Electrospun Conductive Composites with Anisotropic Microstructures and Tunable Mechanical Properties for Wearable Bioelectronics
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Fabrication of Electrospun Membranes
2.3. Fabrication of Composite Conductive Membranes
2.4. Characterization and Testing Methods
3. Results and Discussion
3.1. Microstructural Analysis of Composite Membranes
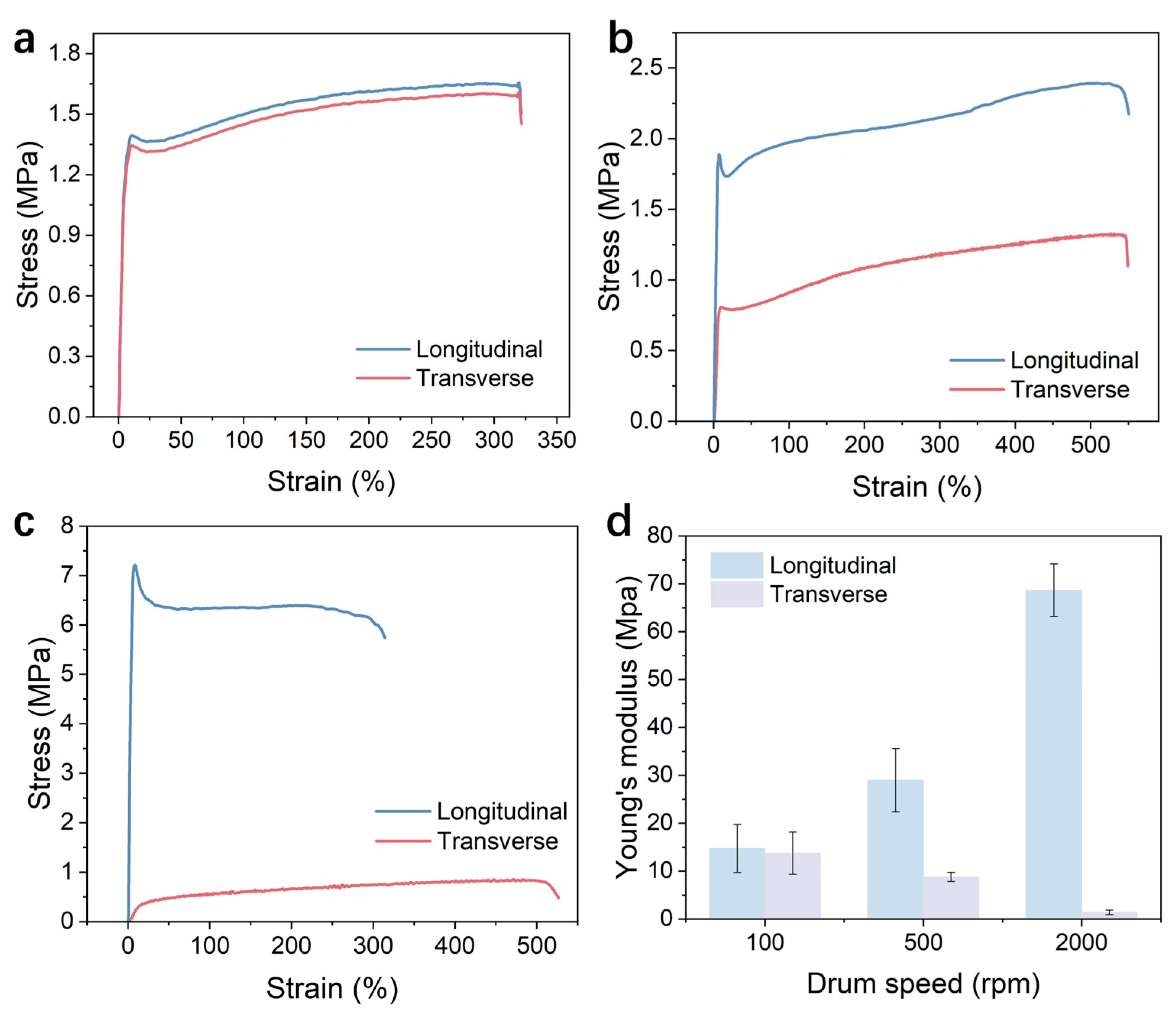
3.2. Mechanical Performance Analysis
3.3. Electrical Performance Analysis
3.4. Electrode-Skin Interface Impedance Characteristics
| Dimension | Material System | Structural Design Strategy | Mechanical Performance | Electrical Performance | Demonstrated Application |
|---|---|---|---|---|---|
| MXene Composite Fibers (2025) [66] | MXene, CNTs, PLA | Static-dynamic densification | Ultrahigh strength (941.5 MPa) | High electronic conductivity | Wireless e-textiles |
| Breathable Textile Electrodes (2026) [67] | Cotton, MXene, PEDOT:PSS | Surface functionalization coating | Intrinsic fabric flexibility | Stable for biopotentials | ECG, EMG, EEG monitoring |
| TPEE/PPy Membrane (2026) [68] | TPEE fibers, PPy, TA | Electrospun substrate in situ polymerization | High stretchability (0.4–1.3 Mpa) | Moderate electronic conductivity | AI diagnosis |
| WADE-Skin (2023) [69] | SBS/PAAND fibers, EGaIn | Multilayer fibrous stack | Skin-like softness (850 kPa) | Low impedance | ECG and HMI |
| PEO/Ionogel Membranes (This Study) | PEO fibers, ([EMIm][DCA]) | Programmable fiber alignment | Programmable anisotropy (1–74 MPa) | Stable ionic conductivity | bioelectronic interfaces for ECG and EMG |
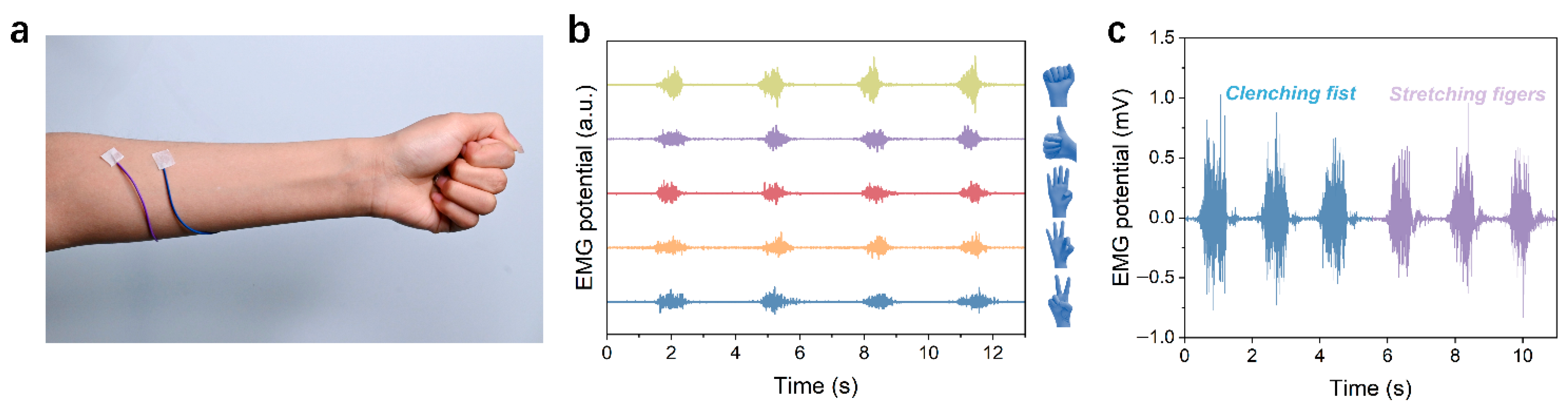
3.5. Physiological Electrical Signal Monitoring Application
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chen, X.G.; Wang, C.H.; Wei, W.; Liu, Y.H.; Ge, S.S.; Zhou, L.Q.; Kong, H. Flexible and sensitive pressure sensor with enhanced breathability for advanced wearable health monitoring. npj Flex. Electron. 2025, 9, 101. [Google Scholar] [CrossRef]
- Wang, Y.Q.; Sun, C.C.; Ahmed, D. A smart acoustic textile for health monitoring. Nat. Electron. 2025, 8, 485–495. [Google Scholar] [CrossRef]
- Yang, M.; Cheng, Y.F.; Yue, Y.; Chen, Y.; Gao, H.; Li, L.; Cai, B.; Liu, W.J.; Wang, Z.Y.; Guo, H.Z. High-performance flexible pressure sensor with a self-healing function for tactile feedback. Adv. Sci. 2022, 9, 2200507. [Google Scholar] [CrossRef]
- Liu, D.J.; Zhu, P.C.; Zhang, F.K.; Li, P.S.; Huang, W.H.; Li, C.; Han, N.N.; Mu, S.R.; Zhou, H.; Mao, Y.C. Intrinsically stretchable polymer semiconductor based electronic skin for multiple perceptions of force, temperature, and visible light. Nano Res. 2023, 16, 1196–1204. [Google Scholar] [CrossRef]
- Lu, L.J.; Wu, J.; Zhang, Y.J.; Liu, C.; Hu, Y.; Chen, B.J.; Zhu, Y.; Mao, Y.C. Noncontact 3D gesture recognition enabled VR human-machine interface via electret-nanofiber-based triboelectric sensor. Nano Res. 2025, 18, 94907924. [Google Scholar] [CrossRef]
- Zhang, W.; Lou, Q.; Sun, J.L.; Liao, J.; Zheng, G.S.; Jiao, F.H.; Chen, W.; Li, X.; Meng, J.J.; Shan, C.X.; et al. Carbon nanodot-based flexible and self-powered white displays. Nano Res. 2025, 18, 94907117. [Google Scholar] [CrossRef]
- Zhu, P.C.; Zhang, B.S.; Wang, H.Y.; Wu, Y.H.; Cao, H.J.; He, L.B.; Li, C.Y.; Luo, X.P.; Li, X.; Mao, Y.C. 3D printed triboelectric nanogenerator as self-powered human-machine interactive sensor for breathing-based language expression. Nano Res. 2022, 15, 7460–7467. [Google Scholar]
- Xu, B.Y.; Yang, M.Y.; Cheng, W.J.; Li, X.Y.; Xu, X.M.; Li, W.M.; Zhang, H.; Zhou, M. Precision aerosol-jet micropatterning of liquid metal for high-performance flexible strain sensors. Nat. Commun. 2025, 16, 7920. [Google Scholar] [CrossRef]
- Feng, T.X.; Ling, D.; Li, C.Y.; Zheng, W.T.; Zhang, S.C.; Li, C.; Emel’yanov, A.; Pozdnyakov, A.S.; Lu, L.J.; Mao, Y.C. Stretchable on-skin touchless screen sensor enabled by ionic hydrogel. Nano Res. 2024, 17, 4462–4470. [Google Scholar] [CrossRef]
- Chen, X.X.; Yang, X.; Lou, Q.; Zhang, Y.; Chen, Y.C.; Lu, Y.C.; Dong, L.; Shan, C.X. Fabry-Perot interference and piezo-phototronic effect enhanced flexible MoS2 photodetector. Nano Res. 2022, 15, 4395–4402. [Google Scholar]
- Zhu, P.C.; Mu, S.R.; Huang, W.H.; Sun, Z.Y.; Lin, Y.Y.; Chen, K.; Pan, Z.F.; Haghighi, M.G.; Sedghi, R.; Wang, J.L.; et al. Soft multifunctional neurological electronic skin through intrinsically stretchable synaptic transistor. Nano Res. 2024, 17, 6550–6559. [Google Scholar]
- Zhu, P.C.; Niu, M.J.; Liang, S.Y.; Yang, W.Q.; Zhang, Y.T.; Chen, K.; Pan, Z.F.; Mao, Y.C. Non-hand-worn, load-free VR hand rehabilitation system assisted by deep learning based on ionic hydrogel. Nano Res. 2025, 18, 94907301. [Google Scholar]
- Chen, B.; Yu, R.; Wang, J.; Feng, Y.; Zhang, Y.; Mao, Y.; Shan, C.; Wang, X. Biomaterials-Based Hydrogel with Superior Bio-Mimetic Ionic Conductivity and Tissue-Matching Softness for Bioelectronics. Adv. Funct. Mater. 2025, 35, e27495. [Google Scholar] [CrossRef]
- Gao, M.Y.; Liu, W.J.; Chen, K.; Sun, H.L.; Liu, X.Q.; Xing, H.N.; Wang, H.T.; Zhu, B.P.; Guo, H.Z. Piezoresistive Effect: A New Concept for Hearing Aids. Adv. Sci. 2025, 12, 2501227. [Google Scholar] [CrossRef]
- Han, S.J.; Liu, C.R.; Xu, H.H.; Yao, D.Y.; Yan, K.H.; Zheng, H.L.; Chen, H.J.; Gui, X.C.; Chu, S.; Liu, C. Multiscale nanowire-microfluidic hybrid strain sensors with high sensitivity and stretchability. npj Flex. Electron. 2018, 2, 16. [Google Scholar] [CrossRef]
- Li, W.W.; Yang, S.; Shamim, A. Screen printing of silver nanowires: Balancing conductivity with transparency while maintaining flexibility and stretchability. npj Flex. Electron. 2019, 3, 13. [Google Scholar] [CrossRef]
- Lu, L.J.; Hu, G.S.; Liu, J.Q.; Yang, B. 5G NB-IoT System Integrated with High-Performance Fiber Sensor Inspired by Cirrus and Spider Structures. Adv. Sci. 2024, 11, 2309894. [Google Scholar]
- Matsuhisa, N.; Inoue, D.; Zalar, P.; Jin, H.; Matsuba, Y.; Itoh, A.; Yokota, T.; Hashizume, D.; Someya, T. Printable elastic conductors by in situ formation of silver nanoparticles from silver flakes. Nat. Mater. 2017, 16, 834–840. [Google Scholar] [CrossRef]
- Matsuhisa, N.; Kaltenbrunner, M.; Yokota, T.; Jinno, H.; Kuribara, K.; Sekitani, T.; Someya, T. Printable elastic conductors with a high conductivity for electronic textile applications. Nat. Commun. 2015, 6, 7461. [Google Scholar] [CrossRef]
- Park, J.; Wang, S.D.; Li, M.; Ahn, C.; Hyun, J.K.; Kim, D.S.; Kim, D.K.; Rogers, J.A.; Huang, Y.G.; Jeon, S. Three-dimensional nanonetworks for giant stretchability in dielectrics and conductors. Nat. Commun. 2012, 3, 916. [Google Scholar] [CrossRef]
- Sunwoo, S.H.; Kim, H.J.; Kim, J.H.; Kim, D.C.; Kim, D.H. Intrinsically soft electronics using conducting nanomaterials and liquid metals. NPG Asia Mater. 2025, 17, 40. [Google Scholar] [CrossRef]
- Tan, P.; Wang, H.F.; Xiao, F.R.; Lu, X.; Shang, W.H.; Deng, X.B.; Song, H.F.; Xu, Z.Y.; Cao, J.F.; Gan, T.S.; et al. Solution-processable, soft, self-adhesive, and conductive polymer composites for soft electronics. Nat. Commun. 2022, 13, 358. [Google Scholar]
- Xie, Z.L.; Zhu, J.N.; Dou, Z.L.; Zhang, Y.Z.; Wang, K.; Wu, K.; Fu, Q. Liquid metal interface mechanochemistry disentangles energy density and biaxial stretchability tradeoff in composite capacitor film. Nat. Commun. 2024, 15, 7817. [Google Scholar] [CrossRef]
- Xu, Q.G.; Chu, N.N.; Wang, Y.; Wang, H.; Xu, T.T.; Li, X.L.; Huang, S.Z.; Li, X.J.; Luo, Y.S.; Yang, H.Y.; et al. 3D Printed Low-Tortuosity and Ultra-Thick Hierarchical Porous Electrodes for High-Performance Wearable Quasi-Solid-State Zn-VOH Batteries. Adv. Sci. 2025, 12, 2401660. [Google Scholar]
- Chang, S.L.; Deng, Y.; Li, N.; Wang, L.J.; Shan, C.X.; Dong, L. Continuous synthesis of ultra-fine fiber for wearable mechanoluminescent textile. Nano Res. 2023, 16, 9379–9386. [Google Scholar] [CrossRef]
- Fatahian, R.; Erfani, R. Surrogate modeling of electrospun PVA/PLA nanofibers using artificial neural network for biomedical applications. Sci. Rep. 2025, 15, 12886. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.; Dong, K.; Ye, C.Y.; Jiang, Y.; Zhai, S.Y.; Cheng, R.W.; Liu, D.; Gao, X.P.; Wang, J.; Wang, Z.L. A breathable, biodegradable, antibacterial, and self-powered electronic skin based on all-nanofiber triboelectric nanogenerators. Sci. Adv. 2020, 6, eaba9624. [Google Scholar] [CrossRef]
- Samadian, H.; Zamiri, S.; Ehterami, A.; Farzamfar, S.; Vaez, A.; Khastar, H.; Alam, M.; Ai, A.; Derakhshankhah, H.; Allahyari, Z.; et al. Electrospun cellulose acetate/gelatin nanofibrous wound dressing containing berberine for diabetic foot ulcer healing: In vitro and in vivo studies. Sci. Rep. 2020, 10, 8312. [Google Scholar] [CrossRef]
- Smoak, M.M.; Hogan, K.J.; Grande-Allen, K.J.; Mikos, A.G. Bioinspired electrospun dECM scaffolds guide cell growth and control the formation of myotubes. Sci. Adv. 2021, 7, eabg4123. [Google Scholar] [CrossRef]
- Sohrabi, M.; Razbin, M. Hybrid modeling for optimizing electrospun polyurethane nanofibrous membranes in air filtration applications. Sci. Rep. 2025, 15, 27306. [Google Scholar] [CrossRef] [PubMed]
- Gilmore, T.S.; Gouma, P.I. Scalable electrospinning using a desktop, high throughput, self-contained system. Sci. Rep. 2024, 14, 25844. [Google Scholar] [CrossRef]
- Hosseinian, H.; Jimenez-Moreno, M.; Sher, M.; Rodriguez-Garcia, A.; Martinez-Chapa, S.O.; Hosseini, S. An origami-based technique for simple, effective and inexpensive fabrication of highly aligned far-field electrospun fibers. Sci. Rep. 2023, 13, 7083. [Google Scholar]
- Persano, L.; Dagdeviren, C.; Su, Y.W.; Zhang, Y.H.; Girardo, S.; Pisignano, D.; Huang, Y.G.; Rogers, J.A. High performance piezoelectric devices based on aligned arrays of nanofibers of poly(vinylidenefluoride-co-trifluoroethylene). Nat. Commun. 2013, 4, 1633. [Google Scholar] [CrossRef] [PubMed]
- Shaker, A.; Khedewy, A.T.; Hassan, M.A.; Abd El-Baky, M.A. Thermo-mechanical characterization of electrospun polyurethane/carbon-nanotubes nanofibers: A comparative study. Sci. Rep. 2023, 13, 17368. [Google Scholar] [CrossRef] [PubMed]
- Tsuboi, K.; Marcelletti, E.; Matsumoto, H.; Ashizawa, M.; Minagawa, M.; Furuya, H.; Tanioka, A.; Abe, A. Preparation of poly(γ-benzyl-L-glutamate) nanofibers by electrospinning from isotropic and biphasic liquid crystal solutions. Polym. J. 2012, 44, 360–365. [Google Scholar] [CrossRef]
- Yu, H.Q.; Li, Y.; Song, Y.; Wu, Y.B.; Lan, X.J.; Liu, S.M.; Tang, Y.; Xu, S.S.; Chen, B.J. Ultralong well-aligned TiO2: Ln3+ (Ln = Eu, Sm, or Er) fibres prepared by modified electrospinning and their temperature-dependent luminescence. Sci. Rep. 2017, 7, 44099. [Google Scholar]
- Bu, X.M.; Ge, Y.X.; Wang, L.; Wu, L.L.; Ma, X.F.; Lu, D.Y. Design of highly stretchable deep eutectic solvent-based ionic gel electrolyte with high ionic conductivity by the addition of zwitterion ion dissociators for flexible supercapacitor. Polym. Eng. Sci. 2021, 61, 154–166. [Google Scholar]
- Choi, S.E.; Oh, S.J.; Yoon, J.M.; Bae, J.W. A Non-Volatile, Low-Voltage, Stretchable Transparent Dielectric Heater for Real-World Autonomous Thermal Management Platform. Small 2025, 22, e10593. [Google Scholar] [CrossRef]
- He, X.N.; Zhang, B.; Liu, Q.J.; Chen, H.; Cheng, J.X.; Jian, B.C.; Yin, H.L.; Li, H.G.; Duan, K.; Zhang, J.W.; et al. Highly conductive and stretchable nanostructured ionogels for 3D printing capacitive sensors with superior performance. Nat. Commun. 2024, 15, 6431. [Google Scholar] [CrossRef]
- Jung, J.; Lee, S.; Kim, H.; Lee, W.; Chong, J.; You, I.; Kang, J. Self-healing electronic skin with high fracture strength and toughness. Nat. Commun. 2024, 15, 9763. [Google Scholar] [CrossRef]
- Lyu, X.L.; Yu, K.; Zhang, H.Q.; Zhou, P.P.; Shen, Z.H.; Zou, Z.G. Tough fiber-reinforced composite ionogels with crack resistance surpassing metals. Nat. Commun. 2025, 16, 4005. [Google Scholar] [CrossRef]
- Marsavelski, A.; Smrecki, V.; Vianello, R.; Zinic, M.; Mogus-Milankovic, A.; Santic, A. Supramolecular Ionic-Liquid Gels with High Ionic Conductivity. Chem.–Eur. J. 2015, 21, 12121–12128. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Feng, W.W.; Liu, Y.C.; Chen, L.L.; Chen, T.; Jin, Z.K.; Wang, C. Elastic and ultra stable ionic conductors for long-life-time soft robots working at extreme environments. Nat. Commun. 2025, 16, 6004. [Google Scholar] [PubMed]
- Wang, H.; Gupta, A.; Lu, Q.C.; Wu, W.T.; Wang, X.Y.; Huang, X.; Hu, X.Y.; Lee, P.S. Ultrasoft and fast self-healing poly(ionic liquid) electrode for dielectric elastomer actuators. Nat. Commun. 2025, 16, 7405. [Google Scholar]
- Wang, J.Q.; Wu, B.H.; Wei, P.; Sun, S.T.; Wu, P.Y. Fatigue-free artificial ionic skin toughened by self-healable elastic nanomesh. Nat. Commun. 2022, 13, 4411. [Google Scholar]
- Wang, S.H.; Liu, H.Y.; Yu, Z.Y.; Ren, X.L.; Hua, Q.; Panahi-Sarmad, M.; Yang, P.; Liu, C.H.; Renneckar, S.; Liu, H.; et al. Cellulose-mediated ionic liquid crystallization enables tough-stiff switchable ionogels. Nat. Commun. 2025, 16, 9007. [Google Scholar]
- Wang, Y.; Wei, Z.X.; Ji, T.T.; Bai, R.B.; Zhu, H.L. Highly Ionic Conductive, Stretchable, and Tough Ionogel for Flexible Solid-State Supercapacitor. Small 2024, 20, 2307019. [Google Scholar]
- Wen, J.Q.; Zhou, L.; Ye, T.L. Polymer ionogels and their application in flexible ionic devices. SmartMat 2024, 5, e1253. [Google Scholar] [CrossRef]
- Ye, Z.W.; Yu, H.M.; Xie, H.X.; Zhu, W.W.; Shi, S.T.; Liu, C.C.; Wang, Y.Y.; Liao, J.Q.; Sun, Q.F.; Zhao, D.W.; et al. Flame-Retardant Ionogel Enabled by Lignin Molecular Networks for Fire Rescue. Adv. Sci. 2025, 12, e06901. [Google Scholar]
- Zhou, X.; Zhou, K.J.; Tang, L.; Chen, Z.Y.; Hu, Q.Y.; Gao, J.; Zhang, Y.; Zhang, J.; Zhang, S.G. A Strong and Highly Transparent Ionogel Electrolyte Enabled by In Situ Polymerization-Induced Microphase Separation for High-Performance Electrochromic Devices. Macromol. Rapid Commun. 2024, 45, 202300736. [Google Scholar] [CrossRef]
- Lv, B.; Zhao, G.R.; Wang, H.G.; Wang, Q.J.; Yang, B.P.; Ma, W.; Li, Z.Y.; Li, J.J. Ionogel Fiber-Based Flexible Sensor for Friction Sensing. Adv. Mater. Technol. 2023, 8, 2201617. [Google Scholar]
- Wang, X.; Gao, Q.S.; Schubert, D.W.; Liu, X.H. Review on Electrospun Conductive Polymer Composites Strain Sensors. Adv. Mater. Technol. 2023, 8, 2300293. [Google Scholar] [CrossRef]
- Meinhold, K.L.; Tankersley, T.; Darlington, R.; Robinson, J.L. Mandrel Diameter Is a Dominating Parameter for Fiber Alignment Control in Rotating Mandrel Electrospinning Systems. J. Appl. Polym. Sci. 2025, 142, e57364. [Google Scholar]
- Mondragón, M.; Arias, E.; Elizalde, L.E.; Castañeda, M.E.; Moggio, I. Luminescence properties of aligned-electrospun fibers of poly(9-vinylcarbazole) doped with a europium (III) complex. J. Lumin. 2017, 192, 745–751. [Google Scholar]
- Sakamoto, H.; Asakawa, H.; Fukuma, T.; Fujita, S.; Suye, S. Atomic force microscopy visualization of hard segment alignment in stretched polyurethane nanofibers prepared by electrospinning. Sci. Technol. Adv. Mater. 2014, 15, 558–571. [Google Scholar] [CrossRef]
- Wang, H.B.; Mullins, M.E.; Cregg, J.M.; McCarthy, C.W.; Gilbert, R.J. Varying the diameter of aligned electrospun fibers alters neurite outgrowth and Schwann cell migration. Acta Biomater. 2010, 6, 2970–2978. [Google Scholar] [CrossRef]
- Croisier, F.; Duwez, A.S.; Jérôme, C.; Léonard, A.F.; van der Werf, K.O.; Dijkstra, P.J.; Bennink, M.L. Mechanical testing of electrospun PCL fibers. Acta Biomater. 2012, 8, 218–224. [Google Scholar] [CrossRef]
- Pouladzadeh, F.; Katbab, A.A.; Haghighipour, N.; Kashi, E. Carbon nanotube loaded electrospun scaffolds based on thermoplastic urethane (TPU) with enhanced proliferation and neural differentiation of rat mesenchymal stem cells: The role of state of electrical conductivity. Eur. Polym. J. 2018, 105, 286–296. [Google Scholar] [CrossRef]
- Stachewicz, U.; Bailey, R.J.; Wang, W.; Barber, A.H. Size dependent mechanical properties of electrospun polymer fibers from a composite structure. Polymer 2012, 53, 5132–5137. [Google Scholar] [CrossRef]
- Sukiman, M.S.; Andriyana, A.; Ang, B.C.; Metselaar, H.S.C. Elastic properties of electrospun PVDF nanofibrous membranes: Experimental investigation and numerical modelling using pixel-based finite element method. Polym. Test. 2020, 81, 106218. [Google Scholar] [CrossRef]
- Zaarour, B.; Zhu, L.; Huang, C.; Jin, X. Enhanced piezoelectric properties of randomly oriented and aligned electrospun PVDF fibers by regulating the surface morphology. J. Appl. Polym. Sci. 2019, 136, 47049. [Google Scholar]
- Huang, L.W.; Bui, N.N.; Manickam, S.S.; McCutcheon, J.R. Controlling Electrospun Nanofiber Morphology and Mechanical Properties Using Humidity. J. Polym. Sci. B Polym. Phys. 2011, 49, 1734–1744. [Google Scholar]
- Sousa, A.S.P.; Noites, A.; Vilarinho, R.; Santos, R. Long-Term Electrode-Skin Impedance Variation for Electromyographic Measurements. Sensors 2023, 23, 8582. [Google Scholar]
- Zhang, Y.Z.; Zhou, J.L.; Yang, H.Y.; Liu, Q.X.; Wang, M.; Xiong, F.; Chen, D.Y.; Du, L.X. Effect of Fabric Electrode Surface Coating Medium on ECG Signal Quality under Dynamic and Static Conditions. Coatings 2023, 13, 108. [Google Scholar] [CrossRef]
- Ni, Q.C.; Lou, Q.; Shen, C.L.; Zheng, G.S.; Song, R.W.; Hao, J.N.; Liu, J.L.; Zhu, J.Y.; Zang, J.H.; Dong, L.; et al. Sensitive humidity sensor based on moisture-driven energy generation. Nano Res. 2024, 17, 5578–5586. [Google Scholar] [CrossRef]
- Zhou, T.; Yan, J.; Cao, C.; He, Q.; Li, W.; Chen, L.; Wu, C.; Feng, Y.; Lau, D.; Cheng, Q.; et al. Ultrastrong MXene composite fibers through static-dynamic densification for wireless electronic textiles. Nat. Commun. 2025, 16, 111703. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Guo, H.; Cai, J.; Wang, F.; Li, J.; Tang, Q.; Yu, J.; Ding, B.; Li, Z. Breathable and reusable fabric epidermal electrodes for personal health monitoring. Nano Energy 2026, 148, 111703. [Google Scholar] [CrossRef]
- Liu, J.; Yi, J.; Shen, W.; Hu, J.; Fu, T.; Jing, M.; Qian, H.; Chen, J.; Li, M.; Lin, Y.; et al. Highly Stretchable and Durable Thermoplastic Poly(ether–ester) Fibrous Membrane for Constructing a Deep Learning-Assisted Knee Deformity Diagnosis System. Adv. Fiber Mater. 2026. [Google Scholar] [CrossRef]
- Chen, F.; Zhuang, Q.; Ding, Y.; Zhang, C.; Song, X.; Chen, Z.; Zhang, Y.; Mei, Q.; Zhao, X.; Huang, Q.; et al. Wet-Adaptive Electronic Skin. Adv. Mater. 2023, 35, e2305630. [Google Scholar] [PubMed]








| Polymer | PEO | PCL | PVA | TPU | PVDF | PAN |
|---|---|---|---|---|---|---|
| Young’s modulus | 1–74 MPa | 3–5 MPa | 1–10 MPa | 1–10 MPa | 3–15 MPa | 5.7–9.4 MPa |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Liu, J.; Liu, C.; Du, A.; Liu, Y.; Feng, Y.; Zhang, Y.; Pan, Z.; Lu, L.; Mao, Y. Electrospun Conductive Composites with Anisotropic Microstructures and Tunable Mechanical Properties for Wearable Bioelectronics. Materials 2026, 19, 684. https://doi.org/10.3390/ma19040684
Liu J, Liu C, Du A, Liu Y, Feng Y, Zhang Y, Pan Z, Lu L, Mao Y. Electrospun Conductive Composites with Anisotropic Microstructures and Tunable Mechanical Properties for Wearable Bioelectronics. Materials. 2026; 19(4):684. https://doi.org/10.3390/ma19040684
Chicago/Turabian StyleLiu, Jing, Chang Liu, Ankang Du, Yiming Liu, Yunxiang Feng, Yujie Zhang, Zhifeng Pan, Lijun Lu, and Yanchao Mao. 2026. "Electrospun Conductive Composites with Anisotropic Microstructures and Tunable Mechanical Properties for Wearable Bioelectronics" Materials 19, no. 4: 684. https://doi.org/10.3390/ma19040684
APA StyleLiu, J., Liu, C., Du, A., Liu, Y., Feng, Y., Zhang, Y., Pan, Z., Lu, L., & Mao, Y. (2026). Electrospun Conductive Composites with Anisotropic Microstructures and Tunable Mechanical Properties for Wearable Bioelectronics. Materials, 19(4), 684. https://doi.org/10.3390/ma19040684









