Influence of Human Blood Contamination on Microhardness of Glass-Ionomer Cements and Glass-Hybrid Material
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Samples
2.2. Exposure of Samples in Experimental Groups to Human Blood
2.3. Vickers Microhardness Test and Thermocycling
2.4. Statistical Analysis
3. Results
3.1. Microhardness of Control Groups Before Thermocycling
3.2. Microhardness of Control Groups After Thermocycling
3.3. Microhardness of Experimental Groups Before Thermocycling
3.4. Microhardness of Experimental Groups After Thermocycling
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wilson, A.D.; Kent, B.E. A new translucent cement for dentistry. The glass ionomer cement. Br. Dent. J. 1972, 132, 133–135. [Google Scholar] [CrossRef]
- Mickenautsch, S. High-viscosity glass-ionomer cements for direct posterior tooth restorations in permanent teeth: The evidence in brief. J. Dent. 2017, 55, 121–123. [Google Scholar] [CrossRef]
- Gok Baba, M.; Kirzioglu, Z.; Ceyhan, D. One-year clinical evaluation of two high-viscosity glass-ionomer cements in class II restorations of primary molars. Aust. Dent. J. 2021, 66, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Borburema Neves, A.; Gracindo Lopes, L.I.; Gomes Bergstrom, T.; Saddock Sá da Silva, A.; Tadeu Lopes, R.; de Almeida Neves, A. Porosity and pore size distribution in high-viscosity and conventional glass ionomer cements: A micro-computed tomography study. Restor. Dent. Endod. 2021, 46, e57. [Google Scholar] [CrossRef] [PubMed]
- Kielbassa, A.M.; Oehme, E.P.; Shakavets, N.; Wolgin, M. In vitro wear of (resin-coated) high-viscosity glass ionomer cements and glass hybrid restorative systems. J. Dent. 2021, 105, 103554. [Google Scholar] [CrossRef]
- Gaviria, J.; Quijano, J.; Ruiz, P. Computational study of the formation reaction of polyacid precursors of glass-ionomer materials. J. Mater. Sci. Chem. Eng. 2021, 9, 1–9. [Google Scholar] [CrossRef]
- Vetromilla, B.M.; Opdam, N.J.; Leida, F.L.; Sarkis-Onofre, R.; Demarco, F.F.; van der Loo, M.P.J.; Cenci, M.S.; Pereira-Cenci, T. Treatment options for large posterior restorations: A systematic review and network meta-analysis. J. Am. Dent. Assoc. 2020, 151, 614–624. [Google Scholar] [CrossRef] [PubMed]
- Brkanović, S.; Ivanišević, A.; Miletić, I.; Mezdić, D.; Jukić Krmek, S. Effect of Nano-Filled Protective Coating and Different pH Environment on Wear Resistance of New Glass Hybrid Restorative Material. Materials 2021, 14, 755. [Google Scholar] [CrossRef]
- Joshi, G.; Heiss, M. Glass-Hybrid Technology for Long-Term Restorations. Compend. Contin. Educ. Dent. 2021, 42 (Suppl. S1), 2–5. [Google Scholar]
- Lohbauer, U.; Krämer, N.; Siedschlag, G.; Schubert, E.W.; Lauerer, B.; Müller, F.A.; Petschelt, A.; Ebert, J. Strength and wear resistance of a dental glass-ionomer cement with a novel nanofilled resin coating. Am. J. Dent. 2011, 24, 124–128. [Google Scholar]
- Chuenarrom, C.; Benjakul, P.; Daosodsai, P. Effect of indentation load and time on Knoop and Vickers microhardness tests for enamel and dentin. Mater. Res. 2009, 12, 473–476. [Google Scholar] [CrossRef]
- Okada, K.; Tosaki, S.; Hirota, K.; Hume, W.R. Surface hardness change of restorative filling materials stored in saliva. Dent. Mater. 2001, 17, 34–39. [Google Scholar] [CrossRef]
- Ben Ghorbal, G.; Tricoteaux, A.; Thuault, A.; Louis, G.; Chicot, D. Comparison of conventional Knoop and Vickers hardness of ceramic materials. J. Eur. Ceram. Soc. 2017, 37, 2531–2535. [Google Scholar] [CrossRef]
- Poggio, C.; Viola, M.; Mirando, M.; Chiesa, M.; Beltrami, R.; Colombo, M. Microhardness of different esthetic restorative materials: Evaluation and comparison after exposure to acidic drink. Dent. Res. J. 2018, 15, 166–172. [Google Scholar] [CrossRef]
- Nekoofar, M.H.; Stone, D.; Caldwell, S.; Bahjri, K.; Dummer, P.M.H. The effect of blood contamination on dentin bonding: A systematic review and meta-analysis. J. Dent. 2021, 113, 103787. [Google Scholar]
- Lund, R.G.; Da Silveira, I.A.; Ribeiro, J.S.; Rubin, D.; Peralta, S.L.; Cuevas-Suárez, C.E.; Piva, E. Influence of blood contamination and decontamination procedures on bond strength of a two-step etch and rinse adhesive system. Eur. J. Gen. Dent. 2019, 8, 71–75. [Google Scholar] [CrossRef]
- Mar, B.; Ekambaram, M.; Li, K.C.; Zwirner, J.; Mei, M.L. The influence of saliva and blood contamination on bonding between resin-modified glass ionomer cements and resin composite. Oper. Dent. 2023, 48, 218–225. [Google Scholar] [CrossRef]
- Freeman, R.; Varanasi, S.; Meyers, I.A.; Symons, A.L. Effect of air abrasion and thermocycling on resin adaptation and shear bond strength to dentin for an etch-and-rinse and self-etch resin adhesive. Dent. Mater. J. 2012, 31, 180–188. [Google Scholar] [CrossRef]
- Cavalcanti, A.N.; Mitsui, F.H.O.; Ambrosano, G.M.B.; Marchi, G.M. Influence of adhesive systems and flowable composite lining on bond strength of class II restorations submitted thermal and mechanical stresses. J. Biomed. Mater. Res. B Appl. Biomater. 2007, 80, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Hickel, R.; Roulet, J.F.; Bayne, S.; Heintze, S.D.; Mjör, I.A.; Peters, M.; Rousson, V.; Randall, R.; Schmalz, G.; Tyas, M.; et al. Recommendations for conducting controlled clinical studies of dental restorative materials. Int. Dent. J. 2007, 57, 300–302. [Google Scholar] [CrossRef]
- Opdam, N.J.M.; Collares, K.; Hickel, R.; Bayne, S.C.; Loomans, B.A.; Cenci, M.S.; Lynch, C.D.; Correa, M.B.; Demarco, F.; Schwendicke, F.; et al. Clinical studies in restorative dentistry: New directions and new demands. Dent. Mater. 2018, 34, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.L.; Kim, Y.K.; Kim, K.H.; Kwon, T.Y. Changes in degree of conversion and microhardness of dental resin cements. Oper. Dent. 2010, 35, 203–210. [Google Scholar] [CrossRef]
- Moshaverinia, M.; Navas, A.; Jahedmanesh, N.; Shah, K.C.; Moshaverinia, A.; Ansari, S. Comparative evaluation of the physical properties of a reinforced glass ionomer dental restorative material. J. Prosthet. Dent. 2019, 122, 154–159. [Google Scholar] [CrossRef] [PubMed]
- Bala, O.; Arisu, H.D.; Yikilgan, I.; Arslan, S.; Gullu, A. Evaluation of surface roughness and hardness of different glass ionomer cements. Eur. J. Dent. 2012, 6, 79–86. [Google Scholar] [CrossRef]
- Alqasabi, S.Y.; Sulimany, A.M.; Almohareb, T.; Alayad, A.; Bawazir, O.A. The Effect of Different Coating Agents on the Microhardness, Water Sorption, and Solubility of EQUIA Forte® HT. Coatings 2024, 14, 751. [Google Scholar] [CrossRef]
- Handoko, M.W.; Tjandrawinata, R. Octarina. The effect of nanofilled resin coating on the hardness of glass ionomer cement. Sci. Dent. J. 2020, 4, 97. [Google Scholar]
- Faraji, F.; Heshmat, H.; Banava, S. Effect of protective coating on microhardness of a new glass ionomer cement: Nanofilled coating versus unfilled resin. J. Conserv. Dent. 2017, 20, 260–263. [Google Scholar] [CrossRef]
- Ryu, W.; Park, H.; Lee, J.; Seo, H. Effect of Nano-Filled Protective Coating on Microhardness and Wear Resistance of Glass-Ionomer Cements. J. Korean Acad. Pediatr. Dent. 2019, 46, 226–232. [Google Scholar] [CrossRef]
- Noppawong, S.; Pratabsingha, J.; Thamsoonthorn, C.; Vichathai, W.; Saikaew, P. Bond Strengths of Universal Adhesives to Dentin Contaminated with a Hemostatic Agent. J. Adhes. Dent. 2022, 24, 421–426. [Google Scholar]
- Patel, S.; Hamer, S. A simple guide to using dental dam. Br. Dent. J. 2021, 230, 644–650. [Google Scholar] [CrossRef]
- Chen, A.M.; Ekambaram, M.; Li, K.C.; Cooper, P.R.; Mei, M.L. A scoping review of the influence of clinical contaminants on bond strength in direct adhesive restorative procedures. J. Dent. 2024, 145, 104985. [Google Scholar] [CrossRef]
- Moinzadeh, A.T.; Aznar Portoles, C.; Schembri Wismayer, P.; Camilleri, J. Bioactivity Potential of EndoSequence BC RRM Putty. J. Endod. 2016, 42, 615–621. [Google Scholar] [CrossRef]
- Nekoofar, M.H.; Davies, T.E.; Stone, D.; Basturk, F.B.; Dummer, P.M.H. Microstructure and chemical analysis of blood-contaminated mineral trioxide aggregate. Int. Endod. J. 2011, 44, 1011–1018. [Google Scholar] [CrossRef] [PubMed]
- Imataki, R.; Shinonaga, Y.; Nishimura, T.; Abe, Y.; Arita, K. Mechanical and Functional Properties of a Novel Apatite-Ionomer Cement for Prevention and Remineralization of Dental Caries. Materials 2019, 12, 3998. [Google Scholar] [CrossRef]
- Nicholson, J.W.; Anstice, H.M. The physical chemistry of resin modified glass ionomers. J. Mater. Sci. Mater. Med. 1994, 5, 119–122. [Google Scholar] [CrossRef]
- Ivica, A.; Šalinović, I.; Jukić Krmek, S.; Garoushi, S.; Lassila, L.; Säilynoja, E.; Miletić, I. Mechanical Properties and Ion Release from Fibre-Reinforced Glass Ionomer Cement. Polymers 2024, 16, 607. [Google Scholar] [CrossRef] [PubMed]
- Shimada, Y.; Hokii, Y.; Yamamoto, K.; Fukushima, K.; Kumagai, T. Evaluation of hardness increase of GIC restorative surface in saliva. In Proceedings of the ConsEuro 2015, London, UK, 14–16 May 2015. [Google Scholar]
- Gale, M.S.; Darvell, B.W. Thermal cycling procedures for laboratory testing of dental restorations. J. Dent. 1999, 27, 89–99. [Google Scholar] [CrossRef]
- Nicholson, J.W. Maturation processes in glass-ionomer dental cements. Acta Biomater. Odontol. Scand. 2018, 4, 63–71. [Google Scholar] [CrossRef]

| Material | Type of Material | Manufacturer | Composition |
|---|---|---|---|
| Fuji II LC | Resin-modified GIC | GC (Tokyo, Japan) | Liquid: 24% polyacrylic acid; 25% distilled water; 35% HEMA; 6% tartaric acid; 0.1% camphorquinone; 2, 2, 4 TMHEDC; TEGDMA. * Powder: fluoroaluminosilicate glass |
| Fuji IX GP Extra | High-viscosity GIC | GC (Tokyo, Japan) | Liquid: 50% distilled water; 40% polyacrylic and tartaric acid; 10% polybasic carboxylic acid. Powder: 95% fluorosilicate glass; 5% polyacrylic acid |
| EQUIA Forte HT | Glass-hybrid material | GC (Tokyo, Japan) | Liquid: 40-aqueous solution of polyacrylic acid. Powder: 95% fluorosilicate glass; 5% polyacrylic acid |
| EQUIA Forte Coat | Low viscosity nanofilled resin | GC (Tokyo, Japan) | 40–50% Methyl methacrylate 10–15% colloidal silica 0.09% camphorquinone 20–40% urethane methacrylate 1–5% phosphate ester monomer |
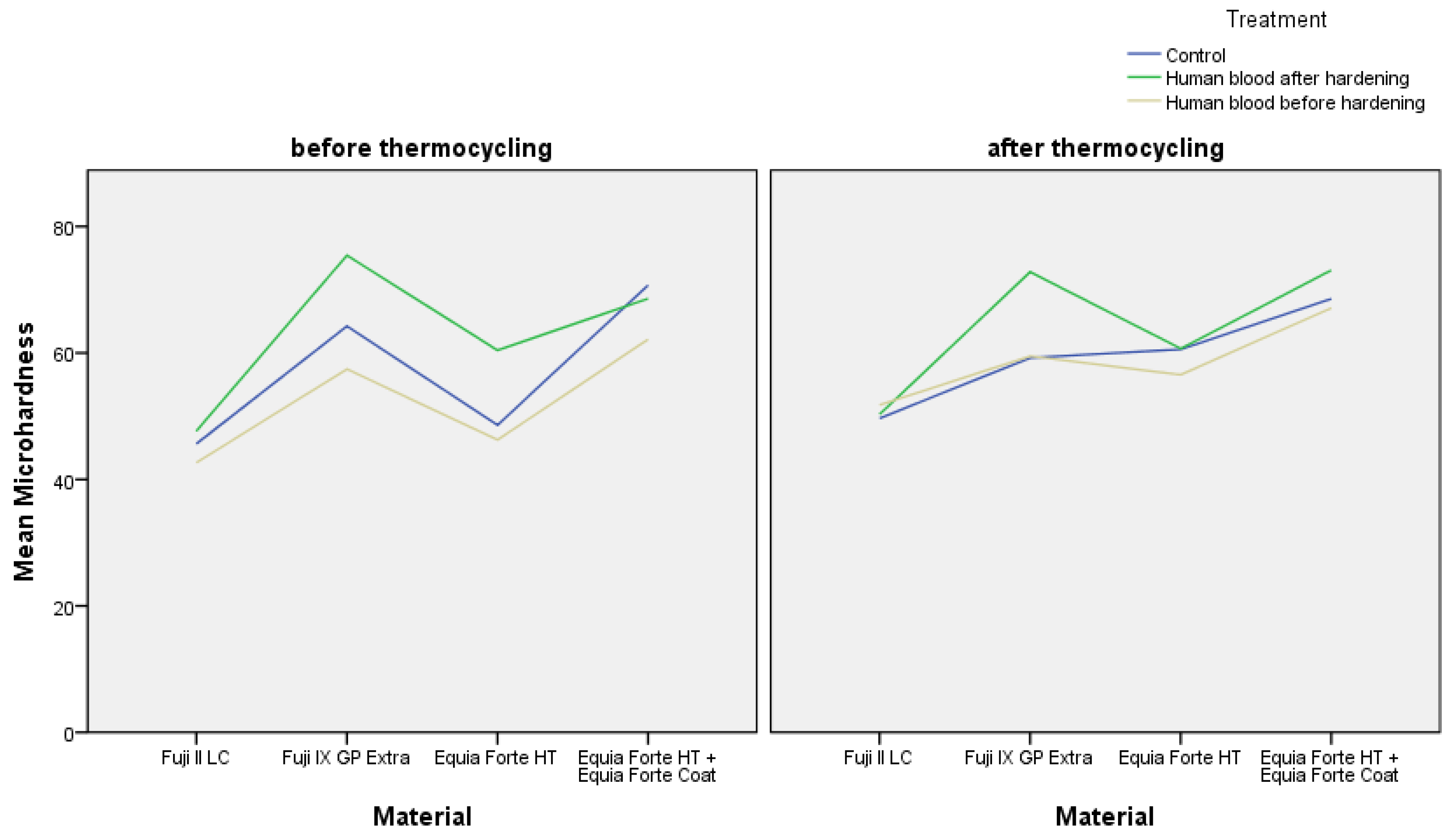
| Material | Treatment | Thermocycling | Mean Value | Standard Deviation |
|---|---|---|---|---|
| Fuji II LC | control group | before | 45.63 | 5.22 |
| after | 49.66 | 8.01 | ||
| blood after hardening | before | 47.65 | 4.97 | |
| after | 50.28 | 7.49 | ||
| blood before hardening | before | 42.65 | 4.21 | |
| after | 51.76 | 6.36 | ||
| Fuji IX GP Extra | control group | before | 64.23 | 9.66 |
| after | 59.23 | 7.45 | ||
| blood after hardening | before | 75.43 | 5.57 | |
| after | 72.81 | 7.36 | ||
| blood before hardening | before | 57.45 | 8.39 | |
| after | 59.49 | 7.36 | ||
| EQUIA Forte HT | control group | before | 48.60 | 9.45 |
| after | 60.57 | 12.63 | ||
| blood after hardening | before | 60.42 | 7.40 | |
| after | 60.69 | 7.55 | ||
| blood before hardening | before | 46.26 | 7.51 | |
| after | 56.56 | 7.16 | ||
| EQUIA Forte HT+ EQUIA Forte Coat | control group | before | 70.71 | 8.79 |
| after | 68.57 | 7.65 | ||
| blood after hardening | before | 68.60 | 6.59 | |
| after | 73.07 | 7.85 | ||
| blood before hardening | before | 62.15 | 7.79 | |
| after | 67.10 | 7.15 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Franić, K.; Brundić, A.; Matijević, J.; Ivanišević, A.; Miletić, I.; Baraba, A. Influence of Human Blood Contamination on Microhardness of Glass-Ionomer Cements and Glass-Hybrid Material. Materials 2025, 18, 4075. https://doi.org/10.3390/ma18174075
Franić K, Brundić A, Matijević J, Ivanišević A, Miletić I, Baraba A. Influence of Human Blood Contamination on Microhardness of Glass-Ionomer Cements and Glass-Hybrid Material. Materials. 2025; 18(17):4075. https://doi.org/10.3390/ma18174075
Chicago/Turabian StyleFranić, Katarina, Ana Brundić, Jurica Matijević, Ana Ivanišević, Ivana Miletić, and Anja Baraba. 2025. "Influence of Human Blood Contamination on Microhardness of Glass-Ionomer Cements and Glass-Hybrid Material" Materials 18, no. 17: 4075. https://doi.org/10.3390/ma18174075
APA StyleFranić, K., Brundić, A., Matijević, J., Ivanišević, A., Miletić, I., & Baraba, A. (2025). Influence of Human Blood Contamination on Microhardness of Glass-Ionomer Cements and Glass-Hybrid Material. Materials, 18(17), 4075. https://doi.org/10.3390/ma18174075







