Corrosion-Induced Stress Concentration Characteristics Study of Steel Strands in Bridge Cables Under Tension-Bending Coupling Loads
Abstract
1. Introduction
2. Salt Spray Accelerated Corrosion Test of Steel Strands
2.1. Steel Strand Specimens Treatment
2.2. Fabrication of the Salt Spray Corrosion Chamber
2.3. Preparation of Corrosive Solution
2.4. Test Conditions for Salt Spray Accelerated Corrosion
2.5. Mass Loss Analysis of Corroded Steel Strands
3. Morphological Characteristics of Corrosion Pits on Steel Strands
3.1. Scanning of Corrosion Pit Depth
3.2. Statistical Analysis of Corrosion Pit Depth
4. Refined Numerical Method of Corroded Steel Strands Under Tension-Bending Coupling Loads
4.1. Numerical Modeling Method
4.2. Numerical Analysis of Equivalent Stress at Corrosion Pits on Steel Strands
5. Parameter Studies of Stress Concentration Characteristics of Corroded Steel Strands Under Tension-Bending Coupling Loads
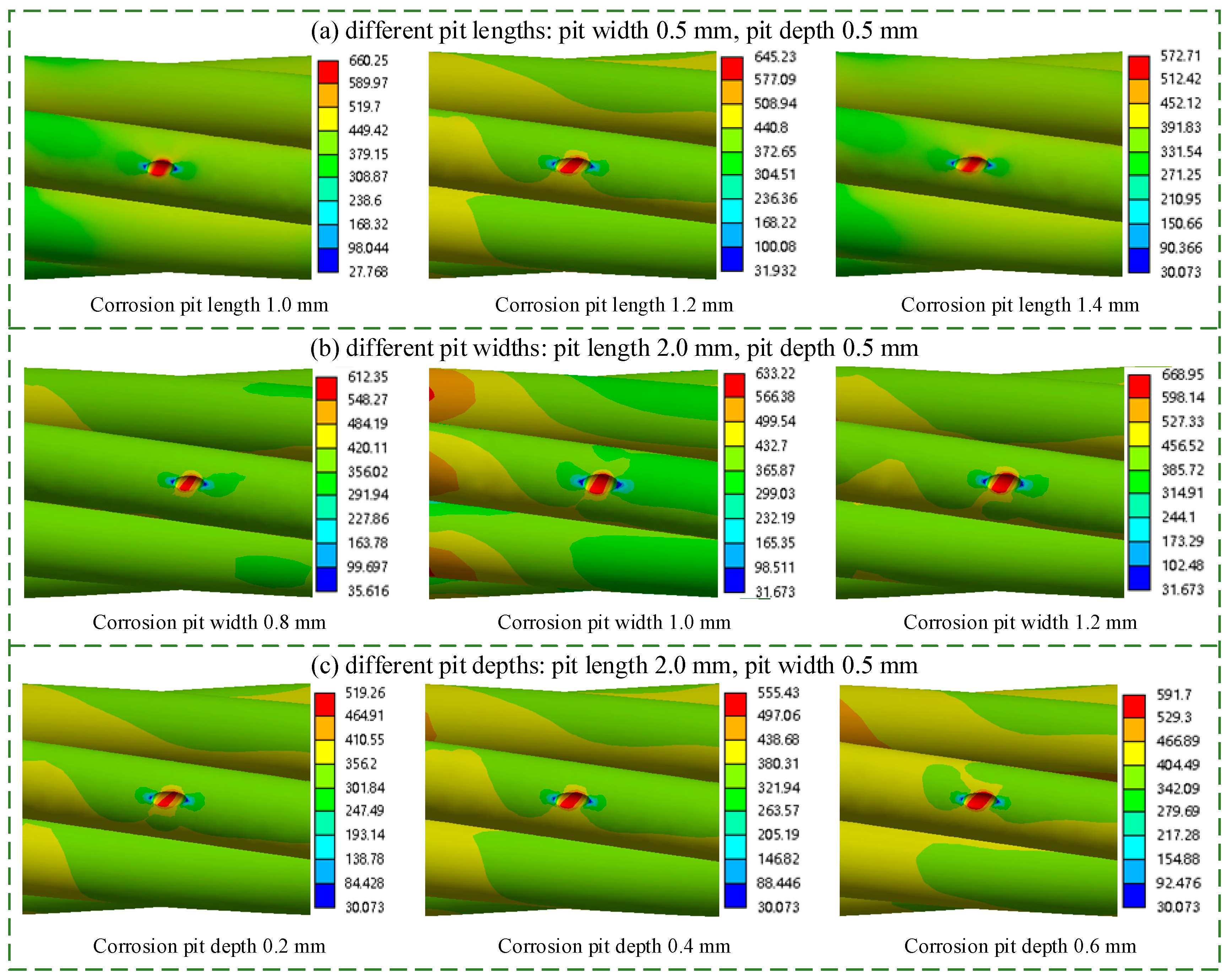
5.1. Effect of Corrosion Pit Length
5.2. Effect of Corrosion Pit Width
5.3. Effect of Corrosion Pit Depth
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wang, H.; Shen, R.L.; Bai, L.H.; Wang, L.; Chen, W. Study on the movements and bending stresses of hangers and control measures in self-anchored rail suspension bridges. Eng. Struct. 2023, 275, 115304. [Google Scholar] [CrossRef]
- Cai, H.J.; Liu, Z.X.; Guo, T.; Cai, C.S.; Sun, Z.; Zong, Y.; Zhou, C. Investigation on nonstationary wind effects and intelligent multi-level warning method for wind-induced vibration of bridges. Structures 2026, 84, 111112. [Google Scholar] [CrossRef]
- Sun, H.H.; Tang, X.T.; Cai, S.Y.; Yan, L.; Liu, H.J. Fatigue of suspender anchorages under axial and bending loads of suspension bridges. J. Constr. Steel Res. 2024, 212, 108252. [Google Scholar] [CrossRef]
- Sun, H.H.; Tang, X.T.; Wang, C.S.; Fan, P.; Wang, L.Y. Stress response behaviors of steel wires at the anchorage part of bridge cables under tension and bending coupling loads. J. Bridge Eng. 2024, 29, 04024016. [Google Scholar] [CrossRef]
- Yuan, Y.G.; Liu, X.D.; Pu, G.N.; Wang, T.; Zheng, D.Z. Temporal and spatial variability of corrosion of high-strength steel wires within a bridge stay cable. Constr. Build. Mater. 2021, 308, 125108. [Google Scholar] [CrossRef]
- Yao, G.W.; Yang, S.C.; Zhang, J.Q.; Leng, Y.L. Analysis of corrosion-fatigue damage and fracture mechanism of in-service bridge cables/hangers. Adv. Civ. Eng. 2021, 2021, 6633706. [Google Scholar]
- Nakamura, S.; Suzumura, K. Experimental Study on Fatigue Strength of Corroded Bridge Wires. J. Bridge Eng. 2013, 18, 200–209. [Google Scholar] [CrossRef]
- Li, S.L.; Xu, Y.; Li, H.; Guan, X.C. Uniform and Pitting Corrosion Modeling for High-Strength Bridge Wires. J. Bridge Eng. 2014, 19, 04014025. [Google Scholar] [CrossRef]
- Wan, S.; Zhou, H.J.; Li, L.X.; Wang, C.; Filippo, M.D.; Gong, F. Degradation of artificially corroded galvanized high-strength steel wires: Corrosion morphology and mechanical behavior. Constr. Build. Mater. 2022, 346, 128387. [Google Scholar] [CrossRef]
- Zhou, H.J.; Wan, S.P.; Li, W.J. Test and simulation of corroded high strength steel wires: From scanned morphology feature to mechanical degradation. Corros. Sci. 2024, 240, 112392. [Google Scholar] [CrossRef]
- Yan, K.; Liu, G.D.; Li, Q.; Jiang, C.Y.; Ren, T.C.; Li, Z.; Xie, L. Corrosion characteristics and evaluation of galvanized high-strength steel wire for bridge cables based on 3D laser scanning and image recognition. Constr. Build. Mater. 2024, 422, 135845. [Google Scholar] [CrossRef]
- Chen, A.R.; Yang, Y.Y.; Ma, R.J.; Li, L.W.; Tian, H.; Pan, Z.C. Experimental study of corrosion effects on high-strength steel wires considering strain influence. Constr. Build. Mater. 2020, 240, 117910. [Google Scholar] [CrossRef]
- Fang, K.; Li, S.L.; Chen, Z.S.; Li, H. Geometric characteristics of corrosion pits on high-strength steel wires in bridge cables under applied stress. Struct. Infrastruct. Eng. 2021, 17, 34–48. [Google Scholar] [CrossRef]
- Ma, R.J.; Su, R.Q.; Cui, C.J.; Liu, Y.; Hu, H.; Chen, A.R. Corrosion characteristics and mechanical properties degradation of stressed high-strength steel wires in acidic environment. Case Stud. Constr. Mater. 2025, 22, e04773. [Google Scholar] [CrossRef]
- Li, R.; Miao, C.Q.; Zhang, Y.L.; Wang, Y.C.; Chen, X.L. Corrosion characteristics and damage constitutive model of galvanized steel wires for bridge cables. Structures 2021, 34, 3414–3426. [Google Scholar] [CrossRef]
- Li, R.; Wang, H.; Miao, C.Q.; Yuan, Z.J.; Ni, Y. Evaluating Stress Corrosion Damage of Steel Wires as Bridge Cables Based on the Corrosion Surface. J. Mater. Civ. Eng. 2024, 36, 04024113. [Google Scholar] [CrossRef]
- Li, R.; Wang, H.; Yuan, Z.J.; Miao, C.Q.; Liu, Y.D. Evolution law of corrosion characteristics and time-dependent reliability analysis for service life prediction of corroded bridge cables. Eng. Struct. 2025, 327, 119667. [Google Scholar] [CrossRef]
- Miao, C.Q.; Yu, J.; Mei, M.X. Distribution law of corrosion pits on steel suspension wires for a tied Bridge. Anti-Corros. Methods Mater. 2016, 63, 166–170. [Google Scholar] [CrossRef]
- Vecchi, F.; Franceschini, L.; Tondolo, F.; Belletti, B.; Montero, J.S.; Minetola, P. Corrosion morphology of prestressing steel strands in naturally corroded PC beams. Constr. Build. Mater. 2021, 296, 123720. [Google Scholar] [CrossRef]
- Wang, L.; Li, T.; Dai, L.Z.; Chen, W.; Huang, K. Corrosion Morphology and Mechanical Behavior of Corroded Prestressing Strands. J. Adv. Concr. Technol. 2020, 18, 545–556. [Google Scholar] [CrossRef]
- Yu, X.R.; Yan, Z.T.; Zhang, G.F.; Liu, J.W. Comprehensive investigation and modeling of pitting corrosion effects on mechanical properties and fatigue performance of steel wires. Case Stud. Constr. Mater. 2024, 20, e02621. [Google Scholar] [CrossRef]
- Peng, W.; Fan, W.J. Experimental Study on Corrosion Development Model and Fatigue Performance of Strand Steel for Coastal Structures. J. Mar. Sci. Eng. 2023, 11, 665. [Google Scholar] [CrossRef]
- Yao, G.W.; Zeng, G.P.; He, X.B.; Wang, Q.L.; Wu, Y.F.; Jiang, E.G.; Zhu, Y.D.; Wang, B.C. Study on corrosion damage mechanisms and fatigue life of corroded steel wires in suspension bridge cable strands. Case Stud. Constr. Mater. 2025, 23, e05042. [Google Scholar] [CrossRef]
- Guo, Z.W.; Chen, H.L.; Yao, G.W. Bayesian prediction of the stress concentration effect on high-strength wires with corrosion pits. Eng. Fail. Anal. 2022, 131, 105827. [Google Scholar] [CrossRef]
- Zhang, H.P.; Liu, H.J.; Deng, Y.; Cao, Y.; He, Y.B.; Liu, Y.; Deng, Y. Fatigue behavior of high-strength steel wires considering coupled effect of multiple corrosion-pitting. Corros. Sci. 2025, 244, 112633. [Google Scholar] [CrossRef]
- Li, R.; Miao, C.Q.; Yu, J. Effect of characteristic parameters of pitting on strength and stress concentration factor of cable steel wire. Constr. Build. Mater. 2020, 240, 117915. [Google Scholar] [CrossRef]
- Li, R.; Miao, C.Q.; Zhuang, M.L. Experimental and Numerical Investigation of Stress Concentration Factor of Cable Steel Wire with Corrosion Pits. KSCE J. Civ. Eng. 2020, 24, 1581–1592. [Google Scholar] [CrossRef]
- Yu, X.R.; Yao, G.W.; Gu, L.F.; Fan, W.Q. Numerical and Experimental Study on the Steel Strands under the Coupling Effect of a Salt Spray Environment and Cyclic Loads. Materials 2020, 13, 736. [Google Scholar] [CrossRef]
- Fan, W.J.; Zhou, Y.Z.; Cheng, P.J.; Peng, W. Study on fatigue properties and life prediction of corroded steel strands. J. Constr. Steel Res. 2024, 217, 108636. [Google Scholar] [CrossRef]
- Deng, N.C.; Xu, J.; Zhu, G.C.; Han, Z.Q. Study on the Mechanical Properties of Corroded Steel Strands at Deflection Angles. Buildings 2023, 13, 795. [Google Scholar] [CrossRef]
- GB/T 10125-2012; Corrosion Tests in Artificial Atmospheres-Salt Spray Tests. Standards Press of China: Beijing, China, 2012.









| Chemical Component | NaCl | H2O | CuCl2·2H2O | CH3COOH |
|---|---|---|---|---|
| Content per 5 L | 250 g | 4718.7 mL | 1.3 g | 30 mL |
| Mass percentage | 5% | 94.37% | 0.03% | 0.60% |
| Test Conditions | Corrosion Durations (Days) | Number of Specimens | Corrosive Solution |
|---|---|---|---|
| C-0 | 0 | 3 | 5% NaCl solution with a mass fraction, and adjust its pH to 3.0~3.3 with acetic acid |
| C-1 | 45 | 3 | |
| C-2 | 75 | 3 | |
| C-3 | 105 | 3 | |
| C-4 | 135 | 3 | |
| C-5 | 165 | 3 |
| Test Conditions | Specimen No. | Mass of the Corroded Part (g) | Mass Loss Proportion η (%) | ||
|---|---|---|---|---|---|
| Before Corrosion m0 | After Corrosion m1 | Individual Value | Average Value | ||
| C-1 | SN-1 | 336.67 | 319.58 | 5.08 | 5.26% |
| SN-2 | 334.66 | 316.59 | 5.40 | ||
| SN-3 | 333.70 | 316.02 | 5.30 | ||
| C-2 | SN-4 | 335.72 | 306.04 | 8.84 | 10.12% |
| SN-5 | 337.32 | 298.67 | 11.46 | ||
| SN-6 | 333.42 | 299.84 | 10.07 | ||
| C-3 | SN-7 | 336.83 | 295.87 | 12.16 | 12.81% |
| SN-8 | 336.55 | 293.04 | 12.93 | ||
| SN-9 | 337.27 | 292.31 | 13.33 | ||
| C-4 | SN-10 | 335.48 | 288.75 | 13.93 | 14.05% |
| SN-11 | 336.07 | 288.26 | 14.23 | ||
| SN-12 | 335.12 | 288.27 | 13.98 | ||
| C-5 | SN-13 | 329.11 | 280.17 | 14.87 | 15.22% |
| SN-14 | 330.79 | 279.85 | 15.40 | ||
| SN-15 | 327.89 | 277.45 | 15.38 | ||
| Test Conditions | Number of Corrosion Pits | Fitting Gauss Model Parameters | Maximum Frequency (%) | RMSE (%) | ||
|---|---|---|---|---|---|---|
| Mean μ | Variance σ | R2 | ||||
| C-1 | 1875 | 0.40 | 0.19 | 0.82 | 19.80 | 5.59 |
| C-2 | 2052 | 0.40 | 0.27 | 0.98 | 20.95 | 7.00 |
| C-3 | 2254 | 0.56 | 0.25 | 0.93 | 20.50 | 5.86 |
| C-4 | 2452 | 0.66 | 0.38 | 0.97 | 19.58 | 6.16 |
| C-5 | 2569 | 0.65 | 0.33 | 0.94 | 21.00 | 6.67 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Ding, S.; Peng, X.; Xu, J.; Ding, D.; Sun, H.; Cai, S. Corrosion-Induced Stress Concentration Characteristics Study of Steel Strands in Bridge Cables Under Tension-Bending Coupling Loads. Materials 2026, 19, 646. https://doi.org/10.3390/ma19040646
Ding S, Peng X, Xu J, Ding D, Sun H, Cai S. Corrosion-Induced Stress Concentration Characteristics Study of Steel Strands in Bridge Cables Under Tension-Bending Coupling Loads. Materials. 2026; 19(4):646. https://doi.org/10.3390/ma19040646
Chicago/Turabian StyleDing, Shaoling, Xiyang Peng, Jian Xu, Dehao Ding, Huahuai Sun, and Shunyao Cai. 2026. "Corrosion-Induced Stress Concentration Characteristics Study of Steel Strands in Bridge Cables Under Tension-Bending Coupling Loads" Materials 19, no. 4: 646. https://doi.org/10.3390/ma19040646
APA StyleDing, S., Peng, X., Xu, J., Ding, D., Sun, H., & Cai, S. (2026). Corrosion-Induced Stress Concentration Characteristics Study of Steel Strands in Bridge Cables Under Tension-Bending Coupling Loads. Materials, 19(4), 646. https://doi.org/10.3390/ma19040646






