Electrospun PVA/CS/HA/BA Nanofiber Scaffolds with Enhanced Mechanical Stability and Antifungal Activity for Bone Tissue Engineering
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Electrospinning Solution
2.3. Preparation of Nanofibers by Electrospinning and Chemical Cross-Linking
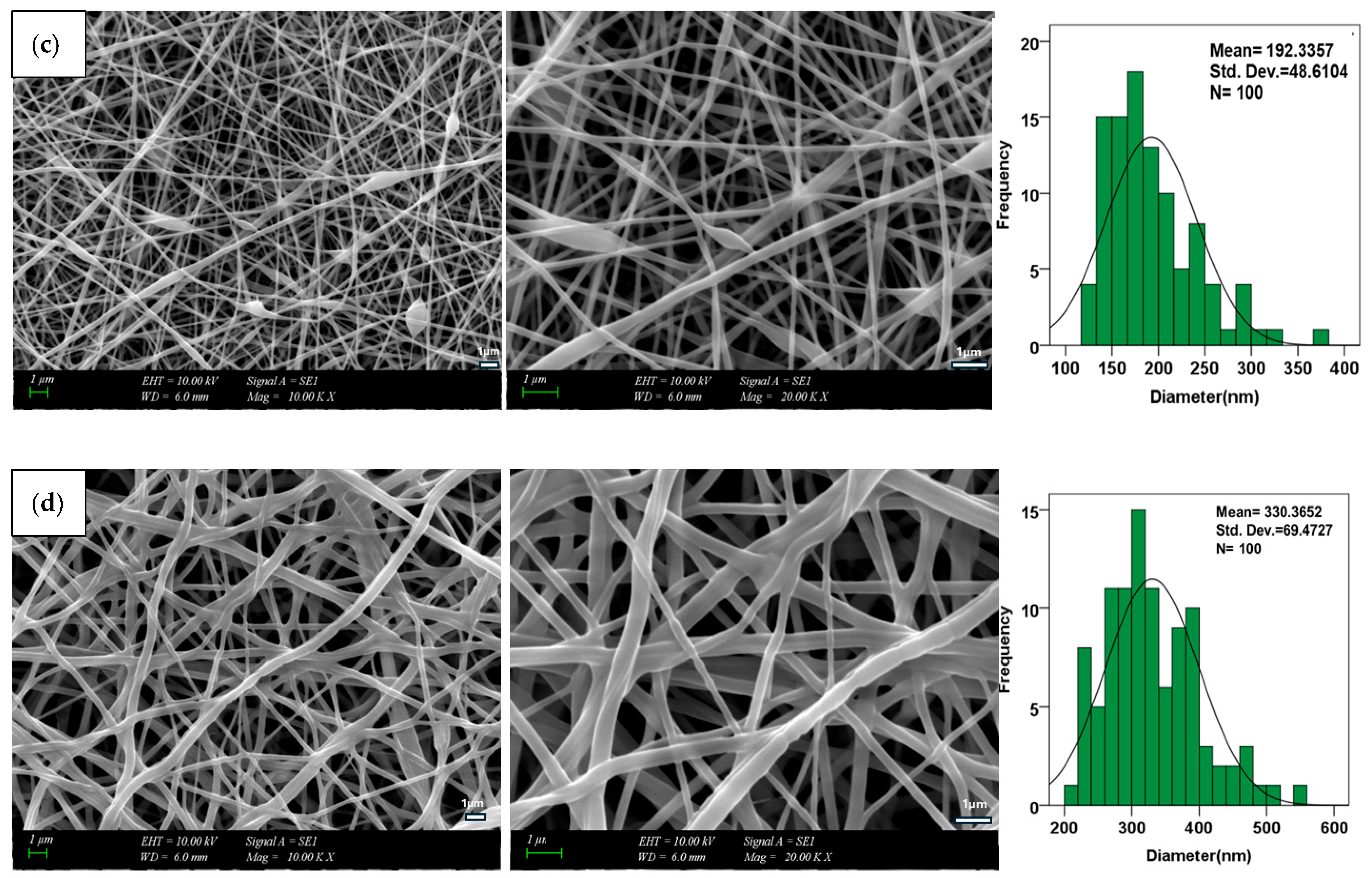
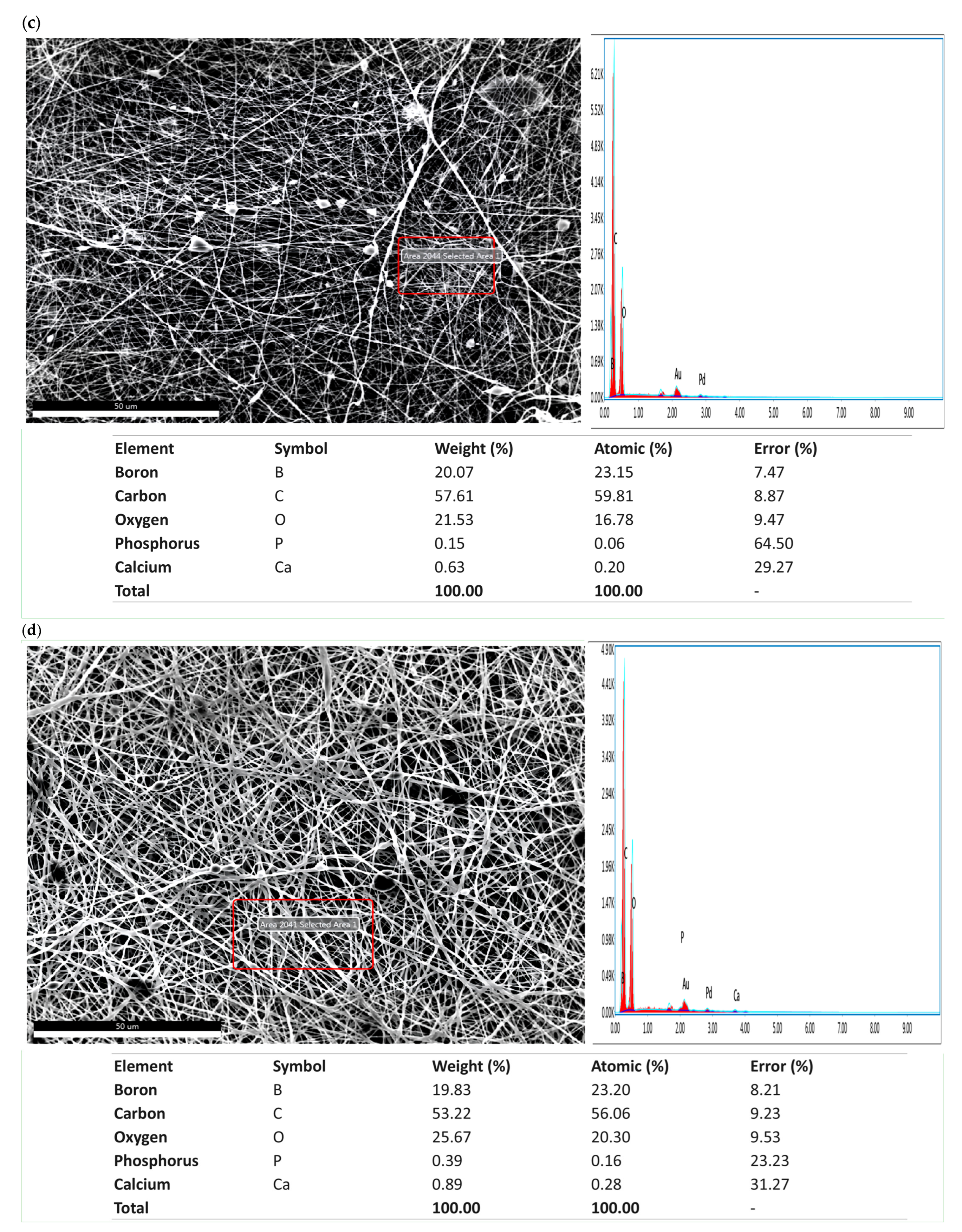
2.4. Scanning Electron Microscopy
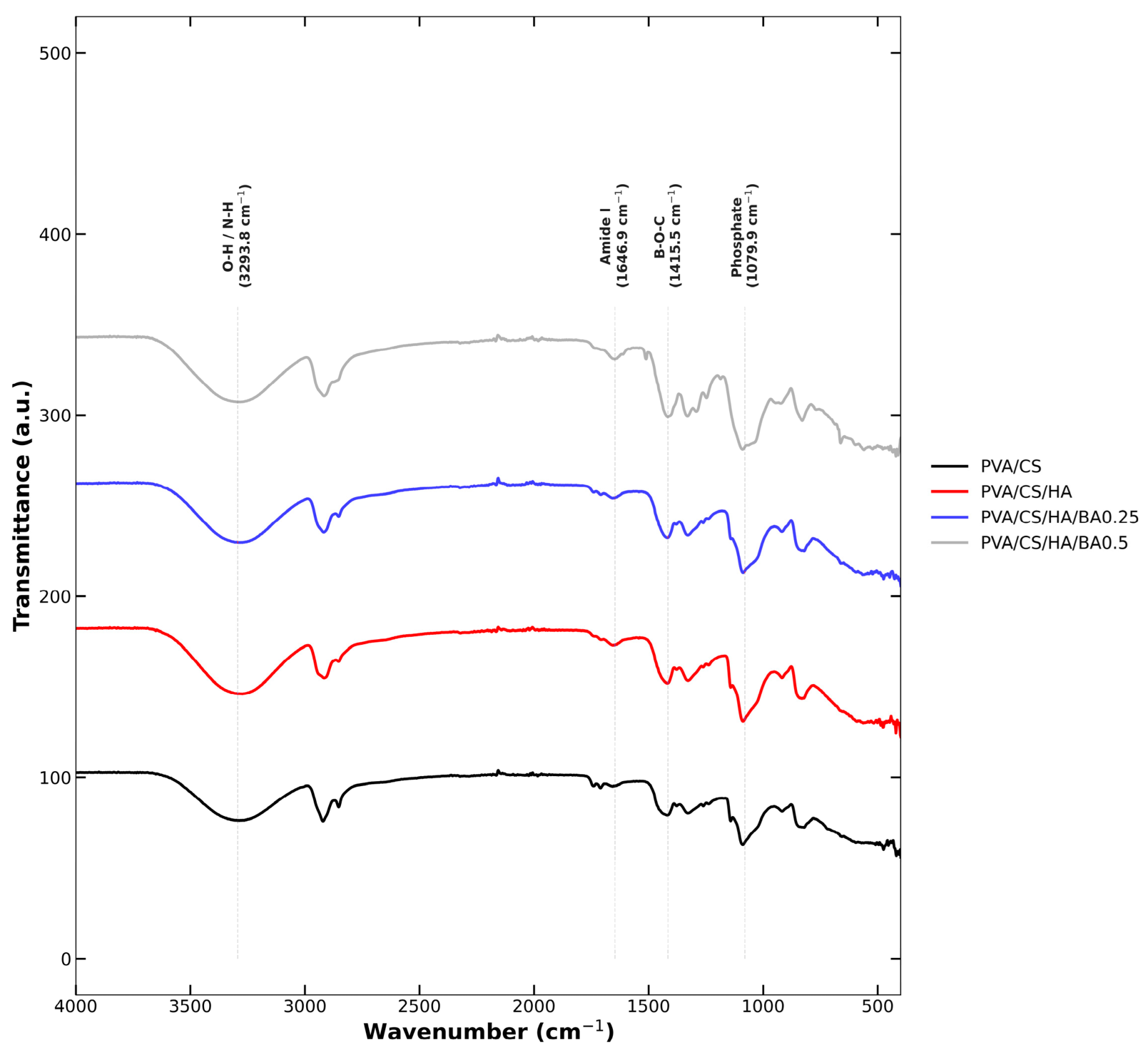
2.5. Fourier Transform Infrared Spectroscopy (FT-IR)
2.6. Differential Scanning Calorimetry (DSC)
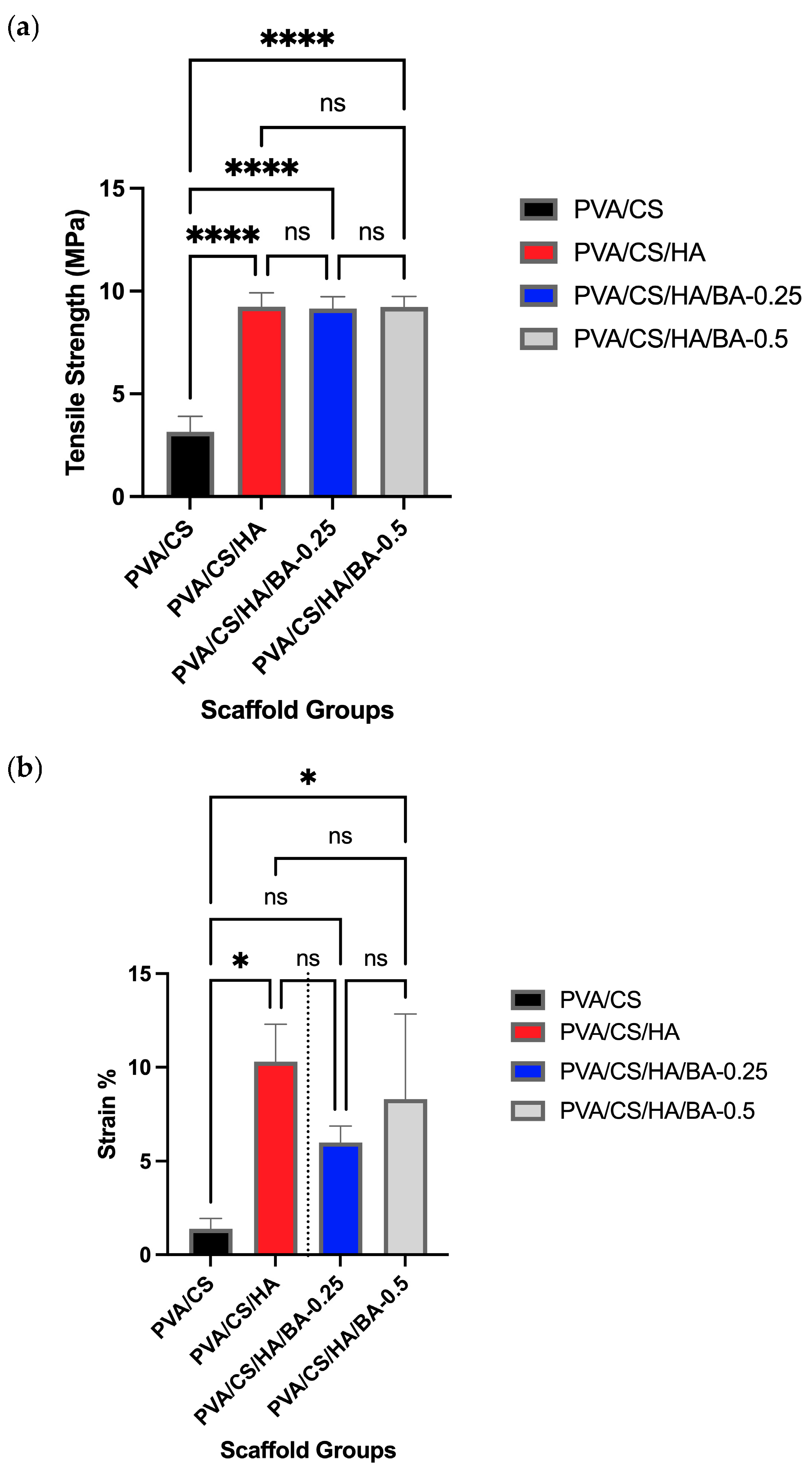
2.7. Mechanical Characterization
2.8. Cell Viability Evaluation of Nanofiber Scaffolds (Indirect MTT Assay)
2.9. Antifungal Activity Evaluation
2.10. Statistical Analysis
3. Results and Discussion
3.1. Structural Morphology of Electrospun Scaffold System
3.2. Spectroscopic Characterization and Molecular Interaction Analysis
3.3. Thermal Stability and Transition Properties of Electrospun Scaffolds
3.4. Mechanical Properties of Nanofibers
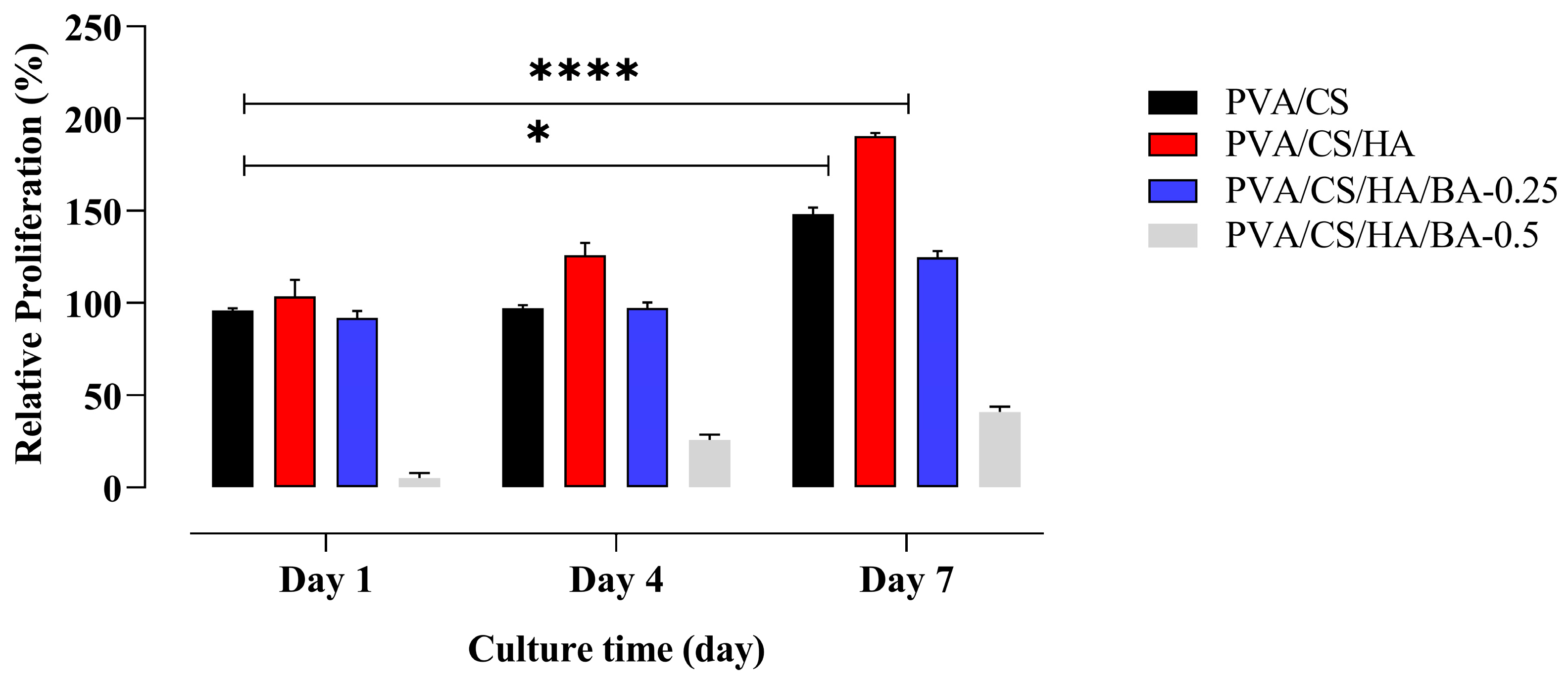
3.5. Evaluation of the Cytocompatibility of Nanofiber Scaffolds
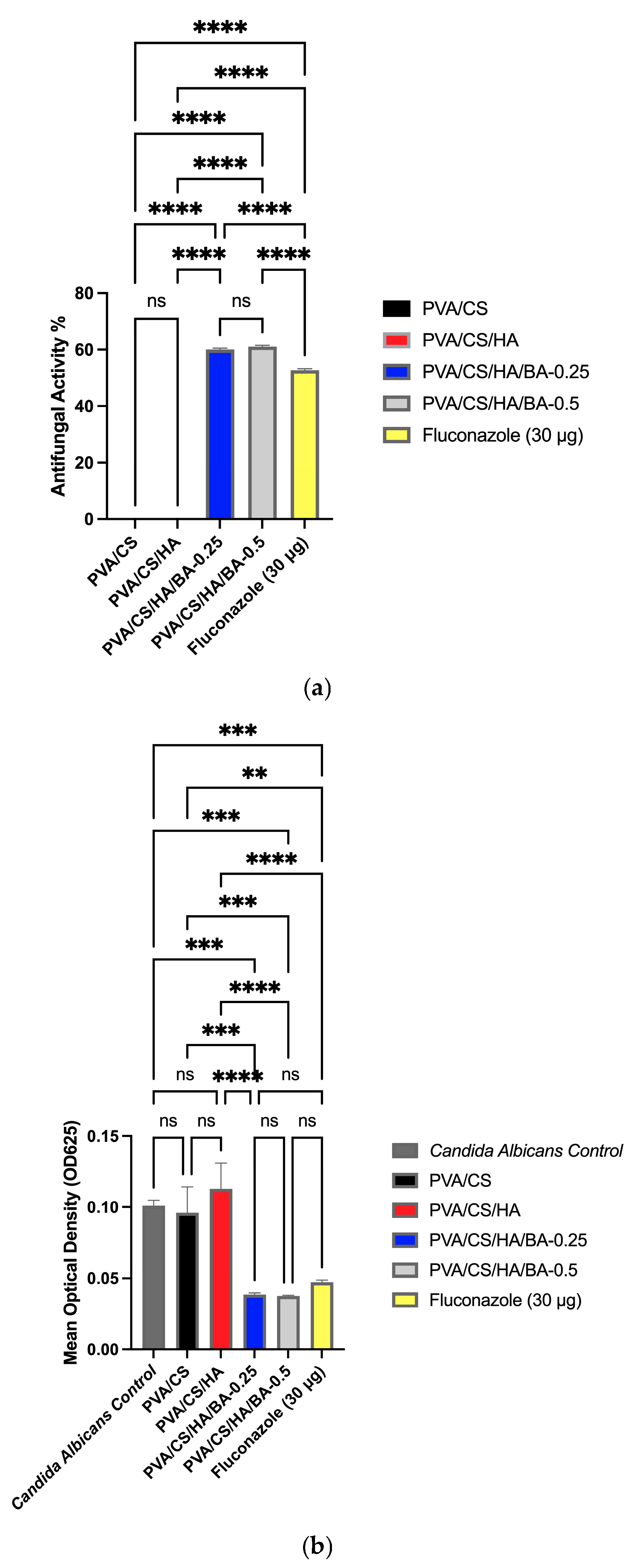
3.6. Evaluation of Antifungal Activity of PVA/CS/HA/BA Scaffolds
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Santin, M. 14—Bone tissue engineering. In Bone Repair Biomaterials; Woodhead Publishing: Cambridge, UK, 2009; pp. 378–422. [Google Scholar] [CrossRef]
- Ma, P.; Wu, W.; Wei, Y.; Ren, L.; Lin, S.; Wu, J. Biomimetic gelatin/chitosan/polyvinyl alcohol/nano-hydroxyapatite scaffolds for bone tissue engineering. Mater. Des. 2021, 207, 109865. [Google Scholar] [CrossRef]
- Salim, S.A.; Loutfy, S.A.; El-Fakharany, E.M.; Taha, T.H.; Hussien, Y.; Kamoun, E.A. Influence of chitosan and hydroxyapatite incorporation on properties of electrospun PVA/HA nanofibrous mats for bone tissue regeneration: Nanofibers optimization and in-vitro assessment. J. Drug Deliv. Sci. Technol. 2021, 62, 102417. [Google Scholar] [CrossRef]
- Wongsawichai, K.; Kingkaew, A.; Pariyaisut, A.; Khondee, S.; Kingkaew, E.; Pariyaisut, E. Porous hydroxyapatite/chitosan/carboxymethyl cellulose scaffolds with tunable microstructures for bone tissue engineering. Key Eng. Mater. 2019, 819, 9–14. [Google Scholar] [CrossRef]
- Gavilanes-Martínez, M.A.; Coral-Garzón, A.; Cáceres, D.H.; García, A.M. Antifungal activity of boric acid, triclosan and zinc oxide against different clinically relevant Candida species. Mycoses 2021, 64, 1045–1052. [Google Scholar] [CrossRef]
- Liu, Q.; Liu, Z.; Zhang, C.; Xu, Y.; Li, X.; Gao, H. Effects of 3% Boric Acid Solution on Cutaneous Candida albicans Infection and Microecological Flora Mice. Front. Microbiol. 2021, 12, 709880. [Google Scholar] [CrossRef]
- De Seta, F.; Schmidt, M.; Vu, B.; Essmann, M.; Larsen, B. Antifungal mechanisms supporting boric acid therapy of Candida vaginitis. J. Antimicrob. Chemother. 2008, 63, 325–336. [Google Scholar] [CrossRef]
- Ahtzaz, S.; Sher Waris, T.; Shahzadi, L.; Anwar Chaudhry, A.; Ur Rehman, I.; Yar, M. Boron for tissue regeneration-it’s loading into chitosan/collagen hydrogels and testing on chorioallantoic membrane to study the effect on angiogenesis. Int. J. Polym. Mater. Polym. Biomater. 2019, 69, 525–534. [Google Scholar] [CrossRef]
- Ghiska, R.; Debie, M.P.; Albab, M.F.; Alfata, R.; Sofyan, N.; Yuwono, H. The effect of HA addition on the mechanical properties of PVA/CS biomaterials for bone scaffold application. In AIP Conference Proceedings; AIP Publishing LLC: Melville, NY, USA, 2018; Volume 1933, p. 020006. [Google Scholar] [CrossRef]
- Abazari, M.F.; Nejati, F.; Nasiri, N.; Khazeni, Z.A.S.; Nazari, B.; Enderami, S.E.; Mohajerani, H. Platelet-rich plasma incorporated electrospun PVA–CS–HA nanofibers accelerate osteogenic differentiation and bone reconstruction. Gene 2019, 720, 144096. [Google Scholar] [CrossRef]
- Satpathy, A.; Pal, A.; Sengupta, S.; Das, A.; Hasan, M.; Ratha, I.; Barui, A.; Bodhak, S. Bioactive nano-HA doped electrospun PVA–CS composite nanofibers for bone tissue engineering applications. J. Indian Inst. Sci. 2019, 99, 289–302. [Google Scholar] [CrossRef]
- Januariyasa, I.K.; Ana, I.D.; Yusuf, Y. Nanofibrous poly(vinyl alcohol)/CS containing carbonated HA nanoparticles scaffold for bone tissue engineering. Mater. Sci. Eng. C 2020, 107, 110347. [Google Scholar] [CrossRef]
- Seeman, E.; Delmas, P.D. Bone quality: The material and structural basis of bone strength and fragility. N. Engl. J. Med. 2006, 354, 2250–2261. [Google Scholar] [CrossRef]
- Feng, W.; Li, D.; Zang, J.; Fu, L. Biomechanical comparison of xenogeneic bone material treated with different methods. Xenotransplantation 2017, 24, e12343. [Google Scholar] [CrossRef] [PubMed]
- Turner, C.H. Biomechanics of bone: Determinants of skeletal fragility and bone quality. Osteoporos. Int. 2002, 13, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Sorushanova, A.; Delgado, L.M.; Wu, Z.; Shologu, N.; Kshirsagar, A.; Raghunath, R.; Mullen, A.M.; Bayon, Y.; Pandit, A.; Raghunath, M.; et al. The collagen superfamily: From biosynthesis to advanced biomaterial development. Adv. Mater. 2019, 31, e1801651. [Google Scholar] [CrossRef]
- Brun, V.; Guillaume, C.; Mechiche-Alami, S.; Josse, J. CS/HA hybrid scaffold for bone tissue engineering. Bio-Medical. Mater. Eng. 2014, 24, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Gaweł, J.; Milan, J.; Żebrowski, J.; Płoch, D.; Stefaniuk, I.; Kus-Liśkiewicz, M. Biomaterial composed of CS, riboflavin, and HA for bone tissue regeneration. Sci. Rep. 2023, 13, 17004. [Google Scholar] [CrossRef]
- Pineda-Castillo, S.; Bernal-Ballén, A.; Bernal-López, C.; Segura-Puello, H.; Nieto-Mosquera, D.; Villamil-Ballesteros, A.; Muñoz-Forero, D.; Munster, L. Synthesis and characterization of poly(vinyl alcohol)–CS–HA scaffolds: A promising alternative for bone tissue regeneration. Molecules 2018, 23, 2414. [Google Scholar] [CrossRef]
- Firnanelty, S.; Sugiarti, S. Charlena Synthesis of HA–CS–PVAcomposite as injectable bone substitute material Rasayan. J. Chem. 2017, 10, 570–576. [Google Scholar] [CrossRef]
- Mu, X.; Zheng, W.; Xiao, L.; Zhang, W.; Jiang, X. Engineering a 3D vascular network in hydrogel for mimicking a nephron. Lab Chip 2013, 13, 1612–1618. [Google Scholar] [CrossRef]
- Gezer, A.H.; Acar, A. Effects of boron compounds and ozonated olive oil on experimental Microsporum canis infection in rats. Turk. J. Veter. Anim. Sci. 2020, 44, 258–265. [Google Scholar] [CrossRef]
- Kapukaya, R.; Ciloglu, O. Treatment of chronic wounds with polyurethane sponges impregnated with BA particles: A randomised controlled trial. Int. Wound J. 2020, 17, 1159–1165. [Google Scholar] [CrossRef] [PubMed]
- Tort, S.; Acartürk, F. Investigation of propolis and boron containing disinfectants and comparison with WHO-recommended formulation against COVID-19. Gazi Med. J. 2020, 31, 532–536. [Google Scholar] [CrossRef]
- Sayin, Z.; Ucan, U.S.; Sakmanoglu, A. Antibacterial and antibiofilm effects of boron on different bacteria. Biol. Trace Elem. Res. 2016, 173, 241–246. [Google Scholar] [CrossRef] [PubMed]
- Price, C.J.; Marr, M.C.; Myers, C.; Seely, J.C.; Heindel, J.J.; Schwetz, B.A. The developmental toxicity of BA in rabbits. Toxicol. Sci. 1996, 34, 176–187. [Google Scholar] [CrossRef]
- Erickson, A.E.; Edmondson, D.; Chang, F.C.; Wood, D.; Gong, A.; Levengood, S.L.; Zhang, M. High-throughput and high-yield fabrication of uniaxially-aligned chitosan-based nanofibers by centrifugal electrospinning. Carbohydr. Polym. 2015, 134, 467–474. [Google Scholar] [CrossRef]
- Sulutaş, R.B.; Cesur, S.; Seyhan, S.A.; Alkaya, D.B.; Şahin, A.; Ekren, N.; Gündüz, O. Electrospun amygdalin and Inula helenium extract-loaded PLA/PVP nanofibrous patches for colon cancer treatment: Fabrication, characterization and antitumour effect. J. Drug Deliv. Sci. Technol. 2024, 99, 105940. [Google Scholar] [CrossRef]
- Das, R.; Burbery, N.J. Trends and developments in the manufacturing of polymer nanofibrils with the electrospinning technique. Appl. Mech. Mater. 2013, 446-447, 1298–1303. [Google Scholar] [CrossRef]
- Li, Y.; Bou-Akl, T. Electrospinning in tissue engineering. In Electrospinning—Material, Techniques, and Biomedical Applications; IntechOpen: London, UK, 2016. [Google Scholar] [CrossRef]
- Paaver, U.; Heinämäki, J.; Laidmäe, I.; Lust, A.; Kozlova, J.; Sillaste, E.; Kirsimäe, K.; Veski, P.; Kogermann, K. Electrospun nanofibers as a potential controlled-release solid dispersion system for poorly water-soluble drugs. Int. J. Pharm. 2015, 479, 252–260. [Google Scholar] [CrossRef]
- Xue, J.; Wu, T.; Dai, Y.; Xia, Y. Electrospinning and electrospun nanofibers: Methods, materials, and applications. Chem. Rev. 2019, 119, 5298–5415. [Google Scholar] [CrossRef]
- Nokhasteh, S.; Molavi, M.A.; Ghayeni, M.K.; Avalshahr, A.S. Preparation of PVA/CS samples by electrospinning and film casting methods and evaluating the effect of surface morphology on their antibacterial behavior. Mater. Res. Express 2019, 7, 015401. [Google Scholar] [CrossRef]
- Koosha, M.; Mirzadeh, H. Electrospinning, mechanical properties, and cell behavior study of CS/PVA nanofibers. J. Biomed. Mater. Res. Part A 2015, 103, 3081–3093. [Google Scholar] [CrossRef]
- Sundhamurti, D.; Vasanthan, S.K.; Kuppan, P.; Krishnan, M.U.; Sethuraman, S. Electrospun nanostructured CS–poly(vinyl alcohol) scaffolds: A biomimetic extracellular matrix as dermal substitute. Biomed. Mater. 2012, 7, 045005. [Google Scholar] [CrossRef]
- Gadhave, R.V.; Mahanwar, P.A.; Gadekar, P.T. Study of cross-linking between BA and different types of PVA adhesive. Open J. Polym. Chem. 2019, 9, 16–26. [Google Scholar] [CrossRef]
- Araújo, E.S.; Nascimento, M.L.F.; de Oliveira, H.P. Influence of Triton X-100 on PVA fibres production by the electrospinning technique. Fibres Text. East. Eur. 2013, 21, 39–43. [Google Scholar]
- Işık, A.F.; San Keskin, N.O.; Ulçay, Y. Synthesis and in vitro antimicrobial characterization of boron–PVA electrospun nanofibers. J. Text. Inst. 2018, 110, 575–580. [Google Scholar] [CrossRef]
- Matabola, K.P.; de Vries, A.R.; Luyt, A.S.; Kumar, R. Studies on single polymer composites of poly(methyl methacrylate) reinforced with electrospun nanofibers with a focus on their dynamic mechanical properties. Express Polym. Lett. 2011, 5, 635–642. [Google Scholar] [CrossRef]
- Zhang, L.; Tsuzuki, T.; Wang, X. Preparation and characterization of cellulose nanofiber film. Mater. Sci. Forum 2010, 654-656, 1760–1763. [Google Scholar] [CrossRef]
- Kimura, N.; Kim, H.; Kim, B.; Lee, K.; Kim, I.S. Molecular orientation and crystalline structure of aligned electrospun nylon-6 nanofibers: Effect of gap size. Macromol. Mater. Eng. 2010, 295, 1090–1096. [Google Scholar] [CrossRef]
- Philip, P.; Jose, E.T. Studies of interaction of methylene blue with pure and structurally modified electrospun poly(methyl methacrylate) nanofibers. Environ. Prog. Sustain. Energy 2025, 44, e14600. [Google Scholar] [CrossRef]
- Kang, H.K.; Oh, H.J.; Kim, J.Y.; Kim, H.Y.; Choi, Y.O. Effect of process control parameters on the filtration performance of PAN–CTAB nanofiber/nanonet web combined with meltblown nonwoven. Polymers 2021, 13, 3591. [Google Scholar] [CrossRef]
- Cho, Y.S.; Yoon, H.; Jin, S.G. Novel Saccharomyces cerevisiae-loaded polyvinylpyrrolidone/SiO2 nanofiber for wound dressing prepared using electrospinning method. Materials 2024, 17, 2903. [Google Scholar] [CrossRef] [PubMed]
- Kai, D.; Prabhakaran, M.P.; Stahl, B.; Eblenkamp, M.; Wintermantel, E.; Ramakrishna, S. Mechanical properties and in vitro behavior of nanofiber–hydrogel composites for tissue engineering applications. Nanotechnology 2012, 23, 095705. [Google Scholar] [CrossRef] [PubMed]
- Venugopal, J.R.; Low, S.; Choon, A.T.; Ramakrishna, S. Interaction of cells and nanofiber scaffolds in tissue engineering. J. Biomed. Mater. Res. Part B Appl. Biomater. 2007, 84B, 34–48. [Google Scholar] [CrossRef] [PubMed]
- Mao, X.; Bai, Y.; Yu, J.; Ding, B. Flexible and highly temperature-resistant polynanocrystalline zirconia nanofibrous membranes designed for air filtration. J. Am. Ceram. Soc. 2016, 99, 2760–2768. [Google Scholar] [CrossRef]
- Uslu, I.; Daştan, H.; Altaş, A.; Yayli, A.; Atakol, O.; Aksu, M.L. Preparation and characterization of PVA/boron polymer produced by an electrospinning technique. e-Polymers 2017, 7, 133. [Google Scholar] [CrossRef]
- Özcan, M.; Kaya, C.; Kaya, F. An optimization study for electrospun borate ester nanofibers as lightweight, flexible, and affordable neutron shields for personal protection. Macromol. Mater. Eng. 2023, 308, 2300150. [Google Scholar] [CrossRef]
- Vu, T.H.N.; Morozkina, S.N.; Uspenskaya, M.V. Study of the nanofibers fabrication conditions from the mixture of poly(vinyl alcohol) and CS by electrospinning method. Polymers 2022, 14, 811. [Google Scholar] [CrossRef]
- Hong, X.; He, J.; Zou, L.; Wang, Y.; Li, Y.V. Preparation and characterization of high-strength and high-modulus PVA fiber via dry-wet spinning with cross-linking of BA. J. Appl. Polym. Sci. 2021, 138, 51394. [Google Scholar] [CrossRef]
- Ilhan, E.; Cesur, S.; Sulutaş, R.B.; Pilavci, E.; Dalbayrak, B.; Kaya, E.; Arisan, E.D.; Tinaz, G.B.; Sengor, M.; Kijeńska-Gawrońska, E.; et al. The role of multilayer electrospun poly(vinyl alcohol)/gelatin nanofibers loaded with fluconazole and cinnamaldehyde in the potential treatment of fungal keratitis. Eur. Polym. J. 2022, 176, 111390. [Google Scholar] [CrossRef]
- Mansur, H.S.; Sadahira, C.M.; Souza, A.N.; Mansur, A.A.P. FTIR spectroscopy characterization of poly(vinyl alcohol) hydrogel with different hydrolysis degrees and chemically crosslinked with glutaraldehyde. Mater. Sci. Eng. C 2008, 28, 539–548. [Google Scholar] [CrossRef]
- Varma, R.; Vasudevan, S. Extraction, characterization and antimicrobial activity of CS from horse mussel Modiolus modiolus. ACS Omega 2020, 5, 20224–20230. [Google Scholar] [CrossRef] [PubMed]
- Rajasekaran, S.; Kungumaraj, K.K.; Mani, D.; Mani, P. CS/guar gum–graphene oxide porous scaffolds for tissue engineering applications. Mater. Today Commun. 2024, 41, 110288. [Google Scholar] [CrossRef]
- Sistani, S.; Asgharzade, S.; Arab, S.; Bahraminasab, M.; Soltani-Fard, E. Fabrication and evaluation of a host–guest polylactic acid/gelatin–HA–blueberry scaffold for bone regeneration. J. Orthop. Surg. Res. 2025, 20, 788. [Google Scholar] [CrossRef] [PubMed]
- Lefèvre, G. Determination of isotopic ratio of boron in BA solutions by ATR–FTIR spectroscopy. J. Radioanal. Nucl. Chem. 2023, 332, 337–342. [Google Scholar] [CrossRef]
- Choe, S.; You, S.; Park, K.; Kim, Y.; Park, J.; Cho, Y.; Seo, J.; Yang, H.; Myung, J. BA-crosslinked poly(vinyl alcohol): Biodegradable, biocompatible, robust, and high-barrier paper coating. Green Chem. 2024, 26, 8230–8241. [Google Scholar] [CrossRef]
- Prosanov, I.Y.; Abdulrahman, S.T.; Thomas, S.; Bulina, N.V.; Gerasimov, K.B. Complex of PVA with BA: Structure and use. Mater. Today Commun. 2018, 14, 77–81. [Google Scholar] [CrossRef]
- da Mata, G.C.; Morais, M.S.; de Oliveira, W.P.; Aguiar, M.L. Composition Effects on the Morphology of PVA/Chitosan Electrospun Nanofibers. Polymers 2022, 14, 4856. [Google Scholar] [CrossRef]
- Sánchez-Téllez, D.A.; Baltierra-Uribe, S.L.; Vidales-Hurtado, M.A.; Valdivia-Flores, A.; García-Pérez, B.E.; Téllez-Jurado, L. Novel PVA–Hyaluronan–Siloxane Hybrid Nanofiber Mats for Bone Tissue Engineering. Polymers 2024, 16, 497. [Google Scholar] [CrossRef]
- Jiamboonsri, P.; Sangkhun, W.; Wanwong, S. Methyl Gallate and Amoxicillin-Loaded Electrospun Poly(vinyl alcohol)/Chitosan Mats: Impact of Acetic Acid on Their Anti-Staphylococcus aureus Activity. Polymers 2024, 17, 7. [Google Scholar] [CrossRef]
- Ceylan, S.; Dimmock, R.; Yang, Y. Development of Boron-Containing PVA-Based Cryogels with Controllable Boron Releasing Rate and Altered Influence on Osteoblasts. Polymers 2023, 15, 1653. [Google Scholar] [CrossRef]
- Çay, A.; Miraftab, M.; Kumbasar, E.P.A. Characterization and swelling performance of physically stabilized electrospun poly(vinyl alcohol)/chitosan nanofibers. Eur. Polym. J. 2014, 61, 253–262. [Google Scholar] [CrossRef]
- Iqbal, N.; Ganguly, P.; Yildizbakan, L.; Raif, E.M.; Jones, E.; Giannoudis, P.V.; Jha, A. Chitosan Scaffolds from Crustacean and Fungal Sources: A Comparative Study for Bone-Tissue-Engineering Applications. Bioengineering 2024, 11, 720. [Google Scholar] [CrossRef] [PubMed]
- Takeno, H.; Shikano, R.; Kikuchi, R. Mechanical performance of corn starch/poly(vinyl alcohol) composite hydrogels reinforced by inorganic nanoparticles and cellulose nanofibers. Gels 2022, 8, 514. [Google Scholar] [CrossRef]
- Srisang, N.; Maikaew, J.; Tambunlertchai, S.; Eiangmee, O.; Kamdenlek, P.; Manaspon, C.; Salem, A.K.; Phetpan, K.; Siriwan, S.; Sokjabok, S. Optimizing Bone Scaffold Design Using Response Surface and Artificial Neural Network-Genetic Algorithm Methods with Biocompatibility Evaluation. Available online: https://ssrn.com/abstract=5340703 (accessed on 15 January 2026).
- Cesur, S.; Ilhan, E.; Tut, T.A.; Kaya, E.; Dalbayrak, B.; Bosgelmez-Tinaz, G.; Arısan, E.D.; Gunduz, O.; Kijeńska-Gawrońska, E. Design of cinnamaldehyde- and gentamicin-loaded double-layer corneal nanofiber patches with antibiofilm and antimicrobial effects. ACS Omega 2023, 8, 28109–28121. [Google Scholar] [CrossRef]
- Lemma, S.M.; Scampicchio, M.; Mahon, P.J.; Sbarski, I.; Wang, J.; Kingshott, P. Controlled release of retinyl acetate from β-cyclodextrin-functionalized poly(vinyl alcohol) electrospun nanofibers. J. Agric. Food Chem. 2015, 63, 3481–3488. [Google Scholar] [CrossRef]
- Peppas, N.A.; Merrill, E.W. Crosslinked poly(vinyl alcohol) hydrogels as swollen elastic networks. J. Appl. Polym. Sci. 1977, 21, 1763–1770. [Google Scholar] [CrossRef]
- Choo, K.; Ching, Y.C.; Chuah, C.H.; Julai, S.; Liou, N.S. Preparation and characterization of PVA–CS composite films reinforced with cellulose nanofiber. Materials 2016, 9, 644. [Google Scholar] [CrossRef]
- Kanimozhi, K.; Khaleel Basha, S.; Sugantha Kumari, V. Processing and characterization of chitosan/PVA and methylcellulose porous scaffolds for tissue engineering. Mater. Sci. Eng. C 2016, 61, 484–491. [Google Scholar] [CrossRef]
- Rinaudo, M. Chitin and CS: Properties and applications. Prog. Polym. Sci. 2006, 31, 603–632. [Google Scholar] [CrossRef]
- Pistone, A.; Iannazzo, D.; Celesti, C.; Piperopoulos, E.; Ashok, D.; Cembran, A.; Tricoli, A.; Nisbet, D. Engineering of CS–HA–magnetite hierarchical scaffolds for guided bone growth. Materials 2019, 12, 2321. [Google Scholar] [CrossRef]
- Radhakrishnan, J.; Gandham, G.S.P.D.; Sethuraman, S.; Subramanian, A. Phase-induced porous composite microspheres sintered scaffold with protein–mineral interface for bone tissue engineering. RSC Adv. 2015, 5, 22005–22014. [Google Scholar] [CrossRef]
- Dorozhkin, S.V. Bioceramics of calcium orthophosphates. Biomaterials 2010, 31, 1465–1485. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Zhao, L.; Zhang, C.; Li, L. Water states and thermal processability of BA-modified poly(vinyl alcohol). J. Appl. Polym. Sci. 2015, 133, 43246. [Google Scholar] [CrossRef]
- Oktay, B.; Ciftci, F.; Erarslan, A.; Ahlatcıoğlu Özerol, E. Dual-layer natamycin and boric-acid reinforced PVA/CS by 3D printing and electrospinning: Characterization and in vitro evaluation. Polymers 2025, 17, 1673. [Google Scholar] [CrossRef]
- Yang, D.; Jin, Y.; Ma, G.; Chen, X.; Lu, F.; Nie, J. Fabrication and characterization of CS/PVA with HA biocomposite nanoscaffolds. J. Appl. Polym. Sci. 2008, 110, 3328–3335. [Google Scholar] [CrossRef]
- Rezaei, A.; Katoueizadeh, E.; Zebarjad, S.M. Investigating the influence of zinc oxide nanoparticles morphology on the properties of electrospun polyvinyl alcohol/chitosan (PVA/CS) nanofibers. J. Drug Deliv. Sci. Technol. 2023, 86, 104712. [Google Scholar] [CrossRef]
- CLSI. Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts, 3rd ed.; Approved Standard; CLSI: Wayne, PA, USA, 2008. [Google Scholar]
- Iavazzo, C.; Gkegkes, I.D.; Zarkada, I.M.; Falagas, M.E. Boric acid for recurrent vulvovaginal candidiasis: The clinical evidence. J. Women’s Heal. 2011, 20, 1245–1255. [Google Scholar] [CrossRef]
- Syvolos, Y.; Salama, O.E.; Gerstein, A.C. Constraint on boric acid resistance and tolerance evolvability in Candida albicans. Can. J. Microbiol. 2024, 70, 384–393. [Google Scholar] [CrossRef]
- Güner, P.; Aşkun, T.; Er, A. Antimicrobial potential of boron-containing compounds: Antibacterial, antifungal, and antimycobacterial activities. J. Boron 2025, 10, 111–120. [Google Scholar] [CrossRef]




| Solution | PVA (w/v%) | HA (w/v) | CS (w/v) | BA (w/vt) | TX-100 (w/vt) |
|---|---|---|---|---|---|
| PVA-CS | 10 | – | 1 | – | – |
| PVA-CS-HA | 10 | 0.5 | 1 | – | – |
| PVA-CS-HA-BA (0.25 BA) | 10 | 0.5 | 1 | 0.25 | 1 |
| PVA-CS-HA-BA (0.5 BA) | 10 | 0.5 | 1 | 0.5 | 1 |
| Solution | Voltage (kV) | Flow Rate (mL·h−1) | Distance (cm) | Spinning Duration (h) | Observation/Justification |
|---|---|---|---|---|---|
| PVA/CS | 25 | 1.0 | 12 | 3 | Stable Taylor cone; Baseline |
| PVA/CS/HA | 25 | 1.0 | 12 | 3 | Stable jet; Homogeneous HA |
| PVA/CS/HA/0.25BA | 27 | 1.6 | 12 | 3 | Increased for viscosity/viscoelasticity adaptation |
| PVA/CS/HA/0.5BA | 27 | 1.6 | 12 | 3 | Increased to prevent needle clogging and stabilize jet |
| Wavenumber (cm−1) | Assignment | Component | Intensity Observation |
|---|---|---|---|
| 3300 | O–H/N–H stretching | PVA/CS | Broadens with BA addition [60,61] |
| 2869–2989 | C–H stretching | PVA/CS | Stable polymer backbone [62,63] |
| 1592–1650 | Amide I and II | CS | Confirms CS presence [64,65] |
| 1365–1367 | B–O (Borate-ester) | BA | Increases from 0.25 to 0.5% BA [48,66] |
| 1030–1134 | PO43− stretching | HA | Validates HA integration [11,12,56] |
| 560–600 | PO43− bonding | HA | Confirms HA crystallinity [11,65,67] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Yavuz, Y.; Kartal, I.; Cesur, S.; Kanli, Z.; Kaya, E.; Tinaz, G.; Gunduz, O. Electrospun PVA/CS/HA/BA Nanofiber Scaffolds with Enhanced Mechanical Stability and Antifungal Activity for Bone Tissue Engineering. Materials 2026, 19, 412. https://doi.org/10.3390/ma19020412
Yavuz Y, Kartal I, Cesur S, Kanli Z, Kaya E, Tinaz G, Gunduz O. Electrospun PVA/CS/HA/BA Nanofiber Scaffolds with Enhanced Mechanical Stability and Antifungal Activity for Bone Tissue Engineering. Materials. 2026; 19(2):412. https://doi.org/10.3390/ma19020412
Chicago/Turabian StyleYavuz, Yagizer, Ilyas Kartal, Sumeyye Cesur, Zehra Kanli, Elif Kaya, Gulgun Tinaz, and Oguzhan Gunduz. 2026. "Electrospun PVA/CS/HA/BA Nanofiber Scaffolds with Enhanced Mechanical Stability and Antifungal Activity for Bone Tissue Engineering" Materials 19, no. 2: 412. https://doi.org/10.3390/ma19020412
APA StyleYavuz, Y., Kartal, I., Cesur, S., Kanli, Z., Kaya, E., Tinaz, G., & Gunduz, O. (2026). Electrospun PVA/CS/HA/BA Nanofiber Scaffolds with Enhanced Mechanical Stability and Antifungal Activity for Bone Tissue Engineering. Materials, 19(2), 412. https://doi.org/10.3390/ma19020412








