Concrete Incorporating a Spent CO2 Absorbent: Comprehensive Assessment of Microstructure, Strength, and Durability
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.1.1. Binder
2.1.2. Spent CO2 Absorbent
2.1.3. ISO Graded Standard Sand
2.1.4. Aggregate
2.1.5. Admixture
2.2. Mix Proportion and Preparation of Mortar Test Specimens
2.2.1. Mix Proportion of Mortar
2.2.2. Preparation of Mortar Specimens
2.2.3. Microstructural and Chemical Characterization
2.2.4. Mortar Test Method
2.3. Mix Proportion and Preparation of Concrete Test Specimens
2.3.1. Mix Proportion of Concrete
2.3.2. Preparation of Concrete Specimens
2.3.3. Compressive and Flexural Strength Test
2.3.4. Flexural Strength Test
2.3.5. Freezing–Thawing Test
2.3.6. Chemical Resistance Test
2.3.7. Accelerated Carbonation Test
3. Experiment Results and Analysis
3.1. SEM Result
3.2. XRD Result
3.3. TGA Result
3.4. Mortar Strength Test Result
3.5. Concrete Strength Test Result
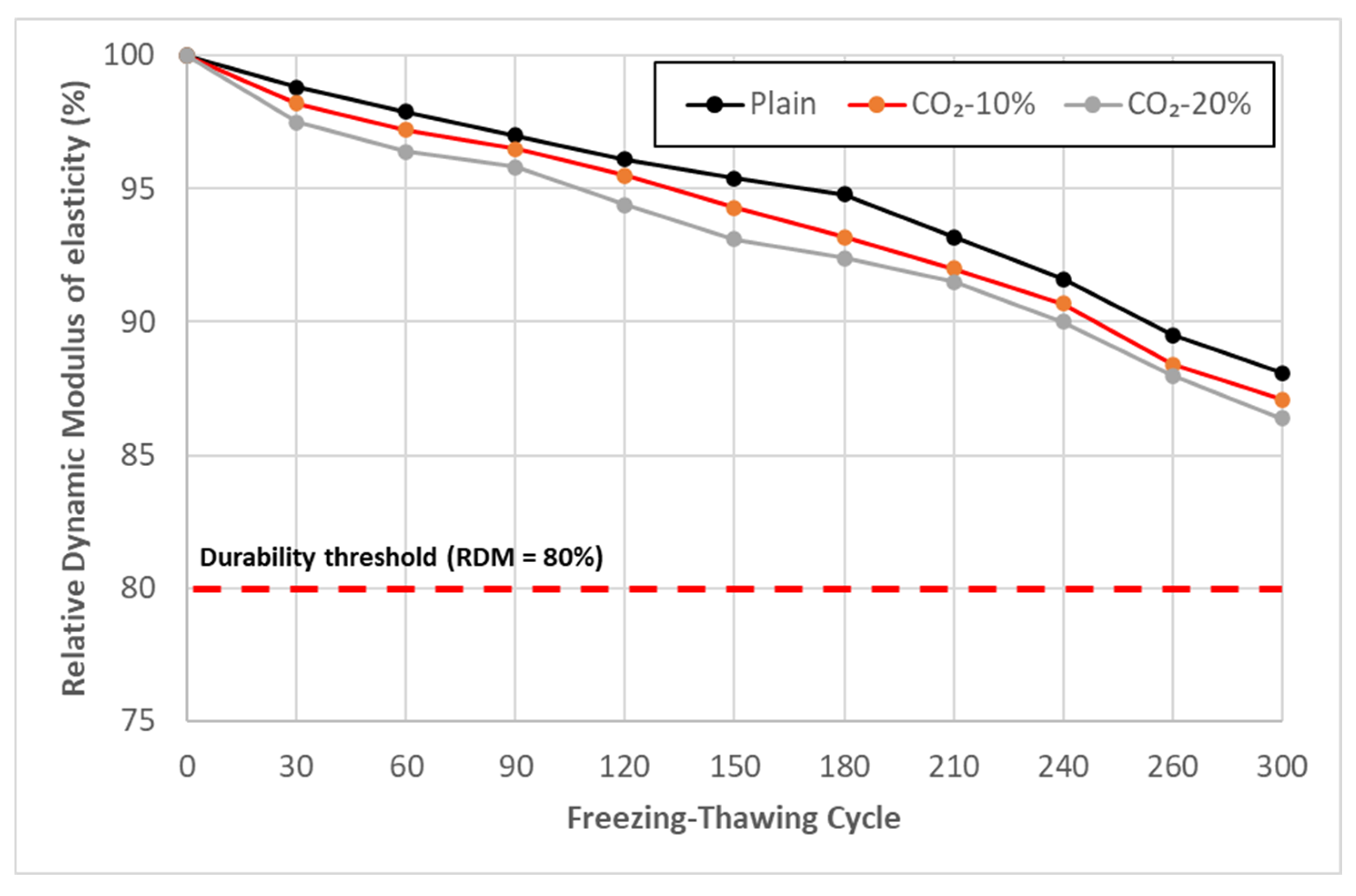
3.6. Freezing–Thawing Test Result
3.7. Chemical Resistance Test Result
3.8. Accelerated Carbonation Test Result
4. Conclusions
- (1)
- CO2 absorbent replacement was observed to be associated with changes in the microstructure, including increased CaCO3 formation due to carbonation and partial decalcification of the C–S–H gel. At replacement ratios of 10–20%, CaCO3-related micro-filling and nucleation effects were found to coincide with a relatively dense pore structure. In contrast, at replacement levels of 30% or higher, SEM observations indicated a higher presence of localized micro-pores and reduced apparent continuity of the gel structure.
- (2)
- Strength evaluation showed that CO2 absorbent replacement at 10–20% was associated with enhanced early-age strength compared to the Plain mixture. This behavior may be related to carbonate-based nucleation effects and the activation of GGBS in the presence of residual alkalinity. Conversely, replacement levels of 30% or higher were accompanied by reductions in compressive and flexural strength, which may be associated with carbonation-related microstructural changes, including partial C–S–H decalcification.
- (3)
- In the durability assessment, CO2-replaced concrete exhibited freezing–thawing resistance comparable to that of the Plain mixture. Accelerated carbonation tests showed a tendency for carbonation resistance to decrease with increasing CO2 replacement ratio; however, at replacement levels of 10–20%, the carbonation depth remained within a range similar to that of Plain. These results suggest that, within this replacement range, durability performance was not critically compromised under the conditions investigated.
- (4)
- Chemical resistance to strong acid solutions (5% HCl and 5% H2SO4) was slightly lower for CO2-replaced concrete compared to Plain. This tendency may be associated with acid dissolution reactions involving CaCO3, as well as the formation of soluble salts such as CaCl2, CaSO4, NaCl, and Na2SO4 originating from carbonation products and Na+ ions in the spent absorbent. Nevertheless, the increase in mass loss remained within a limited range, and mixtures with 10–20% replacement maintained a practically acceptable level of chemical resistance.
- (5)
- Overall, the experimental results indicate that the spent CO2 absorbent can be applied to cementitious materials within a 10–20% replacement range without significant deterioration in mechanical or durability performance. This replacement range may offer potential benefits, including improved early-age strength, pore structure refinement, and the incorporation of carbonated products. While the present findings demonstrate the feasibility of recycling spent DAC absorbents into cementitious materials, further studies are required to quantitatively verify CO2 storage capacity and to evaluate long-term durability under field-relevant exposure conditions.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Hickmann, T.; Widerberg, O.; Lederer, M.; Pattberg, P. The United Nations Framework Convention on Climate Change Secretariat as an Orchestrator in Global Climate Policymaking. Int. Rev. Adm. Sci. 2021, 87, 21–38. [Google Scholar] [CrossRef]
- Falkner, R. The Paris Agreement and the New Logic of International Climate Politics. Int. Aff. 2016, 92, 1107–1125. [Google Scholar] [CrossRef]
- Mon, M.T.; Tansuchat, R.; Yamaka, W. CCUS Technology and Carbon Emissions: Evidence from the United States. Energies 2024, 17, 1748. [Google Scholar] [CrossRef]
- Vishal, V.; Chandra, D.; Singh, U.; Verma, Y. Understanding Initial Opportunities and Key Challenges for CCUS Deployment in India at Scale. Resour. Conserv. Recycl. 2021, 175, 105829. [Google Scholar] [CrossRef]
- Boot-Handford, M.E.; Abanades, J.C.; Anthony, E.J.; Blunt, M.J.; Brandani, S.; Mac Dowell, N.; Fennell, P.S. Carbon Capture and Storage Update. Energy Environ. Sci. 2014, 7, 130–189. [Google Scholar] [CrossRef]
- Bui, M.; Adjiman, C.S.; Bardow, A.; Anthony, E.J.; Boston, A.; Brown, S.; Fennell, P.S.; Fuss, S.; Galindo, A.; Hackett, L.A.; et al. Carbon Capture and Storage (CCS): The Way Forward. Energy Environ. Sci. 2018, 11, 1062–1176. [Google Scholar] [CrossRef]
- Sanz-Pérez, E.S.; Murdock, C.R.; Didas, S.A.; Jones, C.W. Direct Capture of CO2 from Ambient Air. Chem. Rev. 2016, 116, 11840–11876. [Google Scholar] [CrossRef] [PubMed]
- Erans, M.; Sanz-Pérez, E.S.; Hanak, D.P.; Clulow, Z.; Reiner, D.M.; Mutch, G.A. Direct Air Capture: Process Technology, Techno-Economic and Socio-Political Challenges. Energy Environ. Sci. 2022, 15, 1360–1405. [Google Scholar] [CrossRef]
- Kasturi, A.; Gug Jang, G.; Dona-Tella Akin, A.; Jackson, A.; Jun, J.; Stamberga, D.; Custelcean, R.; Sholl, D.S.; Yiacoumi, S.; Tsouris, C. AnEffective Air–Liquid Contactor for CO2 Direct Air Capture Using Aqueous Solvents. Sep. Purif. Technol. 2023, 324, 124398. [Google Scholar] [CrossRef]
- Custelcean, R. Direct Air Capture of CO2 Using Solvents. Annu. Rev. Chem. Biomol. Eng. 2022, 13, 217–234. [Google Scholar] [CrossRef]
- Rackley, S.A. Carbon Capture and Storage; Butterworth-Heinemann: Oxford, UK, 2017. [Google Scholar]
- Mostafa, M.; Antonicelli, C.; Varela, C.; Barletta, D.; Zondervan, E. Capturing CO2 from the Atmosphere: Design and Analysis of a Large-Scale DAC Facility. Carbon Capture Sci. Technol. 2022, 4, 100060. [Google Scholar] [CrossRef]
- Abanades, J.C.; Anthony, E.J.; Wang, J.; Oakey, J.E. Fluidized Bed Combustion Systems Integrating CO2 Capture with CaO. Environ. Sci. Technol. 2005, 39, 2861–2866. [Google Scholar] [CrossRef] [PubMed]
- Keith, D.W.; Holmes, G.; Angelo, D.S.; Heidel, K. A Process for Capturing CO2 from the Atmosphere. Joule 2018, 2, 1573–1594. [Google Scholar] [CrossRef] [PubMed]
- Kaleeckal, B.V.; Gioja, L.; OBrien, K.; Giardinella, S.; Echeto, M.; Prause, S.; Baroi, C.; Larimore, R.; Kenny, M.; Baumeister, A.; et al. Direct Air Capture and Utilization System (DACUS): FEED Study of CarbonCapture Inc. DAC and CarbonCure Utilization Technologies Using United States Steel’s Gary Works Plant Waste Heat. In Proceedings of the 17th Greenhouse Gas Control Technologies Conference (GHGT-17), Zurich, Switzerland, 20–24 October 2024. [Google Scholar] [CrossRef]
- ASTM C150; Standard Specification for Portland Cement. ASTM International: West Conshohocken, PA, USA, 2022.
- Kim, Y.T.; Lee, J.W.; Yang, S.L. Influence of Nano SiO2/Al2O3-TiO2 powder on strength and durability properties of concrete. KSCE J. Civ. Eng. 2026, 30, 100462. [Google Scholar] [CrossRef]
- KS L ISO 679; Methods of Testing Cements—Determination of Strength. Korean Agency for Technology and Standards (KATS): Eumseong County, Republic of Korea, 2022.
- ISO 679; Methods of Testing Cements—Determination of Strength. International Organization for Standardization (ISO): Geneva, Switzerland, 2009.
- ASTM C1437; Standard Test Method for Flow of Hydraulic Cement Mortar. ASTM International: West Conshohocken, PA, USA, 2021.
- KS F 2405; Test Method for Compressive Strength of Concrete. Korean Agency for Technology and Standards (KATS): Eumseong County, Republic of Korea, 2021.
- KS F 2408; Test Method for Flexural Strength of Concrete. Korean Agency for Technology and Standards (KATS): Eumseong County, Republic of Korea, 2021.
- KS F 2456; Testing Method for Resistance of Concrete to Rapid Freezing and Thawing. Korean Agency for Technology and Standards (KATS): Eumseong County, Republic of Korea, 2021.
- ASTM C267-01; Standard Test Method for Chemical Resistance of Mortars, Grouts, and Monolithic Surfacings and Polymer Concretes. ASTM International: West Conshohocken, PA, USA, 2012.
- KS F 2584; Standard Test Method for Accelerated Carbonation of Concrete. Korean Agency for Technology and Standards (KATS): Eumseong County, Republic of Korea, 2020.
- Sevelsted, T.F.; Skibsted, J. Carbonation of C–S–H and C–A–S–H samples studied by 13C, 27Al and 29Si MAS NMR spectroscopy. Cem. Concr. Res. 2015, 71, 56–65. [Google Scholar] [CrossRef]
- Zajac, M.; Hilbig, H.; Bullerjahn, F.; Ben Haha, M. Reactions Involved in Carbonation Hardening of Portland Cement: Effect of Curing Temperature. J. Sustain. Cem.-Based Mater. 2023, 12, 1107–1125. [Google Scholar] [CrossRef]
- Liu, X.; Feng, P.; Cai, Y.; Yu, X.; Liu, Q. Carbonation Behaviors of Calcium Silicate Hydrate (C–S–H): Effects of Aluminum. Constr. Build. Mater. 2022, 325, 126825. [Google Scholar] [CrossRef]
- Moon, E.J.; Kim, S.J.; Park, H.G.; Choi, Y.C. A Study on Cementitious Materials as Carbon Capture Materials—Microstructure Change by Carbonation Curing. J. Korea Inst. Struct. Maint. Insp. 2018, 22, 123–129. [Google Scholar]
- Maddalena, R.; Li, K.; Chater, P.A.; Michalik, S.; Hamilton, A. Direct Synthesis of a Solid Calcium–Silicate–Hydrate (C–S–H). Constr. Build. Mater. 2019, 223, 554–565. [Google Scholar] [CrossRef]
- Stepkowska, E.T.; Blanes, J.M.; Franco, F.; Real, C.; Pérez-Rodríguez, J.L. Calcite, Vaterite and Aragonite Forming on Cement Hydration from Liquid and Gaseous Phase. Cem. Concr. Res. 2003, 33, 191–201. [Google Scholar] [CrossRef]
- Hashim, M.S.; Kaczmarek, S.E. The Transformation of Aragonite to Calcite in the Presence of Magnesium: Implications for Marine Diagenesis. Earth Planet. Sci. Lett. 2021, 574, 117166. [Google Scholar] [CrossRef]
- Na, Y.K.; Moon, J.W.; Oh, T.; Yoo, D.Y. Effect of Calcium Hydroxide Addition on the Promotion of the Pozzolanic Reaction of Excess Silica Fume in Ultra-High Performance Concrete. J. Korean Soc. Hazard Mitig. 2024, 24, 223–229. [Google Scholar] [CrossRef]
- Li, Y.; Liu, W.; Xing, F.; Wang, S.; Tang, L.; Lin, S.; Dong, Z. Carbonation of Synthetic Calcium Silicate Hydrate (C–S–H) under Different CO2 Concentrations: Chemical Phase Analysis and Kinetics. J. CO2 Util. 2020, 35, 303–313. [Google Scholar] [CrossRef]
- Yaseen, S.A.; Yiseen, G.A.; Li, Z. Elucidation of Calcite Structure of Calcium Carbonate Formation Based on Hydrated Cement Mixed with Graphene Oxide and Reduced Graphene Oxide. ACS Omega 2019, 4, 10160–10170. [Google Scholar] [CrossRef]
- Madadi, A.; Wei, J. Characterization of Calcium Silicate Hydrate Gels with Different Calcium-to-Silica Ratios and Polymer Modifications. Gels 2022, 8, 75. [Google Scholar] [CrossRef]
- Kahlouche, R.; Badaoui, A.; Criado, M. Fresh, Hardened and Durability Properties of Sodium Carbonate-Activated Algerian Slag Exposed to Sulfate and Acid Attacks. Mater. Constr. 2023, 73, e321. [Google Scholar] [CrossRef]
- Liang, Q.; Huang, X.; Zhang, L.; Yang, H. A Review on Research Progress of Corrosion Resistance of Alkali-Activated Slag Cement Concrete. Materials 2024, 17, 5065. [Google Scholar] [CrossRef]
- Jang, J.G.; Kim, H.J.; Park, S.M.; Lee, H.K. The Influence of Sodium Hydrogen Carbonate on the Hydration of Cement. Constr. Build. Mater. 2015, 94, 746–749. [Google Scholar] [CrossRef]
- Andrew, O.T.; Omotayo, O.O.; Arum, C.; Ikumapayi, C.M. Effects of Sodium Carbonate Admixture and Mix Design Ratios on the Compressive Strength of Concrete. Niger. J. Technol. 2023, 42, 185–190. [Google Scholar] [CrossRef]
- Zhang, F.; Hu, Z.; Wei, F.; Wen, X.; Li, X.; Dai, L.; Liu, L. Study on Concrete Deterioration in Different NaCl–Na2SO4 Solutions and the Mechanism of Cl− Diffusion. Materials 2021, 14, 5054. [Google Scholar] [CrossRef]
- Yoshida, N.; Nakayama, K. Impact of NaCl on Deterioration of Hardened Cement Paste due to Sulfuric Acid Attack. ce/papers 2023, 6, 1295–1304. [Google Scholar] [CrossRef]
- Azar, P.; Samson, G.; Patapy, C.; Cussigh, F.; Frouin, L.; Idir, R.; Cyr, M. Durability of Sodium Carbonate Alkali-Activated Slag Concrete Assessed by a Performance-Based Approach. Constr. Build. Mater. 2024, 423, 135873. [Google Scholar] [CrossRef]
- Papadakis, V.G.; Vayenas, C.G.; Fardis, M.N. Fundamental Modeling and Experimental Investigation of Concrete Carbonation. ACI Mater. J. 1991, 88, 363–373. [Google Scholar] [CrossRef]
- Morandeau, A.; Thiery, M.; Dangla, P. Investigation of the Carbonation Mechanism of CH and C–S–H in Terms of Kinetics, Microstructure Changes and Moisture Properties. Cem. Concr. Res. 2014, 56, 153–170. [Google Scholar] [CrossRef]
- Thiery, M.; Villain, G.; Dangla, P.; Platret, G. Investigation of the Carbonation Front Shape on Cementitious Materials: Effects of the Chemical Kinetics. Cem. Concr. Res. 2007, 37, 1047–1058. [Google Scholar] [CrossRef]
- Ngala, V.T.; Page, C.L. Effects of Carbonation on Pore Structure and Diffusional Properties of Hydrated Cement Pastes. Cem. Concr. Res. 1997, 27, 995–1007. [Google Scholar] [CrossRef]
- Lothenbach, B.; Scrivener, K.; Hooton, R.D. Supplementary Cementitious Materials. Cem. Concr. Res. 2011, 41, 1244–1256. [Google Scholar] [CrossRef]










| Density (g/cm3) | Fineness (cm2/g) | Chemical Properties (%) | ||||||
|---|---|---|---|---|---|---|---|---|
| SiO2 | Al2O3 | Fe2O3 | CaO | MgO | SO3 | Ig.loss | ||
| 3.14 | 3492 | 21.1 | 4.65 | 3.14 | 62.8 | 2.81 | 2.1 | 2.18 |
| Density (g/cm3) | Blaine Fineness (cm2/g) | Chemical Properties (%) | ||||||
|---|---|---|---|---|---|---|---|---|
| SiO2 | Al2O3 | Fe2O3 | CaO | MgO | SO3 | Ig.loss | ||
| 2.90 | 4570 | 24.7 | 16.4 | 0.18 | 49.1 | 2.73 | 1.52 | 0.68 |
| Sieve size (mm) | 2.0 | 1.6 | 1.0 | 0.5 | 0.16 | 0.08 |
| Cumulative passing (%) | 0 | 7 ± 5 | 33 ± 5 | 67 ± 5 | 87 ± 5 | 99 ± 5 |
| Density (g/cm3) | Absorption (%) | Unit Weight (kg/m3) | Fineness Modulus | Sound (%) |
|---|---|---|---|---|
| 2.59 | 1.08 | 1598 | 2.75 | 2.7 |
| Density (g/cm3) | Absorption (%) | Unit Weight (kg/m3) | Fineness Modulus | Sound (%) |
|---|---|---|---|---|
| 2.70 | 1.82 | 1566 | 7.05 | 3.1 |
| Appearance | Density (g/cm3) | pH | Active Matter (%) |
|---|---|---|---|
| Brownish powder | 0.37 | 6.0 ± 1 | 98 ± 2 |
| Test ID | W/B (%) | Mix Composition (g) | |||||
|---|---|---|---|---|---|---|---|
| Sand | OPC | GGBS | Water | Spent CO2 Absorbent (aq.) | |||
| Plain | 50 | 1350 | 225 | 225 | 225 | - | |
| CO2 | 10% | 202.5 | 22.5 | ||||
| 20% | 180.0 | 45.0 | |||||
| 30% | 157.5 | 67.5 | |||||
| 40% | 135.0 | 90.0 | |||||
| Test ID | W/B (%) | Unit Weight (kg/m3) | ||||||
|---|---|---|---|---|---|---|---|---|
| OPC | GGBS | Fine Aggregate | Coarse Aggregate | Water | Spent CO2 Absorbent (aq.) | Ad. | ||
| Plain | 48.6 | 174 | 174 | 727 | 1015 | 174 | - | 0.8 |
| CO2-10% | 156.6 | 17.4 | 0.92 | |||||
| CO2-20% | 139 | 34.8 | 0.98 | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Yang, S.-L.; Lee, J.-W. Concrete Incorporating a Spent CO2 Absorbent: Comprehensive Assessment of Microstructure, Strength, and Durability. Materials 2026, 19, 577. https://doi.org/10.3390/ma19030577
Yang S-L, Lee J-W. Concrete Incorporating a Spent CO2 Absorbent: Comprehensive Assessment of Microstructure, Strength, and Durability. Materials. 2026; 19(3):577. https://doi.org/10.3390/ma19030577
Chicago/Turabian StyleYang, Sung-Lin, and Jong-Won Lee. 2026. "Concrete Incorporating a Spent CO2 Absorbent: Comprehensive Assessment of Microstructure, Strength, and Durability" Materials 19, no. 3: 577. https://doi.org/10.3390/ma19030577
APA StyleYang, S.-L., & Lee, J.-W. (2026). Concrete Incorporating a Spent CO2 Absorbent: Comprehensive Assessment of Microstructure, Strength, and Durability. Materials, 19(3), 577. https://doi.org/10.3390/ma19030577





