Investigation of Degradation Behavior and Mechanical Performance Deterioration of Magnesium Alloys in Hank’s Solution
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Microstructure Characterization
2.3. Degradation Behavior
2.4. Mechanical Integrity After Degradation
3. Results
3.1. Microstructure
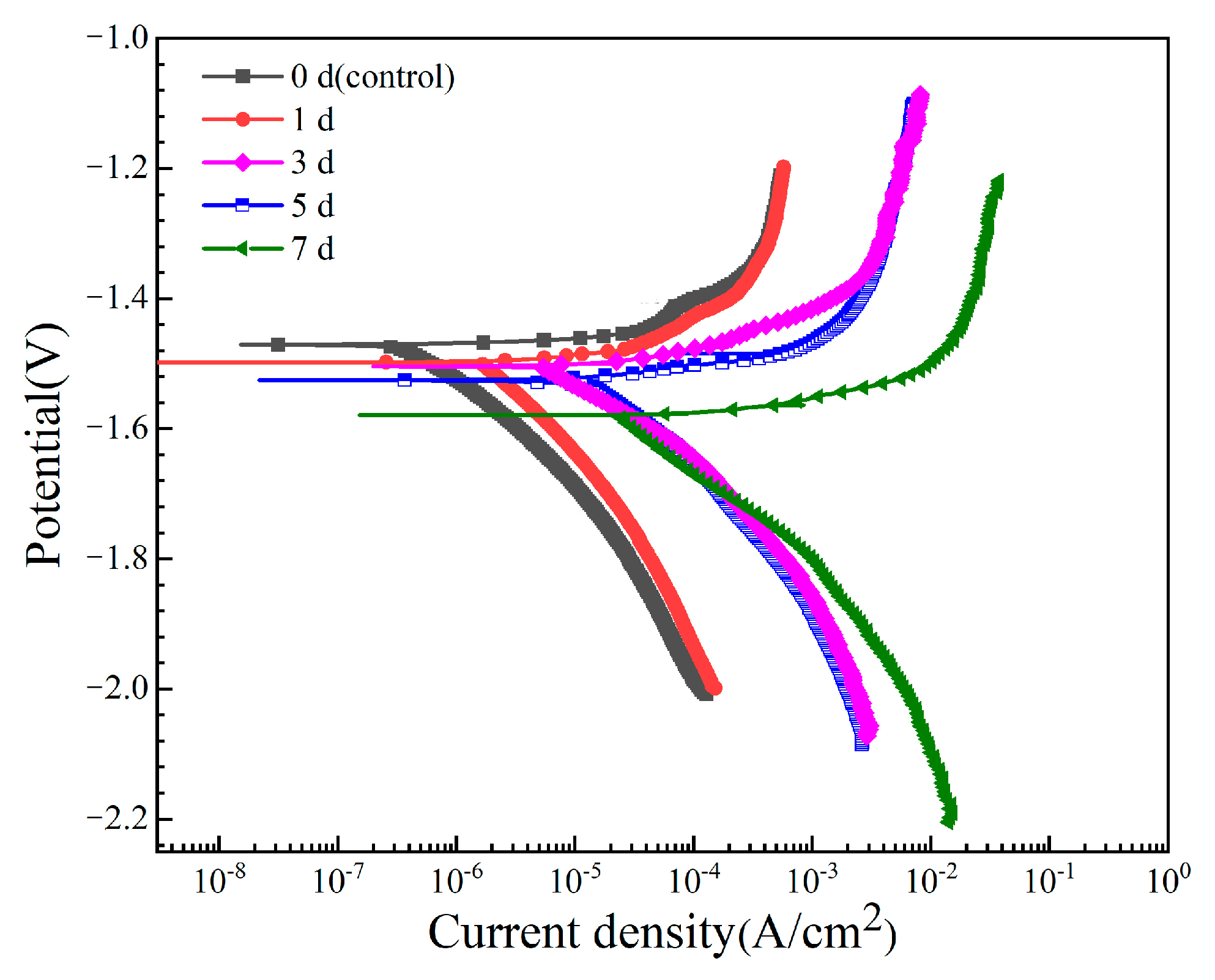
3.2. Degradation Characteristics of Mg-6Zn-0.5Cu Alloy Under Varying Immersion Durations
- (1)
- Electrochemical behavior
- (2)
- Weight loss and corrosion morphologies
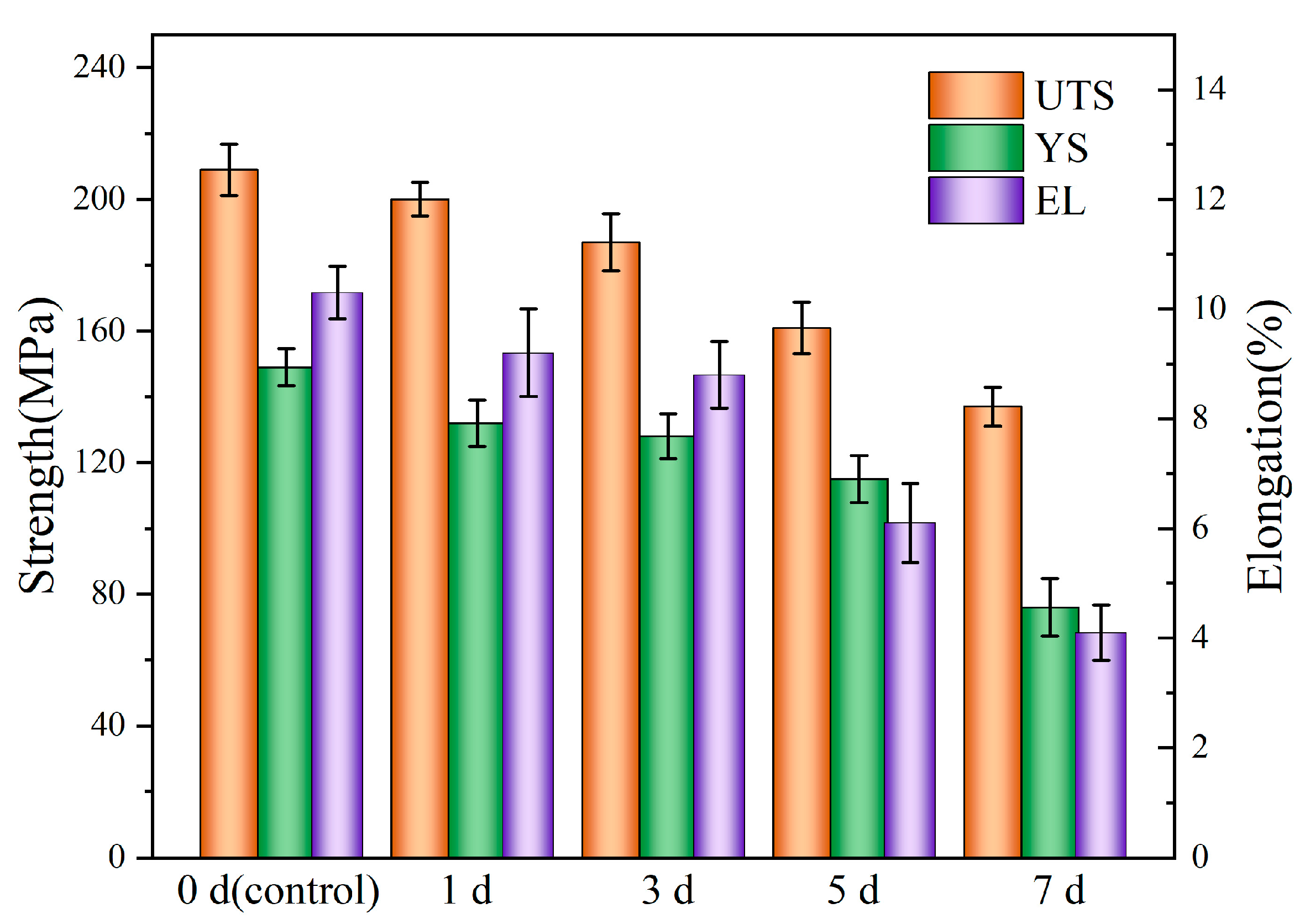
3.3. Mechanical Integrity of Mg-6Zn-0.5Cu Alloy After Varying Immersion Durations
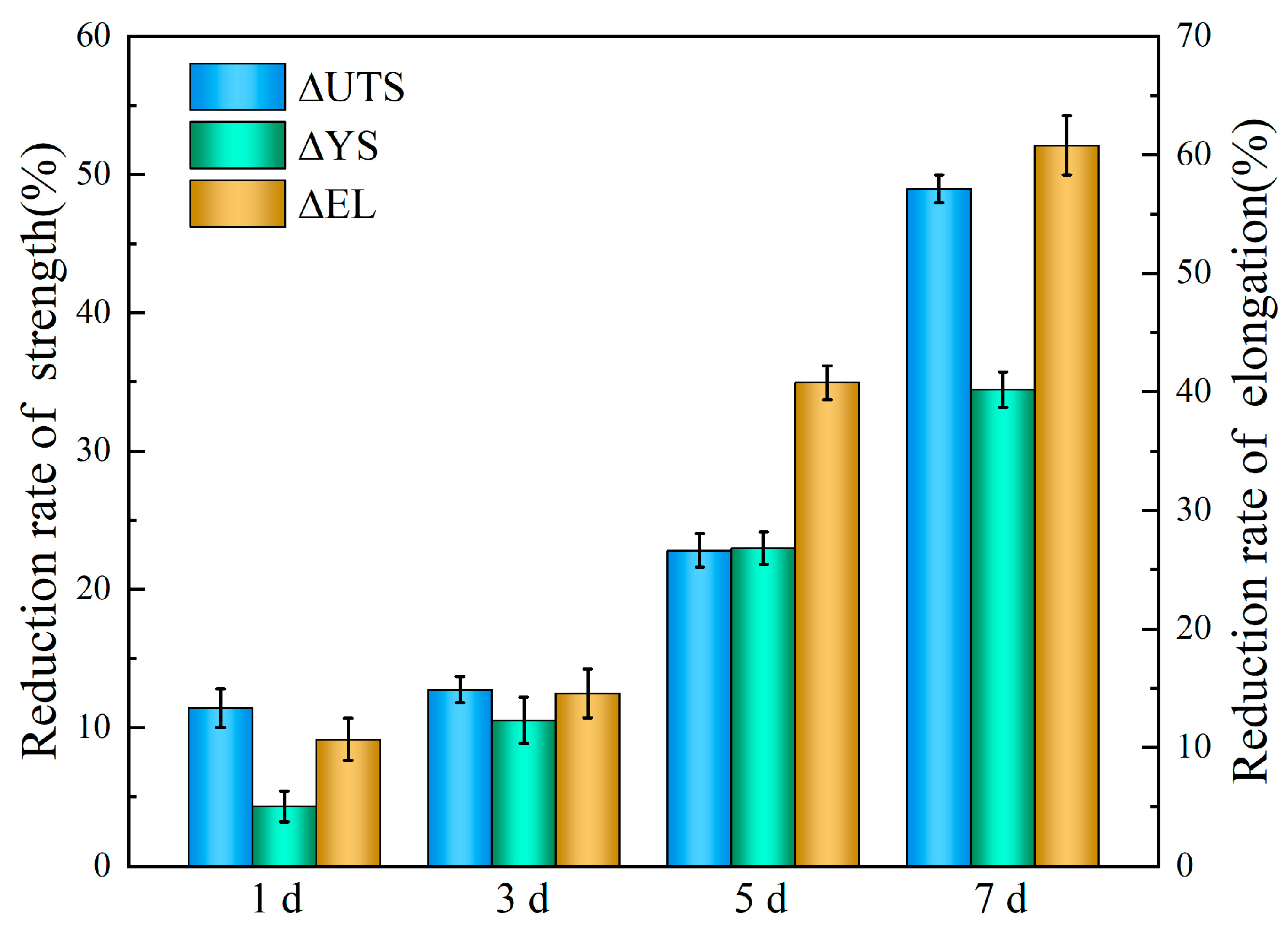
4. Discussion
5. Conclusions
- (1)
- After 7 days’ immersion, the UTS and EL decreased markedly by 34.4% and 60.1%, respectively. While the weight loss of 0.054 mg∙cm−2∙d corresponds to a degradation rate of 0.11 mm/y.
- (2)
- The results indicate that the alloy is susceptible to localized corrosion under this physiological environment with mechanical deterioration occurring at a rate that exceeds the corrosion.
- (3)
- The pronounced loss in mechanical performance is attributed to localized corrosion associated with secondary phases. Under tensile testing, the resulting corrosion pits serve as stress concentrators, promoting microcrack initiation and leading to premature fracture of the alloy.
- (4)
- Even a modest corrosion rate can result in severe mechanical degradation due to localized attack, highlighting the limitation of relying solely on corrosion rate to predict the service life of biodegradable Mg implants.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Mei, D.; Zhang, Q.Y.; Li, Y.Q.; Liu, M.; Li, W.; Jiang, P.; Hou, R.; Zhu, S.; Wang, L.; Guan, S. The misalignment between degradation rate and mechanical integrity of Mg-Zn-Y-Nd alloy during the degradation evaluation in modified Hank’s solutions. J. Magnes. Alloys 2024, 12, 3661–3674. [Google Scholar] [CrossRef]
- She, J.; Chen, J.; Xiong, X.M.; Yang, Y.; Peng, X.; Chen, D.; Pan, F. Research advances of magnesium and magnesium alloys globally in 2023. J. Magnes. Alloys 2024, 12, 3441–3475. [Google Scholar] [CrossRef]
- Shunmugasamy, V.C.; AbdelGawad, M.; Sahail, M.U.; Ibrahim, T.; Khan, T.; Seers, T.D.; Mansoor, B. In vitro and in vivo study on fine-grained Mg-Zn-RE-Zr alloy as a biodegradable orthopedic implant produced by friction stir processing. Bioact. Mater. 2023, 28, 448–466. [Google Scholar]
- Guo, Y.; Yu, Y.J.; Han, L.P.; Ma, S.; Zhao, J.; Chen, H.; Yang, Z.; Zhang, F.; Xia, Y.; Zhou, Y. Biocompatibility and osteogenic activity of guided bone regeneration membrane based on chitosan-coated magnesium alloy. Mater. Sci. Eng. C 2019, 100, 226–235. [Google Scholar] [CrossRef]
- Bian, D.; Zhou, X.C.; Liu, J.N.; Li, W.; Shen, D.; Zheng, Y.; Gu, W.; Jiang, J.; Li, M.; Chu, X.; et al. Degradation behaviors and in-vivo biocompatibility of a rare earth- and aluminum-free magnesium-based stent. Acta Biomater. 2021, 124, 382–397. [Google Scholar] [CrossRef] [PubMed]
- Sahu, M.R.; Yamamoto, A. An overview of the recent developments in biodegradable Mg-Zn alloy. J. Magnes. Alloys 2025, 13, 486–509. [Google Scholar] [CrossRef]
- Niranjan, C.A.; Raghavendra, T.; Rao, M.P.; Siddaraju, C.; Gupta, M.; Jain, V.K.S.; Aishwarya, R. Magnesium alloys as extremely promising alternatives for temporary orthopedic implants-A review. J. Magnes. Alloys 2023, 11, 2688–2718. [Google Scholar] [CrossRef]
- Chen, L.X.; Blawert, C.; Yang, J.; Hou, R.; Wang, X.; Zheludkevich, M.L.; Li, W. The stress corrosion cracking behaviour of biomedical Mg-1Zn alloy in synthetic or natural biological media. Corros. Sci. 2020, 175, 108876. [Google Scholar] [CrossRef]
- Hou, R.Q.; Victoria-Hernandez, J.; Jiang, P.; Willumeit-Römer, R.; Luthringer-Feyerabend, B.; Yi, S.; Letzig, D.; Feyerabend, F. In vitro evaluation of the ZX11 magnesium alloy as potential bone plate: Degradability and mechanical integrity. Acta Biomater. 2019, 97, 608–622. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.H.; Liu, Y.J.; Wang, W.; Zhang, Y. Effects of electrolyte component in simulated body fluid on the corrosion behavior and mechanical integrity of magnesium. Corros. Sci. 2015, 98, 201–210. [Google Scholar] [CrossRef]
- Jiang, P.L.; Blawert, C.; Bohlen, J.; Zheludkevich, M.L. Corrosion performance, corrosion fatigue behavior and mechanical integrity of an extruded Mg4Zn0.2Sn alloy. J. Mater. Sci. Technol. 2020, 59, 107–116. [Google Scholar] [CrossRef]
- Fu, Q.Y.; Liang, W.Q.; Huang, J.X.; Jin, W.; Guo, B.; Li, P.; Xu, S.; Chu, P.K.; Yu, Z. Research perspective and prospective of additive manufacturing of biodegradable magnesium-based materials. J. Magnes. Alloys 2023, 11, 1485–1504. [Google Scholar] [CrossRef]
- Gu, X.N.; Zheng, Y.F.; Cheng, Y.; Zhong, S.; Xi, T. In vitro corrosion and biocompatibility of binary magnesium alloys. Biomaterials 2009, 30, 484–498. [Google Scholar] [CrossRef] [PubMed]
- Hou, L.D.; Li, Z.; Zhao, H.; Pan, Y.; Pavlinich, S.; Liu, X.; Li, X.; Zheng, Y.; Li, L. Microstructure, Mechanical Properties, Corrosion behavior and biocompatibility of as-extruded biodegradable Mg-3Sn-1Zn-0.5Mn alloy. J. Mater. Sci. Technol. 2016, 32, 874–882. [Google Scholar] [CrossRef]
- Zhao, D.L.; Wang, T.T.; Kuhlmann, J.; Dong, Z.; Chen, S.; Joshi, M.; Salunke, P.; Shanov, V.N.; Hong, D.; Kumta, P.N.; et al. In vivo monitoring the biodegradation of magnesium alloys with an electrochemical H2 sensor. Acta Biomater. 2016, 36, 361–368. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.N.; Bian, D.; Zheng, Y.F.; Chu, X.; Lin, Y.; Wang, M.; Lin, Z.; Li, M.; Zhang, Y.; Guan, S. Comparative in vitro study on binary Mg-RE (Sc, Y, La, Ce, Pr, Nd, Sm, Eu, Gd, Tb, Dy, Ho, Er, Tm, Yb and Lu) alloy systems. Acta Biomater. 2020, 102, 508–528. [Google Scholar] [CrossRef]
- Jia, H.M.; Feng, X.H.; Yang, Y.S. Microstructure and mechanical properties of directionally solidified Mg–Zn alloy as a biomaterial. Mater. Sci. Technol. 2019, 35, 2165–2172. [Google Scholar] [CrossRef]
- Dong, Q.S.; Jiang, J.H.; Zhang, J.H.; Hu, Z.; Zhang, X. Clarifying stress corrosion cracking behavior of biomedical Mg-Gd-Zn-Zr alloy. J. Magnes. Alloys 2025, 13, 3450–3465. [Google Scholar] [CrossRef]
- Jia, H.M.; Li, J.R.; Li, Y.J.; Wang, M.; Luo, S.; Zhang, Z. Mechanical properties and stress corrosion cracking behavior of a novel Mg-6Zn-1Y-0.5Cu-0.5Zr alloy. J. Alloys Compd. 2022, 911, 164995. [Google Scholar] [CrossRef]
- Liu, M.Y.; Wang, J.F.; Zhu, S.J.; Zhang, Y.; Sun, Y.; Wang, L.; Guan, S. Corrosion fatigue of the extruded Mg-Zn-Y-Nd alloy in simulated body fluid. J. Alloys Compd. 2020, 8, 231–240. [Google Scholar] [CrossRef]
- Xiong, Y.; Shen, Y.S.; He, L.Y.; Yang, Z.; Song, R. Stress corrosion cracking behavior of LSP/MAO treated magnesium alloy during SSRT in a simulated body fluid. J. Alloys Compd. 2020, 822, 153707. [Google Scholar] [CrossRef]
- GB/T 228.1-2021; Standardization Administration of China. Metallic Materials-Tensile Testing-Part 1: Method of Test at Room Temperature. China Standard Press: Beijing, China, 2021.
- Khan, S.A.; Bhuiyan, M.S.; Miyashita, Y.; Mutoh, Y.; Koike, T. Corrosion fatigue behavior of die-cast and shot-blasted AM60 magnesium alloy. Mater. Sci. Eng. A 2011, 528, 1961–1966. [Google Scholar] [CrossRef]
- Jia, H.M.; Guo, W.Z. Anisotropic tensile and degradation properties of as-extruded Mg-6Zn-2Sn-0.5Mn alloy. Mater. Today Commun. 2024, 38, 108044. [Google Scholar] [CrossRef]
- Jia, H.M.; Ren, X.W.; Wang, M.R. Effect of asymmetric rolling on corrosion behavior of AZ31B magnesium alloy sheet. Mater. Sci. Technol. 2022, 38, 987–994. [Google Scholar] [CrossRef]
- Kirkland, N.T.; Birbilis, N.; Staiger, M.P. Assessing the corrosion of biodegradable magnesium implants: A critical review of current methodologies and their limitations. Acta Biomater. 2012, 8, 925–936. [Google Scholar] [CrossRef]
- Shi, Z.M.; Liu, M.; Atrens, A. Measurement of the corrosion rate of magnesium alloys using Tafel extrapolation. Corros. Sci. 2010, 52, 579–588. [Google Scholar] [CrossRef]
- Wang, H.X.; Song, Y.W.; Shan, D.Y.; Han, E.-H. Effects of corrosive media on the localized corrosion forms of Mg-3Zn alloy. Corros. Commun. 2021, 2, 24–32. [Google Scholar] [CrossRef]
- Li, D.H.; Hu, H.X.; Pan, H.D.; Wang, Z.; An, W.; Zheng, Y. Corrosion of Ti75 alloy in a 3.5 wt% NaCl solution containing different concentrations of Cu2+. Corros. Sci. 2025, 243, 112567. [Google Scholar] [CrossRef]
- Baek, S.M.; Kim, B.; Park, S.S. Influence of intermetallic particles on the corrosion properties of extruded ZK60 Mg alloy containing Cu. Metals 2018, 8, 323. [Google Scholar] [CrossRef]
- Erinc, M.; Sillekens, W.H.; Mannens, R.; Werkhoven, R.J. Applicability of existing magnesium alloys as biomedical implant materials. Magnes. Technol. 2009, 8, 209–214. [Google Scholar]
- Thirumalaikumarasamy, D.; Shanmugam, K.; Balasubramanian, V. Influence of chloride ion concentration on immersion corrosion behavior of plasma sprayed alumina coatings on AZ31B magnesium alloy. J. Magnes. Alloys 2014, 2, 325–334. [Google Scholar] [CrossRef]
- Jia, H.M.; Guo, W.Z.; Li, Y.F.; Xi, Y.; Xu, S.; Wang, L. Microstructure and degradation behavior of Mg-6Zn-1Y-0.5Zr-0.5Cu alloy for fracturing ball applications. Mater. Today Commun. 2025, 44, 112100. [Google Scholar] [CrossRef]

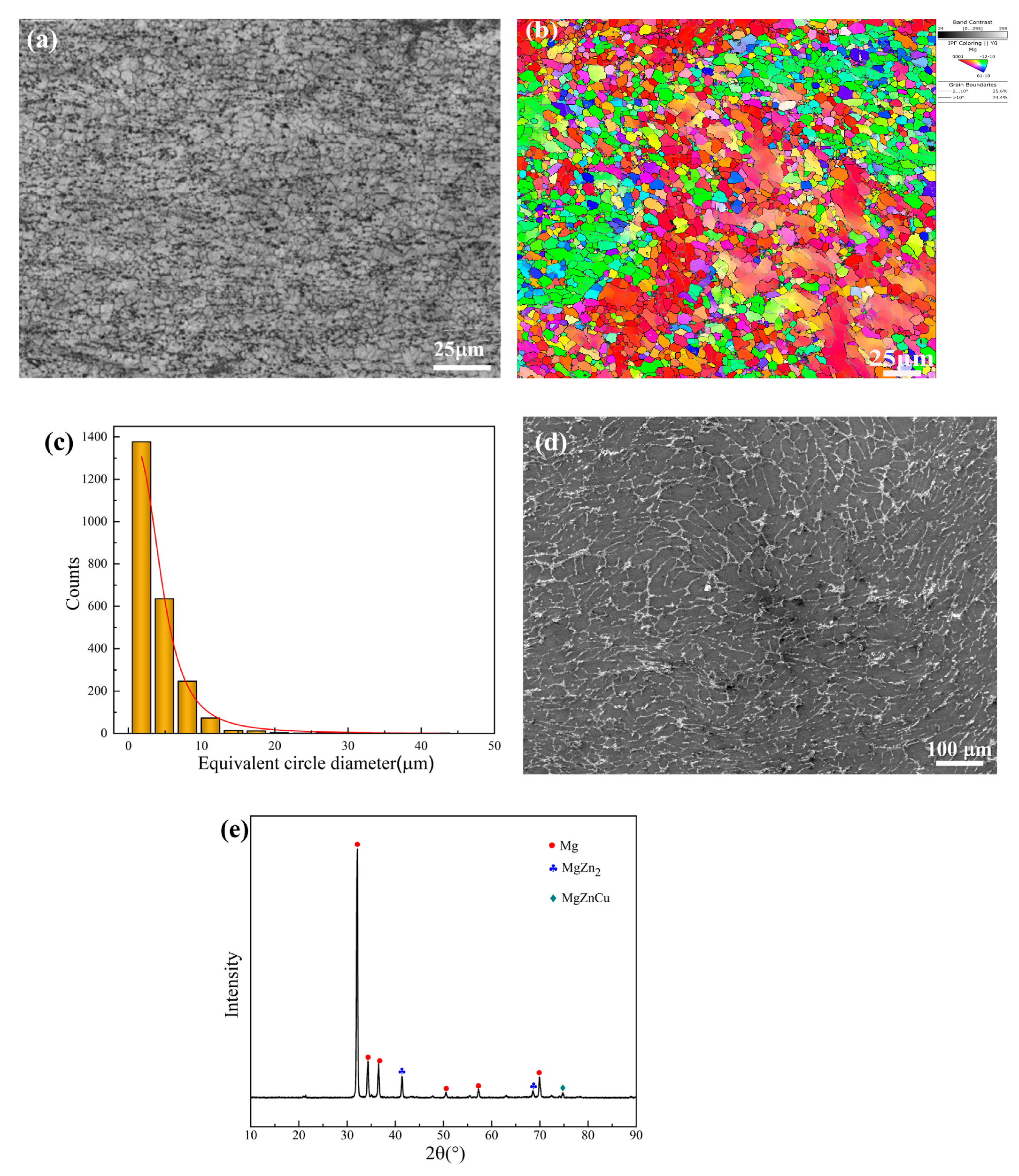







| Element | Zn | Cu | Mg |
|---|---|---|---|
| Content | 6 | 0.5 | Bal. |
| NaCl | KCl | CaCl2 | Na2HPO4∙7H2O | MgSO4∙7H2O | NaHCO3 | KH2PO4 | C6H12O6 |
|---|---|---|---|---|---|---|---|
| 8 | 0.4 | 0.14 | 0.09 | 0.2 | 0.35 | 0.06 | 1 |
| Time (d) | Ecorr (V) | icorr (A/cm2) |
|---|---|---|
| 0 | −1.482 ± 0.016 | (6.28 ± 0.153) × 10−7 |
| 1 | −1.501 ± 0.018 | (2.05 ± 0.129) × 10−6 |
| 3 | −1.507 ± 0.019 | (6.19 ± 0.190) × 10−6 |
| 5 | −1.528 ± 0.027 | (1.71 ± 0.341) × 10−5 |
| 7 | −1.591 ± 0.044 | (3.06 ± 0.553) × 10−5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jia, H.; Li, Y.; Xu, S.; Xi, Y.; Gui, W. Investigation of Degradation Behavior and Mechanical Performance Deterioration of Magnesium Alloys in Hank’s Solution. Materials 2025, 18, 5102. https://doi.org/10.3390/ma18225102
Jia H, Li Y, Xu S, Xi Y, Gui W. Investigation of Degradation Behavior and Mechanical Performance Deterioration of Magnesium Alloys in Hank’s Solution. Materials. 2025; 18(22):5102. https://doi.org/10.3390/ma18225102
Chicago/Turabian StyleJia, Hongmin, Yifan Li, Shanna Xu, Yuntao Xi, and Weimin Gui. 2025. "Investigation of Degradation Behavior and Mechanical Performance Deterioration of Magnesium Alloys in Hank’s Solution" Materials 18, no. 22: 5102. https://doi.org/10.3390/ma18225102
APA StyleJia, H., Li, Y., Xu, S., Xi, Y., & Gui, W. (2025). Investigation of Degradation Behavior and Mechanical Performance Deterioration of Magnesium Alloys in Hank’s Solution. Materials, 18(22), 5102. https://doi.org/10.3390/ma18225102






