A Rapid Spheroidizing Annealing Process for High-Carbon Steel
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Microstructures
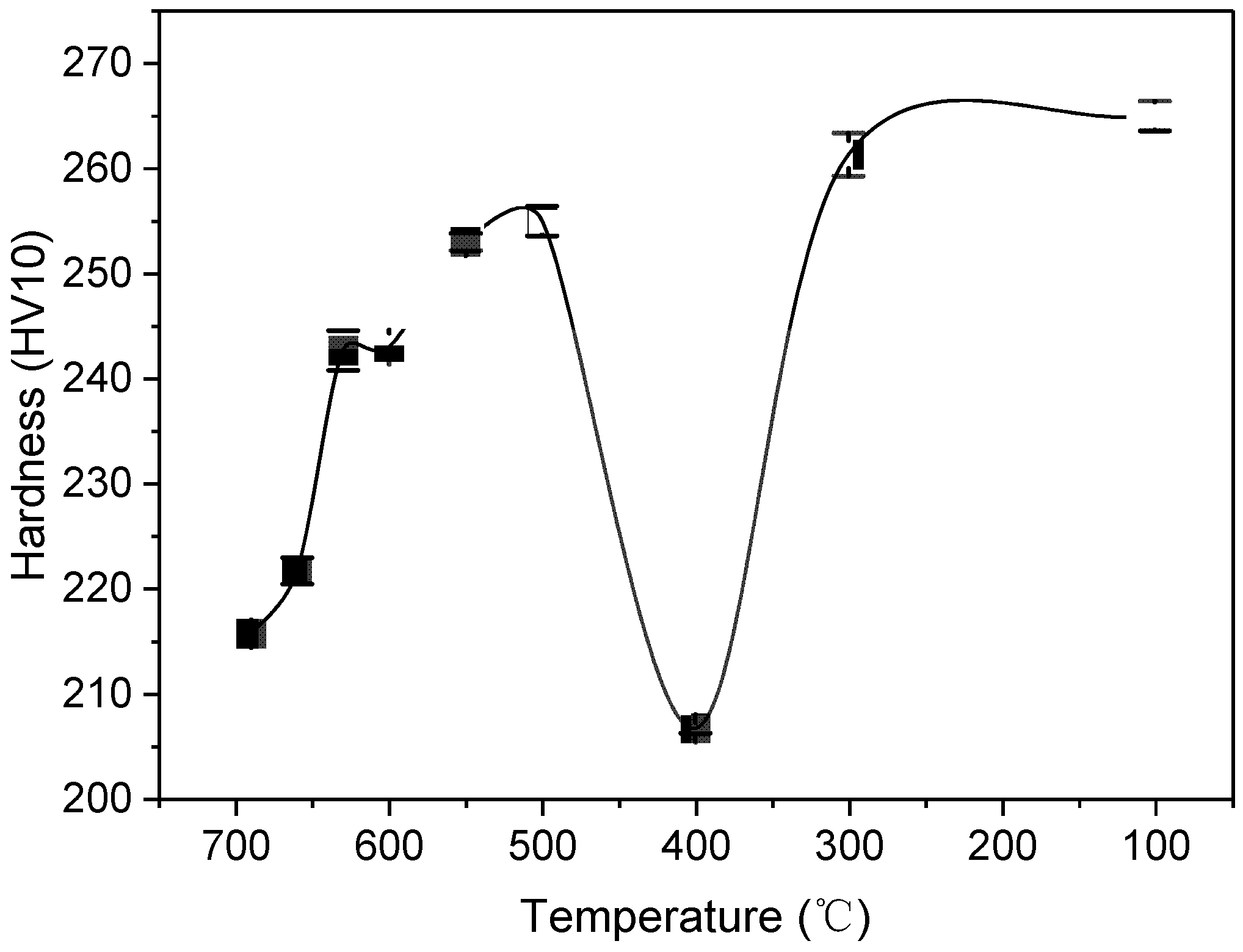
3.2. Hardness Tests
3.3. Spheroidization Mechanism
4. Conclusions
- (1)
- Controlling the pretreatment temperature within a lower range (e.g., 400 °C) promotes uniform precipitation of fine carbides during subsequent reheating, which facilitates the formation of a spherical or near-spherical pearlite microstructure. When the pretreatment temperature is too low, the carbides precipitated from martensite decomposition are excessively fine, requiring additional energy to drive their Ostwald ripening process. This consequently leads to a significant extension of the process duration needed to obtain spherical carbides of suitable size.
- (2)
- As the pretreatment temperature decreases from 690 °C to 100 °C, the hardness exhibits an initial increase, followed by a decrease at 400 °C, and then a subsequent increase. The significant hardness reduction to 206.7 HV at 400 °C represents an ideal hardness level comparable to that achieved through conventional spheroidizing annealing.
- (3)
- The decomposition of induced non-equilibrium phases (martensite/lower bainite) establishes an efficient spheroidization pathway. The fine carbides precipitated during phase transformation provide nucleation sites for spheroidized pearlite growth, while the excess vacancies and high-density dislocation networks released during decomposition enhance carbon mobility, ultimately promoting the spheroidization process. This process achieves significant microstructure refinement while significantly shortening the processing time. It will help save energy and time in industrial production.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhao, X.-Y.; Zhao, X.-M.; Dong, C.-Y.; Yang, Y.; Han, H.-B. Effect of Prior Microstructures on Cementite Dissolution Behavior During Subcritical Annealing of High Carbon Steels. Met. Mater. Int. 2022, 28, 1315–1327. [Google Scholar] [CrossRef]
- Spindola, M.O.; Buono, V.T.L. Effect of Intercritical Annealing with High Cooling Rate Associated with Cold Deformation and Subcritical Annealing on Microstructure and Mechanical Properties of SAE 52100. J. Mater. Res. Technol. 2020, 9, 9136–9141. [Google Scholar] [CrossRef]
- Gökmeşe, H.; Karadağ, H.B.; Taşdöğen, H. Effect of Various Spheroidizing Methods on Microstructure–Mechanical Properties and Wear Performance of PM High-Carbon Steel. Trans. Indian Inst. Met. 2022, 75, 435–447. [Google Scholar] [CrossRef]
- Yin, F.; Han, P.; Han, Q.; Wang, H.; Hua, L.; Cheng, G.J. Ultrastrong Gradient M50 Bearing Steel with Lath-Shape Nano-Martensite by Ultrasonic Shot Peening and Its Enhanced Wear Resistance at Elevated Temperature. Mater. Des. 2024, 239, 112786. [Google Scholar] [CrossRef]
- Kumar, R.; Dwivedi, R.K.; Arya, R.K.; Sonia, P.; Yadav, A.S.; Saxena, K.K.; Khan, M.I.; Moussa, S.B. Current Development of Carbide Free Bainitic and Retained Austenite on Wear Resistance in High Silicon Steel. J. Mater. Res. Technol. 2023, 24, 9171–9202. [Google Scholar] [CrossRef]
- Wang, S.; Cao, L.; Zhang, Z. Influence of Carbide Morphology on the Deformation and Fracture Mechanisms of Spheroidized 14CrMoR Steel. Metals 2019, 9, 1221. [Google Scholar] [CrossRef]
- Pandit, A.S.; Bhadeshia, H.K.D.H. Divorced Pearlite in Steels. Proc. R. Soc. A 2012, 468, 2767–2778. [Google Scholar] [CrossRef]
- Huo, X.D.; Guo, L.; He, K.N.; Huang, R. Effect of Spheroidizing Annealing Time on Microstructure and Hardness of GCr15 Bearing Steel. Adv. Mater. Res. 2012, 581–582, 928–931. [Google Scholar] [CrossRef]
- Oyama, T.; Sherby, O.D.; Wadsworth, J.; Walser, B. Application of the Divorced Eutectoid Transformation to the Development of Fine-Grained, Spheroidized Structures in Ultrahigh Carbon Steels. Scr. Metall. 1984, 18, 799–804. [Google Scholar] [CrossRef]
- Chen, Q.W.; Zhu, G.H.; Cao, S.M.; Zhao, A.M. Quick Spherodizing in GCr15 Steel by Mechanism of Divorced Eutectoid. Adv. Mater. Res. 2011, 295–297, 515–519. [Google Scholar] [CrossRef]
- Verhoeven, J.D.; Gibson, E.D. The Divorced Eutectoid Transformation in Steel. Metall. Mater. Trans. A 1998, 29, 1181–1189. [Google Scholar] [CrossRef]
- Verhoeven, J.D. The Role of the Divorced Eutectoid Transformation in the Spheroidization of 52100 Steel. Metall. Mater. Trans. A 2000, 31, 2431–2438. [Google Scholar] [CrossRef]
- Li, Z.-X.; Li, C.-S.; Kim, S.-H.; Suh, D.-W. Influence of Initial Pearlite Morphology on the Microstructure Evolution During Heat Treatment of 1.0C–1.5Cr Steel. Met. Mater. Int. 2019, 25, 9–17. [Google Scholar] [CrossRef]
- Li, Z.-X.; Li, C.-S.; Zhang, J.; Li, B.-Z.; Pang, X.-D. Microstructure of Hot Rolled 1.0C-1.5Cr Bearing Steel and Subsequent Spheroidization Annealing. Metall. Mater. Trans. A 2016, 47, 3607–3621. [Google Scholar] [CrossRef]
- Li, Y.; Chen, C.; Fan, Z.; Jiang, D.; Shuai, S.; Tu, T.; Ren, Z. Effects of Cooling Rate on Isothermal Spheroidizing Annealing of Hot-Rolled GCr15 Bearing Steel. J. Mater. Res. Technol. 2024, 31, 329–337. [Google Scholar] [CrossRef]
- Han, D.-X.; Du, L.-X.; Zhang, B.; Misra, R.D.K. Effect of Deformation on Deformation-Induced Carbides and Spheroidization in Bearing Steel. J. Mater. Sci. 2019, 54, 2612–2627. [Google Scholar] [CrossRef]
- Qian, D.; Chen, B.; Wang, F.; Wu, L. Rapid Spheroidizing Annealing via Combining Warm Deformation with Divorced Eutectoid Transformation in M50 Steel. Metals 2022, 12, 359. [Google Scholar] [CrossRef]
- Ji, H.; Liu, B.-S.; Ma, D.-S.; Chen, Z.-Z.; Li, D.-L.; Kang, A.-J.; Sun, L.-J. Behavior of Spheroidizing Annealing and Carbides of 7CrMn2Mo Steel. Trans. Mater. Heat Treat. 2011, 32, 115. [Google Scholar] [CrossRef]
- GB/T 4340.1; Metallic Materials—Vickers Hardness Test. National Standards of the People’s Republic of China: Beijing, China, 2005.
- Monia, S.; Varshney, A.; Gouthama; Sangal, S.; Kundu, S.; Samanta, S.; Mondal, K. Development of Highly Ductile Spheroidized Steel from High C (0.61 wt.% C) Low-Alloy Steel. J. Mater. Eng. Perform. 2015, 24, 4527–4542. [Google Scholar] [CrossRef]
- Li, Z.-X.; Li, C.-S.; Ren, J.-Y.; Li, B.-Z.; Suh, D.-W. Design of Online Spheroidization Process for 1.0C-1.5Cr Bearing Steel and Microstructure Analysis. Metall. Mater. Trans. A 2018, 49, 1782–1794. [Google Scholar] [CrossRef]
- Li, Z.-X.; Li, C.-S.; Zhang, J.; Qiao, B.; Li, Z.-Z. Effects of Annealing on Carbides Size and Distribution and Cold Formability of 1.0C-1.5Cr Bearing Steel. Metall. Mater. Trans. A 2015, 46, 3220–3231. [Google Scholar] [CrossRef]
- Legros, M.; Dehm, G.; Arzt, E.; Balk, T.J. Observation of Giant Diffusivity Along Dislocation Cores. Science 2008, 319, 1646–1649. [Google Scholar] [CrossRef]
- Nematollahi, G.A.; Grabowski, B.; Raabe, D.; Neugebauer, J. Multiscale Description of Carbon-Supersaturated Ferrite in Severely Drawn Pearlitic Wires. Acta Mater. 2016, 111, 321–334. [Google Scholar] [CrossRef]
- Sun, H.; Bi, P.; Hashimoto, N.; Oka, H.; Xue, Q.; Isobe, S. Controlling the Diffusion Behavior in Diffusion Bonded Reduced Activation Cr0.8FeMn1.3Ni1.3 High Entropy Alloy/316SS Joints through Dislocations. Mater. Des. 2025, 257, 114447. [Google Scholar] [CrossRef]





| C | Si | Mn | Cr | Fe |
|---|---|---|---|---|
| ≤0.80 | ≤0.3 | ≤0.80 | ≤0.20 | - |
| Temperature/°C | 690 | 660 | 630 |
|---|---|---|---|
| Interlamellar Spacing/nm | 480~1350 | 304~660 | 207~500 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Li, B.; Tong, Z.; Zhao, M.; Wu, X.; Zheng, W. A Rapid Spheroidizing Annealing Process for High-Carbon Steel. Materials 2026, 19, 249. https://doi.org/10.3390/ma19020249
Li B, Tong Z, Zhao M, Wu X, Zheng W. A Rapid Spheroidizing Annealing Process for High-Carbon Steel. Materials. 2026; 19(2):249. https://doi.org/10.3390/ma19020249
Chicago/Turabian StyleLi, Bei, Zhi Tong, Mengying Zhao, Xinlang Wu, and Wenyue Zheng. 2026. "A Rapid Spheroidizing Annealing Process for High-Carbon Steel" Materials 19, no. 2: 249. https://doi.org/10.3390/ma19020249
APA StyleLi, B., Tong, Z., Zhao, M., Wu, X., & Zheng, W. (2026). A Rapid Spheroidizing Annealing Process for High-Carbon Steel. Materials, 19(2), 249. https://doi.org/10.3390/ma19020249






