Radiation-Induced Synthesis of Asymmetric Porous PVDF-g-PIL Membranes via β-Cyclodextrin Leaching for Vanadium Redox Flow Battery
Abstract
1. Introduction
2. Experimental
2.1. Materials
2.2. Preparation of PVDF-IL Powder
2.3. Preparation of PVDF-IL Porous Membranes
2.4. Characterization
2.5. Ion Exchange Capacity, Porosity, Water Uptake, Swelling Ratio and Ion Conductivity of the PVDF-IL-CD
2.6. Vanadium Permeability
2.7. Antioxidant Testing
3. Results and Discussion
3.1. Grafting Pattern of IL on PVDF
3.2. Characterization of the Prepared PVDF-IL-CD IEM
3.3. Mechanical Characteristics
3.4. Porosity, Water Uptake and Swelling Ratio
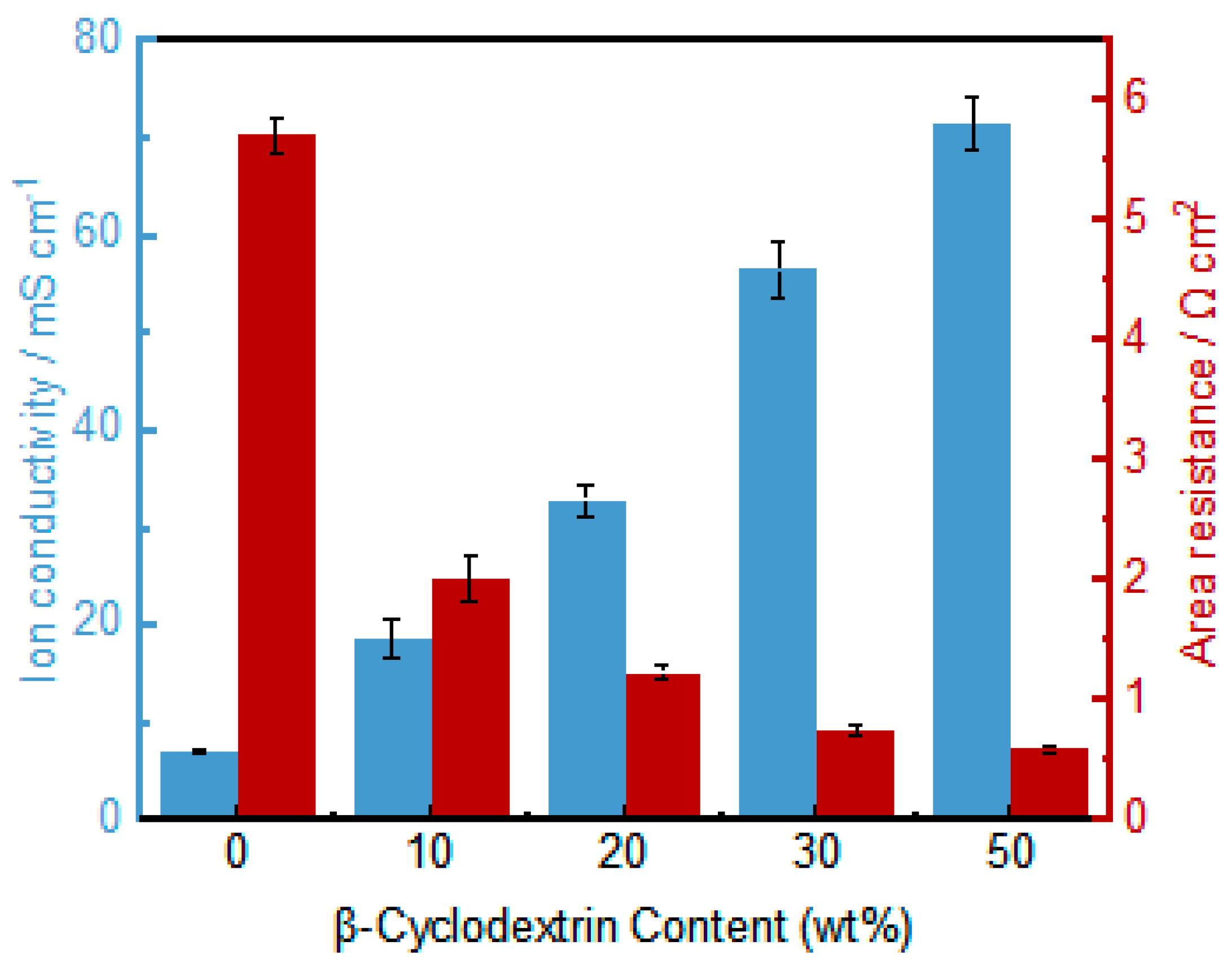
3.5. Ion Exchange Capacity and Conductivity Testing of Asymmetric Porous PVDF-IL Membranes
3.6. Permeability of Vanadium Ions
3.7. Antioxidant Properties
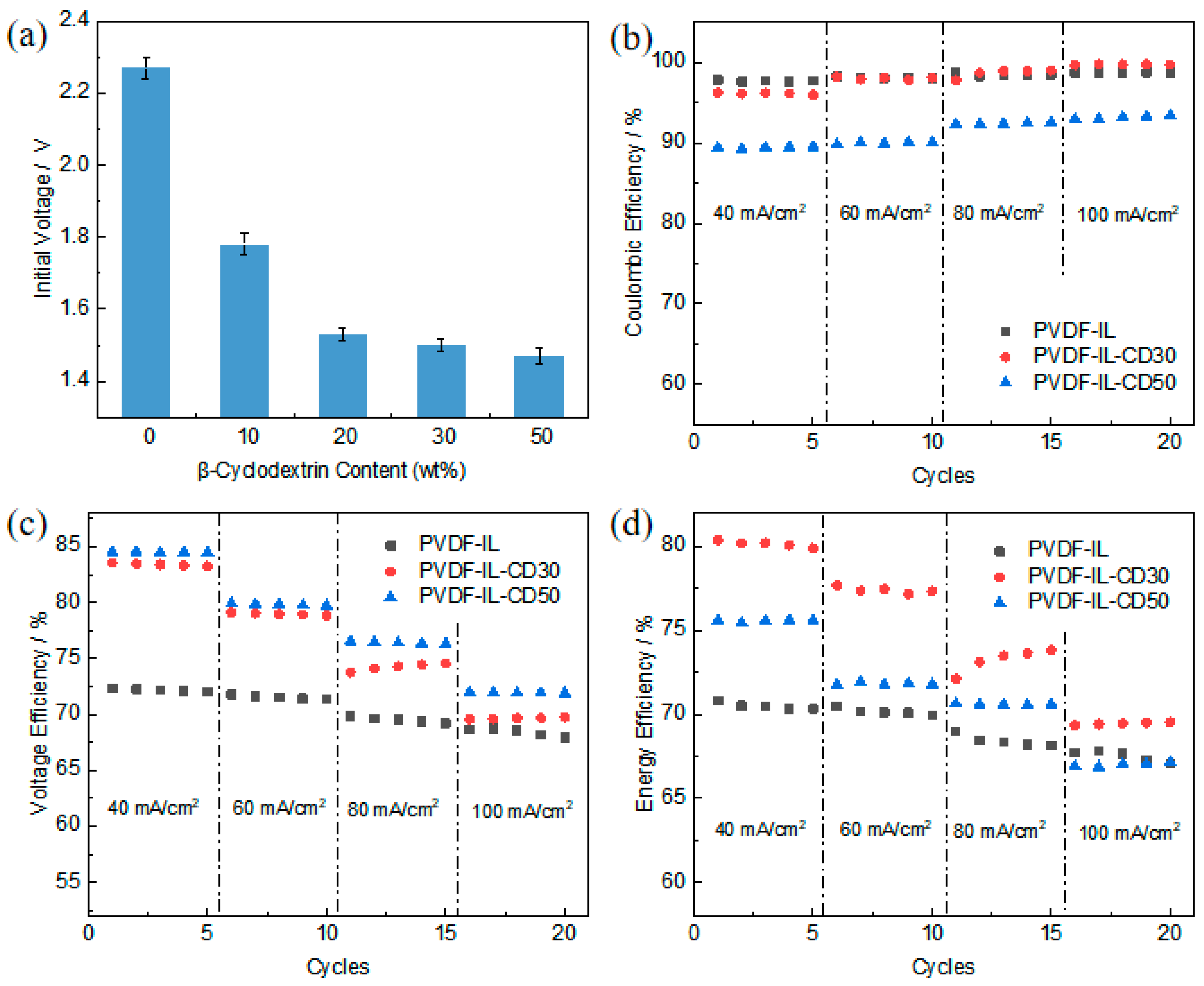
3.8. Single-Cell Testing of Asymmetric Porous PVDF-IL Separators
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Ge, G.; Zhang, C.; Li, X. Multi-Electron Transfer Electrode Materials for High-Energy-Density Flow Batteries. Next Energy 2023, 1, 100043. [Google Scholar] [CrossRef]
- Lu, Y.; Wang, C.; Li, J. Covalent Organic Frameworks Mixed-matrix Membrane Fabricated via Hot-rolling for Vanadium Redox Flow Battery. Chem. Eng. J. 2025, 525, 170709. [Google Scholar] [CrossRef]
- Bamati, N.; Raoofi, A. Development Level and the Impact of Technological Factor on Renewable Energy Production. Renew. Energy 2020, 151, 946–955. [Google Scholar] [CrossRef]
- Aramendia, I.; Fernandez-Gamiz, U.; Martinez-San-vicente, A.; Zulueta, E.; Lopez-Guede, J.M. Vanadium Redox Flow Batteries: A Review Oriented to Fluid-Dynamic Optimization. Energies 2021, 14, 176. [Google Scholar] [CrossRef]
- Arevalo-Cid, P.; Dias, P.; Mendes, A.; Azevedo, J. Redox Flow Batteries: A New Frontier on Energy Storage. Sustain. Energy Fuels 2021, 5, 5366–5419. [Google Scholar] [CrossRef]
- Liu, A.; Chen, K.; Li, J.; Ren, Q.; Meng, X.; Shen, M.; Wang, C. Highly ion-selective sulfonated poly(biphenyl pyridinium) zwitterionic membranes for vanadium redox flow batteries. J. Memb. Sci. 2025, 733, 124315. [Google Scholar] [CrossRef]
- Jiang, K.; Zhang, B.; Liu, Q.; Sun, Z.; Yang, C.; Pang, X.; Zhang, X.; Li, T.; Yang, Y.; Zhang, S.; et al. Selective Swelling-Induced Tricyclohexylphosphine-Functionalized Adamantane-Containing Poly(aryl ether ketone) Membranes for Vanadium Redox Flow Batteries. ACS Appl. Energy Mater. 2025, 8, 18078–18088. [Google Scholar] [CrossRef]
- Thiam, B.G.; Vaudreuil, S. Review—Recent Membranes for Vanadium Redox Flow Batteries. J. Electrochem. Soc. 2021, 168, 070553. [Google Scholar] [CrossRef]
- MacHado, C.A.; Brown, G.O.; Yang, R.; Hopkins, T.E.; Pribyl, J.G.; Epps, T.H. Redox Flow Battery Membranes: Improving Battery Performance by Leveraging Structure-Property Relationships. ACS Energy Lett. 2021, 6, 158–176. [Google Scholar] [CrossRef]
- Tempelman, C.H.L.; Jacobs, J.F.; Balzer, R.M.; Degirmenci, V. Membranes for All Vanadium Redox Flow Batteries. J. Energy Storage 2020, 32. [Google Scholar] [CrossRef]
- Jung, M.; Lee, W.; Nambi Krishnan, N.; Kim, S.; Gupta, G.; Komsiyska, L.; Harms, C.; Kwon, Y.; Henkensmeier, D. Porous-Nafion/PBI Composite Membranes and Nafion/PBI Blend Membranes for Vanadium Redox Flow Batteries. Appl. Surf. Sci. 2018, 450, 301–311. [Google Scholar] [CrossRef]
- Chai, S.; He, S.; Li, Z.; Zhu, Y.; Li, H. Supramolecular modification of Nafion ion channels for high-performance vanadium redox flow batteries. Chem. Eng. J. 2025, 514, 163174. [Google Scholar] [CrossRef]
- Gubler, L. Membranes and Separators for Redox Flow Batteries. Curr. Opin. Electrochem. 2019, 18, 31–36. [Google Scholar] [CrossRef]
- Ikhsan, M.M.; Abbas, S.; Do, X.H.; Ha, H.Y.; Azizi, K.; Henkensmeier, D. Sulfonated Polystyrene/Polybenzimidazole Bilayer Membranes for Vanadium Redox Flow Batteries. Adv. Energy Mater. 2025, 15, 2400139. [Google Scholar] [CrossRef]
- Suhag, S.; Kumar, P.; Mandal, J.R.; Kulshrestha, V.; Shahi, V.K. A New Strategy for Crosslinking of PVDF-Co-HFP by Grafting of High Molality of Amine Groups via Side Carbon Chain Tuned AEMs for VRFB. J. Energy Storage 2025, 128, 117246. [Google Scholar] [CrossRef]
- Xiao, Z.; Yuan, S.; Wu, J.; Mao, D.; Li, X. Influence of Typical Cation Structures of the Functionalized Poly(Phenylene Ether)s as Anion Exchange Membranes in Vanadium Redox Flow Batteries. ACS Appl. Energy Mater. 2025, 8, 5174–5189. [Google Scholar] [CrossRef]
- Xing, Y.; Geng, K.; Chu, X.; Wang, C.; Liu, L.; Li, N. Chemically Stable Anion Exchange Membranes Based on C2-Protected Imidazolium Cations for Vanadium Flow Battery. J. Memb. Sci. 2021, 618, 118696. [Google Scholar] [CrossRef]
- Chen, Y.; Li, Y.; Xu, J.; Chen, S.; Chen, D. Densely Quaternized Fluorinated Poly(Fluorenyl Ether)s with Excellent Conductivity and Stability for Vanadium Redox Flow Batteries. ACS Appl. Mater. Interfaces 2021, 13, 18923–18933. [Google Scholar] [CrossRef]
- Zhang, D.; Su, L.; Peng, S.; Pang, B.; Shi, D.; Zhao, L.; Meng, T.; Guo, X.; Dai, Y.; Yan, X.; et al. Ultra-thin Spin-coated Polybenzimidazole/Nafion Composite Membranes for All-vanadium Redox Flow Batteries. J. Memb. Sci. 2026, 738, 124856. [Google Scholar] [CrossRef]
- Liu, F.; Wang, S.; Li, J.; Tian, X.; Wang, X.; Chen, H.; Wang, Z. Polybenzimidazole/Ionic-Liquid-Functional Silica Composite Membranes with Improved Proton Conductivity for High Temperature Proton Exchange Membrane Fuel Cells. J. Memb. Sci. 2017, 541, 492–499. [Google Scholar] [CrossRef]
- Lu, D.; Wen, L.; Nie, F.; Xue, L. Synthesis and Investigation of Imidazolium Functionalized Poly(Arylene Ether Sulfone)s as Anion Exchange Membranes for All-Vanadium Redox Flow Batteries. RSC Adv. 2016, 6, 6029–6037. [Google Scholar] [CrossRef]
- Upadhyay, P.; Sharma, J.; Kulshrestha, V. Advancing Polybenzimidazole Membrane by Knitting Soft Poly(vinyl imidazolium) for Vanadium Redox Flow Batteries. Batter. Supercaps 2025, e202500714. [Google Scholar] [CrossRef]
- Yang, J.; Li, Q.; Cleemann, L.N.; Jensen, J.O.; Pan, C.; Bjerrum, N.J.; He, R. Crosslinked Hexafluoropropylidene Polybenzimidazole Membranes with Chloromethyl Polysulfone for Fuel Cell Applications. Adv. Energy Mater. 2013, 3, 622–630. [Google Scholar] [CrossRef]
- Zhou, J.; Liu, Y.; Zuo, P.; Li, Y.; Dong, Y.; Wu, L.; Yang, Z.; Xu, T. Highly Conductive and Vanadium Sieving Microporous Tröger’s Base Membranes for Vanadium Redox Flow Battery. J. Memb. Sci. 2021, 620, 118832. [Google Scholar] [CrossRef]
- Devrim, Y.; Bulanık Durmuş, G.N. Composite Membrane by Incorporating Sulfonated Graphene Oxide in Polybenzimidazole for High Temperature Proton Exchange Membrane Fuel Cells. Int. J. Hydrogen Energy 2022, 47, 9004–9017. [Google Scholar] [CrossRef]
- Sizov, V.E.; Kondratenko, M.S.; Gallyamov, M.O. Ion Transport Properties of Porous Polybenzimidazole Membranes for Vanadium Redox Flow Batteries Obtained via Supercritical Drying of Swollen Polymer Films. J. Appl. Polym. Sci. 2018, 135, 46262. [Google Scholar] [CrossRef]
- Luo, T.; David, O.; Gendel, Y.; Wessling, M. Porous Poly(Benzimidazole) Membrane for All Vanadium Redox Flow Battery. J. Power Sources 2016, 312, 45–54. [Google Scholar] [CrossRef]
- Wang, G.; Wang, L.; You, J.; Yang, Y.; Wang, Y.; Wang, W.; Liu, L.; Huang, J.; Li, Y.; Yu, W.; et al. Preparation of Hollow Silica/PTFE Fiber Membrane with Excellent Thermal Insulation Performance by Electrospinning. Appl. Therm. Eng. 2024, 255, 123959. [Google Scholar] [CrossRef]
- Matsuyama, H.; Maki, T.; Teramoto, M.; Kobayashi, K. Effect of PVP Additive on Porous Polysulfone Membrane Formation by Immersion Precipitation Method. Sep. Sci. Technol. 2003, 38, 3449–3458. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, H.; Li, X.; Zhang, H.; Wei, W. Porous Poly (Ether Sulfone) Membranes with Tunable Morphology: Fabrication and Their Application for Vanadium Flow Battery. J. Power Sources 2013, 233, 202–208. [Google Scholar] [CrossRef]
- Wang, Z.; Jiang, J.; Dong, Z.; Song, Y.; Zhao, L. Radiation Synthesis of Imidazolium Ionic Liquid Grafted PVDF as the Anion Exchange Membrane for Vanadium Redox Flow Batteries. New J. Chem. 2023, 47, 8013–8021. [Google Scholar] [CrossRef]
- Wang, Z.; Song, Y.; Jiang, J.; Zhao, W.; Zhang, M.; Dong, Z.; Zhao, L. Promoting Ion Conductivity of Imidazolium Grafted PVDF Membranes through Blending PVP for Vanadium Redox Flow Batteries. Ionics 2024, 30, 901–911. [Google Scholar] [CrossRef]
- Paramasivaganesh, K.; Govindasamy, C.; Shanmugasundaram, E.; Krishnan, N.K.; Saravanan, C.; Thanusu, J.; Thambusamy, S. Structural and Antibacterial Assessment of Two Distinct Dihydroxy Biphenyls Encapsulated with β-Cyclodextrin Supramolecular Complex. J. Mol. Struct. 2025, 1320, 139701. [Google Scholar] [CrossRef]






| Thickness/μm | Porosity/% | Water Uptake/% | Swelling Ratio/% | |
|---|---|---|---|---|
| Nafion 115 | 130 | - | 25.96 ± 1.48 | 10.06 ± 1.44 |
| PVDF-IL | 58 | 13.41 ± 1.13 | 7.76 ± 0.71 | 3.33 ± 0.55 |
| PVDF-IL-CD10 | 55 | 27.61 ± 1.54 | 15.95 ± 0.85 | 5.45 ± 0.34 |
| PVDF-IL-CD20 | 59 | 28.51 ± 1.32 | 17.68 ± 1.04 | 7.68 ± 0.81 |
| PVDF-IL-CD30 | 62 | 32.44 ± 1.44 | 26.89 ± 1.42 | 14.70 ± 1.26 |
| PVDF-IL-CD50 | 62 | 40.24 ± 1.63 | 31.79 ± 1.11 | 17.80 ± 1.31 |
| Membrane | Sfm (%) |
|---|---|
| PVDF-IL | 0.96 ± 0.04 |
| PVDF-IL-CD10 | 1.35 ± 0.08 |
| PVDF-IL-CD20 | 1.72 ± 0.11 |
| PVDF-IL-CD30 | 2.88 ± 0.09 |
| PVDF-IL-CD50 | 3.36 ± 0.15 |
| Nafion 115 | 0.78 ± 0.05 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Yu, J.; Li, W.; Niu, W.; Zhang, M.; Bai, J.; Li, P.; Wang, L.; Cui, Y.; Cui, S.; Que, X.; et al. Radiation-Induced Synthesis of Asymmetric Porous PVDF-g-PIL Membranes via β-Cyclodextrin Leaching for Vanadium Redox Flow Battery. Materials 2026, 19, 583. https://doi.org/10.3390/ma19030583
Yu J, Li W, Niu W, Zhang M, Bai J, Li P, Wang L, Cui Y, Cui S, Que X, et al. Radiation-Induced Synthesis of Asymmetric Porous PVDF-g-PIL Membranes via β-Cyclodextrin Leaching for Vanadium Redox Flow Battery. Materials. 2026; 19(3):583. https://doi.org/10.3390/ma19030583
Chicago/Turabian StyleYu, Jiangtao, Wenkang Li, Wei Niu, Manman Zhang, Junqing Bai, Pengtao Li, Liang Wang, Yuqing Cui, Shuanfang Cui, Xueyan Que, and et al. 2026. "Radiation-Induced Synthesis of Asymmetric Porous PVDF-g-PIL Membranes via β-Cyclodextrin Leaching for Vanadium Redox Flow Battery" Materials 19, no. 3: 583. https://doi.org/10.3390/ma19030583
APA StyleYu, J., Li, W., Niu, W., Zhang, M., Bai, J., Li, P., Wang, L., Cui, Y., Cui, S., Que, X., Ma, J., & Zhao, L. (2026). Radiation-Induced Synthesis of Asymmetric Porous PVDF-g-PIL Membranes via β-Cyclodextrin Leaching for Vanadium Redox Flow Battery. Materials, 19(3), 583. https://doi.org/10.3390/ma19030583






