Molecular Dynamics Study on the Compressive Behavior of Intermetallic Compounds in 3xxx Aluminum Alloys
Abstract
1. Introduction
2. Methodology
2.1. Interatomic Potential
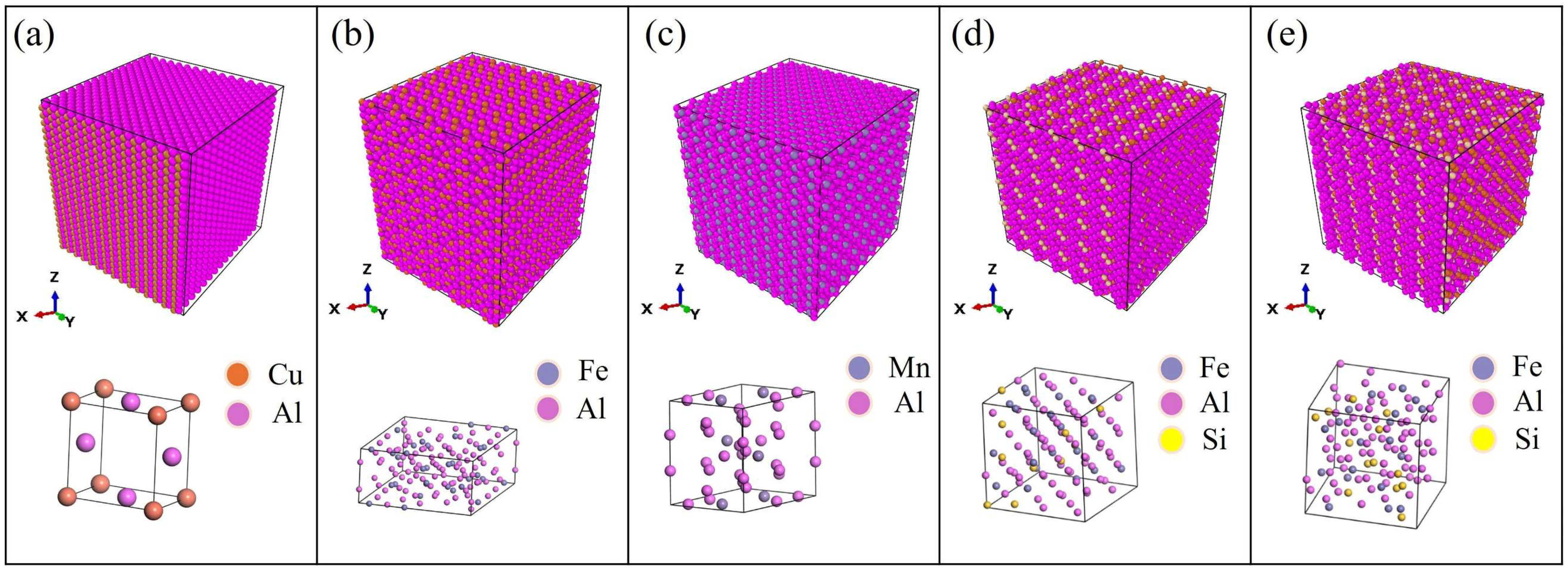
2.2. Structure
2.3. LAMMPS Environment
2.4. Method Validation
3. Results and Discussions
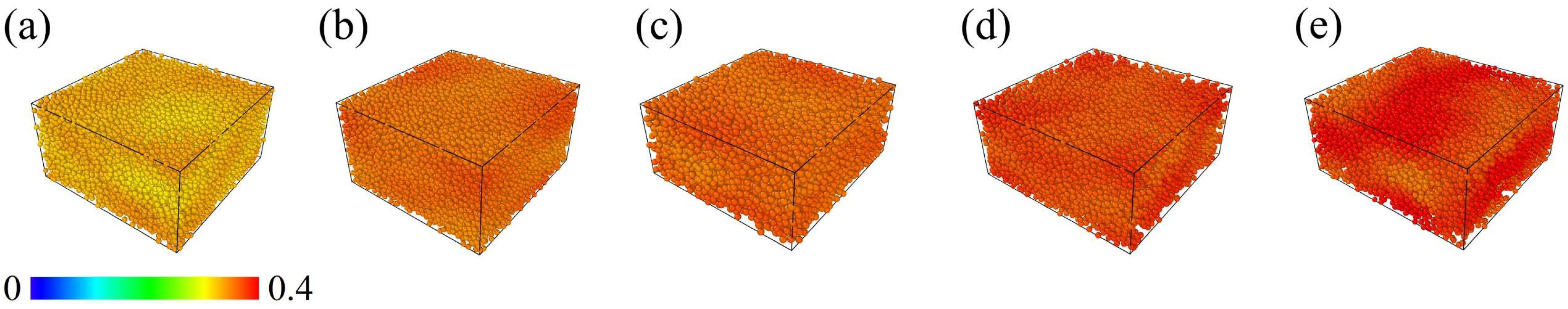
3.1. Relaxation Results
3.2. Stress–Strain Behavior
3.3. Strain Evolution
3.4. Temperature Effects on Compressive Properties
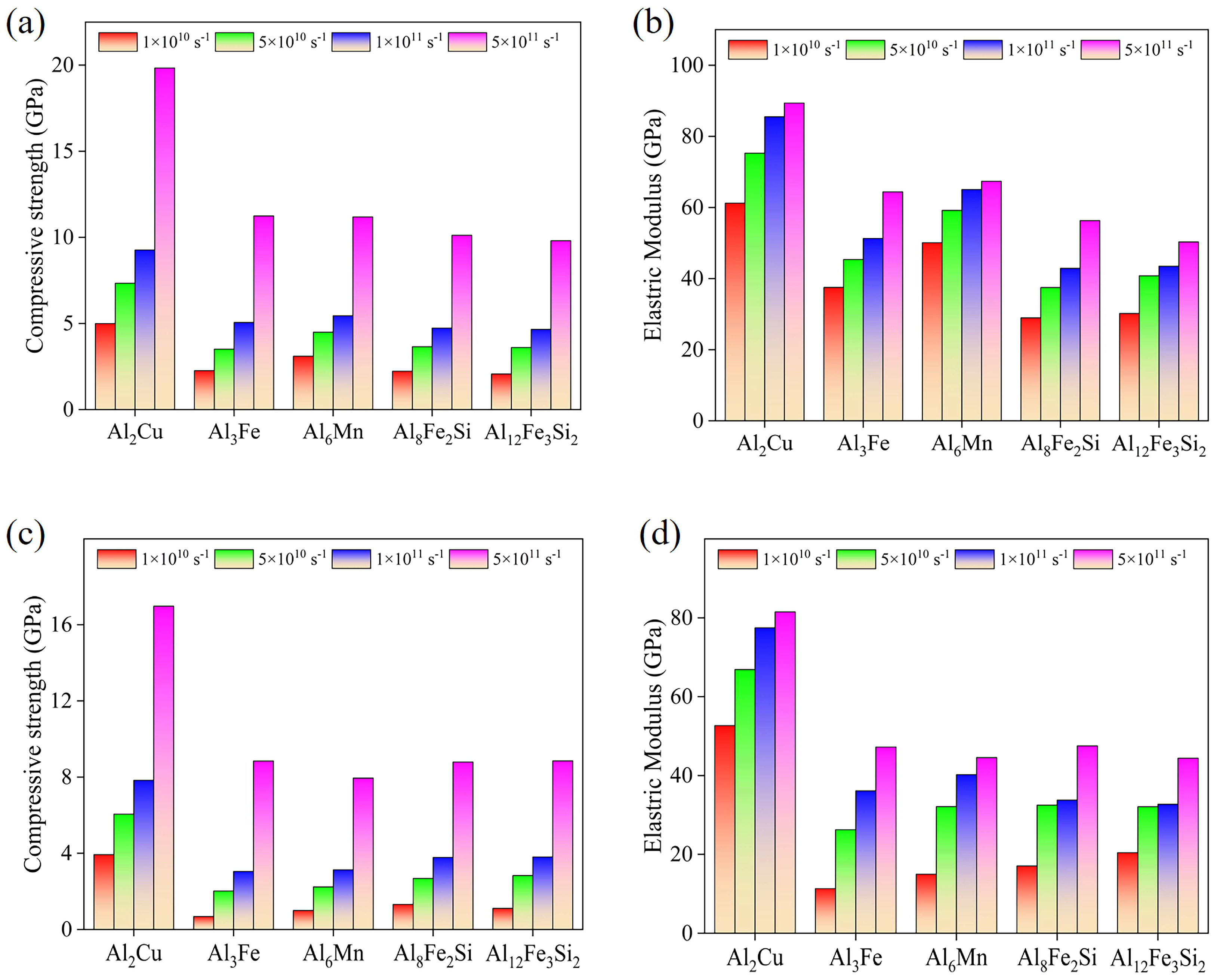
3.5. The Strain Rate Effect on Compressive Properties
3.6. The Strain Rate Sensitivity of Compressive Strength
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Tan, Y.; Wang, X.; Ma, M.; Zhang, J.; Liu, W.; Fu, R.; Xiang, S. A study on microstructure and mechanical properties of AA 3003 aluminum alloy joints by underwater friction stir welding. Mater. Charact. 2017, 127, 41–52. [Google Scholar] [CrossRef]
- Yang, H.; Gao, Y.; Qin, W.; Li, Y. Microstructure and corrosion behavior of electroless Ni–P on sprayed Al–Ce coating of 3003 aluminum alloy. Surf. Coat. Technol. 2015, 281, 176–183. [Google Scholar] [CrossRef]
- Chen, G.; Fu, G.; Yan, W.; Cheng, C.; Zou, Z. Mathematical Model of Dynamic Recrystallization of Aluminum Alloy 3003. Met. Sci. Heat Treat. 2013, 55, 220–225. [Google Scholar] [CrossRef]
- Chen, G.; Fu, G.; Lin, X.; Wang, J.; Cheng, C.; Yang, K. Effect of Efficient Purification on Mechanical Properties of 3003 Aluminum Alloy. Key Eng. Mater. 2018, 777, 402–407. [Google Scholar] [CrossRef]
- Gao, Z.; Jin, X.; Zhang, Z.; Niu, J.; Brnic, J. Study on Microstructure and Mechanical Properties of 3003 Aluminum Alloy Joints Brazed with Al–Si–Cu–Ni Paste Brazing. Sci. Rep. 2014, 14, 10648. [Google Scholar] [CrossRef]
- Khakbz, F.; Kazeminezhad, M. Work hardening and mechanical properties of severely deformed AA3003 by constrained groove pressing. J. Manuf. Process. 2012, 14, 20–25. [Google Scholar] [CrossRef]
- Wang, T.; Wang, Y.; Zhang, Y.; Jiang, S.; Zhang, L. Influence of an AlSi5 Filler Wire on Microstructures and Mechanical Properties of EBW-brazed CP-Al to 304 SS Joint. J. Manuf. Process. 2020, 56, 12–18. [Google Scholar] [CrossRef]
- Dong, J.; Li, H.; Zhu, Z.; Yang, C.; Miao, Y.; Xu, C.; Chen, B.; Liu, Z. Microstructure Evolution and Tensile Behavior of Balanced Al-Mg-Si Alloy with Various Homogenization Parameters. Trans. Nonferrous Met. Soc. China 2024, 34, 3536–3553. [Google Scholar] [CrossRef]
- Wang, X.; Guan, R.; Misra, R.D.K.; Wang, Y.; Li, H.; Shang, Y. The Mechanistic Contribution of Nanosized Al3Fe Phase on the Mechanical Properties of Al-Fe Alloy. Mater. Sci. Eng. A 2018, 724, 452–460. [Google Scholar] [CrossRef]
- Beygi, R.; Carbas, R.G.C.; Margues, E.A.S.; Barbosa, A.Q.; Kasaei, M.M.; Silva, L.F.M.D. Mechanism of Toughness Enhancement of Brittle Fracture by Intermittent η-Intermetallic in Al/Cu Joint Made by FSW. Mater. Sci. Eng. A 2024, 890, 145907. [Google Scholar] [CrossRef]
- Gao, Z.; Qin, Z.; Lu, Q. Controlled Atmosphere Brazing of 3003 Aluminum Alloy Using Low-Melting-Point Filler Metal Fabricated by Melt-Spinning Technology. Materials 2022, 15, 6080. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, F.; Sugamoto, J.; Nakashima, H.; Yoshinaga, H. Threshold Stress and Its Temperature Dependence for High-Temperature Deformation of a Precipitation-Hardened Al-0.7 at% Mn Alloy. J. Jpn. Inst. Met. 1995, 59, 1–7. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, A.; Xie, J.; Guo, Y. Deformation Mechanisms in Al/Al2Cu/Cu Multilayer under Compressive Loading. J. Alloys Compd. 2021, 885, 160921. [Google Scholar] [CrossRef]
- Dong, L.; Jones, H. The Dependence of Growth Temperature on Growth Velocity for Primary Al3Fe in Steady State Solidification of Hypereutectic AlFe Alloys. Scr. Metall. Mater. 1991, 25, 2855–2859. [Google Scholar] [CrossRef]
- Yang, Q.; Zhou, Y.; Tan, Y.; Xiang, S.; Ma, M.; Zhao, F.; Microstructure, E.O. Texture Evolution and Strengthening Mechanisms on Mechanical Properties of 3003 Aluminum Alloy During Cryogenic Rolling. J. Alloys Compd. 2021, 884, 161135. [Google Scholar] [CrossRef]
- Fang, X.; Li, Y.; Zheng, Q.; Guo, J.; Yang, Y.; Ding, W.; Ma, C.; He, K.; Su, N.; Jiang, J.; et al. Theoretical Prediction of Structural, Mechanical, and Thermophysical Properties of the Precipitates in 2xxx Series Aluminum Alloy. Metals 2022, 12, 2178. [Google Scholar] [CrossRef]
- Chen, C.; Richter, A.; Thomson, R.C. Mechanical Properties of Intermetallic Phases in Multi-Component Al–Si Alloys Using Nanoindentation. Intermetallics 2009, 17, 634–641. [Google Scholar] [CrossRef]
- Ogura, T.; Ueda, K.; Saito, Y.; Hirose, A. Nanoindentation Measurement of Interfacial Reaction Layers in 6000 Series Aluminum Alloys and Steel Dissimilar Metal Joints with Alloying Elements. Mater. Trans. 2011, 52, 979–984. [Google Scholar] [CrossRef]
- Backofen, W.A. Superplasticity in an Al-Zn Alloy. Trans. Am. Soc. Met. 1964, 57, 980–990. [Google Scholar]
- Lee, B.J.; Baskes, M.I. Second nearest-neighbor modified embedded-atom-method potential. Phys. Rev. 2000, 62, 8564–8567. [Google Scholar] [CrossRef]
- Lee, B.J.; Ko, W.S.; Kim, H.K.; Kim, E.H. The modified embedded-atom method interatomic potentials and recent progress in atomistic simulations. Calphad 2010, 34, 510–522. [Google Scholar] [CrossRef]
- Rose, J.H.; Smith, J.R.; Guinea, F.; Ferrante, J. Universal features of the equation of state of metals. Phys. Rev. B 1984, 29, 2963–2969. [Google Scholar] [CrossRef]
- Baskes, M.I. Modified Embedded-Atom Potentials for Cubic Materials and Impurities. Phys. Rev. B 1992, 46, 2727–2742. [Google Scholar] [CrossRef]
- Sharifi, H.; Wick, C. Developing interatomic potentials for complex concentrated alloys of Cu, Ti, Ni, Cr, Co, Al, Fe, and Mn. Comput. Mater. Sci. 2025, 248, 113595. [Google Scholar] [CrossRef]
- Barlock, J.G.; Mondolfo, L.F. Structure of Some Aluminium-Iron-Magnesium-Manganese-Silicon Alloys. Int. J. Mater. Res. 1975, 66, 605–611. [Google Scholar] [CrossRef]
- Owen, E.A.; Preston, G.D. X-ray analysis of solid solutions. Proc. Phys. Soc. Lond. 1923, 36, 14–30. [Google Scholar] [CrossRef]
- Black, P.J. The structure of FeAl3. II. Acta. Cryst. 1955, 8, 175–182. [Google Scholar] [CrossRef]
- Corby, R.N.; Black, P.J. The Structure of A-(AlFeSi) by Anomalous-Dispersion Methods. Acta Cryst. B 1973, 29, 2669–2677. [Google Scholar] [CrossRef]
- Zhou, M. A New Look at the Atomic Level Virial Stress: On Continuum-Molecular System Equivalence. Proc. R. Soc. A 2003, 459, 2347–2392. [Google Scholar] [CrossRef]
- Yang, C.; Li, W.; Wen, Z. Study on mechanical behavior and electronic structures of Al–Cu intermetallic compounds based on first-principles calculations. Solid State Commun. 2011, 151, 1270–1274. [Google Scholar] [CrossRef]
- Zhang, J.; Huang, Y.; Mao, C.; Peng, P. Structural, Elastic and Electronic Properties of θ (Al2Cu) and S (Al2CuMg) Strengthening Precipitates in Al–Cu–Mg Series Alloys: First-principles Calculations. Solid State Commun. 2012, 152, 2100–2104. [Google Scholar] [CrossRef]
- Gu, J.; Bai, J.; Zhu, Y.; Qin, Y.; Gu, H.; Zhai, Y.; Ma, P. First-principles study of the influence of doping elements on phase stability, crystal and electronic structure of Al2Cu (θ) phase. Comput. Mater. Sci. 2016, 111, 328–333. [Google Scholar] [CrossRef]
- Li, C.; Zeng, S.; Chen, Z.; Cheng, N.; Chen, T. First-principles Calculations of Elastic and Thermodynamic Properties of the Four Main Intermetallic Phases in Al–Zn–Mg–Cu Alloys. Comput. Mater. Sci. 2014, 93, 210–220. [Google Scholar] [CrossRef]
- Xia, Z.; Wen, B.; Fan, C. Structure and Stability of the Stoichiometric Al3Fe Phase. Metals 2019, 9, 1322. [Google Scholar] [CrossRef]
- Shang, S.; Wang, J.; Wang, Y.; Du, Y.; Liu, Z. Phonon and thermodynamic properties of Al–Mn compounds: A first-principles study. Comput. Mater. Sci. 2011, 50, 2096–2103. [Google Scholar] [CrossRef]
- Ogura, T.; Saito, Y.; Ueda, K.; Hirose, A. Evaluation of Interfacial Microstructures in Dissimilar Joints of Aluminum Alloys to Steel Using Nanoindentation Technique. J. Phys. Conf. Ser. 2009, 165, 012016. [Google Scholar] [CrossRef]
- Chen, G.; Fu, G.; Wei, T.; Cheng, C.; Wang, J.; Wang, H. Establishment of dynamic-recrystallization-state diagram for hot deformation of 3003 aluminum alloy. Mater. Technol. 2018, 52, 341–347. [Google Scholar] [CrossRef]
- Xiao, X.; Guo, Y.; Zhang, R.; Bayoumy, D.; Shen, H.; Li, J.; Gan, K.; Zhang, K.; Zhu, Y.; Huang, A. Achieving Uniform Plasticity in a High Strength Al-Mn-Sc Based Alloy Through Laser-Directed Energy Deposition. Addit. Manuf. 2022, 60, 103273. [Google Scholar] [CrossRef]
- Griffith, A.A. The Phenomena of Rupture and Flow in Solids. R. Soc. 1921, 221, 582–593. [Google Scholar] [CrossRef]
- Huh, M.Y.; Jee, J.C. Formation of A Random Texture and Ultrafine Grains in Aa 3003 Aluminium Alloy During the Repeated Shear Deformation Introduced by Continuous Confined Strip Shearing. Mater. Sci. Technol. 2004, 20, 819–824. [Google Scholar] [CrossRef]
- Cheng, C.; Meng, X.; Wu, Y.; Shi, L.; Wu, H.; Cao, T.; Zhang, S.; Zhao, J. On Dynamic Mechanical Properties of 3003 Aluminum Alloy Based on Generalized Incremental Stress-State-Dependent Damage Model with Modified Johnson–Cook Equation. J. Mater. Eng. Perform. 2022, 32, 451–461. [Google Scholar] [CrossRef]








| Element | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Al | 3.36 | 4.05 | 4.53 | 0.90 | 1.89 | 0.10 | 6.97 | 0.007 | −0.74 | −1.89 | 9.121 | 1.0 |
| Cu | 3.54 | 3.62 | 5.10 | 0.93 | 3.75 | 5.58 | 5.74 | 3.14 | 6.23 | 2.70 | 0.87 | 1.0 |
| Fe | 4.29 | 2.83 | 5.02 | 0.43 | 1.89 | 0 | 0.12 | 0.11 | 10.74 | −4.26 | −4.30 | 1.0 |
| Mn | 2.91 | 2.47 | 7.87 | 0.53 | 8.58 | 0.001 | 0.32 | 4.99 | 8.28 | 19.00 | −17.34 | 1.0 |
| Al2Cu | 3.28 | 2.45 | 2.80 | -- | -- | -- | -- | -- | -- | -- | -- | -- |
| Al3Fe | 3.30 | 2.59 | 5.00 | -- | -- | -- | -- | -- | -- | -- | -- | -- |
| Al6Mn | 3.03 | 2.28 | 5.00 | -- | -- | -- | -- | -- | -- | -- | -- | -- |
| Lattice Parameter | Structure Dimension | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| IMC | |||||||||
| Al2Cu | 4.11 | 4.11 | 2.88 | 90.0 | 90.0 | 90.0 | 61.6 | 61.6 | 66.5 |
| Al3Fe | 15.49 | 8.08 | 12.47 | 90.0 | 107.7 | 90.0 | 62.0 | 64.6 | 74.8 |
| Al6Mn | 6.38 | 7.46 | 8.76 | 90.0 | 90.0 | 90.0 | 70.2 | 67.2 | 70.1 |
| Al8Fe2Si | 11.78 | 11.77 | 11.79 | 90.0 | 90.0 | 90.0 | 70.7 | 70.6 | 70.7 |
| Al12Fe3Si2 | 11.77 | 11.81 | 11.81 | 90.0 | 90.0 | 90.0 | 70.6 | 70.8 | 70.9 |
| Properties | Previous Experimental Study (0 K–100 K) | From This Study (623 K–823 K) |
|---|---|---|
| 99 GPa~120 GPa [30,31,32,33] | 52.68 GPa~89.38 GPa | |
| 133 GPa~140.9 GPa [34,36] | 11.33 GPa~64.39 GPa | |
| 126.6 GPa [16] | 14.94 GPa~67.38 GPa | |
| 102.6 GPa~121 GPa [35] | ||
| 148.6 GPa [7] | 17.05 GPa~56.31 GPa | |
| -- | 20.42 GPa~50.31 GPa |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Li, Y.; Bai, J.; Yang, Z.; Chen, Z.; Wang, C.; Zheng, Q.; Tie, D. Molecular Dynamics Study on the Compressive Behavior of Intermetallic Compounds in 3xxx Aluminum Alloys. Materials 2026, 19, 535. https://doi.org/10.3390/ma19030535
Li Y, Bai J, Yang Z, Chen Z, Wang C, Zheng Q, Tie D. Molecular Dynamics Study on the Compressive Behavior of Intermetallic Compounds in 3xxx Aluminum Alloys. Materials. 2026; 19(3):535. https://doi.org/10.3390/ma19030535
Chicago/Turabian StyleLi, Yexin, Jingyuan Bai, Zhou Yang, Zhongjie Chen, Chuanyang Wang, Quanfeng Zheng, and Di Tie. 2026. "Molecular Dynamics Study on the Compressive Behavior of Intermetallic Compounds in 3xxx Aluminum Alloys" Materials 19, no. 3: 535. https://doi.org/10.3390/ma19030535
APA StyleLi, Y., Bai, J., Yang, Z., Chen, Z., Wang, C., Zheng, Q., & Tie, D. (2026). Molecular Dynamics Study on the Compressive Behavior of Intermetallic Compounds in 3xxx Aluminum Alloys. Materials, 19(3), 535. https://doi.org/10.3390/ma19030535






