Reusable NiCo/Cu Catalysts for Sustainable Hydrogen Generation
Highlights
- Electroless deposition provides a simple and cost-effective route to Ni–Co coatings on Cu.
- Catalytic activity strongly varies with Co content in the 4–90 wt.% range.
- Ni10Co90/Cu retains high stability, with only 12% activity loss after reuse.
- Synergistic Ni–Co interaction significantly enhances hydrogen generation efficiency.
- Co-rich Ni–Co coatings exhibit superior hydrogen generation activity.
- The Ni–Co/Cu system is a viable, low-cost catalyst for sustainable H2 production.
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemical Reagents
2.2. Catalyst Preparation and Processing
2.3. Structural and Surface Characterization Methods
2.4. Hydrolysis Measurements of NaBH4
3. Results and Discussion
3.1. Morphology and Composition of Coatings
3.2. Structural Characterization of the Catalysts (XRD)
3.3. Surface Chemistry and Oxidation States (XPS)
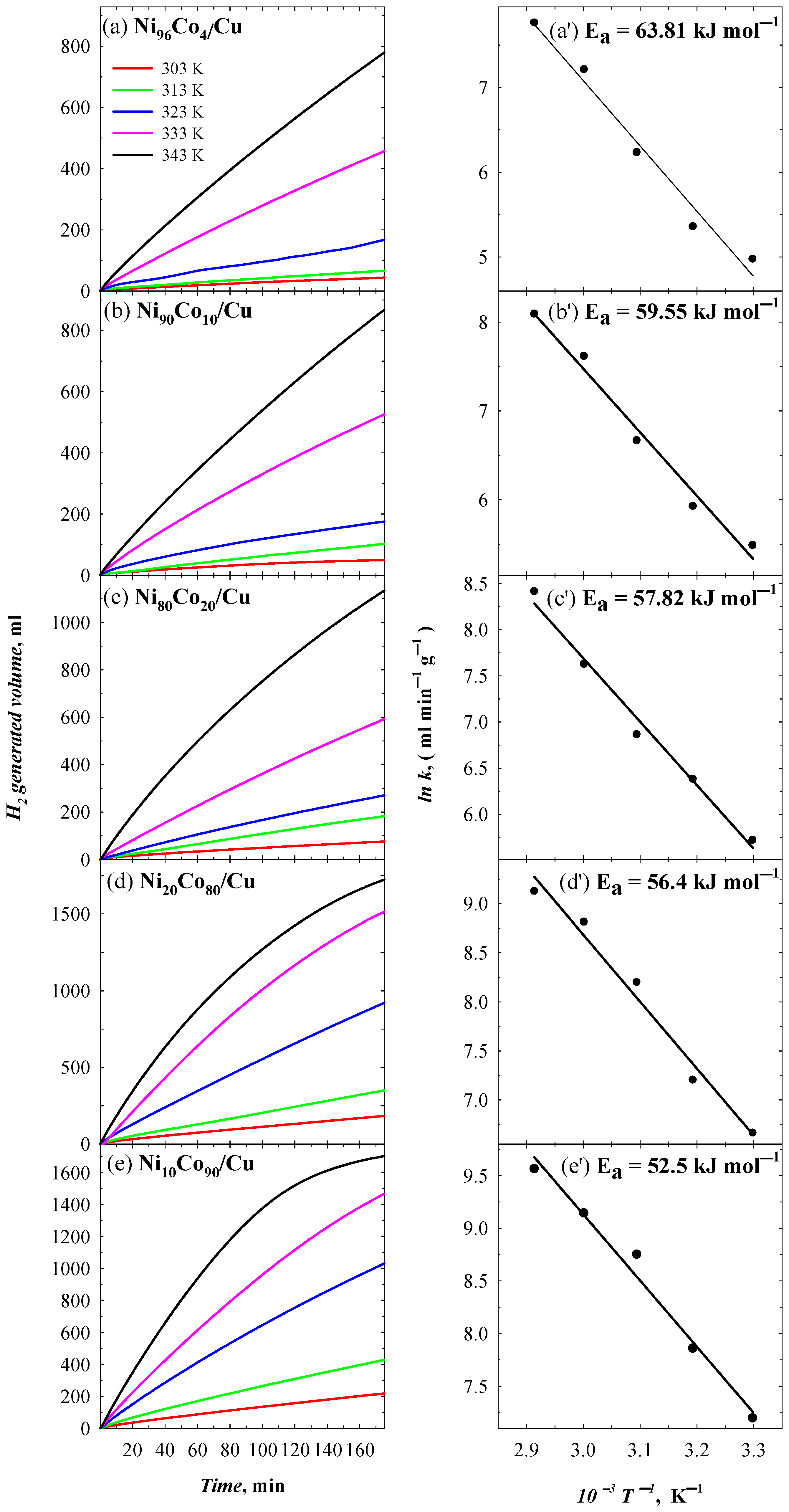
3.4. Catalytic Activity of NiCo/C Toward NaBH4 Hydrolysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Available online: https://atomfair.com/hydrogen-primer/article.php?id=G8-146 (accessed on 23 October 2025).
- Yan, X.L.; Hino, R. (Eds.) Nuclear Hydrogen Production Handbook, 1st ed.; CRC Press: Boca Raton, FL, USA, 2011. [Google Scholar] [CrossRef]
- Available online: https://www.scribd.com/document/234534909/Cre?utm_source (accessed on 23 October 2025).
- Available online: https://www.iea.org/reports/net-zero-by-2050 (accessed on 23 October 2025).
- Available online: https://airandspace.si.edu/collection-objects/fuel-cell-gemini/nasm_A19660646000 (accessed on 23 October 2025).
- Yusaf, T.; Mahamude, A.S.F.; Kadirgama, K.; Ramasamy, D.; Farhana, K.; Dhahad, H.A.; Talib, A.R.A. Sustainable hydrogen energy in aviation—A narrative review. Int. J. Hydrogen Energy 2024, 52, 1026–1045. [Google Scholar] [CrossRef]
- Virdi, P.S.; Guo, W.; Cattafesta, L.; Cheetham, P.; Cooley, L.; Gladin, J.; He, J.; Ionel, D.M.; Kim, C.; Li, H.; et al. Liquid hydrogen storage and transfer-control system for integrated zero emission aviation (IZEA). IOP Conf. Ser. Mater. Sci. Eng. 2024, 1302, 012024. [Google Scholar] [CrossRef]
- Jaramillo, D.E.; Moreno-Blanco, J.; Aceves, S.M. Evaluation of cryo-compressed hydrogen for heavy-duty trucks. Int. J. Hydrogen Energy 2024, 87, 928–938. [Google Scholar] [CrossRef]
- Jaffary, Z.; Wiedemann, M. Towards net zero aviation: Exploring safe hydrogen refuelling at airports. J. Air Transp. Res. Soc. 2025, 4, 100048. [Google Scholar] [CrossRef]
- Maciorowski, D.; Ludwiczak, A.; Kozakiewicz, A. Hydrogen, the future of aviation. Combust. Engines 2024, 197, 126–131. [Google Scholar] [CrossRef]
- Zhou, X.; Meng, X.F.; Wang, J.M.; Shang, N.Z.; Feng, T.; Gao, Z.Y.; Zhang, H.X.; Ding, X.L.; Gao, S.T.; Feng, C.; et al. Boron nitride supported NiCoP nanoparticles as noble metal-free catalyst for highly efficient hydrogen generation from ammonia borane. Int. J. Hydrogen Energy 2019, 44, 4764–4770. [Google Scholar] [CrossRef]
- Dabaro, M.D.; Kim, H. Improved hydrogen generation via NaBH4 hydrolysis: Synergistic role of sulfur and C4+-Doping unveils (440) high-index facets and modulates Co2+/Co3+ ratios in the Co3O4 lattice. Int. J. Hydrogen Energy 2024, 69, 660–674. [Google Scholar] [CrossRef]
- Zhao, L.; Wei, Q.; Zhang, L.; Zhao, Y.; Zhang, B. NiCo alloy decorated on porous N-doped carbon derived from ZnCo ZIF as highly efficient and magnetically recyclable catalyst for hydrogen evolution from ammonia borane. Renew. Energy 2021, 173, 273–282. [Google Scholar] [CrossRef]
- Arshad, F.; ul Haq, T.; Khan, A.; Haik, Y.; Hussain, I.; Sher, F. Multifunctional porous NiCo bimetallic foams toward water splitting and methanol oxidation-assisted hydrogen production. Energy Convers. Manag. 2022, 254, 115262. [Google Scholar] [CrossRef]
- Herraiz-Cardona, I.; Ortega, E.; Vazquez-Gomez, L.; Perez-Herranz, V. Electrochemical characterization of a NiCo/Zn cathode for hydrogen generation. Int. J. Hydrogen Energy 2011, 36, 11578–11587. [Google Scholar] [CrossRef]
- Serhat Akyuz, E.; Telli, E.; Farsak, M. Hydrogen generation electrolyzers: Paving the way for sustainable energy. Int. J. Hydrogen Energy 2024, 81, 1338–1362. [Google Scholar] [CrossRef]
- Wang, W.; Li, T.; Komarneni, S.; Lu, X.; Liu, B. Recent advances in Co-based co-catalysts for efficient photocatalytic hydrogen generation. J. Colloid Interface Sci. 2022, 608, 1553–1575. [Google Scholar] [CrossRef]
- Zhou, Z.; Zeng, L.; Xiong, G.; Yang, L.; Yuan, H.; Yu, J.; Xu, S.; Wang, D.; Zhang, X.; Liu, H.; et al. Multifunctional electrocatalyst of NiCo-NiCoP nanoparticles embedded into P-doped carbon nanotubes for energy-saving hydrogen production and upgraded conversion of formaldehyde. Chem. Eng. J. 2021, 426, 129214. [Google Scholar] [CrossRef]
- Wen, S.; Yang, T.; Zhao, N.; Ma, L.; Liu, E. Ni-Co-Mo-O nanosheets decorated with NiCo nanoparticles as advanced electrocatalysts for highly efficient hydrogen evolution. Appl. Catal. B Environ. 2019, 258, 117953. [Google Scholar] [CrossRef]
- Mondal, K.; Malode, S.J.; Shetti, N.P.; Alqarni, S.A.; Pandiaraj, S.; Alodhayb, A. Porous nanostructures for hydrogen generation and storage. J. Energy Storage 2024, 76, 109719. [Google Scholar] [CrossRef]
- Liu, T.; Li, N.; Wu, B.; Han, X.; Ding, Q.; Zhao, Q.; Cheng, Q.; Wang, R.; Li, D. Electroless deposition of Ni–Co–B–P coating as a durable electrode for hydrogen evolution reaction in alkaline media. Int. J. Hydrogen Energy 2025, 102, 900–908. [Google Scholar] [CrossRef]
- Wu, M.; Gao, M.; Qu, S.; Liu, Y.; Sun, W.; Liang, C.; Zhang, X.; Li, Z.; Yang, Y.; Pan, H. LiBH4 hydrogen storage system with low dehydrogenation temperature and favorable reversibility promoted by metallocene additives. J. Energy Storage 2023, 72, 108679. [Google Scholar] [CrossRef]
- Li, X.; Yan, Y.; Jensen, T.R.; Filinchuk, Y.; Dovgaliuk, I.; Chernyshov, D.; He, L.; Li, Y.; Li, H.W. Magnesium borohydride Mg(BH4)2 for energy applications: A review. J. Mater. Sci. Technol. 2023, 161, 170–179. [Google Scholar] [CrossRef]
- Shen, J.; Chen, W.; Lv, G.; Yang, Z.; Yan, J.; Liu, X.; Dai, Z. Hydrolysis of NH3BH3 and NaBH4 by graphene quantum dots-transition metal nanoparticles for highly effective hydrogen evolution. Int. J. Hydrogen Energy 2021, 46, 796–805. [Google Scholar] [CrossRef]
- Wang, Y.; Mao, X.; Yang, J.; Wang, J.; Guan, W.; Chen, J.; Han, B.; Tian, Z. One-step synthesis of Ni/yttrium-doped barium zirconates catalyst for on-site hydrogen production from NH3 decomposition. Int. J. Hydrogen Energy 2022, 47, 2608–2621. [Google Scholar] [CrossRef]
- Ridwan, M.; Suhanda, D.; Aziz, I.; Abdullah, I. Dehydrogenation of hydrazine hydrate using NiCo bimetallic catalyst supported on natural zeolite (ZA), Z-NaY, Z-HY, Al2O3 and TiO2. Rasayan J. Chem. 2021, 14, 1821–1828. [Google Scholar] [CrossRef]
- Shin, H.; Park, S.Y.; Megersa, D.D.; Bae, J.K.; Cho, I.S.; Yu, H.K. Fabrication of self-rolled Ni catalyst using water soluble ceramics for NaBH4 dehydrogenation. Int. J. Hydrogen Energy 2024, 51, 1028–1037. [Google Scholar] [CrossRef]
- Sheng, L.; Qi, G.; Jin, K.; Chen, A.; Huang, X.; Liu, G.; Zhou, M.; Wang, H.; Li, Y.; Wang, K.; et al. Pressure strategy to improve H atomic utilization via optimized decomposition pathway in solid hydrazine borane. J. Phys. Chem. Lett. 2024, 15, 9939–9944. [Google Scholar] [CrossRef]
- Ekinci, A.; Genli, N.; Sahin, O.; Baytar, O. Facile “Green” synthesis of a novel Co-W-B catalyst from Rheum ribes shell extract and its effect on sodium borohydride hydrolysis: Kinetic mechanism. Int. J. Hydrogen Energy 2024, 51, 796–808. [Google Scholar] [CrossRef]
- Shiferraw, B.T.; Baye, A.F.; Kim, H. Mechanistic insights into the deprotonation of methanol by facile synthesized 3D flower-like BiOX (X = Cl, Br, I) catalysts for rapid hydrogen generation from NaBH4 methanolysis: The ‘X’ factor. Int. J. Hydrogen Energy 2024, 51, 435–450. [Google Scholar] [CrossRef]
- Megersa, D.D.; Kim, Y.; Kim, N.; Lee, J.; Bae, J.S.; Choi, J.Y.; Yu, H.K. Ru/graphene hybrid film catalyst for NaBH4 hydrolysis reaction. Int. J. Hydrogen Energy 2022, 47, 15687–15694. [Google Scholar] [CrossRef]
- Ugale, A.D.; Ghodke, N.P.; Kang, G.S.; Nam, K.B.; Bhoraskar, S.V.; Mathe, V.L.; Yoo, J.B. Cost-effective synthesis of carbon loaded Co3O4 for controlled hydrogen generation via NaBH4 hydrolysis. Int. J. Hydrogen Energy 2022, 47, 16–29. [Google Scholar] [CrossRef]
- Li, X.; Yang, N.; Cen, X.; Li, S.; Zhang, L.; Lu, Z.H. Exceptional activity of hollow porphyrin frameworks-confined Ni nanoparticles for hydrogen production from NaBH4 methanolysis. Fuel 2023, 354, 129332. [Google Scholar] [CrossRef]
- Wang, H.; Xu, F.; Sun, L.; Wu, J.; Zhang, G.; Zhu, Y.; Shao, Q.; Luo, Y.; Peng, X.; Wang, Y.; et al. Novel MOF/COF dual carrier anchoring Ru nanoparticles for improved hydrogen production by hydrolysis of NaBH4. J. Alloys Compd. 2024, 978, 173415. [Google Scholar] [CrossRef]
- Jia, J.; Deng, K.; Li, J.; Zhang, Z.; Duan, X.; Huang, H. In situ transformation of 3D Co3O4 nanoparticles to 2D nanosheets with rich surface oxygen vacancies to boost hydrogen generation from NaBH4. Chem. Eng. J. 2021, 424, 130350. [Google Scholar] [CrossRef]
- Nunes, H.X.; Silva, D.L.; Rangel, C.M.; Pinto, A.M.F.R. Rehydrogenation of sodium borates to close the NaBH4-H2 cycle: A review. Energies 2021, 14, 3567. [Google Scholar] [CrossRef]
- Minkina, V.G.; Shabunya, S.I.; Kalinin, V.I.; Martynenko, V.V. Hydrogen generation from hydrolysis of concentrated NaBH4 solutions under adiabatic conditions. Int. J. Hydrogen Energy 2022, 47, 21772–21781. [Google Scholar] [CrossRef]
- Tai, T.; Cao, H.; Feng, W.; Yin, Z.; Zhang, H.; Zheng, G. High-efficient synthesis of NaBH4 by solid-phase electrolysis process on a core-shell-type cathode. Int. J. Hydrogen Energy 2024, 51, 172–183. [Google Scholar] [CrossRef]
- Li, H.; Hu, X.; Wang, L.; Shi, L.; Isimjan, T.T.; Yang, X. Kinetically promoted hydrogen generation by Ru nanoparticles decorated CoB2O4 on mesoporous carbon spheres with rich oxygen vacancies for NaBH4 hydrolysis. Chem. Eng. J. 2024, 481, 148547. [Google Scholar] [CrossRef]
- Gokkus, K.; Ozbal, A.; Gur, M.; Akalin, S.A.; Butun, V. Hyperbranched novel functional polymeric catalysts offering new approaches in hydrogen production via NaBH4 methanolysis. Int. J. Hydrogen Energy 2025, 98, 106–126. [Google Scholar] [CrossRef]
- Bu, Y.; Liu, J.; Cai, D.; Huang, P.; Wei, S.; Luo, X.; Liu, Z.; Xu, F.; Sun, L.; Wei, X. Magnetic recyclable catalysts with dual protection of hollow Co/N/C framework and surface carbon film for hydrogen production from NaBH4 hydrolysis. J. Alloys Compd. 2023, 938, 168495. [Google Scholar] [CrossRef]
- Abutaleb, A. Synthesis of copper/sulfur Co-doped TiO2-carbon nanofibers as catalysts for H2 production via NaBH4 hydrolysis. Inorganics 2023, 11, 352. [Google Scholar] [CrossRef]
- Doherty, S.; Knight, J.G.; Alharbi, H.Y.; Paterson, R.; Wills, C.; Dixon, C.; Šiller, L.; Chamberlain, T.W.; Griffiths, A.; Collins, S.M.; et al. Efficient hydrolytic hydrogen evolution from sodium borohydride catalyzed by polymer immobilized ionic liquid-stabilized platinum nanoparticles. ChemCatChem 2022, 14, e202101752. [Google Scholar] [CrossRef]
- Su, C.-C.; Lu, M.-C.; Wang, S.-L.; Huang, Y.-H. Ruthenium immobilized on Al2O3 pellets as a catalyst for hydrogen generation from hydrolysis and methanolysis of sodium borohydride. RSC Adv. 2012, 2, 2073–2079. [Google Scholar] [CrossRef]
- Ecer, U.; Zengin, A.; Sagan, T. Synthesis and catalytic performance of Pd NPs-doped polymer brushes for optimization and modeling of NaBH4 hydrolysis. J. Energy Inst. 2025, 119, 101974. [Google Scholar] [CrossRef]
- Yang, F.; Xiang, C.; Fang, S.; Xu, F.; Sun, L.; Shen, C.Y.; Zou, Y. Synthesis and catalytic performance of nanoflower-like Ru@CoAl-LDH composite catalyst for NaBH4 hydrolysis. J. Alloys Compd. 2023, 945, 169280. [Google Scholar] [CrossRef]
- Biehler, E.; Quach, Q.; Abdel-Fattah, T.M. Gold nanoparticles AuNP decorated on fused graphene-like materials for application in a hydrogen generation. Materials 2023, 16, 4779. [Google Scholar] [CrossRef] [PubMed]
- Tiri, R.N.E.; Gulbagca, F.; Aygun, A.; Cherif, A.; Sen, F. Biosynthesis of Ag–Pt bimetallic nanoparticles using propolis extract: Antibacterial effects and catalytic activity on NaBH4 hydrolysis. Environ. Res. 2022, 206, 112622. [Google Scholar] [CrossRef] [PubMed]
- Lima da Silva, B.S.; Amorim, M.K.M.; Souza, E.S.; Alaide de Oliveira, M.; Madeiro da Costa, O.M.M.; Barros, B.S.; Kulesza, J. Sodium borohydride hydrolysis for hydrogen generation over Mn-BDC and MnCo-BDC (BDC–1,4-benzene-dicarboxylate) coordination polymers. Polyhedron 2025, 273, 117493. [Google Scholar] [CrossRef]
- Yurderi, M. PtCo/g-C3N4 bimetallic catalyst: Boosting hydrogen production in sodium borohydride hydrolysis. Int. J. Hydrogen Energy 2025, 143, 105–111. [Google Scholar] [CrossRef]
- Yuan, C.; Xu, T.; Guo, M.; Sun, Y.; Zhang, T.; Yu, X. Unveiling the bifunctional modulation evoked by bromine doping of CoP towards efficient hydrolytic hydrogen generation. Appl. Catal. B Environ. 2024, 343, 123562. [Google Scholar] [CrossRef]
- Saleem, F.; Abid, M.Z.; Rafiq, K.; Rauf, A.; Ahmad, K.; Iqbal, S.; Jin, R.; Hussain, E. Synergistic effect of Cu/Ni cocatalysts on CdS for sun-light driven hydrogen generation from water splitting. Int. J. Hydrogen Energy 2024, 52, 305–319. [Google Scholar] [CrossRef]
- Hansu, T.A.; Sahin, O.; Caglar, A.; Kivrak, H. A remarkable Mo doped Ru catalyst for hydrogen generation from sodium borohydride: The effect of Mo addition and estimation of kinetic parameters. React. Kinet. Mech. Catal. 2020, 131, 661–676. [Google Scholar] [CrossRef]
- Jiang, J.; Qiu, Y.; Dong, H.; Yang, L.; Miao, Y.; Xiong, L.; Gao, B.; Zhang, X.; Chu, P.K.; Peng, X. Enhancing hydrogen evolution by heterointerface engineering of Ni/MoN catalysts. J. Colloid Interface Sci. 2025, 686, 681–690. [Google Scholar] [CrossRef]
- Amber, H.; Vjūnova, K.; Sukackienė, Z.; Šimkūnaitė, D.; Vaičiūnienė, J.; Tamašauskaitė-Tamašiūnaitė, L.; Norkus, E. Hydrogen production on CoFe, CoFeMn and CoFeMo coatings deposited on Ni foam via electroless metal plating. Chemija 2023, 34, 155–162. [Google Scholar] [CrossRef]
- Jasrotia, R.; Verma, A.; Ahmed, J.; Khanna, V.; Fazil, M.; Alshehri, S.M.; Kumari, S.; Kumar, P.; Ahmad, T.; Kandwal, A. Mg1-xNixGayFe2-yO4 nano catalysts for green hydrogen generation with highly efficient photo/electro catalytic water splitting applications. Int. J. Hydrogen Energy 2024, 52, 1228–1240. [Google Scholar] [CrossRef]
- Erat, N.; Bozkurt, G.; Ozer, A. Co/CuO-NiO-Al2O3 catalyst for hydrogen generation from hydrolysis of NaBH4. Int. J. Hydrogen Energy 2022, 47, 24255–24267. [Google Scholar] [CrossRef]
- Santos, F.S.; Giroto, A.S.; Torres, J.A.; Oliveira, A.V.P.S.; Santos, V.M.F.; Nogueira, A.E. Hydrogen generation via NaBH4 oxide catalysts. Int. J. Hydrogen Energy 2024, 92, 113–123. [Google Scholar] [CrossRef]
- Shen, J.; Xu, D.; Ji, J.; Zhang, Q.; Fan, X. In situ evolved defective TiO2 as robust support for CoB-catalyzed hydrolysis of NaBH4. Int. J. Hydrogen Energy 2023, 48, 1001–1010. [Google Scholar] [CrossRef]
- Liu, Y.; Zheng, X.; Xu, B.; Xia, Z.; Wang, Y. A novel hydrogen storage material: The hydrolysis products of sodium borohydride. J. Phys. Chem. Solids 2024, 188, 111923. [Google Scholar] [CrossRef]
- Han, Y.; Ma, J.; Wang, X.; Wang, Y.; Zhand, K.; Cao, Z.; Wu, S.; Li, G. Preparation of Co–P–B/foam Ni catalyst and its catalytic performance for hydrogen production by hydrolysis of NaBH4 solution. Fuel 2024, 372, 132262. [Google Scholar] [CrossRef]
- Ekinci, A.; Sahin, O.; Turgut, S.; Baytar, O. Eco-friendly nanocatalysts for hydrogen generation via sodium borohydride hydrolysis and enhanced fuel cell applications. Int. J. Hydrogen Energy 2025, 143, 791–801. [Google Scholar] [CrossRef]
- Li, S.; Qui, S.; Chua, Y.S.; Xia, Y.; Zou, Y.; Xu, F.; Sun, L.; Chu, H. Co–B supported on waxberry-like hierarchical porous carbon microspheres: An efficient catalyst for hydrogen generation via sodium borohydride hydrolysis. Mater. Chem. Phys. 2024, 319, 129399. [Google Scholar] [CrossRef]
- Onat, E.; Ekinci, S.; Sahin, O.; Izgi, M.S. Effective and environmentally friendly Co nanocatalyst on sodium borohydride hydrolysis in different solvents. Int. J. Hydrogen Energy 2025, 142, 864–874. [Google Scholar] [CrossRef]
- Wang, Y.; Zheng, X.; Wang, Y.; Li, X.; Liu, Y.; Xia, Z. Effect of loaded Co–B on hydrogen release performance of sodium borohydride. J. Phys. Chem. Solids 2025, 204, 112761. [Google Scholar] [CrossRef]
- Jeong, S.U.; Kim, R.K.; Cho, E.A.; Kim, H.J.; Nam, S.W.; Oh, I.H.; Hong, S.A.; Kim, S.H. A study on hydrogen generation from NaBH4 solution using the high-performance Co-B catalyst. J. Power Sources 2005, 144, 129–134. [Google Scholar] [CrossRef]
- Earl, C.; Shah, I.H.; Cook, S.; Cheeseman, C.R. Environmental sustainability and supply resilience of cobalt. Sustainability 2022, 14, 4124. [Google Scholar] [CrossRef]
- Domingo, J.L. Cobalt in the environment and its toxicological implications. In Reviews of Environmental Contamination and Toxicology; Springer: New York, NY, USA, 1989; Volume 108, pp. 105–132. [Google Scholar] [CrossRef]
- Izgi, M.S.; Sahin, O.; Faal, S.; Celik, F.A.; Onat, E.; Karabulut, E. Green synthesis of highly efficient and stable Ni@CQD nanoparticles: Experimental and theoretical approach for hydrogen production from dimethyl aminborane and sodium borohydride hydrolysis. Fuel 2025, 397, 135412. [Google Scholar] [CrossRef]
- Ekinci, A.; Cengiz, E.; Kuncan, M.; Sahin, O. Hydrolysis of sodium borohydride solutions both in the presence of Ni–B catalyst and in the case of microwave application. Int. J. Hydrogen Energy 2020, 45, 34749–34760. [Google Scholar] [CrossRef]
- Liang, Z.; Li, Q.; Li, F.; Zhao, S.; Xi, X. Hydrogen generation from hydrolysis of NaBH4 based on high stable NiB/NiFe2O4 catalyst. Int. J. Hydrogen Energy 2017, 42, 3971–3980. [Google Scholar] [CrossRef]
- Lee, J.K.; Ann, H.; Yi, Y.; Lee, K.W.; Uhm, S.; Lee, J. A stable Ni–B catalyst in hydrogen generation via NaBH4 hydrolysis. Catal. Commun. 2011, 16, 120–123. [Google Scholar] [CrossRef]
- Lakhali, H.; Kocaman, S.; Ahmetli, G.; Ceyhan, A.A. Enhanced hydrogen generation in borohydride hydrolysis using an efficient and reusable IA-CNT supported Co-Mo-B catalyst. Diam. Relat. Mater. 2025, 155, 112364. [Google Scholar] [CrossRef]
- Lakhali, H.; Ceyhan, A.A.; Sahin, O. Novel Fe3O4@SiO2/Co-Mo-B core-shell magnetic nanocatalyst: A reusable system for high-performance hydrogen evolution in borohydride hydrolysis. Inorg. Chem. Commun. 2025, 177, 114406. [Google Scholar] [CrossRef]
- Akkus, M.S. Investigating the efficiency of NiFe catalysts in potassium borohydride hydrolysis for hydrogen production. Fuel 2026, 403, 136099. [Google Scholar] [CrossRef]
- Sajad Ghasemi Vajargah, S.G.; Gilani, N. Enhancing the activity of Ni-B catalyst via Cu doping towards hydrogen evolution from NaBH4 hydrolysis. Renew. Energy 2024, 235, 121322. [Google Scholar] [CrossRef]
- Mu, J.; Ye, Y.; Wang, Y.; Zhu, Z.; Wang, Y.; Zhang, H. Dislocation-driven high catalytic performance of FeCoNiCrMn high-entropy alloy for the hydrolysis of NaBH4. Appl. Phys. Lett. 2021, 119, 121901. [Google Scholar] [CrossRef]
- Liu, C.H.; Chen, B.H.; Hsueh, C.L.; Ku, J.R.; Tsau, F.; Hwang, K.J. Preparation of magnetic cobalt-based catalyst for hydrogen generation from alkaline NaBH4 solution. Appl. Catal. B Environ. 2009, 91, 368–379. [Google Scholar] [CrossRef]
- Yue, C.; Yang, P.; Wang, J.; Zhao, X.; Wang, Y.; Yang, L. Facile synthesis and characterization of nano-Pd loaded NiCo microfibers as stable catalysts for hydrogen generation from sodium borohydride. Chem. Phys. Lett. 2020, 743, 137170. [Google Scholar] [CrossRef]
- Wang, L.; Li, Z.; Liu, X.; Zhang, P.; Xie, G. Hydrogen generation from alkaline NaBH4 solution using electroless-deposited Co–W–P supported on γ-Al2O3. Int. J. Hydrogen Energy 2015, 40, 7965–7973. [Google Scholar] [CrossRef]
- Li, Z.; Li, H.; Wang, L.; Liu, T.; Zhang, T.; Wang, G.; Xie, G. Hydrogen generation from catalytic hydrolysis of sodium borohydride solution using supported amorphous alloy catalysts (Ni–Co–P/γ-Al2O3). Int. J. Hydrogen Energy 2014, 39, 14935–14941. [Google Scholar] [CrossRef]
- Rakap, M.; Kalu, E.E.; Ozkar, S. Cobalt–nickel–phosphorus supported on Pd-activated TiO2 (Co–Ni–P/Pd-TiO2) as cost-effective and reusable catalyst for hydrogen generation from hydrolysis of alkaline sodium borohydride solution. J. Alloys Compd. 2011, 509, 7016–7021. [Google Scholar] [CrossRef]
- Guo, J.; Hou, Y.; Li, B. Facile synthesis of a hollow Ni–Fe–B nanochain and its enhanced catalytic activity for hydrogen generation from NaBH4 hydrolysis. RSC Adv. 2018, 8, 25873–25880. [Google Scholar] [CrossRef]
- Nie, M.; Zou, Y.C.; Huang, Y.M.; Wang, J.Q. Ni–Fe–B catalysts for NaBH4 hydrolysis. Int. J. Hydrogen Energy 2012, 37, 1568–1576. [Google Scholar] [CrossRef]
- Wang, Y.; Li, G.; Wu, S.; Wei, Y.; Meng, W.; Xie, Y.; Cui, Y.; Lian, X.; Chen, Y.; Zhang, X. Hydrogen generation from alkaline NaBH4 solution using nanostructured Co–Ni–P catalysts. Int. J. Hydrogen Energy 2017, 42, 16529–16537. [Google Scholar] [CrossRef]
- Yajima, S.; Matsushita, S.; Togawa, Y.; Kanbe, T.; Saito, H. Electroless nickel plating from a glycine bath. J. Met. Finish. Soc. Jpn. 1986, 37, 255–260. [Google Scholar] [CrossRef]
- Norkus, E.; Stankevičienė, I.; Jagminienė, A.; Balčiūnaitė, A.; Stalnionis, G.; Pakštas, V.; Tamašauskaitė-Tamašiūnaitė, L. Application of diethylenetriamine for electroless deposition of cobalt alloys with controllable amount of boron using morpholine borane as reducing agent. Coatings 2025, 15, 1081. [Google Scholar] [CrossRef]
- Saito, M.; Fujiwara, H.; Mizuno, J.; Homma, T. Preparation of nano-structured CoCu films by electrodeposition. ECS Meet. Abstr. 2006, MA2005-01, 1278. [Google Scholar] [CrossRef]
- Li, D.Y.; Li, N.; Xiao, N.; Zhu, Z.Y.; Wang, F.F. Kinetic study of electroless copper with glycine as complexing agent. Asian J. Chem. 2013, 25, 3679–3681. [Google Scholar] [CrossRef]
- Mardiansyah, D.; Badloe, T.; Triyana, K.; Mehmood, M.Q.; Raeis-Hosseini, N.; Lee, Y.; Sabarman, H.; Kim, K.; Rho, J. Effect of temperature on the oxidation of Cu nanowires and development of an easy to produce, oxidation-resistant transparent conducting electrode using a PEDOT: PSS coating. Sci. Rep. 2018, 8, 10639. [Google Scholar] [CrossRef]
- Cui, L.; Wang, Y.; Han, X.; Xu, P.; Wang, F.; Liu, D.; Zhao, H.; Du, Y. Phenolic resin reinforcement: A new strategy for hollow NiCo@C microboxes against electromagnetic pollution. Carbon 2021, 174, 673–682. [Google Scholar] [CrossRef]
- Li, B.; Li, C.; Gao, Y.; Guo, H.; Kang, Y.; Zhao, S. Microstructure and tribocorrosion properties of Ni-based composite coatings in artificial seawater. Coatings 2019, 9, 822. [Google Scholar] [CrossRef]
- Tesfaye, R.M.; Das, G.; Park, B.J.; Kim, J.; Yoon, H.H. Ni-Co bimetal decorated carbon nanotube aerogel as an efficient anode catalyst in urea fuel cells. Sci. Rep. 2019, 9, 479. [Google Scholar] [CrossRef]
- Kytsya, A.; Berezovets, V.; Verbovytskyy, Y.; Bazylyak, L.; Kordan, V.; Zavaliy, I.; Yartys, V.A. Bimetallic Ni-Co nanoparticles as an efficient catalyst of hydrogen generation via hydrolysis of NaBH4. J. Alloys Compd. 2022, 908, 164484. [Google Scholar] [CrossRef]
- X-Ray Photoelectron Spectroscopy (XPS) Reference Pages. Available online: http://www.xpsfitting.com/search/label/Nickel (accessed on 24 October 2025).
- Biesinger, M.C.; Payne, B.P.; Lau, L.W.M.; Gerson, A.R.; Smart, R.S.C. X-ray photoelectron spectroscopic chemical state quantification of mixed nickel metal, oxide and hydroxide systems. Surf. Interface Anal. 2009, 41, 324. [Google Scholar] [CrossRef]
- Biesinger, M.C.; Payne, B.P.; Grosvenor, A.P.; Lau, L.W.M.; Gerson, A.R.; Smart, R.S.C. Resolving surface chemical states in XPS analysis of first row transition metals, oxides and hydroxides: Cr, Mn, Fe, Co and Ni. Appl. Surf. Sci. 2011, 257, 2717. [Google Scholar] [CrossRef]
- X-ray Photoelectron Spectroscopy (XPS) Reference Pages. Available online: http://www.xpsfitting.com/search/label/Oxygen (accessed on 24 October 2025).
- HarwellXPS guru. Available online: https://www.harwellxps.guru/xpskb/nickel/#1755794317174-e9df6303-1cf1 (accessed on 23 October 2025).
- X-ray Photoelectron Spectroscopy (XPS) Reference Pages. Available online: http://www.xpsfitting.com/search/label/Cobalt (accessed on 24 October 2025).
- Yang, J.; Liu, H.; Martens, W.N.; Frost, R.L. Synthesis and characterization of cobalt hydroxide, cobalt oxyhydroxide, and cobalt oxide nanodiscs. J. Phys. Chem. C 2010, 114, 111. [Google Scholar] [CrossRef]
- Hsieh, P.-T.; Chen, Y.-C.; Kao, K.S.; Wang, C.M. Luminescence mechanism of ZnO thin film investigated by XPS measurement. Appl. Phys. A 2008, 90, 317. [Google Scholar] [CrossRef]
- Cakanyildirim, C. An effective trimetalic crystalline catalyst for sodium borohydride hydrolysis. Energy Sources A Recovery Util. Environ. Eff. 2025, 47, 2684–2695. [Google Scholar] [CrossRef]
- Demirci, U.B.; Miele, P. Cobalt in NaBH4 hydrolysis. Phys. Chem. Chem. Phys. 2010, 12, 14651–14665. [Google Scholar] [CrossRef] [PubMed]
- Zouli, N.; Maafa, I.M.; Abutaleb, A.; Yousef, A.; El-Halwany, M.M. Membrane nanofiber-supported cobalt–nickel nanoparticles as an effective and durable catalyst for H2 evolution via sodium borohydride hydrolysis. Polymers 2023, 15, 814. [Google Scholar] [CrossRef] [PubMed]
- Akduman, H.O.; Ozdemir, E. Zirconia supported bimetallic Co–Mn–B catalyst with superior catalytic activity for hydrolysis of sodium borohydride. Int. J. Hydrogen Energy 2025, 100, 67–78. [Google Scholar] [CrossRef]
- Ingersoll, J.C.; Mani, N.; Thenmozhiyal, J.C.; Muthaiah, A. Catalytic hydrolysis of sodium borohydride by a novel nickel–cobalt–boride catalyst. J. Power Sources 2007, 173, 450–457. [Google Scholar] [CrossRef]
- Yun, S.M.; Lee, T.H.; Kim, M.J.; Oh, T.H. Adurable cobalt catalyst supported on Ni foam coated with Al2O3 for hydrogen generation from NaBH4 hydrolysis. Mater. Chem. Phys. 2024, 328, 130015. [Google Scholar] [CrossRef]
- Sukackienė, Z.; Valeckytė, G.; Kepenienė, V.; Vaičiūnienė, J.; Tamašauskaitė-Tamašiūnaitė, L.; Stalnionienė, I.; Jasulaitienė, V.; Stalnionis, G.; Norkus, E. Non-Precious metals catalysts for hydrogen generation. Coatings 2023, 13, 1740. [Google Scholar] [CrossRef]
- Didehban, A.; Zabihi, M.; Shahrouzi, J.R. Experimental studies on the catalytic behavior of alloy and core-shell supported Co-Ni bimetallic nano-catalysts for hydrogen generation by hydrolysis of sodium borohydride. Int. J. Hydrogen Energy 2018, 43, 20645–20660. [Google Scholar] [CrossRef]
- Sukackienė, Z.; Valeckytė, G.; Kepenienė, V.; Vaičiūnienė, J.; Tamašauskaitė-Tamašiūnaitė, L.; Stalnionienė, I.; Jasulaitienė, V.; Pakštas, V.; Norkus, E. The dependence of NiMo/Cu catalyst composition on its catalytic activity in sodium borohydride hydrolysis reactions. Materials 2024, 17, 4353. [Google Scholar] [CrossRef] [PubMed]
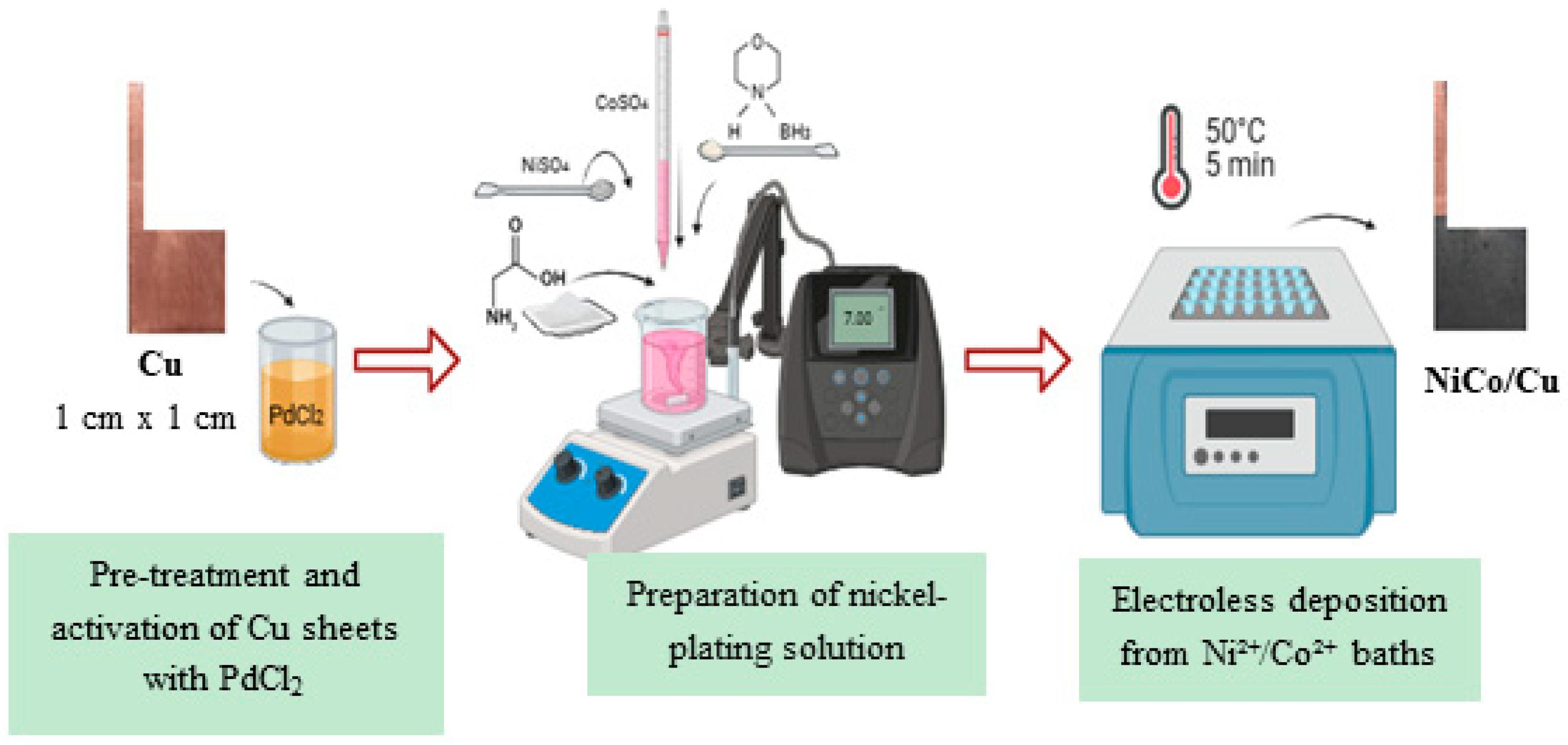
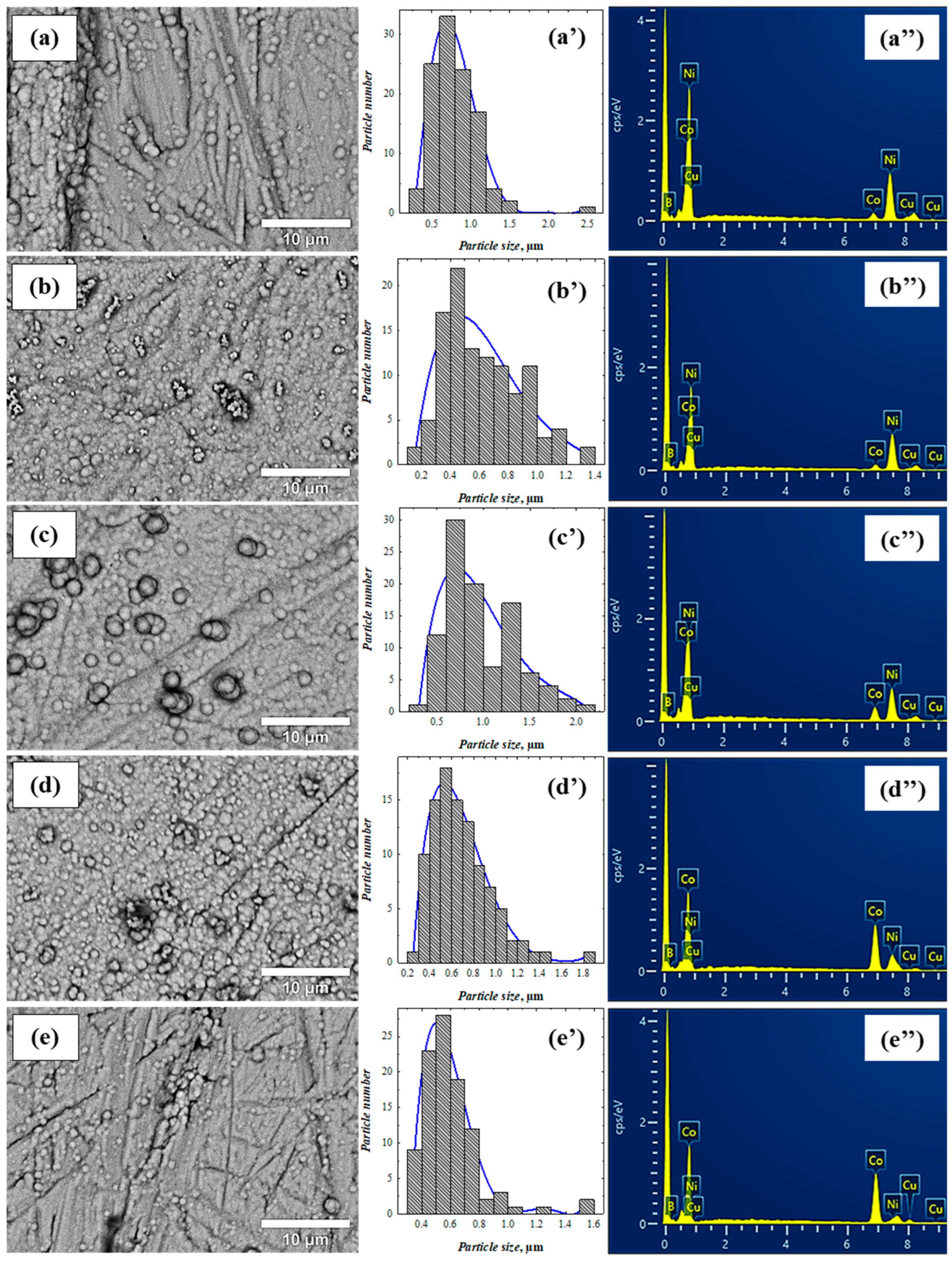




| Catalyst | Composition of Solution (mol L−1) | Deposition Conditions | |||||
|---|---|---|---|---|---|---|---|
| NiSO4 | Glycine | MB | CoSO4 | pH | T, °C | t, min | |
| Ni96Co4/Cu | 0.1 | 0.2 | 0.2 | 0.0025 | 7 | 50 | 5 |
| Ni90Co10/Cu | 0.005 | ||||||
| Ni80Co20/Cu | 0.01 | ||||||
| Ni20Co80/Cu | 0.1 | ||||||
| Ni10Co90/Cu | 0.05 | 0.2 | |||||
| Catalyst | Element, wt.% | Element Loadings, µg cm−2 | Deposition Rate, µm h−1 | |||
|---|---|---|---|---|---|---|
| Ni | Co | Ni | Co | Ni + Co | ||
| Ni96Co4/Cu | 96.45 | 3.55 | 1047.0 | 38.5 | 1085.5 | 14.6 |
| Ni90Co10/Cu | 89.95 | 10.05 | 789.0 | 88.2 | 877.2 | 11.8 |
| Ni80Co20/Cu | 79.58 | 20.42 | 732.9 | 188.1 | 921.0 | 12.4 |
| Ni20Co80/Cu | 22.61 | 77.39 | 179.4 | 614.0 | 793.4 | 10.7 |
| Ni10Co90/Cu | 9.56 | 90.44 | 52.5 | 496.3 | 548.8 | 7.4 |
| Co (wt.%) | 2θ (111) | a, Å |
|---|---|---|
| 0 | 44.50 | 3.5235 |
| 10 | 44.65 | 3.5123 |
| 20 | 44.80 | 3.5012 |
| 80 | 45.05 | 3.4828 |
| 90 | 45.15 | 3.4754 |
| Catalyst | Ea, kJ mol−1 | T, K | v, mL min−1 | v, L min−1 gcat−1 |
|---|---|---|---|---|
| Ni96Co4/Cu | 63.81 | 303 | 0.31 | 0.14 |
| 313 | 0.46 | 0.21 | ||
| 323 | 1.10 | 0.51 | ||
| 333 | 2.94 | 1.35 | ||
| 343 | 5.09 | 2.34 | ||
| Ni90Co10/Cu | 59.55 | 303 | 0.42 | 0.24 |
| 313 | 0.66 | 0.38 | ||
| 323 | 1.38 | 0.79 | ||
| 333 | 3.56 | 2.03 | ||
| 343 | 5.73 | 3.27 | ||
| Ni80Co20/Cu | 57.82 | 303 | 0.56 | 0.30 |
| 313 | 1.09 | 0.59 | ||
| 323 | 1.76 | 0.96 | ||
| 333 | 3.77 | 2.05 | ||
| 343 | 8.30 | 4.51 | ||
| Ni20Co80/Cu | 56.4 | 303 | 1.24 | 0.78 |
| 313 | 2.13 | 1.34 | ||
| 323 | 5.77 | 3.64 | ||
| 333 | 10.66 | 6.72 | ||
| 343 | 14.59 | 9.19 | ||
| Ni10Co90/Cu | 52.5 | 303 | 1.46 | 1.33 |
| 313 | 2.84 | 2.59 | ||
| 323 | 6.93 | 6.31 | ||
| 333 | 10.24 | 9.33 | ||
| 343 | 15.61 | 14.22 |
| Catalyst | Ea, kJ mol−1 | T, K | v, mL min−1 | v, L min−1 gcat−1 |
|---|---|---|---|---|
| Ni/Cu | 67.9 | 303 K | 0.18 | 0.24 |
| 313 K | 0.39 | 0.52 | ||
| 323 K | 0.77 | 1.03 | ||
| 333 K | 1.64 | 2.19 | ||
| 343 K | 3.99 | 5.34 | ||
| Co/Cu | 55.5 | 303 K | 0.94 | 0.75 |
| 313 K | 1.89 | 1.51 | ||
| 323 K | 3.71 | 2.97 | ||
| 333 K | 7.13 | 5.71 | ||
| 343 K | 11.92 | 9.55 |
| Catalyst | Electrolyte | Ea, kJ mol−1 | Reference |
|---|---|---|---|
| Co–Ni–P/Pd-TiO2 | 0.30 M NaBH4 + 10 wt.% NaOH | 57.0 | [82] |
| Ni-B | 10 wt.% NaBH4 + 5 wt.% NaOH | 61.84 | [72] |
| NiCoB | 2.7 wt.% NaBH4 + 15 wt.% NaOH | 62.0 | [107] |
| Ni9Co1 | 5 wt.% NaBH4 + 2 M NaOH | 64.2 | [79] |
| Co/Al2O3–Ni foam | 20 wt.% NaBH4 + 5 wt.% NaOH | 64.3 | [108] |
| Co-B | 20 wt.% NaBH4 + 5 wt.% NaOH | 64.87 | [66] |
| Co | 5 wt.% NaBH4 + 5 wt.% NaOH | 66.67 | [78] |
| Ni/Cu | 5 wt.% NaBH4 + 0.4 wt.% NaOH | 67.9 | [109] |
| Nickel–Cobalt (Alloy) | 5 wt.% NaBH4 + 1 wt.% NaOH | 68.84 | [110] |
| NiB/NiFe2O4 | 5 wt.% NaBH4 + 10 wt.% NaOH | 72.52 | [71] |
| Ni96Co4/Cu | 5 wt.% NaBH4 + 0.4 wt.% NaOH | 63.81 | this study |
| Ni90Co10/Cu | 5 wt.% NaBH4 + 0.4 wt.% NaOH | 59.55 | this study |
| Ni80Co20/Cu | 5 wt.% NaBH4 + 0.4 wt.% NaOH | 57.82 | this study |
| Ni20Co80/Cu | 5 wt.% NaBH4 + 0.4 wt.% NaOH | 56.4 | this study |
| Ni10Co90/Cu | 5 wt.% NaBH4 + 0.4 wt.% NaOH | 52.5 | this study |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Valeckytė, G.; Sukackienė, Z.; Kepenienė, V.; Šakickaitė, R.; Vaičiūnienė, J.; Tamašauskaitė-Tamašiūnaitė, L.; Stupakova, J.; Norkus, E. Reusable NiCo/Cu Catalysts for Sustainable Hydrogen Generation. Materials 2026, 19, 852. https://doi.org/10.3390/ma19050852
Valeckytė G, Sukackienė Z, Kepenienė V, Šakickaitė R, Vaičiūnienė J, Tamašauskaitė-Tamašiūnaitė L, Stupakova J, Norkus E. Reusable NiCo/Cu Catalysts for Sustainable Hydrogen Generation. Materials. 2026; 19(5):852. https://doi.org/10.3390/ma19050852
Chicago/Turabian StyleValeckytė, Gitana, Zita Sukackienė, Virginija Kepenienė, Raminta Šakickaitė, Jūratė Vaičiūnienė, Loreta Tamašauskaitė-Tamašiūnaitė, Jolanta Stupakova, and Eugenijus Norkus. 2026. "Reusable NiCo/Cu Catalysts for Sustainable Hydrogen Generation" Materials 19, no. 5: 852. https://doi.org/10.3390/ma19050852
APA StyleValeckytė, G., Sukackienė, Z., Kepenienė, V., Šakickaitė, R., Vaičiūnienė, J., Tamašauskaitė-Tamašiūnaitė, L., Stupakova, J., & Norkus, E. (2026). Reusable NiCo/Cu Catalysts for Sustainable Hydrogen Generation. Materials, 19(5), 852. https://doi.org/10.3390/ma19050852








