synthesis of Magnetic Biochar Derived from Burley Tobacco Stems towards Enhanced Cr(VI) Removal: Performance and Mechanism. Nanomaterials 2022, 12, 678. [Google Scholar] [CrossRef]
- Leone, V.O.; Pereira, M.C.; Aquino, S.F.; Oliveira, L.C.A.; Correa, S.; Ramalho, T.C.; Gurgel, L.V.A.; Silva, A.C. Adsorption of Diclofenac on a Magnetic Adsorbent Based on Maghemite: Experimental and Theoretical Studies. New J. Chem. 2018, 42, 437–449. [Google Scholar] [CrossRef]
- Zhao, A.; Tang, Q.; Chen, Y.; Qiu, C.; Huang, X. Magnetic Adsorbent Fe3O4/ZnO/LC for the Removal of Tetracycline and Congo Red from Aqueous Solution. Molecules 2023, 28, 6499. [Google Scholar] [CrossRef]
- Revellame, E.D.; Fortela, D.L.; Sharp, W.; Hernandez, R.; Zappi, M.E. Adsorption kinetic modeling using pseudo-first order and pseudo-second order rate laws: A review. Clean. Eng. Technol. 2020, 1, 100032. [Google Scholar] [CrossRef]
- Scaffaro, R.; Maio, A.; Gammino, M. Electrospun Polymeric Nanohybrids with Outstanding Pollutants Adsorption and Electroactivity for Water Treatment and Sensing Devices. Adv. Compos. Hybrid Mater. 2024, 7, 1–19. [Google Scholar] [CrossRef]
- Patil, A.G.; Vivekanandan, A.K.; Hemavathi, A.B.; Suri, S.; Pande, R.; Kagale, S.; Yadav, S.; Patil, G. Sustainable Wastewater Treatment Using Magnetic Nanohybrids. In IWA Publishing eBooks; UWA Publishing: Nedlands, Australia, 2023; p. 113. [Google Scholar] [CrossRef]
- Zheng, Z.; He, J.; Zhang, Z.; Kumar, A.; Khan, M.; Lung, C.W.; Lo, I.M.C. Magnetically Recyclable Nanophotocatalysts in Photocatalysis-Involving Processes for Organic Pollutant Removal from Wastewater: Current Status and Perspectives. Environ. Sci. Nano 2024, 11, 1784–1816. [Google Scholar] [CrossRef]


| Method | Merits | Demerits | Ref. |
|---|---|---|---|
| Ball milling | Low cost; widely used; scalable process | Requires elevated temperatures; potential contamination from milling media | [58] |
| Laser evaporation | Chemical-free process; high-purity products; good stoichiometric control | High equipment cost; limited scalability; complex instrumentation | [59] |
| Wire explosion | Environmentally friendly; low energy consumption; high product purity | Produces polydisperse nanoparticles; limited control over size distribution | [60] |
| Electron beam lithography | Enables fabrication of nanoscale patterns and two-dimensional geometries with high precision | Requires complex instrumentation; limited throughput; high operational cost | [61] |
| Coprecipitation | Simple and reproducible; produces ultrafine powders without intermediate phases; cost-effective | Requires strict pH control; possible residual impurities if insufficiently washed | [62] |
| Sol–gel | Versatile; low-temperature processing; high crystallinity and purity | Limited production yield; relatively long processing time | [63] |
| Spray pyrolysis | Compatible with various precursors; high purity; good control of particle size distribution | Requires high processing temperatures; possible minor product contamination | [64] |
| Self-combustion | Simple and rapid; produces single-phase powders; energy-efficient | Limited control over particle size; high local reaction temperatures | [65] |
| Thermal decomposition | Excellent control over particle size and morphology; high crystallinity | Requires elevated reaction temperatures; high energy consumption; organic solvent use | [66] |
| Microemulsion | Thermodynamically stable system; uniform particle dispersion; narrow size distribution | Limited production yield; requires surfactants and organic solvents | [67] |
| Hydrothermal | Simple process; high crystallinity; good control of morphology | Requires elevated temperature and pressure conditions; specialized autoclave equipment | [68] |
| Solvothermal | Minimizes adsorption of anions and cations; reduces contamination; good size control | Requires elevated temperatures; organic solvent handling | [69] |
| Pechini | Low toxicity precursors; fine powder formation; good compositional control; high purity | Long processing time; limited material yield; possible minor contamination | [70] |
| Polyol | Precise control over nucleation and growth; nanometric particle size; low agglomeration | Slow reaction kinetics; limited scalability. | [71] |
| Self-assembly | Enables fabrication of sub-20 nm molecular patterns; atomically precise nanostructures | Difficult design and fabrication; limited large-scale applicability | [72] |
| Laser pyrolysis | Produces very small particles; rapid synthesis | Poor crystallinity due to short residence time; broad size distribution | [73] |
| Microwave | Energy-efficient; controllable reaction parameters; short processing time | Possible variation in physicochemical properties; limited scalability | [74] |
| Arc discharge | Cost-effective; catalyst-free process | Limited control over particle size and shell thickness; not suitable for industrial scale | [75] |
| Sonochemical | Rapid synthesis; ultrafine powder formation | Possible structural defects; impurity incorporation; limited uniformity | [76] |
| Oxidation reduction | Fast reaction rates; simple chemistry | Gas contamination risk; high capital and operational costs | [77] |
| Chemical vapor deposition | Controllable deposition rate; suitable for industrial-scale production; high surface-to-volume ratio | Requires complex equipment; possible structural defects | [78] |
| Combustion synthesis | Simple and rapid; energy-efficient; scalable | Generates gaseous emissions (CO2, CO); limited size control | [79] |
| Adsorbent Material | Synthesis Method | Removal Mechanism | Pollutant(s) | Isotherm Model | Removal Efficiency (%, mg/g) | Ref. |
|---|---|---|---|---|---|---|
| TiO2/Fe3O4 | - | Adsorption | Sulfamethazine | L; F | 88.00% | [83] |
| 0.67BiFeO3–0.33BaTiO3 | Sol–Gel | Catalysis | Methylene Blue, Rhodamine and Crystal violet. | L | Mb: 98.00% Rb: 91.00% Cv: 88.00% | [84] |
| Graphene-magnetite functionalized diatomite | - | Adsorption | Organochlorine-pesticides | L | 97.00% | [85] |
| CoFe2O4/PMS | Hydrothermal and Coprecipitation | Catalysis | Phenol sulfonic acid | - | 90.00% | [86] |
| Fly-AsFe3O4 | Precipitation | Adsorption | Red Dye | L; F | 153 mg/g | [87] |
| NiFe2O4 | Coprecipitation | Photocatalysis | Titan Yellow | L | 98.80% | [88] |
| COFe2O4/SnO2 | Sol–Gel | Photocatalysis | Indigo carmine dye | - | 85% | [89] |
| Fe3O4/HMIL | - | Adsorption | Coomassie brilliant blue R-250 | L; F | ~93.00–98.00% | [90] |
| Magnetic MPANI@La | Oxidation-polymerization | Adsorption | Phosphate ions | L | 92.49% | [91] |
| NiO/Co@Cmagnetic | Solvothermal | Adsorption | Organic nitrogen pesticides | L | 62.20 mg/g | [92] |
| CAF@Fe3O4 | Green | Photocatalysis | Methyl orange | L | 100.00% | [93] |
| Fe/Zr-MOFs | Solvothermal | Adsorption | Doxycycline hydrochloride | F | 87.50% | [94] |
| CoFe2O4@ZnMOF/Graphene | Precipitation | Photocatalysis | Diazinón | L | 97.38% | [95] |
| Fe3O4@IL | Coprecipitation | Adsorption | Ionic silver | F | 100.00% | [96] |
| Fe3O4@MgO | Microwave | Photocatalysis | Rhodamine B | L | 99.00% | [97] |
| Zr-doped Fe3O4 | Solvothermal | Photocatalysis | Diazinon Phosphorus | - | 97.50% | [98] |
| a-Fe2O3/Cu2O | Hydrothermal | Photocatalysis | Benzotriazoles | - | 100.00% | [99] |
| SBH-Fe3O4 | Co-precipitation | Adsorption | Chloroquine | L | 98.84 mg/g | [100] |
| Fe3O4/CA | Green/coprecipitation | Photocatalyst | Metylene Blue | - | 93.14% | [101] |
| NdFeO3 | Sol–gel-citrate | Photacatalyst | Lomefloxacin and methylene blue | L; F | Lf: 88.00% Mb: 95% | [102] |
| BiOI-Fe3O4 | Coprecipitation | Photocatalytic | Polystyrene | - | 73.00% | [103] |
| AHA-Fe3O4 | Hydrothermal | Chemisorption | Tetracycline | L; F | 91.36% | [104] |
| Fe-doped TiO2@Fe3O4 | Sol–Gel | Photocatalysis | Metronidazole | - | 99.37% | [105] |
| TiO2–Fe3O4 | Hydrothermal and Microwave | Photocatalysis | Metronidazole | - | 90.00% | [106] |
| Fe3O4@TiO2-P25 | Oxidative precipitation | Photocatalytic | Metoprolol | L | 87.00% | [107] |
| HC-FeNPs | Microwave-assisted | Adsorption | Ethoprophos Terbufos and Diazinon (DIA) | L | 95.00% | [108] |
| CTS@Fe3O4 | Coprecipitation | Adsorption | Caffeic acid Gallic acid Melanoidin | L | Ca: 185 mg/g Ga: 160 mg/g Mel: 580 mg/g | [109] |
| Nd and Mn co-doped SrFe12O19 | Microemulsion | Photocatalysis | Crystal violet | - | 90.70% | [110] |
| Fe3O4@Phe | Green | Adsorption | Ciprofloxacin | L | 49.27 mg/g | [111] |
| Co0.5Cu0.5Fe2O4 | Coprecipitation | Photocatalisys | Tetracycline | - | 86.00% | [112] |
| Fe3O4@SiO2@Salg | - | Adsorption | Organo phosphorus | L | 80.50–100.00% | [113] |
| CoFe2O4/WS2/PMS | Coprecipitation | Catalyst | Sulfathiazole | L | 97.12% | [114] |
| Mn2O3 and Co3O4 | Thermal decomposition | Catalysts | Xylene | - | 90.00% | [115] |
| BC/Fe3O4 | Coprecipitation | Photocatalyst | Dye mixture | - | 92.19% | [116] |
| BC/Fe3O4 | Coprecipitation | Adsorption | Methyl orange(MO) | L; F | 83.50% | [117] |
| α-Fe2O3/TiO2 | Sonochemical | Photocatalyst | Tetracycline | - | 97.50% | [118] |
| TiO2 doped Fe | Sol–Gel | Photodegradation | Congo red dye | - | 99.00% | [119] |
| CoFe2O4@MOF-5) | Coprecitation | Photodegradation | Metronidazole and penicillin-G | L; F | MTZ: 91.71% PCG: 89.31% | [120] |
| SnFe2O4@ZIF-8 | Precipitation | Photodegradation | sulfamethoxazole, ciprofloxacin, ampicillin, erythromycin | L | 90.00% | [121] |
| Fe3O4 | Hydrothermal | Photodegradation | Methylene Blue | L; F | 90.00% | [122] |
| Fe3O4-HKUST-1 | Hydrothermal | Photodegradation | Azo | L; F | 44.65 mg/g | [123] |
| Base cross-linked magnetic resin CH-EP@Fe3O4/AC | Coprecipitation | Adsorption | Malachite green (MG), Reactive red 120 (RR120), | F | MG: 146.30 mg/g RR120: 140.70 mg/g | [124] |
| Cit-Fe3O and @TiO2 | Coprecipitation and solvothermal | Photocatalysis | Eosin-Methylene Blue | L | Eosin: 96.00% Blue: 82.00% | [125] |
| Fe2O4@AC | Coprecipitation | Photocatalysis | Methylene Blue Congo Red | - | 99.90% | [126] |
| VFe2O4@g-C3N4 | Coprecipitation | Photodegradation | Sulfamethoxazole, Chloramphenicol Ciprofloxacin | - | SUF: 100.00% CIP: 94.00% CIF: 90.00% | [127] |
| Prosopis juliflora, impregnated with magnetic nanoparticles | Coprecipitation | Photodegradation | Congo red (CR) | F | 98.55% | [128] |
| Fe3O4 | Coprecipitation | Photodegradation | Anionic azo | L | 99.99% | [129] |
| Si@Fe | Green Method | Biodegradation | Malachite Green and polyethylene | L | Vm: 98.11 Poly: 82.92% | [130] |
| Mg0.5Co0.5Fe2O4 | Green Method | Photodegradation | Congo Red | L | 93.00% | [131] |
| Zn0.5Ni0.5FeCrO4 | Sol–gel/Green | Photodegradation | 4-nitrophenol and aniline | L | Nit: 80.00% Anil: 95.00% | [132] |
| NiFe2O4 | Green Method | Photodegradation | Methylene blue (MB); Azo | L; F | 97.00% | [133] |
| PANI/GO/MOF-Fe3O4 | - | Photodegradation | MO and Naproxen Sodium (NAP) | L; F | 239.78 mg/g 40.64 mg/g | [134] |
| α-Fe2O3@MgO | Hydrothermal | Photocatalysis | Crystal violet (CV) | L | 99.00% | [135] |
| MoS2 NPs Fe3O4/Cs/MoS2/Lac NPs | Hydrothermal | Photocatalysis | Laccase | F | 96.80% | [136] |
| CMC/Ge/citrate@Fe3O4 | Ionotropic Gelation | Adsorption/photocatalysis | Ciprofloxacin | L | 96.00% | [137] |
| MIL-101(Fe)@NiFe2O4 | Hydrothermal | Photocatalysis | Levofloxacin | F | 90.00% | [138] |
| CoFe2O4 | Solvothermal | Photocatalysis | Glycolysis | L | 100.00% | [139] |
| Fe2O3/TiO2 | Sol–Gel | Photocatalysis | Methylene Blue | L | MB: 97.71% | [140] |
| TiO2/Fe2O3 | Sol–Gel | Photocatalysis | Methyl Orange | - | Mo: 94.00% | [141] |
| g-C3N4@Fe3O4 and BNNS@Fe3O4 | Coprecipitation | Adsorption | Polyethylene, polystyrene | - | Polyet: 93.70% Polys: 86.56% | [142] |
| Fe3O4-Ce@BC/PS | Hydrothermal Coprecipitation | Photocatalysis | Soil Polycyclic aromatic hydrocarbons (PAHs) | - | 89.34% | [143] |
| SrFe12O19-Fe3O4 | Polymeric precursor | Photocatalysis | Remazol Red ultra Red/Green/Blue (RGB) dye | L | 100.00% | [144] |
| Ag@CoFe2O4/ h-BN | Hydrothermal and Microwave-assisted | Photocatalysis | Nitrophenols | - | 90.00% | [145] |
| ZnFe2O4 | Green hydrothermal | Photocatalyst | Tetracycline | - | 94.00% | [146] |
| rGO/AK/Fe3O4 | Coprecipitation and ultrasonication | Photocatalyst | Methylene blue | L, F, T, & D-R | 98.20% | [147] |
| LP-CDs@Fe3O4 | Hydrothermal | Photocatalyst | Methylene blue (MB) | - | 98.00% | [148] |
| Corn cobs (CC), iron oxide (IO) | Coprecipitation | Photocatalyst | Triclosan | L; S | 94.20% | [149] |
| ZnFe2O4/ZnO | Combustion | Photocatalyst | Congo Red | - | 90.00% | [150] |
| ZnFe2O4/TiO2 p-n | Sol–gel | Photocatalyst | Ammonia nitrogen | - | 98.52% | [151] |
| Adsorbent Material | Synthesis Method | Adsorption Mechanism | Pollutant(s) | Isotherm Model | Removal Efficiency (%, mg/g) | Ref. |
|---|---|---|---|---|---|---|
| GO/Fe–Mn | Precipitation | Adsorption | Lead (II) | L | 99.00% | [153] |
| Fe3O4-Bentonite | Coprecipitation | Adsorption | Chrome (VI) | L | 96.50% | [154] |
| Inulin-Fe3O4 | Ultrasonic | Adsorption | Co2+ Cu2+ Hg2+ | L | 152.5 mg/g 167.7 mg/g 19.8 mg/g | [155] |
| Fe@NSC | Coprecipitation | Adsorption | Arsenic (III) As(V) | L | 129.54 mg/g 178.65 mg/g | [156] |
| mPAC-SH magnetic | Hydrothermal | Adsorption | Mercury (II) | L | 99.44% | [157] |
| Fe3O4-OP-CS | Hydrothermal carbonization | Adsorption | Copper (II) Lead (II) | L | Cu:92.40% Pb: 94.10% | [158] |
| γ-Fe2O3 and Fe3O4 | Coprecipitation | Adsorption | Chromium and Cupper | L | 92.00% | [159] |
| Ni Fe2O4-TiO2 | Pechini | Photocatalyc | Arsenic (III) | L | 97.50% | [160] |
| Fe3O4-OP-CS | Chemical Precipitation | Adsorption | Cadmium (II) | L | 92.00% | [161] |
| Fe3O4@TpPa-NO2 | - | Adsorption | Lead (II) | L | 909.1 mg/g | [162] |
| Fe3O4 | Thermal -descomposition | Adsorption | Lead (II), Nickel(II), and Cadmium(II) | L | 27.18 mg/g | [163] |
| Fe3O4NPs and Fe/CuNPs AgNPs | Coprecipitation | adsorption | Lead (II), Copper(II), Cadmium (II) and Nickel (II) | D-R, F, L, and T | Pb: 98.39% Cu: 75.52% Cd: 51.54% Ni: 45.34% | [164] |
| Fe–Mn/GO | Hydrothermal | Oxidation and Absorption | As (III), As (V) | - | 90.00~97.00% | [165] |
| Fe/Zr | Hydrothermal | Adsorption | As(III) and As(V) | L | AS (III) 99.00% As(V) 99.80% | [166] |
| Si-Fe-GO | Sol–Gel | Adsorption | Uranium | L | 90.20% | [167] |
| Fe3O4@PSBC | Pyrolysis | Absorption | Chrome (VI) | L | 209 mg/g | [168] |
| (SiO2) with Fe3O4 | Sol–gel | Adsorption | Chrome (VI) | L | 64.80% | [169] |
| Co3O4 | Co-precipitation | Adsorption | Lead (II) | L | 99.44% | [170] |
| MnFe2O4@SBA-15-(CH2)3 | Hydrothermal | Adsorption | As(V), Cd(II), and Lead (II) | R-P | 96.00% | [171] |
| MoS2/Fe3O4 | Solvothermal | Adsorption | Mercury (II) | L; F | 97.00% | [172] |
| Cu Fe2O4 | Pechini-Sol–gel | Adsorption | Cu (II) | L | 377.36 mg/g | [173] |
| CS-Fe3O4/GO | Coprecipitation | Adsorption | Niquel (II) | L | 81.21% | [174] |
| Fe3O4-ACH | Coprecipitation | Adsorption | Cr(VI) | L | 94.10% | [175] |
| Fe3O4@NR-TMD-G1, Fe3O4@NR-TMD-G2 | Co-precipitation | Adsorption | Lead (II) and Cadmium(II) | F, L and D-R | 98.50%, 93.60% | [176] |
| NTs/PEI@alginate@NiFe2O4 | Hydrothermal | Adsorption | Zn2+and Pb2+ | L | Zn: 74.70% Pb: 97.09% | [177] |
| Fe3O4/ZnO | Pyrolisis | Adsorption | Chrome(VI) and Lead (II) | L | 66.23% 384.62 mg/g | [178] |
| ZIF-7 and MnFe2O4 | Precipitation and hydrothermal | Adsorption | Co2+ | L | 99.05% | [179] |
| Fe3O4/Mg-Al LDOs/AlS (SMA) | Coprecipitation | Adsorption | Chrome (VI) Cu (II) | L | 235.3 mg/g 669.2 mg/g | [180] |
| ZnO@Fe3O4 | Hydrothermal | Adsorption | Lead (II) and Cadmium (II) | - | Lead (II): 99.20–100.00% Cad: 99.60–100.00% | [181] |
| Fe3O4@AHA MNPs | Chemical Coprecipitation hydrothermal | Adsorption | La (III) | L | 90.00% | [182] |
| (mGO/CS) and mGO/PA | Coprecipitation | Adsorption | Cr(VI) Pb(II) | L | 95.00% | [183] |
| Cr0.5CoFe1.5O4 | Hydrothermal | Adsorption | La(III) Ce(III) Sm(III) Eu(III) | L | La: 11.51 mg/g Ce: 11.51 mg/g Sm: 14.62 mg/g Eu: 14.62 mg/g | [184] |
| BC/Al(OH)3-Fe3O4-NC | - | Adsorption | Co(II), Cd(II), Sr(II) | L | Co: 99.45%, Cd: 99.65% Sr: 99.78% | [185] |
| AC@Fe3O4 | Coprecipitation | Adsorption | Cd2+, Cu2+, Pb2+ As3+ | L; F | Pb: 89.70% Cu: 83.80% Cd: 82.80% As: 80.90% | [186] |
| Fe3O4@MCLS | Coprecipitation | Adsorption | Cr(VI) | L | 90.00% | [187] |
| Method | Advantages | Disadvantages | Ref. |
|---|---|---|---|
| Electrospinning | Fibers with nanometric sizes, low-cost technology, high surface ratio, high porosity, and improved mechanical properties. | Jet instability, limited control of pore size | [195] |
| Self-Assembly | It is a direct method for making multifunctional nanofibers. | Complex process, high cost, low productivity. | [196] |
| Phase Separation | Well-defined pore architecture and size with low equipment demands | Restricted to certain polymer systems, unsuitable for the fabrication of long continuous fibers | [197] |
| Template Synthesis | Templates of different sizes are used to create fibers of different diameters. | Problem in removing the pavilion | [198] |
| Drawing | Minimum equipment requirement | Nanofibers smaller than 100 nm cannot be obtained | [199] |
| Centrifugal Spinning | Versatile, low-cost, and high-production-rate process. | Difficulty in collecting the manufactured material | [200] |
| Adsorbent Material | NP Synthesis Method | Nanofiber Synthesis Method | Pollutant(s) | Adsorption Mechanism | Isotherm Model | Capacity Adsorption (%, mg/g) | Ref. |
|---|---|---|---|---|---|---|---|
| Contaminants | Organic | ||||||
| Bi2WO6/BiOBr/PAN | Solvothermal | Electrospinning | Levofloxacin | Photocatalysis | L | 95.25% | [201] |
| Magnetic PVA-CNF | Coprecipitation | Electrospinning | Methyl Orange | Adsorption | - | 60.00% | [202] |
| Fe-doped TiO2 | Solvothermal | Electrsopinning | Methyl Blue | Adsorption and Photocatalysis | - | 94.00% | [203] |
| BiFeO3@CdS | Hydrothermal | Electrsopinning | bisphenol A | Catalysis | L | 99.70% | [204] |
| IOC (Act-IOC)Fe3O4 | Pyrolysis | Electrsopinning | Tetracycline and ciprofloxacin | Catalysis | L | Tc: 92.00% Cip: 95.00% | [205] |
| CoFe2O4/BiOCl | Solvothermal | Electrsopinning | Rhodamine B, Norfloxacin. | Photocatalysis | L | RhB: 92.90% Nor:75.50% | [206] |
| α-Fe2O3/PAN/CaCO3/CTA (FPCC) | - | Electrospinning | Methylene Blue, Methyl Orange | Photocatalysis | L | Mb: 96.00% Mo: 95.00% | [207] |
| WO2.72@Fe3O4@cellulose | Solvothermal | Free-drying | Methyl Orange | Photocatalysis | - | 85.00% | [208] |
| FeCuOx/eggshell | Coprecipitation | Electrospinning | Carbamazepine | Catalysis | - | 85.90% | [209] |
| Fe doped TiO2 | Sol–gel | Electrospinning | Rhodamine B, methylene blue, Congo red and methyl orange | Photocatalysis | L | 97.00% 99.00% | [210] |
| Polyacrylonitrile@carbon/MIL-101(Fe) | Hydrothermal | Electrsopinning | Tetracycline | Adsorption | F | 392.64 mg/g | [211] |
| PDA-IL NFsM | - | Electrospinning | Mycotoxins | Adsorption | L | 83.00% | [212] |
| Fe3O4@GA/PVC | Coprecipitación | Phase separation | Reactive Red-195, Reactive Blue (RB19), and Rifampicin (Rif) antibiotic | Adsorption | L | RR195: 98.30% Rif: 96.50% RB19: 95.60% | [213] |
| Cel/α-Fe2O3-ZnO | Hydrothermal | Electrospinning | Reactive black 5 Rb5 | Adsorption | L | Rb5: 99.30 mg/g | [214] |
| g-C3N4/PAN/PANI@LaFeO3 | - | Electrospinning | Methylene blue, Methyl violet, Ciprofloxacin and Acetamiprid, Escherichia coli, Staphylococcus aureus | Adsorption | L | MB: 97.00% MV: 94.30% CIP: 87.60% AP: 88.90% E. coli 100.00%, S.aur: 80.00% | [215] |
| MnxFe2-xO4 | Sonochemical | Electrospinning | Methylene Blue | Catalysis | L | >80.00% | [216] |
| SiO2@Fe3O4@ PS. | - | Electrospinning | Methylene blue | Photocatalytic | - | 99.90% | [217] |
| FeCo-CoFe2O4 | Sol–gel | Electrospinning | Norfloxacin | Photocatalytic | - | 93.8% | [218] |
| ZnO/NiFe2O4/BiOBr 3D | Solvothermal | Electrospinning | Rhodamine B | Photocatalytic | L | 99.61% | [219] |
| SiO2/Ti3C2 MXene/Fe3O4 | Sol–gel | Electrospinning | Doxorubicin and Meyltne Blue | Photocatalytic degradation | - | >90.00% | [220] |
| PCL-PEI-Fe3O4 | Hydrothermal | Electrospinning | Congo-Red | Adsorption | L | 397.43 mg/g | [221] |
| Sn doped α-Fe2O3 | - | Electrospinning | Ciprofloxacin and Methylene blue | Photodegradation | L | Cip: 79.80% Mb: 82.70% | [222] |
| PA6/PANI/α-Fe2O3-x | Ultrasonication & hydrothermal | Electrospinning | Tetracycline | Photodegradation | - | 94.89% | [223] |
| Contaminants | Inorganic | ||||||
| La2O3–CeO2–Fe3O4 | - | Template-Electrospinning | Fluoride | Adsorption | L | 229.89 mg/g | [224] |
| Fe3O4@NC@MnO2 | Hydrothermal | Electrospinning | Re (VII) As (V) | Adsorption | L | 10.9 mg/g 236.3 mg/g | [225] |
| MnFe-LDH/MnFe2O3@3DNF | Hydrothermal | Electrospinning | Cr(VI), Pb(II) and As(III) | Adsorption | L and F | Cr: 79.00% Pb: 84.00% As: 73.00% | [226] |
| GO/CMC/FeNPs | Hydrothermal | Electrospiining | Pb | Adsorption | D-R | 1850 mg/g | [227] |
| ONPs in a PVA | - | Electrospinning | As(V) | Adsorption | L | 80.00% | [228] |
| PAN/gCN-NH2/Fe3O4: PCNFe | Solvothermal | Electrospinning | As(III) and As(V) | Adsorption | L | As(III): 97.00% As(V): 99.00% | [229] |
| AOP/ZIF-90@TA/ZVI (AZ@TZ) | - | Electrospinning | Uranium | Catalysis | L | 140.06 mg/g | [230] |
| PB/SiO2-NH2 NFs | - | Electrospnning | Cs+ | Adsorption | F | 75.36% | [231] |
| Poly-Fe modified GCE | - | Electrospinning | Cr (VI) | electrochemically | - | 90.00% | [232] |
| Bi2WO6 nanosheet/CuFe2O4 | Hydrothermal | Electrospinning | Cr (VI) to Cr (III) | Photocatalysis | - | 90.30- 96.04% | [233] |
| KC/TiO2-Fe3O4 | Precipitation | Nanoparticles | As (III) | Adsorption | L | 92.00–90.00% | [234] |
| AF-Fe3O4 NPs | - | Electrospinning | Cr (VI) | Adsorption | F | 212.1 mg/g | [235] |
| PAN/Fe3O4@CTAB | - | Electrospinning | As (V) | Adsorption | L | 97.00% | [236] |
| Hematite/poly(ε-caprolactone) and chitosan | Coprecipitation | Electrospinning | Cr6+ and Cs+ | Adsorption | L and F | Cr6+: 89.84% Cs+: 94.71% | [237] |
| PET-Fe3O4 | Coprecipitation | Electrospinning | Cu2+ | Adsorption | - | 19.8 m/mg | [238] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Murillo-Ortíz, R.; Martínez-Carreón, M.J.; Herrera-Rivera, R.; Nocedo-Mena, D.; Pérez-Tijerina, E.G. Magnetic Nanostructures for the Removal of Emerging Organic and Inorganic Pollutants: An Overview of Applications in Contaminated Water. Materials 2026, 19, 1057. https://doi.org/10.3390/ma19061057
Murillo-Ortíz R, Martínez-Carreón MJ, Herrera-Rivera R, Nocedo-Mena D, Pérez-Tijerina EG. Magnetic Nanostructures for the Removal of Emerging Organic and Inorganic Pollutants: An Overview of Applications in Contaminated Water. Materials. 2026; 19(6):1057. https://doi.org/10.3390/ma19061057
Chicago/Turabian StyleMurillo-Ortíz, Raquel, María J. Martínez-Carreón, Rosario Herrera-Rivera, Deyani Nocedo-Mena, and Eduardo G. Pérez-Tijerina. 2026. "Magnetic Nanostructures for the Removal of Emerging Organic and Inorganic Pollutants: An Overview of Applications in Contaminated Water" Materials 19, no. 6: 1057. https://doi.org/10.3390/ma19061057
APA StyleMurillo-Ortíz, R., Martínez-Carreón, M. J., Herrera-Rivera, R., Nocedo-Mena, D., & Pérez-Tijerina, E. G. (2026). Magnetic Nanostructures for the Removal of Emerging Organic and Inorganic Pollutants: An Overview of Applications in Contaminated Water. Materials, 19(6), 1057. https://doi.org/10.3390/ma19061057








