Study on the Evolution Mechanism of Carbon Impurities in Polysilicon Production Based on HSC Simulation
Abstract
1. Introduction
2. Materials and Methods
2.1. Thermodynamic Calculation Modeling Methods
2.2. Materials and Experiments
3. Results and Discussion
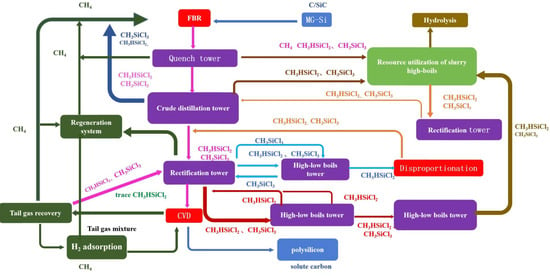
3.1. Technological Route for Polysilicon Preparation via the Modified Siemens Process
3.2. Existence Forms of Carbon Impurities in Metallurgical Silicon and Polysilicon
3.3. Evolution of Carbon Impurities and HSC Simulation of Reaction Thermodynamics
3.4. Sampling and Analysis Results of Carbon-Containing Materials at Different Sites
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yang, D.R. Materials for Solar Cells; Chemical Industry Press: Beijing, China, 2018. [Google Scholar]
- Han, Z.S. Semiconductor Manufacturing Technology; Publishing House of Electronics Industry: Beijing, China, 2015. [Google Scholar]
- O’Mara, W.C.; Herring, R.B.; Hunt, L.P. Handbook of Semiconductor Silicon Technology; Noyes Publications: New York, NY, USA, 1990. [Google Scholar]
- Zhou, J.Z.; Zhan, B.; Lv, B.; Guo, Y.; Jiang, B. Contribution Rates and Simulation Model Refinement for Polysilicon Films Deposited by Large-Sized Tubular Low-Pressure Chemical Vapor Deposition Reactors. Materials 2024, 17, 5952. [Google Scholar] [CrossRef] [PubMed]
- Que, D.L. Silicon Materials Science and Technology; Zhejiang University Press: Hangzhou, China, 2000. [Google Scholar]
- Yuan, X.P.; Guo, L.J.; Lv, Q.H.; Zhao, D.; Liao, H.; Ma, W.H.; Hou, Y.Q.; Shen, J.L. Product quality research in the industrialized preparation of electronic grade polysilicon: A review of impurity sources and control strategies. Renew. Sustain. Energy Rev. 2025, 214, 115569. [Google Scholar] [CrossRef]
- Cao, L.L.; Cai, Y.G.; Zong, B.; Wei, D.L. Influencing factors and control measures of carbon content in polysilicon products. Mod. Chem. Ind. 2019, 39, 183–186. [Google Scholar]
- Chen, Y.M.; Weng, B.F.; Liu, Y.N. Analyses of Polychlorosilanes and Methylchlorosilanes Containing in Synthesized Trichlorosilane. Chem. Newsl. 2014, 77, 274–277. [Google Scholar]
- Yang, H. Study on gas chromatography-mass spectrometry method for determination of 4 kind of methylchlorosilane in high purity trichlorosilane. Anal. Instrum. 2019, 1, 88–92. [Google Scholar]
- Lewis, K.M.; Rethwisch, D.G. Catalyzed Direct Reactions of Silicon; Elsevier: New York, NY, USA, 1993. [Google Scholar]
- Coleman, L.; Buffasultant, E.C. Optimize Your Chlorosilane Distillation Columns. In Proceedings of the Silicon For the Chemical and Solar Industry XIII, Kristiansand, Norway, 13–16 June 2016; Norwegian University of Science and Technology: Trondheim, Norway, 2016. [Google Scholar]
- Vaisarová, V.; Bažant, V.; Chvalovský, V. Organosilicon compounds. LV. Dipole moments of substituted allylsilanes and benzylsilanes. Collect. Czechoslov. Chem. Commun. 1968, 33, 850–858. [Google Scholar] [CrossRef]
- Lai, G.Q.; Xin, S.M. Youjigui Chanpin Hecheng Gongyi Ji Yingyong; Chemical Industry Press: Beijing, China, 2009. [Google Scholar]
- Yang, D.; Wang, F.; Wan, Y. Research progress on separation process of methyl dichlorosilane from trichlorsilane. Inorg. Chem. Ind. 2021, 53, 30–34. [Google Scholar]
- Lepage, J.L.; Soula, G. Preparation of Silane from Methyldichlorosi-Lane and Chlorosilanes. U.S. Patent 4605543, 12 August 1986. [Google Scholar]
- Tanaka, S. Method for Producing Trichlorosilane. WO Patent 2011111335A, 15 September 2011. [Google Scholar]
- Atkins, P.W.; Paula, J.D. Physical Chemistry; Oxford University Press: New York, NY, USA; Oxford, UK, 2006. [Google Scholar]
- Zou, B.S.; Jiang, Y.F.; Liao, G.A.; Shen, Y.Q.; Wang, X.P.; Wang, N.; Wang, Y.; He, C.; Gao, F. Preparation of Al-Ti-Sc master alloys and refining effects on the 6016 aluminum alloy. J. Alloys Compd. 2024, 985, 174094. [Google Scholar] [CrossRef]
- GB_T 14849.6-2014; Methods for Chemical Analysis of Silicon Metal. Part 6: Determination of Carbon. Infrared Absorption Method. Standardization Administration of the People’s Republic of China: Beijing, China, 2014.
- YS/T 1300-2019; Determination of Methyldichlorosilane, Trimethylchlorosilane and Methyltrichlorosilane in Chlorosilanes-Gas Chromatography-Mass Spectrometry Method. Ministry of Industry and Information Technology of the People’s Republic of China: Beijing, China, 2019.
- GB/T 3634.2-2011; Hydrogen—Part 2: Pure Hydrogen, High Pure Hydrogen and Ultra-Pure Hydrogen. Standardization Administration of the People’s Republic of China: Beijing, China, 2011.
- Gu, Y.J.; Gong, S.K. Analysis and Testing Technology on Materials; Central South University Press: Changsha, China, 2011. [Google Scholar]
- GB/T 29057-2023; Practice for Evaluation of Polocrystalline Silicon Rods by Float-Zone Crystal Growth and Spectroscopy. Standardization Administration of the People’s Republic of China: Beijing, China, 2023.
- GB/T 35306-2023; Determination of Carbon and Oxygen Content in Silicon Single Crystal Low Temperature Fourier Transform Infrared Spectroscopy. Standardization Administration of the People’s Republic of China: Beijing, China, 2023.
- Dalaker, H.; Tangstad, M. Time and Temperature Dependence of the Solubility of Carbon in Liquid Silicon Equilibrated with Silicon Carbide and Its Dependence on Boron Levels. Mater. Trans. 2009, 50, 1152–1156. [Google Scholar] [CrossRef]
- Tan, Y.; Qin, S.Q.; Shi, S.; Jiang, D.C. Research Progress on Light Elements (C, N, O) in Solar-grade Silicon. J. Mater. Eng. 2017, 45, 112–118. [Google Scholar]
- Svanem, J.P.; Røe, T. Analytical Method to Measure Different Forms of Carbon in Silicon. In Proceedings of the Silicon For the Chemical and Solar Industry XI, Bergen-Ulvik, Norway, 25–29 June 2012; Norwegian University of Science and Technology: Trondheim, Norway, 2012. [Google Scholar]
- Ding, W.J. Mechanism and Kinetic Studies of a Two-Step Reactions Process for Metallurgical Slicon to Silane. Ph.D. Thesis, Shanghai Jiao Tong Unversity, Shanghai, China, March 2015. [Google Scholar]
- Xing, Q.Y.; Xu, R.Q.; Zhou, Z.; Pei, W.W. Basic Organic Chemistry; Higher Education Press: Beijing, China, 1993. [Google Scholar]
- Chernyavsky, L.I.; Titov, V.A.; Sysoev, S.V. Thermodynamic Simulation of Silicon Deposition from the Gas Phase of Si-Cl-H System. Inoorganic Mater. 2009, 45, 517–521. [Google Scholar] [CrossRef]
- Jong, F.d.; Meyyappan, M. Numerical siulation of silicon carbide chemicai vapor deposition. Diam. Relat. Mater. 1996, 5, 141–150. [Google Scholar] [CrossRef]





| Metallurgical-Grade Silicon | Sample | 1 | 2 | 3 | 4 | 5 | 6 |
| Concentration (%) | 0.015 | 0.023 | 0.012 | 0.018 | 0.015 | 0.021 | |
| Polysilicon | Sample | 1 | 2 | 3 | 4 | 5 | 6 |
| Concentration (ppb) | 26 | 36 | 29 | 34 | 30 | 22 |
| No. | Reaction Equation | ΔG (kcal/mol) | ||||||
| 25 °C | 280 °C | 320 °C | 540 °C | 560 °C | 1000 °C | 1100 °C | ||
| 1 | Si + 4C + 6H2(g) = Si(CH3)4(g) | −35.39 | 10.53 | 16.05 | 26.91 | 29.44 | 97.24 | 107.47 |
| 2 | Si + C + 3HCl(g) = Si(CH3)Cl3(g) | −43.54 | −29.10 | −26.82 | −14.34 | −13.20 | 11.53 | 17.11 |
| 3 | Si + C + 2HCl(g) + H2(g) = SiH(CH3)Cl2(g) | −44.03 | −31.51 | −29.50 | −18.46 | −17.45 | 4.16 | 8.92 |
| 4 | Si + 3C + HCl(g) + 4H2(g) = Si(CH3)3Cl(g) | −35.28 | −10.99 | −7.07 | 14.68 | 16.67 | 59.87 | 69.46 |
| 5 | Si + 6C + 3HCl(g) + H2(g) = Si(C6H5)Cl3(g) | −22.75 | −2.63 | 0.55 | 18.09 | 19.69 | 54.47 | 62.45 |
| 6 | SiCl4(g) + C + 3H2(g) = SiH(CH3)Cl2(g) + 2HCl(g) | 13.61 | 15.87 | 16.31 | 18.95 | 19.20 | 24.69 | 25.84 |
| 7 | SiCl4(g) + 3C + 6H2(g) = Si(CH3)3Cl(g) + 3HCl(g) | 22.37 | 36.40 | 38.80 | 52.55 | 53.84 | 80.40 | 86.36 |
| 8 | SiH2Cl2(g) + C + H2(g) = SiH(CH3)Cl2(g) | −19.10 | −13.29 | −12.32 | −7.13 | −6.69 | 3.44 | 5.61 |
| 9 | SiH2Cl2(g) + 3C + 4H2(g) = Si(CH3)3Cl(g) + HCl(g) | −10.35 | 7.26 | 10.11 | 26.01 | 27.47 | 59.15 | 66.14 |
| 10 | SiHCl3(g) + C + H2(g) = Si(CH3)Cl3(g) | −0.77 | 5.45 | 6.45 | 12.03 | 12.54 | 23.87 | 26.45 |
| 11 | SiHCl3(g) + C + 2H2(g) = SiH(CH3)Cl2(g) + HCl(g) | −1.26 | 3.04 | 3.78 | 7.91 | 8.29 | 16.51 | 18.42 |
| 12 | SiHCl3(g) + 3C + 5H2(g) = Si(CH3)3Cl(g) + 2HCl(g) | 7.49 | 23.56 | 26.21 | 41.05 | 42.42 | 72.88 | 78.8 |
| 13 | SiHCl3(g) + CH4(g)= SiH(CH3)Cl2(g) + HCl(g) | 10.81 | 9.59 | 9.38 | 8.90 | 7.97 | 4.80 | 3.94 |
| 14 | SiCl4(g) + CH4(g) = Si(CH3)Cl3(g) + HCl(g) | 26.18 | 24.82 | 24.58 | 23.26 | 23.13 | 20.36 | 19.72 |
| 15 | SiCl4(g) + 3CH4(g) = Si(CH3)3Cl(g) + 3HCl(g) | 58.60 | 56.04 | 55.56 | 52.65 | 52.37 | 45.30 | 43.43 |
| 16 | SiH(CH3)Cl2(g) + H2(g) = CH4(g) + SiH2Cl2(g) | 7.03 | 6.71 | 6.71 | 6.94 | 6.98 | 8.26 | 8.71 |
| 17 | Si(CH3)Cl3(g) + H2(g) = CH4(g) + SiHCl3(g) | −11.31 | −12.00 | −12.05 | −12.22 | −12.23 | −12.17 | −12.14 |
| 18 | Si(CH3)Cl3(g) + SiHCl3(g) = SiH(CH3)Cl2(g) + SiCl4(g) | −15.37 | −15.22 | −15.21 | −15.16 | −15.16 | −15.56 | −15.78 |
| 19 | Si(CH3)Cl3(g) + SiH2Cl2(g) = SiH(CH3)Cl2(g) + SiHCl3(g) | −18.33 | −18.71 | −18.77 | −19.16 | −19.20 | −20.43 | −20.85 |
| 20 | 2Si(CH3)Cl3(g) + SiH2Cl2(g) = 2SiH(CH3)Cl2(g) + SiCl4(g) | −33.71 | −33.93 | −33.98 | −34.32 | −34.36 | −35.98 | −36.62 |
| 21 | SiH(CH3)Cl2(g) + HCl(g) = CH4(g) + SiHCl3(g) | −10.81 | −9.60 | −9.38 | −8.09 | −7.97 | −4.84 | −3.94 |
| 22 | 3Si(CH3)Cl3(g) = Si(CH3)3Cl(g) + 2SiCl4(g) | −19.96 | −18.42 | −18.21 | −17.12 | −17.03 | −15.76 | −15.73 |
| 23 | 3SiH(CH3)Cl2(g) = Si(CH3)3Cl(g) + SiHCl3(g) + SiH2Cl2(g) | 29.12 | 30.73 | 30.98 | 32.37 | 32.50 | 35.76 | 36.67 |
| 24 | CH4(g) = C + 2H2(g) | 12.08 | 6.55 | 5.60 | 0.18 | −0.32 | −11.70 | −14.31 |
| 25 | 2CH4(g) = C2H6(g) + H2(g) | 16.30 | 16.81 | 16.86 | 16.95 | 16.95 | 16.80 | 16.75 |
| 26 | CO(g) + H2(g) = C + H2O(g) | −21.85 | −13.42 | −12.07 | −4.59 | −3.90 | 11.16 | 14.57 |
| 27 | CO2(g) + 2H2(g) = C + 2H2O(g) | −15.01 | −9.03 | −8.04 | −2.43 | −1.93 | 9.82 | 12.54 |
| 28 | CO(g) + 3H2(g) = CH4(g) + H2O(g) | −33.92 | −19.97 | −17.68 | −4.78 | −3.59 | 22.86 | 28.90 |
| 29 | CO2(g) + 4H2(g) = CH4(g) + 2H2O(g) | −27.08 | −15.58 | −13.65 | −3.65 | −2.62 | 21.52 | 26.86 |
| 30 | CO2(g) + Si = C + SiO2 | −110.44 | −99.15 | −97.39 | −87.85 | −86.99 | −68.61 | −64.52 |
| 31 | 2CO(g) + Si = 2C + SiO2 | −139.12 | −116.96 | −113.50 | −94.59 | −92.89 | −56.11 | −47.90 |
| Sample No | CH3SiHCl2 | CH3SiCl3 | (CH3)3SiCl | (CH3)2SiCl2 | ||||
| DC | HC | DC | HC | DC | HC | DC | HC | |
| 1 | 1.02 × 103 | 1.65 | 121 | 30.8 | / | / | / | / |
| 2 | 1.03 × 103 | 1.18 | 131 | 242 | / | / | / | / |
| 3 | 1.17 × 103 | 1.40 | 192 | 176 | / | / | / | / |
| 4 | 1.14 × 103 | 1.33 | 150 | 83.1 | / | / | / | / |
| 5 | 1.17 × 103 | 2.52 | 82.3 | 24.5 | / | / | / | / |
| 6 | 901 | 3.22 | 71.2 | 47.5 | / | / | / | / |
| 7 | 927 | 14.5 | 87.7 | 255 | / | / | / | / |
| 8 | 1.09 × 103 | 8.14 | 92.5 | 2.04 | / | / | / | / |
| 9 | 1.39 × 103 | 6.04 | 121 | 107 | / | / | / | / |
| 10 | 1.36 × 103 | 5.31 | 44.4 | 84.5 | / | / | / | / |
| Sample No | CH3SiHCl2 | CH3SiCl3 | (CH3)3SiCl | (CH3)2SiCl2 | ||||
| Purified TCS | STC | Purified TCS | STC | Purified TCS | STC | Purified TCS | STC | |
| 1 | 7.22 | / | / | 195 | / | / | / | / |
| 2 | 9.03 | 1.17 | / | 544 | / | / | / | / |
| 3 | 8.21 | / | / | 214 | / | / | / | / |
| 4 | 2.82 | / | / | 254 | / | / | / | / |
| 5 | 5.84 | 1.73 | / | 649 | / | / | / | / |
| 6 | 3.18 | / | / | 175 | / | / | / | / |
| 7 | 7.87 | 1.48 | / | 766 | / | / | / | / |
| 8 | 3.51 | 1.22 | / | 735 | / | / | / | / |
| 9 | 5.03 | 1.07 | / | 699 | / | / | / | / |
| 10 | 3.91 | 1.54 | / | 764 | / | / | / | / |
| Sample No | Reduction-Recovered Hydrogen | Hydrogenation-Recovered Hydrogen | ||||||
| CO | CO2 | CH4 | Methylchlorosilane | CO | CO2 | CH4 | Methylchlorosilane | |
| 1 | / | / | 0.92 | / | / | / | 371 | / |
| 2 | / | / | 1.17 | / | / | / | 339 | / |
| 3 | / | / | 0.84 | / | / | / | 397 | / |
| 4 | / | / | 1.08 | / | / | / | 381 | / |
| 5 | / | / | 1.08 | / | / | / | 318 | / |
| 6 | / | / | 0.93 | / | / | / | 337 | / |
| 7 | / | / | 0.70 | / | / | / | 341 | / |
| 8 | / | / | 0.95 | / | / | / | 319 | / |
| 9 | / | / | 0.60 | / | / | / | 329 | / |
| 10 | / | / | 0.66 | / | / | / | 329 | / |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Hou, Y.; Lv, X.; Huang, G. Study on the Evolution Mechanism of Carbon Impurities in Polysilicon Production Based on HSC Simulation. Materials 2026, 19, 798. https://doi.org/10.3390/ma19040798
Hou Y, Lv X, Huang G. Study on the Evolution Mechanism of Carbon Impurities in Polysilicon Production Based on HSC Simulation. Materials. 2026; 19(4):798. https://doi.org/10.3390/ma19040798
Chicago/Turabian StyleHou, Yu, Xueqian Lv, and Guoqiang Huang. 2026. "Study on the Evolution Mechanism of Carbon Impurities in Polysilicon Production Based on HSC Simulation" Materials 19, no. 4: 798. https://doi.org/10.3390/ma19040798
APA StyleHou, Y., Lv, X., & Huang, G. (2026). Study on the Evolution Mechanism of Carbon Impurities in Polysilicon Production Based on HSC Simulation. Materials, 19(4), 798. https://doi.org/10.3390/ma19040798