3.1. Optimization of High-Temperature Precipitation Parameters
Figure 1 shows the scanning electron microscopy (SEM) microstructure of alloy 2618 after solution treatment at 535 °C for 30 min and high-temperature precipitation for 10 min. At a precipitation temperature of 500 °C, a small amount of precipitate forms within the grain and at the grain boundaries of alloy 2618 (
Figure 1a). When the precipitation temperature is lowered to 485 °C, the quantity and particle size of precipitated phases at the grain boundaries increase (
Figure 1b). When the precipitation temperature is lowered to 470 °C, the precipitates with the grain and at the grain boundaries undergo significant growth (
Figure 1c). At a precipitation temperature of 455 °C, a large number of precipitated phases form within the grain and at the grain boundaries (
Figure 1d). These precipitated phases are continuously distributed at grain boundaries.
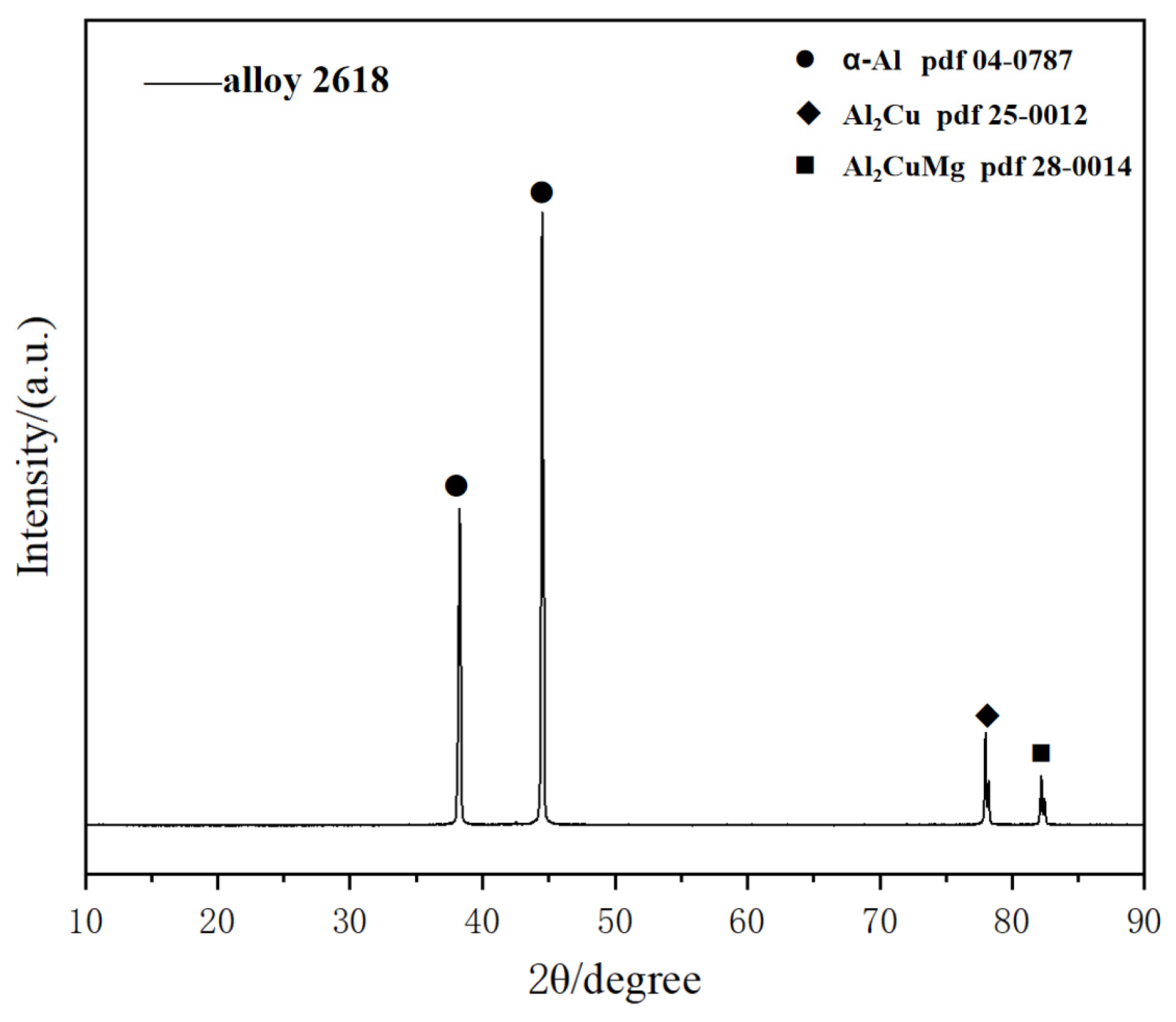
The energy spectrum analysis of the precipitated phase at the grain boundary in
Figure 1c is summarized in
Table 2, revealing the presence of Al
2Cu and Al
2CuMg phases. XRD images of alloy 2618 after precipitation at 470 °C for 10 min can also prove it (
Figure 2).
Figure 3 shows the transmission electron microscopy (TEM) microstructure of alloy 2618 after solution treatment at 535 °C for 30 min and high-temperature precipitation for 10 min. This allows for clearer observation of the distribution, size, and morphology of precipitates within the grain and at the grain boundaries.
When the precipitation temperature is 500 °C, numerous fine needle-shaped precipitates are present within the grains of alloy 2618, with granular precipitates appearing at the grain boundaries (
Figure 3a). These precipitated phases occupy a relatively narrow region. When the precipitation temperature is lowered to 485 °C, the needle-shaped precipitates within the grain become coarser (
Figure 3b). The number and particle size of granular precipitates at the grain boundaries increase significantly. In addition, the width of the grain boundary region occupied by these precipitated phases increases markedly. As shown in
Figure 3c, when the precipitation temperature is lowered to 470 °C, the long needle-shaped precipitates within the grain transform into short needle-shaped or granular phases. The precipitated phases at the grain boundaries undergo coarsening but remain intermittently distributed along the grain boundaries. The width of the grain boundary region occupied by these precipitated phases begins to decrease. When the precipitation temperature is further lowered to 455 °C, most of the long needle-shaped precipitates within the grain transform into short needle-shaped or granular phases, while particularly coarse granular precipitates simultaneously appear (
Figure 3d). In addition, the precipitated phases at the grain boundaries undergo significant coarsening, and the precipitates become nearly end-to-end connected, resulting in a continuous distribution at the grain boundaries. Due to the formation of coarse precipitates, solute atoms in the surrounding region are depleted and PFZ appear near the grain boundary.
High-temperature precipitation utilizes the tendency of alloy grain boundaries to preferentially precipitate phases, allowing solute atoms within the grains to diffuse towards the grain boundaries. During the heating process, the precipitated phase undergoes re-dissolution, increasing the solute concentration at and near the grain boundaries and reducing the width of the PFZ. The quantity, size, and morphology of grain boundary precipitates have a significant influence on subsequent dissolution during heating. Only a limited number of precipitated phases is required at the grain boundaries to ensure their rapid dissolution into the matrix during heating and holding. Therefore, considering the control of grain boundary precipitate quantity, size, and morphology, the optimal precipitation temperature for alloy 2618 was identified as 470 °C.
Grain boundaries are regions where various defects are concentrated and exhibit many characteristics distinct from those within the grains. Atoms at the grain boundaries deviate from their equilibrium positions and possess high potential energy. By slightly adjusting the position of atoms on the grain boundary, a new phase interface can be formed. The Gibbs free energy barrier for the nucleation of precipitated phases at the grain boundaries is lower than that within the grain. In addition, solute atoms diffuse more rapidly along grain boundaries, facilitating nucleation and the growth of new phases at the grain boundaries compared to within the grain. During the high-temperature precipitation of alloy 2618, the driving force for the formation of new phases at the grain boundaries is small, which limits precipitation within the grain. This creates favorable conditions for the preferential precipitation of new phases at the grain boundaries. In addition, solute atoms can continuously diffuse to the grain boundary precipitates at high temperatures, resulting in the formation of coarse granular precipitates at the grain boundaries.
When alloy 2618 undergoes high-temperature precipitation, the nucleation and growth of new phases are not significantly constrained by atomic diffusion kinetics. The thermodynamic driving force and solute supersaturation in the matrix are the main factors influencing the nucleation and growth of new phases. As the precipitation temperature decreases, the driving force for the formation of precipitates and the degree of supersaturation in the matrix increase, promoting the continuous growth and coarsening of precipitates.
Figure 4 shows the relationship between the impact toughness of alloy 2618 and its precipitation temperature. As the precipitation temperature decreases, the impact toughness of alloy 2618 initially increases rapidly, reaching a maximum at 470 °C, and then gradually decreases (
Figure 4). The impact energy of an alloy reflects a combination of its tensile strength and elongation at break. If the tensile strength and elongation at break are both high, the impact energy of the alloy will be relatively high [
17]. Alloy 2618 is strengthened mainly through two mechanisms: solid solution strengthening and precipitation strengthening [
18]. Since the strengthening of alloy 2618 primarily occurs through aging phases, the effect of precipitation strengthening is significantly greater than that of solid solution strengthening. When alloy 2618 is solution-treated at 535 °C for 30 min, copper and magnesium are largely dissolved in the matrix. At this stage, the primary strengthening mechanism is solid solution strengthening. When alloy 2618 is cooled to 500 °C for precipitation, the number of precipitates within the grain and at the grain boundaries remains relatively small, as the temperature is only slightly below the solid solution line. At this stage, the alloy is still mainly strengthened by solid solution, so the impact energy of alloy 2618 is relatively low. When alloy 2618 is cooled to 485 °C for precipitation, the supersaturation of solute atoms in the matrix increases and a greater number of precipitated phases appear within the grain and at the grain boundaries. Due to the increased strengthening effect of the precipitated phases, the impact energy of the alloy is significantly increased. When alloy 2618 is cooled to 470 °C for precipitation, the number of precipitated phases within the grain and at the grain boundaries further increases, leading to a corresponding rise in the impact energy of the alloy. At this stage, the impact toughness of the alloy reaches its maximum. As the precipitation temperature decreases to 455 and 440 °C, the number of precipitates within the grain and at grain boundaries increases; however, the coarsening of precipitates and the continuous distribution of grain boundary precipitates result in a continuous decrease in the impact toughness of the alloy. In addition, PFZs appeared near the grain boundaries. The presence of PFZs at the grain boundaries also contributed to the reduced impact toughness of the alloy [
19]. Therefore, based on the impact toughness of alloy 2618, the optimal precipitation temperature is 470 °C.
Figure 5 shows the SEM microstructure of alloy 2618 after precipitation at 470 °C for different durations. When the temperature of alloy 2618 was decreased from 535 to 470 °C, almost no precipitated phases were observed at the grain boundaries (
Figure 5a). When the alloy was held at 470 °C for 5 min, only a small amount of discontinuous, fine precipitates formed at the grain boundaries. Extending the precipitation time to 10 min led to an increase in the number and size of the precipitated phases at the grain boundaries. When the precipitation time was increased to 20 min, a large number of precipitation phases formed at the grain boundaries of the alloy. Furthermore, the precipitation phases were nearly continuously distributed along the grain boundaries. Therefore, to control the number of grain boundary precipitates and prevent their continuous distribution, the optimal precipitation time for alloy 2618 at 470 °C was set at 10 min.
3.3. Microstructure and Mechanical Properties of Alloy 2618
Figure 7 shows the TEM images of peak-aged alloy 2618 after different solution treatments. Conventional solid followed by peak aging involves solution treatment at 535 °C for 30 min, water quenching, and aging at 200 °C for 11 h. High-temperature precipitation and heating dissolution solution treatment followed by peak aging includes solid solution treatment at 535 °C for 30 min, high-temperature precipitation at 470 °C for 10 min, heating dissolution at 535 °C for 10 min, water quenching, and aging at 200 °C for 11 h.
As shown in
Figure 7a, the number of precipitated phases near the grain boundaries of alloy 2618 subjected to the conventional solid solution and peak aging process is relatively small, which are coarse and intermittently distributed. A wide PFZ of approximately 754 nm is observed near the grain boundary. As shown in
Figure 7b, the precipitates near the grain boundaries of alloy 2618, subjected to high-temperature precipitation, solution treatment, and aging, are relatively dense. A thin layer of continuous precipitated phase forms at the grain boundary, and the width of the PFZ is significantly reduced to approximately 263 nm.
To identify the precipitated phase at the grain boundary, energy dispersive analysis was conducted on the adjacent region. As shown in
Figure 8, the precipitate regions along the grain boundaries exhibited decreased aluminum content, increased iron and nickel contents, and markedly increased copper and magnesium contents. The precipitates at the grain boundaries are likely primarily Al
2Cu and Al
2CuMg, with a small amount of Al
9FeNi. In addition, compared with the matrix region far from the grain boundary, the region adjacent to the grain boundary precipitates lacks copper and magnesium, leading to the formation of PFZs. The formation and variation in width of PFZs at the grain boundaries of 2618 aluminum alloy can be explained by solute depletion theory.
Figure 9 shows the solute concentration distribution near the grain boundary of alloy 2618 after solid solution treatment, high-temperature precipitation, and heating dissolution. As shown in
Figure 9a, when alloy 2618 is subjected to solution treatment, the solute concentration within the grains and at the grain boundaries is essentially uniform. During the aging process, the S (Al
2CuMg) phase preferentially nucleates and grows at the grain boundaries, significantly lowering the solute concentration in the surrounding regions. The reduction in solute supersaturation results in the formation of a relatively wide PFZ, as shown in
Figure 7a. After high-temperature precipitation, the second phases in alloy 2618 preferentially nucleate and grow along the alloy grain boundaries (
Figure 9b). At this stage, the solute concentration near the grain boundary will decrease. As shown in
Figure 9c, during solution treatment, the second phases at the grain boundary will redissolve, allowing for the redistribution of solute atoms [
20]. By controlling the re-dissolution time, the solute concentration near the grain boundary becomes higher than in regions farther away, creating a solute distribution that helps reduce the width of PFZs and aids in further improving the grain boundary structure of the alloy [
21]. Therefore, the combination of high-temperature precipitation, solution treatment, and aging resulted in a significant reduction in the width of the PFZ in alloy 2618, as shown in
Figure 7b.
As shown in
Figure 10, curves A and B have similar slopes in the initial stage, indicating that the two samples have comparable elastic moduli in the elastic phase, meaning their resistance to elastic deformation is similar. Curve B exhibits a slightly higher yield stress than curve A, suggesting that sample B requires greater stress to initiate plastic deformation. The figure reveals that curve B shows a steeper stress increase during the strengthening phase compared to curve A, indicating more pronounced work hardening and greater strength improvement in sample B. Both curves A and B exhibit a necking stage, but the stress decline in curve B is relatively gentler, implying better toughness in sample B during necking, allowing it to withstand deformation without immediate fracture. Ultimately, the curves drop to zero as the material fractures. The figure demonstrates that curves A and B have different fracture strains, with curve B exhibiting a larger fracture strain, indicating better ductility in sample B and the ability to sustain greater deformation.
Table 3 presents the mechanical properties of peak-aged alloy 2618 after different solid solution treatments. As shown in
Table 3, the tensile strength, elongation at break, and impact toughness of aged alloy 2618 treated with the conventional solution were 462 MPa, 10.6%, and 30.3 J/cm
2, respectively. In comparison, these values increased by 5%, 1.9%, and 23.8%, respectively, following high-temperature precipitation and heating dissolution. These results indicate that the comprehensive mechanical properties of the alloy were significantly improved.
Figure 11 shows the tensile fracture morphologies of aged alloy 2618 after different solid solution treatments. As shown in
Figure 11a, the tensile fracture surface of aged alloy 2618 subjected to conventional solid solution treatment exhibits numerous ductile dimples and tearing edges. In addition, many protruding particles are observed on the fracture surface, which adversely affect the toughness of the alloy [
22]. In contrast, the tensile fracture surface of aged alloy 2618 after high-temperature precipitation and solution treatment showed numerous small dimples and fine tearing edges (
Figure 11b). Compared with the tensile fracture of the aged alloy 2618 after conventional solution treatment, a greater number of smaller ductile dimples and tearing edges were observed. In addition, the number of protruding particles on the fracture surface was significantly reduced, as the extended solution treatment time allowed the second phases to dissolve sufficiently into the alloy matrix. The tensile fracture characteristics of aged alloy 2618 indicate that high-temperature precipitation and solution treatment improved its comprehensive mechanical properties [
23].