3.1. Structural and Morphological Characteristics
Before refinement, the crystal systems and space groups of each TiO
2 phase were defined as references. Anatase belongs to the tetragonal system (I4
1/AMD, No.141), and rutile also has a tetragonal structure (P4
2/mnm, No.136). Both are typical structural parameters of TiO
2 [
24,
25]. For Gd-doped samples, crystal system models of Gd-based impurities (e.g., Gd
2O
3 with cubic system Ia-3) were not introduced throughout the refinement process. Subsequent attempts to incorporate Gd
2O
3 into the fitting showed its diffraction peak contribution was <1%. It also increased the Rwp (weighted residual variance) from 7.2% to 10.5%, exceeding the literature-recognized standard of <8% [
26,
27]. This confirms the absence of independent Gd-based impurities in the samples, laying a model foundation for “lattice doping” rather than “phase segregation”.
Figure 2, showing quantitative Rietveld refinement analysis, revealed only two TiO
2 phases (anatase and rutile) in the Gd-doped sample. No Gd-based impurities such as Gd
2O
3 or Gd(OH)
3 were detected, with a fitted mass fraction of 0. Compared with the undoped sample, the anatase mass fraction increased from 78.3% to 89.6% after Gd doping. Meanwhile, the rutile proportion decreased from 21.7% to 10.4%. This indicates that the introduction of Gd
3+ inhibits the anatase-to-rutile phase transformation. It further confirms that Gd is incorporated into TiO
2 in the form of “lattice doping” rather than forming independent impurities to consume Gd sources.
Meanwhile, the lattice parameters, lattice volumes, and volume change rates of anatase and rutile TiO2 before and after Gd doping were obtained via XRD Rietveld refinement, respectively. For the anatase phase TiO2, the lattice parameters of the undoped sample were a = 3.785 ± 0.002 Å and c = 9.514 ± 0.003 Å, with a corresponding lattice volume of 339.2 ± 0.5 Å3. After Gd doping, the lattice parameter a increased to 3.798 ± 0.002 Å and c increased to 9.552 ± 0.003 Å. The lattice volume rose to 343.8 ± 0.5 Å3, with a volume change rate of +1.36%. For the rutile phase TiO2, the lattice parameters of the undoped sample were a = 4.594 ± 0.001 Å and c = 2.958 ± 0.001 Å, with a lattice volume of 62.4 ± 0.1 Å3. After Gd doping, the lattice parameter increased to 4.601 ± 0.001 Å and c increased to 2.965 ± 0.001 Å. The lattice volume increased to 62.8 ± 0.1 Å3, with a volume change rate of +0.64%.
The above results indicate that Gd doping can induce lattice expansion of TiO2, characterized by synchronous increases in lattice parameters a, c and lattice volume. This phenomenon can be attributed to the radius difference between doped and matrix ions. The ionic radius of Gd3+ (0.938 Å) is larger than that of Ti4+ (0.605 Å). When Gd3+ substitutes for Ti4+ sites in the lattice, it exerts a compressive effect on the surrounding lattice, leading to lattice distortion and volume expansion.
Figure 3 presents the macroscopic surface morphology of the TiO
2 and TiO
2-Gd coatings. The TiO
2-Gd sample exhibits a light blue, smooth surface (
Figure 3a), in contrast to the silver-white appearance of the undoped TiO
2 coating (
Figure 3b). This color variation is attributed to the optical properties inherent to the materials. Pure TiO
2 is a wide-bandgap semiconductor (≈3.2 eV for anatase), which primarily absorbs ultraviolet light (wavelength < ~387 nm) and thus appears transparent or silver-white in the visible region [
1,
6]. Although Gd
3+ ions are colorless, their incorporation into the TiO
2 lattice induces significant structural modifications [
28]. The substitution of Ti
4+ (ionic radius ≈ 0.605 Å) with larger Gd
3+ ions (ionic radius ≈ 0.94 Å) creates lattice distortion [
29]. To compensate for charge imbalance and alleviate structural strain, oxygen vacancies are generated [
30]. These vacancies form defect energy levels within the bandgap, which may facilitate weak absorption in the blue-light region, resulting in the observed faint yellowish-blue tint in the Gd-doped sample [
31].
Figure 4a and
Figure 4b present the surface morphologies of undoped and Gd-doped TiO
2 nanotube arrays, respectively. A clear comparison shows that the TiO
2-Gd arrays possess a rougher surface texture than their undoped counterpart. This increased roughness is expected to provide a larger specific surface area, thereby contributing to enhanced photocatalytic activity, This is basically consistent with the viewpoint mentioned in the literature [
32].
Figure 4c presents EDS elemental mapping and spectrum analysis of the TiO
2-Gd sample. The upper (red) map shows Ti (Ti Kα1) distribution, and the lower (green) map shows Gd (Gd Lα1) distribution, both with a 2.5 μm scale bar. Overlapping, agglomeration-free distributions confirm uniform Gd dispersion in the TiO
2 matrix at the micrometer scale. The right-side total spectrum displays only characteristic X-ray peaks of Ti and Gd, with no impurity interference, verifying the sample’s primary composition. Elemental content data (table) shows Ti at 93.14 wt% (98.77 at.%) as the matrix and Gd at 6.86 wt% (1.23 at.%) as the dopant, confirming low-concentration, uniform Gd doping. Collectively, these results confirm Gd is uniformly incorporated into the TiO
2 matrix without forming independent Gd-based impurities.
3.2. XPS Analysis
XPS analysis was performed to examine the oxidation states and chemical coordination in Gd-doped TiO
2.
Figure 5a displays characteristic peaks of Ti 2p, O 1s, C 1s, and notably Gd 4d, confirming the successful incorporation of Gd into the TiO
2 structure [
33]. Compared with pure TiO
2, the Ti 2p region in the doped sample exhibits altered peak intensity along with a slight binding-energy shift, indicating that Gd doping modifies the local chemical environment of Ti [
34]. The presence of Gd-related signals and the shift in Ti 2p collectively demonstrate effective doping and suggest changes in the electronic structure of TiO
2 induced by Gd [
11,
35]. Thus, the XPS survey provides direct evidence of Gd doping and establishes a basis for understanding its influence on the photocatalytic properties of TiO
2.
The Gd 4d XPS spectrum of the TiO
2-Gd composite is presented in
Figure 5b. Deconvolution of the spectrum reveals two primary contributions, centered at binding energies of approximately 148.38 eV and 142.68 eV, which are attributed to distinct electronic states of Gd [
36]. These features indicate that Gd adopts two different chemical environments or oxidation states within the TiO
2 matrix, likely arising from varied doping configurations such as differences in coordination geometry or local charge distribution [
31]. The presence of multiple Gd states is significant, as it can directly modulate the physical and chemical properties of the material, including its optical response and surface catalytic behavior [
37]. Furthermore, the incorporation of Gd introduces new electronic states associated with the Gd 4d orbitals, thereby modifying the host TiO
2 electronic structure [
11]. These induced states, located near the band edges, are expected to influence key processes such as photon absorption and the separation of photogenerated charge carriers [
38]. Collectively, the XPS analysis confirms the successful doping of Gd into TiO
2 and elucidates the specific chemical nature of the incorporated Gd species, providing insight into the structure–property relationships in this modified photocatalyst.
Figure 5c displays the Ti 2p XPS spectrum of pristine TiO
2. Following spectral deconvolution, the spectrum is resolved into three constituent peaks. Consistent with spin–orbit coupling, the Ti 2p orbital manifests as the characteristic doublet, comprising the Ti 2p
3/
2 and Ti 2p
1/
2 components, observed at binding energies of approximately 458.58 eV and 464.38 eV, respectively [
39]. An additional feature at approximately 471.48 eV is identified, which is typically ascribed to a satellite structure associated with the primary Ti 2p transitions [
36]. The peak positions and spectral line shape are characteristic of Ti
4+ in the TiO
2 lattice, aligning well with established reference data [
25]. In contrast, the Ti 2p spectrum of the Gd-doped sample (TiO
2-Gd) exhibits a systematic positive shift upon deconvolution, with corresponding peaks located at approximately 459.38 eV (Ti 2p
3/
2), 465.08 eV (Ti 2p
1/
2), and 472.28 eV (satellite). This uniform shift to higher binding energy signifies an altered chemical environment for Ti following Gd incorporation [
36]. The observed increase in Ti 2p binding energy is indicative of a reduced electron density around the Ti nuclei. This phenomenon can be attributed to the introduction of Gd, which, owing to its distinct electronic structure, engages in electronic interactions with the TiO
2 host, effectively withdrawing electron density from neighboring Ti atoms [
11]. Such a modification in the local electronic structure is a direct consequence of Gd integration into the TiO
2 lattice and its interaction with the Ti–O framework [
39]. The measurable shift in the Ti 2p core level thus provides direct evidence for the successful doping of Gd and its pivotal role in modulating the electronic properties of TiO
2, which is intrinsically linked to the material’s functional performance, such as its catalytic activity [
40].
Figure 5e shows the regulatory effect of Gd doping on oxygen defects. Gd substitutes for Ti sites in the lattice in the form of Gd
3+ (ionic radius of Gd
3+, 0.94 Å > 0.605 Å of Ti
4+). To balance the charge imbalance, oxygen vacancies (oxygen defects) need to be generated in the lattice for compensation. Through the comparison of peak areas, the ratio of oxygen defects to adsorbed oxygen increased from 29% to 38%. This change also laid a structural foundation for the performance optimization of the samples.
3.3. Optical Properties
The optical absorption properties of the synthesized composites were investigated by UV-Vis absorbance spectra, as presented in
Figure 6a,b. Pristine TiO
2 displays a fundamental absorption edge near 400 nm, exhibiting strong absorption in the UV region but negligible absorption at wavelengths longer than 400 nm. This characteristic is attributed to the intrinsic wide bandgap of TiO
2 (≈3.2 eV), which permits only the excitation of electrons by high-energy UV photons [
1,
6]. In contrast, a distinct redshift in the absorption edge and enhanced absorption in the visible-light range are observed for all rare-earth-doped samples. La-doped TiO
2 demonstrates moderate redshift and improved visible-light absorption, indicating a reduction in the effective bandgap due to La incorporation, which extends the photoresponse range [
41]. A more pronounced effect is seen for Ce-doped TiO
2, which exhibits a further redshift and stronger visible-light absorption, suggesting that Ce doping mediates a more substantial bandgap modification. Notably, Gd-doped TiO
2 presents the most significant optical alteration among the doped series. It possesses the broadest and most intense absorption profile across the entire visible spectrum (400–800 nm), implying that Gd doping induces the most effective bandgap narrowing and electronic structure modulation, thereby granting the composite superior visible-light harvesting capability. This enhanced visible-light absorption is of paramount importance for photocatalysis, as visible light constitutes a major portion of the solar spectrum [
42]. The extended photoresponse in these doped TiO
2 composites enables more efficient utilization of solar energy, which is anticipated to yield superior photocatalytic performance [
43]. The increased photon absorption promotes the generation of a greater number of photogenerated electron-hole pairs, consequently supplying more charge carriers to drive surface redox reactions [
44]. The optical band gaps of the samples were estimated via the Tauc relation based on the data from UV-Vis diffuse reflectance spectroscopy (DRS, diffuse reflectance mode) (
Figure 6c). As TiO
2 is an indirect band gap semiconductor, its Tauc relation requires modification in combination with the characteristics of diffuse reflectance data. The samples in this study were TiO
2 nanotube films grown on opaque titanium substrates, making the absorption coefficient calculation method in transmission mode inapplicable. Thus, the diffuse reflectance (R) was converted to an equivalent absorption parameter F(R) (i.e., Kubelka-Munk function value) using the Kubelka-Munk function, which can replace the absorption coefficient α for band gap analysis. The expression of the Kubelka-Munk function is as follows: F(R) = (1 − R)
2/2R, where R is the diffuse reflectance of the sample (directly measured by UV-Vis DRS), and F(R) reflects the combined effects of light absorption and scattering by the material.
The Tauc relation for indirect band gap semiconductors is modified to: (F(R)⋅hν)
1/2∝(hν − Eg), where hν is the photon energy (converted from the wavelength λ via hν = 1240/λ, unit: eV) and Eg is the optical band gap. A plot of (F(R)⋅hν)
1/2 versus hν was constructed (i.e., the Tauc plot in
Figure 6c). Tangents were drawn to the linear segments of the curve, and the hν value corresponding to the intersection of the two tangents was defined as the optical band gap of the sample: the band gap of pure TiO
2 was 2.89 eV, while that of Gd-doped TiO
2 (Gd-TiO
2) was narrowed to 2.46 eV.
This band gap narrowing is consistent with the enhanced visible-light absorption of Gd-TiO
2 in
Figure 6a. The reason is that Gd doping introduces impurity energy levels into the TiO
2 lattice, reducing the energy threshold for electron transitions. This structural change not only narrows the band gap but also boosts the material’s visible-light absorption capacity, laying a structural foundation for the visible-light-driven photocatalytic performance of Gd-TiO
2.
3.4. Photocatalytic Performance
An orthogonal experimental design was employed to identify appropriate electrolyte parameters. In this study, the concentration gradient of Gd(NO3)3·6H2O was set as 0 (not added), 0.01 mol/L, and 0.03 mol/L. This gradient design is based on the following scientific bases.
It serves as an experimental benchmark. It clearly distinguishes the differences in lattice structure, oxygen defect evolution, and final performance between doped and undoped samples. It is a core reference for verifying the regulatory effect of Gd doping. It can directly quantify the impact of doping behavior on the material. From the perspective of doping mechanism, the ionic radius of Gd
3+ (0.94 Å) is significantly larger than that of Ti
4+ (0.605 Å). Low-concentration doping can introduce controllable lattice distortion and oxygen vacancies. Meanwhile, it avoids the risk of lattice collapse or phase separation caused by high-concentration doping. From the perspective of academic relevance, 0.01 mol/L is a classic low-concentration range in TiO
2 rare earth doping research. It is highly consistent with the concentration range (0.001–0.05 mol/L) reported in existing studies [
16]. This facilitates the horizontal comparison of experimental results and academic exchanges. From the analysis of dosage effect, this concentration forms a continuous gradient with 0.01 mol/L. It allows systematic study of the correlation between doping concentration, lattice expansion, and oxygen defect ratio. It helps identify the doping concentration range with optimal performance. From the perspective of stability balance, the medium concentration can ensure sufficient Gd
3+ to replace Ti
4+ lattice sites. This effectively introduces oxygen vacancies. It also avoids the increase in carrier recombination centers caused by excessive defects. It is a key concentration for balancing defect regulation and lattice stability. From the perspective of synthesis feasibility, in the anodic oxidation synthesis system, the concentration of 0.03 mol/L can ensure the uniform dispersion of the precursor solution. It avoids local agglomeration or precipitation caused by excessively high concentration [
34]. This guarantees the uniformity of the samples.
This concentration gradient covers the typical range of “undoped—low doping- medium doping”. It can clearly reveal the structure-activity relationship between Gd doping concentration, material structure, defects, and performance. It provides a reliable experimental basis for the subsequent optimization of doping strategies. As detailed in
Table 1 and
Table 2, the photocatalytic activity was assessed by monitoring the photodegradation efficiency of methylene blue (MB) under simulated sunlight irradiation using a CEL-HXF300 photocatalytic reaction system. Compared to pristine TiO
2, the Gd-doped sample (TiO
2-Gd) exhibited markedly enhanced degradation performance. As depicted in
Figure 7d, the normalized concentration (Cₜ/C
0) of MB decreased rapidly with irradiation time. A particularly sharp decline was observed within the initial 15 min, followed by a continued rapid reduction. After 60 min of reaction, Cₜ/C
0 reached approximately 0.1, indicating significantly faster and more effective pollutant degradation by TiO
2-Gd [
44].
Consistent with the UV-Vis analysis, Gd doping extends the light absorption of TiO
2 into the visible region. This enhanced photon harvesting capability enables TiO
2-Gd to utilize a broader spectrum of solar energy, thereby generating a greater number of photogenerated electron–hole pairs [
45]. These charge carriers serve as active species that drive subsequent degradation reactions [
46]. Furthermore, the incorporation of Gd modifies the electronic structure of TiO
2, which facilitates the separation of photogenerated charges and suppresses their recombination. The increased availability of these electrons and holes significantly improves the overall efficiency of the redox processes involved in pollutant degradation [
47].
Surface morphology plays a critical role in catalytic performance. While smooth surfaces offer limited active area, rough surfaces characterized by nanoscale pores, steps, and edges provide a substantially increased specific surface area. This is consistent with the SEM results presented in the previous section. This enlarged surface offers more active sites for the adsorption of reactant molecules (e.g., pollutants and water), thereby establishing more venues for photocatalytic reactions and forming the physical basis for enhanced efficiency [
48].
Moreover, rough surfaces promote the “light-trapping effect.” In contrast to the specular reflection prevalent on smooth surfaces, which leads to considerable photon loss, rough surfaces induce multiple internal reflections and scattering of incident light [
49]. This phenomenon prolongs the optical path length within the material, markedly boosting photon absorption and utilization. Consequently, more electron–hole pairs are generated, directly amplifying the driving force for photocatalysis [
50]. (
Table 1 and
Table 2)
Figure 8a shows the variation in MB concentration over time in a dark environment. In the initial stage (0–20 min), the MB concentration decreases slightly due to adsorption on the catalyst surface. After 25 min, the curve levels off, with the C/C
0 value stabilizing at around 0.95 without significant fluctuations. This trend indicates that 30 min of dark stirring is sufficient for MB molecules to establish a dynamic adsorption–desorption equilibrium between the TiO
2-Gd catalyst surface and the solution. The impact of the adsorption process on MB concentration reaches a steady state. At equilibrium, the adsorption removal rate of MB by TiO
2-Gd is only about 5%, much lower than the 90% degradation rate after 60 min of photocatalytic reaction. This suggests that the significant decrease in MB concentration during the subsequent light irradiation stage is mainly attributed to photocatalytic degradation rather than the continuous contribution of adsorption. It eliminates the interference of unbalanced adsorption on the evaluation of photocatalytic activity.
As shown in
Figure 8b, throughout the 60 min light irradiation cycle, the C/C
0 value of the “light-only condition” curve remains above 0.98. No obvious decrease in MB concentration is observed, with a degradation rate of less than 2%. This result fully confirms that MB molecules exhibit excellent photostability under the simulated sunlight irradiation used in the experiment and do not undergo photodegradation on their own.
Figure 9a depicts the time-normalized concentration (C
t/C
0) profiles of methylene blue (MB) degradation over three consecutive cycles using Gd-doped TiO
2 (TiO
2-Gd) as the photocatalyst.
The degradation curves exhibit highly consistent trends across all three cycles: the MB concentration declines sharply within the initial 15 min of light irradiation, followed by a relatively slow and steady degradation stage. After 60 min of illumination, the C
t/C
0 value stabilizes at approximately 0.1 for each cycle, corresponding to a degradation efficiency of ~90%. Notably, no obvious upward shift in the curves is observed in subsequent cycles, indicating that the photocatalytic activity of TiO
2-Gd does not undergo significant decay after washing treatments, and its degradation kinetic behavior remains stable. This favorable stability can be ascribed to the homogeneous dispersion of Gd
3+ ions within the TiO
2 lattice via the in situ anodization process. Specifically, no discrete Gd-based crystalline phases are formed; instead, Gd ions establish strong chemical bonds with the Ti–O framework, which effectively inhibits the leaching or agglomeration of active components during cyclic testing [
36].
Figure 9b presents the histogram of MB degradation efficiency for TiO
2-Gd over three consecutive cycles, which intuitively quantifies the extent of performance attenuation during cyclic operation. Statistical results reveal that the degradation efficiency of TiO
2-Gd only decreases slightly from the initial 90% to 84% after three cycles, corresponding to a total attenuation of merely 6%. This result confirms the excellent cyclic stability of TiO
2-Gd. The underlying mechanism is associated with the defect energy levels and oxygen vacancies introduced by Gd doping: these structural modifications can continuously modulate the electronic structure of TiO
2, promote the separation of photogenerated electron–hole pairs, and ensure the sustained generation of hydroxyl radicals (·OH), the dominant active species responsible for MB degradation. Consequently, the attenuation of catalytic efficiency induced by the reduced yield of active species is effectively mitigated [
41].
Figure 10 compares the degradation performance of samples doped by the one-step method and the traditional impregnation method by tracking the variation trend of C
t/C
0.
As can be seen from the curve trends in
Figure 10, in the initial stage of the reaction, the C
t/C
0 value of the sample prepared by the one-step method decreases significantly faster than that of the sample prepared by the impregnation method. This indicates that the sample doped via the one-step method can exert its effect rapidly at the initial degradation stage and exhibits superior initial degradation efficiency.
Throughout the entire reaction cycle, the Ct/C0 value of the one-step method sample remains consistently lower than that of the impregnation method sample, and the gap between the two gradually widens with the passage of time. This result directly demonstrates that the degradation performance of the sample prepared by the one-step method is superior to that of the sample prepared by the traditional impregnation method during the whole reaction process, which also highlights the advantage of the one-step method in enhancing the degradation activity of the samples.
3.5. Simulation Calculations
Titanium dioxide (TiO
2), a widely studied semiconductor, exists primarily in three crystalline phases: brookite, rutile, and anatase. These phases exhibit distinct unit cell structures and bandgap energies, which fundamentally govern their physical and chemical properties [
36,
39]. To elucidate the mechanism by which Gd doping enhances the photocatalytic performance of TiO
2, computational simulations of these three phases were performed using Materials Studio [
23]. The calculations were based on the Perdew-Burke-Ernzerhof (PBE) exchange-correlation functional within the GGA framework. To address the intrinsic underestimation of bandgaps by pure GGA and properly describe the localized d/f orbitals, Hubbard U corrections were applied: U(Ti-3d) = 4.2 eV (a widely adopted value for anatase TiO
2 to match experimental structural and electronic properties [
36,
39]) and U(Gd-4f) = 6.0 eV (selected to account for the strong correlation of Gd-4f electrons, consistent with previous rare-earth doping studies [
28,
35]). The electron-ion interactions were described using ultrasoft pseudopotentials, with a cutoff energy of 400 eV set for the plane-wave basis set to ensure numerical convergence. For geometric optimization, the conjugate gradient algorithm was used, with convergence criteria set to 1 × 10
−5 eV per atom for total energy and 0.02 eV/Å for atomic forces. For electronic structure calculations, a Monkhorst-Pack k-point grid of 3 × 3 × 1 was sampled for the anatase TiO
2 unit cell (and corresponding supercell for Gd doping) to ensure sufficient sampling of the Brillouin zone [
51].
Figure 11a–d show the unit cells of four crystals, respectively. As illustrated in
Figure 11a–d, brookite crystallizes in the orthorhombic system, while both rutile and anatase adopt tetragonal structures [
36]. The calculated bandgap energies, derived from MS simulations and presented in
Figure 11e–g, are 2.390 eV for brookite, 1.853 eV for rutile, and 2.122 eV for anatase [
36]. Notably, following the construction of a supercell and the introduction of Gd, the bandgap of anatase TiO
2 decreased substantially to 0.278 eV (
Figure 11h). This significant reduction is attributed to the introduction of new energy states within the intrinsic bandgap of TiO
2 upon Gd doping, effectively narrowing the bandgap and inducing a redshift in the optical absorption edge [
11,
39]. Consequently, the material gains enhanced capacity to harvest visible light.
As reported by Reference [
11], the incorporation of Gd modifies the electron density distribution surrounding atoms within the TiO
2 lattice, thereby reducing the energy required for electron excitation from the valence band to the conduction band. Notably, this effect not only enhances the yield of photogenerated charge carriers but also accelerates their generation kinetics, which collectively contributes to improved photocatalytic efficiency. Furthermore, the impurity energy levels associated with Gd can act as trapping sites for either electrons or holes, facilitating the spatial separation of photogenerated electron–hole pairs while suppressing their recombination. Consequently, electrons localized at Gd-related states exhibit a substantially reduced probability of recombination with holes, enabling a greater number of charge carriers to migrate to the surface and participate in redox reactions [
38]. Structurally, Gd doping may also modify the local crystal arrangement and surface atomic configuration of TiO
2, creating additional defects such as oxygen vacancies and increasing the density of active sites. These sites often exhibit stronger affinity for pollutant molecules, improving adsorption on the catalyst surface [
48]. The enhanced adsorption increases the contact probability between target pollutants and photogenerated carriers, thereby accelerating degradation rates [
9].
As indicated by the EDS results presented above, the doping concentration of Gd atoms was approximately 1.23 at.%. We first optimized the three unit cells of pristine TiO2 (brookite, rutile, anatase) to obtain their ground-state structures. Subsequently, a 2 × 2 × 1 anatase supercell (containing 32 Ti atoms and 64 O atoms) was constructed for Gd doping. One Ti atom in the supercell was substituted with a Gd atom, corresponding to a doping concentration of approximately 1.04 at. The minor error ensures good agreement with the practical doping condition. The supercell was then re-optimized to relax the lattice distortion induced by the substitution of Ti4+ (ionic radius: 0.605 Å) with the larger-sized Gd3+ (ionic radius: 0.94 Å).
Despite these systematic deviations, the pronounced reduction in the calculated bandgap following Gd doping consistently indicates that the enhanced photocatalytic activity of TiO
2-Gd is closely correlated with effective bandgap narrowing [
19].
Notably, a clear discrepancy exists between first-principles calculated and experimentally measured band gaps, a common observation in semiconductor research. The key origins of this deviation are as follows.
First, DFT calculations with the GGA functional systematically underestimate semiconductor band gaps due to its approximate treatment of electron exchange-correlation energy. This leads to the calculated band gaps of undoped TiO2 (2.122 eV) and Gd-doped TiO2 (0.278 eV) being significantly lower than the experimental values of 2.89 eV and 2.46 eV, respectively. Furthermore, theoretical calculations rely on an idealized periodic supercell model assuming uniform dopant distribution and a defect-free lattice, whereas real samples contain oxygen vacancies, surface states, and grain boundaries. Local lattice distortion or Gd segregation induced by doping is also omitted in the model, contributing to electronic structure deviations. Finally, experimental band gaps derived from Tauc plot extrapolation are subject to errors from sample thickness, surface roughness, and diffuse scattering. Additionally, complex defect states (e.g., oxygen vacancies, hydroxyl groups) that introduce impurity levels in the band gap are simplified or neglected in calculations, further widening the theory-experiment gap.
Overall, this discrepancy arises from the inherent limitations of the GGA functional and the contrast between the idealized computational model and the structural complexity of real materials.
3.6. Reaction Mechanism
To further investigate the contribution of active species in the degradation process, active free radical trapping experiments were carried out. In the reaction system for the degradation of methylene blue (MB) solution by TiO
2-Gd under simulated sunlight, disodium ethylenediaminetetraacetate (EDTA-2Na), L-histidine, 2,2,6,6-tetramethylpiperidin-1-oxyl (TEMPO) and isopropanol (IPA) were added separately, which selectively quench photogenerated holes (h
+), singlet oxygen, superoxide radicals (·O
2−) and hydroxyl radicals (·OH), respectively. As shown in
Figure 12, the degradation rate of MB solution by TiO
2-Gd only decreased slightly after the addition of EDTA-2Na, L-histidine and TEMPO, indicating that h
+ and ·O
2− are not the primary active species in this photocatalytic process. In contrast, the photocatalytic degradation capacity was significantly reduced with the addition of IPA, which demonstrates that ·OH plays a pivotal role in the photocatalytic degradation process. This result is consistent with the high MB degradation efficiency observed in the oxygen-annealed TiO
2 nanotube system.
This aligns with the high MB degradation efficiency observed in oxygen-annealed TiO
2 nanotube systems. Specific reaction conditions (e.g., bias potential, electrolyte) were optimized to enhance active species generation, which is basically consistent with the viewpoint mentioned in the literature [
13].
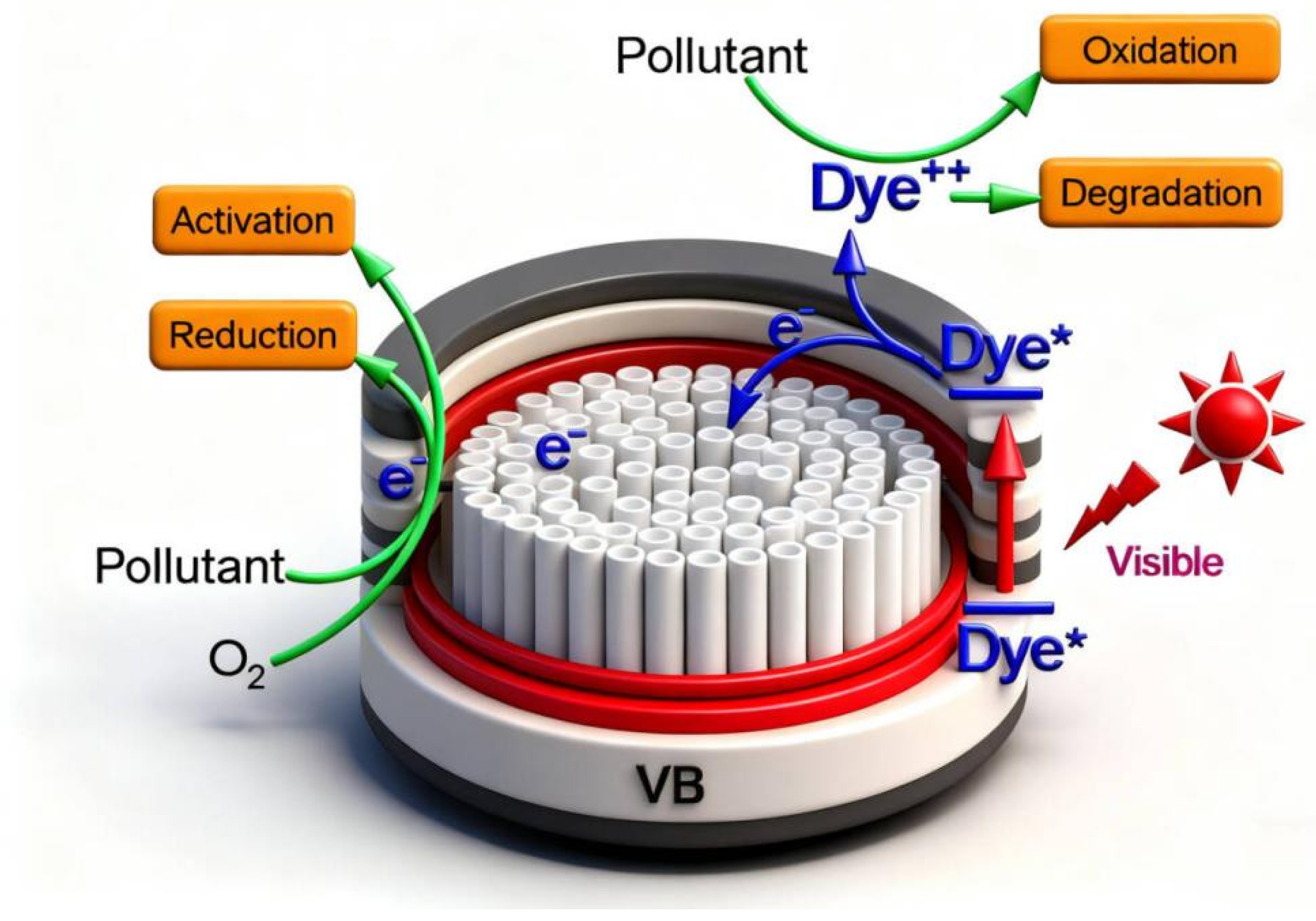
Based on this, a possible mechanism for the TiO
2-Gd photocatalyst was proposed, as illustrated in
Figure 13. The process of TiO
2 photocatalytic degradation of pollutants begins with its light absorption characteristics as a semiconductor. When incident photons have energy equal to or greater than the bandgap of TiO
2 (≈3.2 eV, corresponding to ultraviolet light) [
1,
6], electrons (e
−) in the valence band (VB) are excited to the conduction band (CB). Positively charged holes (h
+) are left behind in the valence band. This generates highly active electron–hole pairs (e
−-h
+ pair). This initiates the entire reaction, described by the equation [
47]:
The separation and migration of photogenerated electrons and holes are crucial for subsequent reactions. Separated carriers migrate to the surface of TiO
2 particles. During this process, most recombine and deactivate [
49]. Only a few successfully reach the surface to participate in reactions. So inhibiting recombination is key to improving photocatalytic efficiency [
46]. Holes (h
+) reaching the surface possess strong oxidizing ability. They can directly oxidize organic pollutants (R) adsorbed on the catalyst surface, degrading them: h
+ + R → R
+ → … → degradation products. However, the primary pathway involves holes reacting with water molecules (H
2O) or hydroxyl groups (OH
−) adsorbed on the catalyst surface. This generates hydroxyl radicals (·OH):
Simultaneously, electrons (e
−) in the conduction band have strong reducing ability. They combine with oxygen (O
2) molecules adsorbed on the TiO
2 surface [
2,
50]. This reduces them to superoxide radical anions (·O
2−):
The generated ·O
2− can be protonated under acidic conditions. It forms hydroperoxyl radicals (·HO
2), and ultimately converts into hydrogen peroxide (H
2O
2) and more hydroxyl radicals (·OH):
Thus, pollutant degradation is primarily accomplished through highly reactive free radicals (·OH) and a small amount of direct hole oxidation. Hydroxyl radicals can non-selectively attack chemical bonds in organic compounds. Through a series of complex chain reactions (e.g., decarboxylation, ring opening, bond breaking), they ultimately mineralize organics into carbon dioxide (CO
2), water (H
2O), and small inorganic ions [
8,
45]. The entire process of photocatalytic degradation is a complex redox reaction chain driven by light on the semiconductor surface. Its efficiency highly depends on the generation [
2,
8], separation, migration of photogenerated carriers, and the competition and synergy of surface reactions.
In summary, under light irradiation, TiO
2-Gd absorbs photons. Valence band electrons are excited to the conduction band, generating photogenerated electrons and holes. Due to Gd doping, the recombination of photogenerated electron–hole pairs is effectively suppressed [
11,
38]. More photogenerated holes can migrate to the surface of TiO
2-Gd. These photogenerated holes have strong oxidizing ability. On one hand, they can directly oxidize methylene blue (MB) molecules adsorbed on the catalyst surface [
1,
6]. On the other hand, they react with hydroxyl groups or water molecules on the TiO
2-Gd surface, generating a large number of hydroxyl radicals [
46]. The significant decline in degradation capability after adding isopropanol confirms that ·OH is the key active species in photocatalytic degradation of MB. It can decompose MB molecules into harmless small molecules through strong oxidation. Although photogenerated electrons also react with oxygen to form superoxide anion radicals, the degradation rate only slightly decreased after adding BQ. This indicates that superoxide radicals are not the primary active species. While photogenerated holes have direct oxidizing effects, the degradation rate only slightly decreased after adding EDTA-2Na and L-histidine. This suggests that direct electron–hole interactions are relatively minor [
45]. Therefore, the photocatalytic degradation of MB by TiO
2-Gd is mainly achieved through the strong oxidation of hydroxyl radicals induced by photogenerated holes. Gd doping significantly enhances photocatalytic performance by optimizing carrier separation and promoting the generation of hydroxyl radicals [
12,
31].