The Behavior of Industrial Wastes as a Replacement for Metakaolin Before Geopolymerization: A Comparative Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
3. Results
3.1. Geopolymer Stability
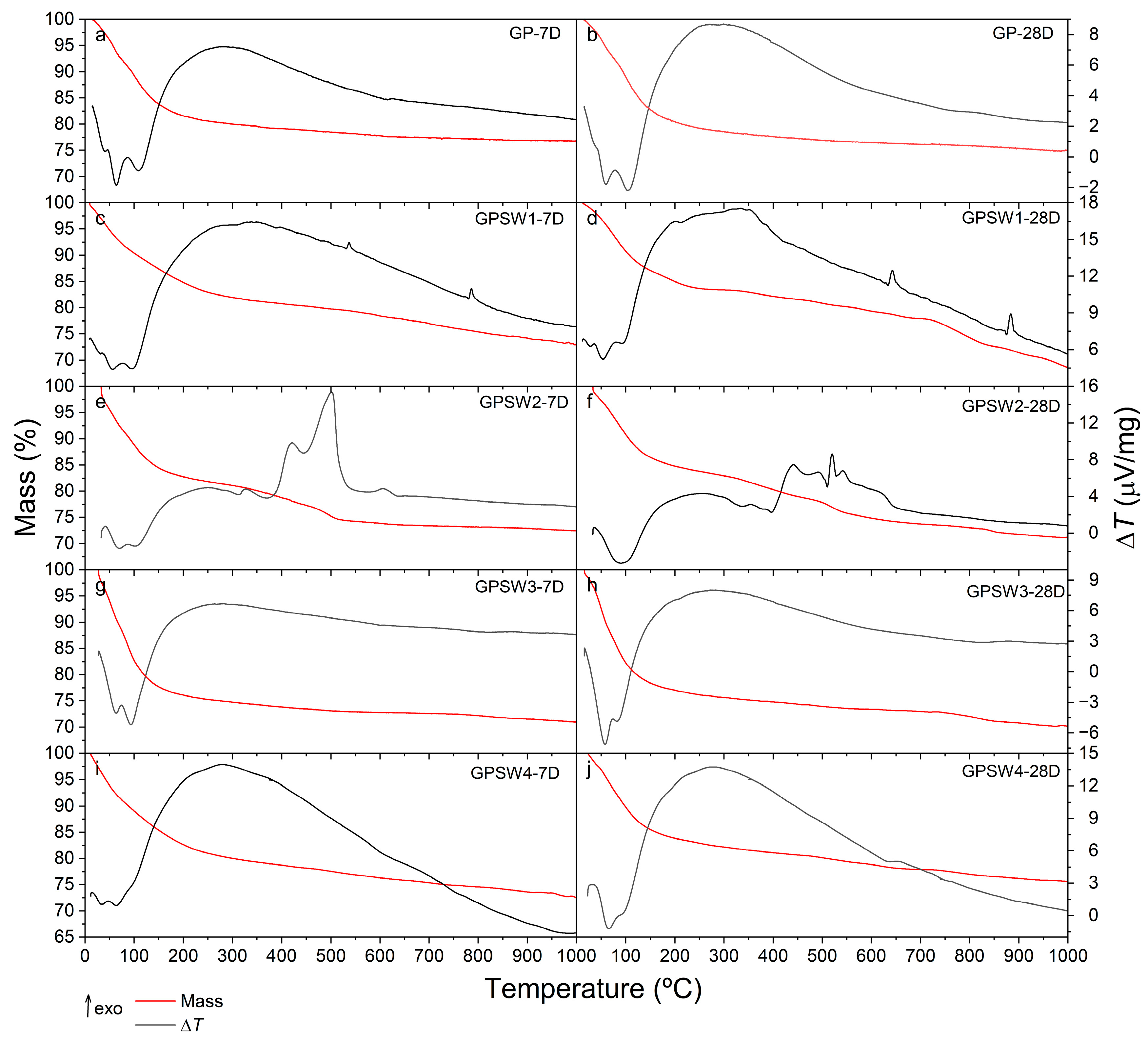
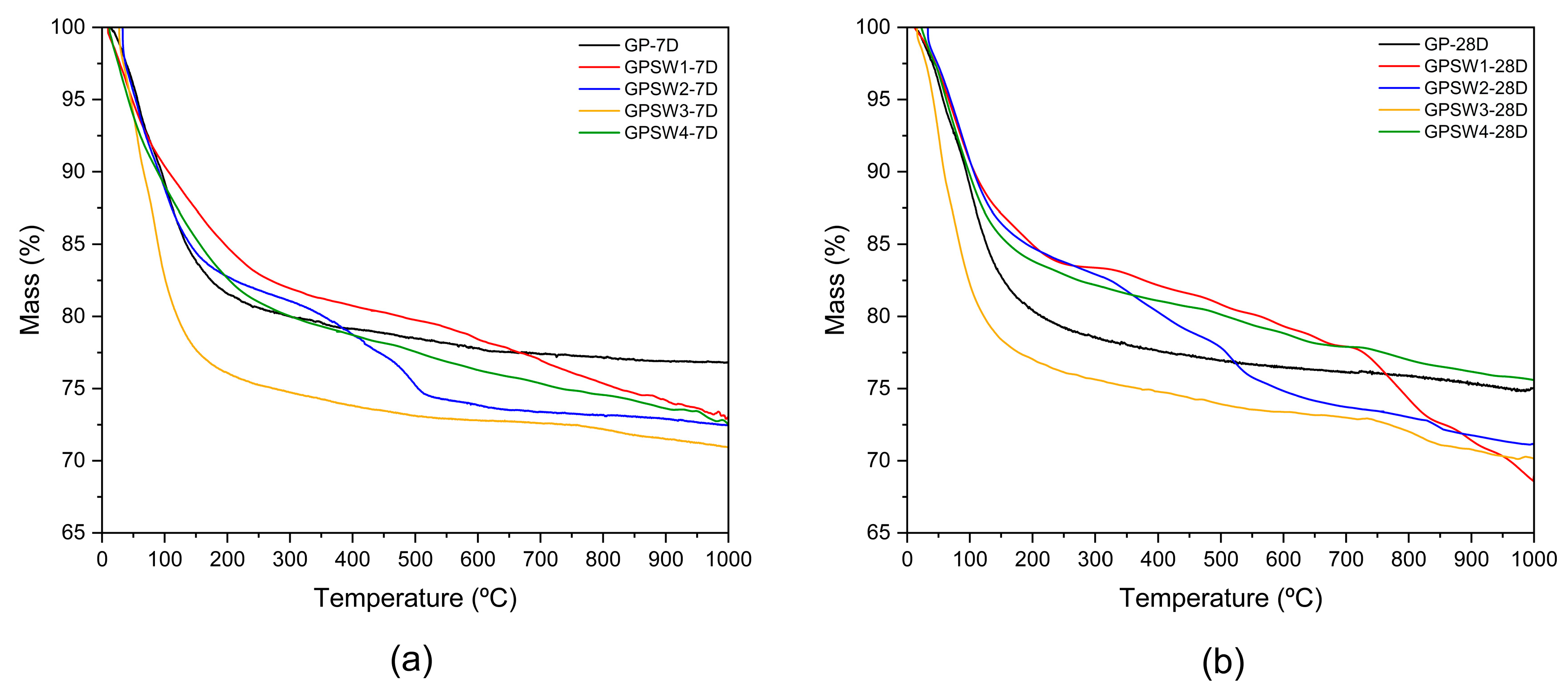
3.2. Thermal Behavior and Thermal Stability
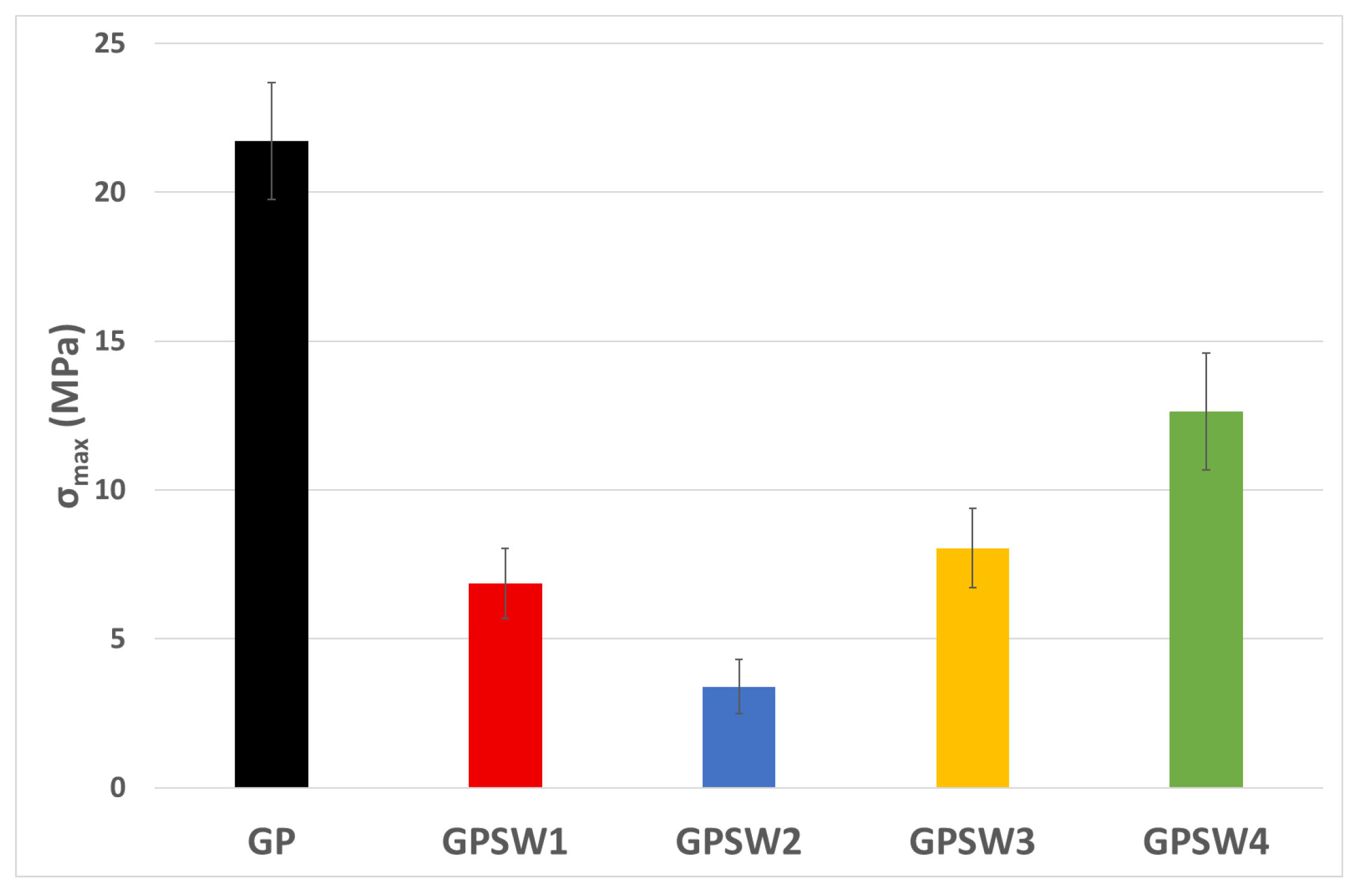
3.3. Mechanical Behavior
3.4. Leaching Test
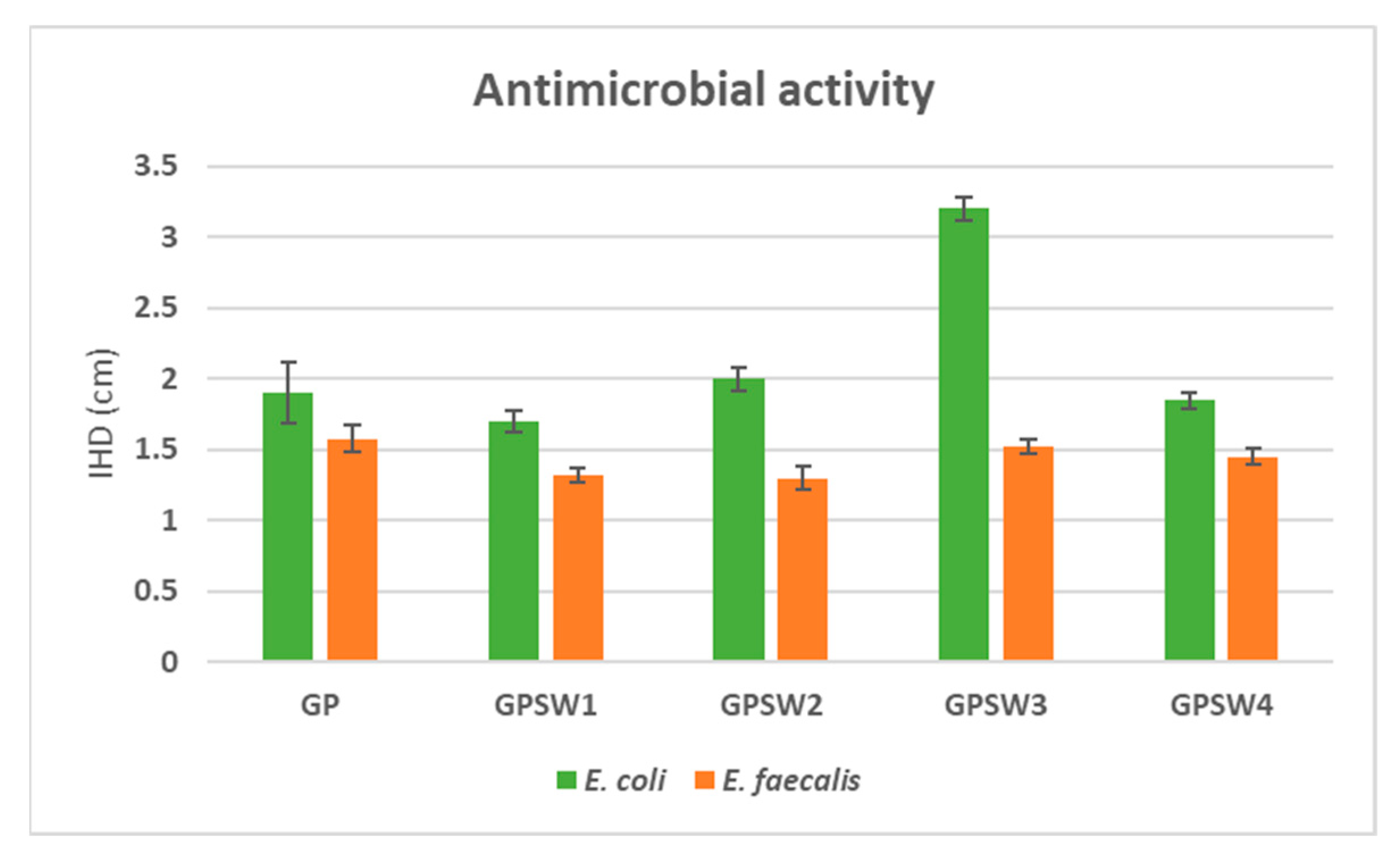
3.5. Antimicrobial Analysis
4. Discussion
5. Conclusions
- None of the wastes had a negative influence on geopolymerization, as confirmed by the redshift of the Si-O-T transmittance band in FT-IR spectra;
- Macroscopically, all geopolymers showed structural stability, indeed, none of them underwent degradation during or after boiling and integrity tests. However, after these tests, GPSW2 showed huge white spots due to efflorescence phenomena;
- Leaching water from integrity tests revealed an alkaline pH (10.9–11.8) for all samples, while the IC values were high for GSPW3 (12.7 mS/m after 28 days of aging);
- Thermal analysis revealed that only GPSW4 showed a high thermal stability, with a mass loss of 17.5% at 28 days of aging;
- Leaching tests underlined that geopolymers containing SW1 and SW4 released antimony (127.5 and 0.128 ppm, respectively) levels;
- Antimicrobial properties revealed enhanced activity against E. coli, particularly in GPSW3 (IHD = 3.2 cm), but limited activity against E. faecalis due to its greater resistance to alkaline conditions.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MK | Metakaolin |
| GP | Geopolymer |
| SW | Solid Waste |
| SEM | Scanning Electron Microscopy |
| FT-IR | Fourier-Transform Infrared |
| TG/DTA | Thermogravimetry–differential thermal analysis |
| RT | Room Temperature |
| IC | Ionic Conductivity |
| IHD | Inhibition Halo Diameter |
References
- Arias, A.; Feijoo, G.; Moreira, M.T.; Tukker, A.; Cucurachi, S. Advancing Waste Valorization and End-of-Life Strategies in the Bioeconomy through Multi-Criteria Approaches and the Safe and Sustainable by Design Framework. Renew. Sustain. Energy Rev. 2025, 207, 114907. [Google Scholar] [CrossRef]
- Tawo, O.E.; Mbamalu, M.I. Advancing Waste Valorization Techniques for Sustainable Industrial Operations and Improved Environmental Safety. Int. J. Sci. Res. Arch. 2025, 14, 127–149. [Google Scholar] [CrossRef]
- Tejaswini, M.S.S.R.; Pathak, P.; Gupta, D.K. Sustainable Approach for Valorization of Solid Wastes as a Secondary Resource through Urban Mining. J. Environ. Manag. 2022, 319, 115727. [Google Scholar] [CrossRef]
- Van Deventer, J.S.J.; Provis, J.L.; Duxson, P.; Brice, D.G. Chemical Research and Climate Change as Drivers in the Commercial Adoption of Alkali Activated Materials. Waste Biomass Valor. 2010, 1, 145–155. [Google Scholar] [CrossRef]
- Elgarahy, A.M.; Maged, A.; Eloffy, M.G.; Zahran, M.; Kharbish, S.; Elwakeel, K.Z.; Bhatnagar, A. Geopolymers as Sustainable Eco-Friendly Materials: Classification, Synthesis Routes, and Applications in Wastewater Treatment. Sep. Purif. Technol. 2023, 324, 124631. [Google Scholar] [CrossRef]
- Jwaida, Z.; Dulaimi, A.; Mashaan, N.; Othuman Mydin, M.A. Geopolymers: The Green Alternative to Traditional Materials for Engineering Applications. Infrastructures 2023, 8, 98. [Google Scholar] [CrossRef]
- Sarkar, M.; Dana, K. Partial Replacement of Metakaolin with Red Ceramic Waste in Geopolymer. Ceram. Int. 2021, 47, 3473–3483. [Google Scholar] [CrossRef]
- Adesanya, E.; Perumal, P.; Luukkonen, T.; Yliniemi, J.; Ohenoja, K.; Kinnunen, P.; Illikainen, M. Opportunities to Improve Sustainability of Alkali-Activated Materials: A Review of Side-Stream Based Activators. J. Clean. Prod. 2021, 286, 125558. [Google Scholar] [CrossRef]
- Kiventerä, J.; Perumal, P.; Yliniemi, J.; Illikainen, M. Mine Tailings as a Raw Material in Alkali Activation: A Review. Int. J. Miner. Metall. Mater. 2020, 27, 1009–1020. [Google Scholar] [CrossRef]
- Geisendorf, S.; Pietrulla, F. The Circular Economy and Circular Economic Concepts—A Literature Analysis and Redefinition. Thunderbird Int. Bus. Rev. 2018, 60, 771–782. [Google Scholar] [CrossRef]
- Singh, N.B.; Middendorf, B. Geopolymers as an Alternative to Portland Cement: An Overview. Constr. Build. Mater. 2020, 237, 117455. [Google Scholar] [CrossRef]
- Sá Ribeiro, R.A.; Sá Ribeiro, M.G.; Kutyla, G.P.; Kriven, W.M. Amazonian Metakaolin Reactivity for Geopolymer Synthesis. Adv. Mater. Sci. Eng. 2019, 2019, 8950764. [Google Scholar] [CrossRef]
- Provis, J.L. Alkali-Activated Materials. Cem. Concr. Res. 2018, 114, 40–48. [Google Scholar] [CrossRef]
- Papa, E.; Minelli, M.; Marchioni, M.C.; Landi, E.; Miccio, F.; Natali Murri, A.; Benito, P.; Vaccari, A.; Medri, V. Metakaolin-Based Geopolymer—Zeolite NaA Composites as CO2 Adsorbents. Appl. Clay Sci. 2023, 237, 106900. [Google Scholar] [CrossRef]
- Duxson, P. Geopolymer Precursor Design. In Geopolymers; Elsevier: Amsterdam, The Netherlands, 2009; pp. 37–49. ISBN 978-1-84569-449-4. [Google Scholar]
- Albidah, A.; Alghannam, M.; Abbas, H.; Almusallam, T.; Al-Salloum, Y. Characteristics of Metakaolin-Based Geopolymer Concrete for Different Mix Design Parameters. J. Mater. Res. Technol. 2021, 10, 84–98. [Google Scholar] [CrossRef]
- Khalifa, A.Z.; Cizer, Ö.; Pontikes, Y.; Heath, A.; Patureau, P.; Bernal, S.A.; Marsh, A.T.M. Advances in Alkali-Activation of Clay Minerals. Cem. Concr. Res. 2020, 132, 106050. [Google Scholar] [CrossRef]
- Liu, J.; Liu, B.; Li, Q.; Zhang, Q.; An, C.; Sun, Z.; Wei, S.; Fan, J. Review of Solid Waste-Based Geopolymers: Preparation, Deterioration and Durability. Mater. Today Commun. 2025, 43, 111820. [Google Scholar] [CrossRef]
- Ren, X.; Wang, F.; He, X.; Hu, X. Resistance and Durability of Fly Ash Based Geopolymer for Heavy Metal Immobilization: Properties and Mechanism. RSC Adv. 2024, 14, 12580–12592. [Google Scholar] [CrossRef]
- Nemaleu, J.G.D.; Kaze, R.C.; Tome, S.; Alomayri, T.; Assaedi, H.; Kamseu, E.; Melo, U.C.; Sglavo, V.M. Powdered Banana Peel in Calcined Halloysite Replacement on the Setting Times and Engineering Properties on the Geopolymer Binders. Constr. Build. Mater. 2021, 279, 122480. [Google Scholar] [CrossRef]
- Amar, M.; Ladduri, B.; Alloul, A.; Benzerzour, M.; Abriak, N.-E. Microstructure and Mechanical Properties of Geopolymers Utilizing Excavated Soils, Metakaolin and Slags. J. Build. Eng. 2024, 86, 108755. [Google Scholar] [CrossRef]
- Korniejenko, K.; Mikuła, J.; Brudny, K.; Aruova, L.; Zhakanov, A.; Jexembayeva, A.; Zhaksylykova, L. A Review of Industrial By-Product Utilization and Future Pathways of Circular Economy: Geopolymers as Modern Materials for Sustainable Building. Sustainability 2025, 17, 4536. [Google Scholar] [CrossRef]
- Bellum, R.R.; Venkatesh, C.; Madduru, S.R.C. Influence of Red Mud on Performance Enhancement of Fly Ash-Based Geopolymer Concrete. Innov. Infrastruct. Solut. 2021, 6, 215. [Google Scholar] [CrossRef]
- Guo, L.; Xu, X.; Wang, Q.; Dong, X.; Liu, X.; Lei, H. Synergistic Utilization of Industrial Waste Red Mud and Rice Husk Ash for Eco-Friendly Geopolymer Preparation: Enhancing Strength and Mitigating Hazardous Leaching. Environ. Sci. Pollut. Res. 2023, 31, 2745–2758. [Google Scholar] [CrossRef]
- Ricciotti, L.; Occhicone, A.; Ferone, C.; Cioffi, R.; Tarallo, O.; Roviello, G. Development of Geopolymer-Based Materials with Ceramic Waste for Artistic and Restoration Applications. Materials 2022, 15, 8600. [Google Scholar] [CrossRef]
- Catauro, M.; Bollino, F.; Dell’Era, A.; Ciprioti, S.V. Pure Al2O3·2SiO2 Synthesized via a Sol-Gel Technique as a Raw Material to Replace Metakaolin: Chemical and Structural Characterization and Thermal Behavior. Ceram. Int. 2016, 42, 16303–16309. [Google Scholar] [CrossRef]
- Long, L.; Zhao, Y.; Lv, G.; Duan, Y.; Liu, X.; Jiang, X. Improving Stabilization/Solidification of MSWI Fly Ash with Coal Gangue Based Geopolymer via Increasing Active Calcium Content. Sci. Total Environ. 2023, 854, 158594. [Google Scholar] [CrossRef]
- De Oliveira, L.B.; De Azevedo, A.R.G.; Marvila, M.T.; Pereira, E.C.; Fediuk, R.; Vieira, C.M.F. Durability of Geopolymers with Industrial Waste. Case Stud. Constr. Mater. 2022, 16, e00839. [Google Scholar] [CrossRef]
- Dal Poggetto, G.; D’Angelo, A.; Blanco, I.; Piccolella, S.; Leonelli, C.; Catauro, M. FT-IR Study, Thermal Analysis, and Evaluation of the Antibacterial Activity of a MK-Geopolymer Mortar Using Glass Waste as Fine Aggregate. Polymers 2021, 13, 2970. [Google Scholar] [CrossRef]
- Viola, V.; D’Angelo, A.; Vertuccio, L.; Catauro, M. Metakaolin-Based Geopolymers Filled with Industrial Wastes: Improvement of Physicochemical Properties through Sustainable Waste Recycling. Polymers 2024, 16, 2118. [Google Scholar] [CrossRef] [PubMed]
- D’Angelo, A.; Vertuccio, L.; Leonelli, C.; Alzeer, M.I.M.; Catauro, M. Entrapment of Acridine Orange in Metakaolin-Based Geopolymer: A Feasibility Study. Polymers 2023, 15, 675. [Google Scholar] [CrossRef] [PubMed]
- Silverstein, R.M.; Webster, F.X.; Kiemle, D.J.; Bryce, D.L. Spectrometric Identification of Organic Compounds, 5th ed.; Wiley: Hoboken, NJ, USA, 2015; ISBN 978-0-470-61637-6. [Google Scholar]
- Kaviyarasu, K.; Sajan, D.; Devarajan, P.A. A Rapid and Versatile Method for Solvothermal Synthesis of Sb2O3 Nanocrystals under Mild Conditions. Appl. Nanosci. 2013, 3, 529–533. [Google Scholar] [CrossRef]
- López Morales, F.; Zayas, T.; Contreras, O.E.; Salgado, L. Effect of Sn Precursor on the Synthesis of SnO2 and Sb-Doped SnO2 Particles via Polymeric Precursor Method. Front. Mater. Sci. 2013, 7, 387–395. [Google Scholar] [CrossRef]
- Singh, S.; Aswath, M.U.; Das Biswas, R.; Ranganath, R.V.; Choudhary, H.K.; Kumar, R.; Sahoo, B. Role of Iron in the Enhanced Reactivity of Pulverized Red Mud: Analysis by Mössbauer Spectroscopy and FTIR Spectroscopy. Case Stud. Constr. Mater. 2019, 11, e00266. [Google Scholar] [CrossRef]
- Nakamoto, K. Infrared and Raman Spectra of Inorganic and Coordination Compounds. In Handbook of Vibrational Spectroscopy; Chalmers, J.M., Griffiths, P.R., Eds.; Wiley: Hoboken, NJ, USA, 2001; ISBN 978-0-471-98847-2. [Google Scholar]
- Mihaylov, M.Y.; Zdravkova, V.R.; Ivanova, E.Z.; Aleksandrov, H.A.; Petkov, P.S.; Vayssilov, G.N.; Hadjiivanov, K.I. Infrared Spectra of Surface Nitrates: Revision of the Current Opinions Based on the Case Study of Ceria. J. Catal. 2021, 394, 245–258. [Google Scholar] [CrossRef]
- Abdullah, M.M.A.B.; Hussin, K.; Bnhussain, M.; Ismail, K.N.; Yahya, Z.; Abdul Razak, R. Fly Ash-Based Geopolymer Lightweight Concrete Using Foaming Agent. Int. J. Mol. Sci. 2012, 13, 7186–7198. [Google Scholar] [CrossRef]
- Koczón, P.; Lewandowski, W.; Mazurek, A.P. Vibrational (FT-IR and FT-Raman) and NMR Studies on Selected Metal (Ca, Mn, Zn) Complexes with Ortho-, Meta-, and Para-Iodobenzoic Acids. Vib. Spectrosc. 1999, 20, 143–149. [Google Scholar] [CrossRef]
- Ji, H.; Liu, D.; Cheng, H.; Zhang, C.; Yang, L.; Ren, D. Infrared Thermochromic Properties of Monoclinic VO2 Nanopowders Using a Malic Acid-Assisted Hydrothermal Method for Adaptive Camouflage. RSC Adv. 2017, 7, 5189–5194. [Google Scholar] [CrossRef]
- Zheng, M.; Zhang, H.; Gong, X.; Xu, R.; Xiao, Y.; Dong, H.; Liu, X.; Liu, Y. A Simple Additive-Free Approach for the Synthesis of Uniform Manganese Monoxide Nanorods with Large Specific Surface Area. Nanoscale Res. Lett. 2013, 8, 166. [Google Scholar] [CrossRef]
- Sgarlata, C.; Formia, A.; Siligardi, C.; Ferrari, F.; Leonelli, C. Mine Clay Washing Residues as a Source for Alkali-Activated Binders. Materials 2021, 15, 83. [Google Scholar] [CrossRef] [PubMed]
- Davidovits, J. Geopolymer: Chemistry & Applications, 5th ed.; Institut Géopolymère: Saint-Quentin, France, 2020; ISBN 978-2-9544531-1-8. [Google Scholar]
- Catauro, M.; Viola, V.; D’Amore, A. Mosses on Geopolymers: Preliminary Durability Study and Chemical Characterization of Metakaolin-Based Geopolymers Filled with Wood Ash. Polymers 2023, 15, 1639. [Google Scholar] [CrossRef]
- Catauro, M.; Dal Poggetto, G.; Sgarlata, C.; Vecchio Ciprioti, S.; Pacifico, S.; Leonelli, C. Thermal and Microbiological Performance of Metakaolin-Based Geopolymers Cement with Waste Glass. Appl. Clay Sci. 2020, 197, 105763. [Google Scholar] [CrossRef]
- Sabbah, R.; An, X.-w.; Chickos, J.S.; Leitão, M.L.P.; Roux, M.V.; Torres, L.A. Reference Materials for Calorimetry and Differential Thermal Analysis. Thermochim. Acta 1999, 331, 93–204. [Google Scholar] [CrossRef]
- EN 12457-2:2004; Characterisation of Waste—Leaching—Compliance Testing for Leaching of Granular Wastes and Sludges—Part 2: Single Stage Test at a Liquid/Solid Ratio of 10 L/kg for Materials with Particle Sizes Smaller than 4 mm (with or without Size Reduction). Ente Nazionale di Unificazione: Milan, Italy, 2004; p. 27.
- Hudzicki, J. Kirby-Bauer Disk Diffusion Susceptibility Test Protocol; American Society for Microbiology: Washington, DC, USA, 2009. [Google Scholar]
- Sikora, A.; Zahra, F. Nosocomial Infections. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Catauro, M.; Ciprioti, S.V. Characterization of Hybrid Materials Prepared by Sol-Gel Method for Biomedical Implementations. A Critical Review. Materials 2021, 14, 1788. [Google Scholar] [CrossRef]
- Chen, L.; Wang, Z.; Wang, Y.; Feng, J. Preparation and Properties of Alkali Activated Metakaolin-Based Geopolymer. Materials 2016, 9, 767. [Google Scholar] [CrossRef] [PubMed]
- Duxson, P.; Lukey, G.C.; Van Deventer, J.S.J. Evolution of Gel Structure during Thermal Processing of Na-Geopolymer Gels. Langmuir 2006, 22, 8750–8757. [Google Scholar] [CrossRef]
- Kamath, M.; Prashant, S.; Ralegaonkar, R. Microstructure Properties of Popular Alkali-Activated Pastes Cured in Ambient Temperature. Buildings 2023, 13, 858. [Google Scholar] [CrossRef]
- Inaty, F.E.; Nasreddine, H.; Djerbi, A.; Gautron, L.; Marchetti, M.; Quiertant, M.; Metalssi, O.O. Mechanical and Durability Performance of Metakaolin and Fly Ash-Based Geopolymers Compared to Cement Systems. Results Eng. 2025, 27, 105788. [Google Scholar] [CrossRef]
- Rihan, M.A.M.; Onchiri, R.O.; Gathimba, N.; Sabuni, B. Assessing the Durability Performance of Geopolymer Concrete Utilizing Fly Ash and Sugarcane Bagasse Ash as Sustainable Binders. Open Ceram. 2024, 20, 100687. [Google Scholar] [CrossRef]
- Simão, L.; Fernandes, E.; Hotza, D.; Ribeiro, M.J.; Montedo, O.R.K.; Raupp-Pereira, F. Controlling Efflorescence in Geopolymers: A New Approach. Case Stud. Constr. Mater. 2021, 15, e00740. [Google Scholar] [CrossRef]
- Longhi, M.A.; Zhang, Z.; Walkley, B.; Rodríguez, E.D.; Kirchheim, A.P. Strategies for Control and Mitigation of Efflorescence in Metakaolin-Based Geopolymers. Cem. Concr. Res. 2021, 144, 106431. [Google Scholar] [CrossRef]
- Tesovnik, A.; Horvat, B. Rapid Immobilisation of Chemical Reactions in Alkali-Activated Materials Using Solely Microwave Irradiation. Minerals 2024, 14, 1219. [Google Scholar] [CrossRef]
- Genua, F.; Giovini, M.; Santoni, E.; Berrettoni, M.; Lancellotti, I.; Leonelli, C. Factors Affecting Consolidation in Geopolymers for Stabilization of Galvanic Sludge. Materials 2025, 18, 3015. [Google Scholar] [CrossRef]
- Aly, Z.; Vance, E.R.; Perera, D.S.; Hanna, J.V.; Griffith, C.S.; Davis, J.; Durce, D. Aqueous Leachability of Metakaolin-Based Geopolymers with Molar Ratios of Si/Al = 1.5–4. J. Nucl. Mater. 2008, 378, 172–179. [Google Scholar] [CrossRef]
- Callister, W.D. Materials Science and Engineering: An Introduction, 7th ed.; Wiley: New York, NY, USA, 2007; ISBN 978-0-471-73696-7. [Google Scholar]
- Brown, M. Differential Scanning Calorimetry—An Introduction for Practitioners. Thermochim. Acta 1997, 303, 117. [Google Scholar] [CrossRef]
- Duxson, P.; Provis, J.L.; Lukey, G.C.; Mallicoat, S.W.; Kriven, W.M.; Van Deventer, J.S.J. Understanding the Relationship between Geopolymer Composition, Microstructure and Mechanical Properties. Colloids Surf. A Physicochem. Eng. Asp. 2005, 269, 47–58. [Google Scholar] [CrossRef]
- Castillo, H.; Collado, H.; Droguett, T.; Sánchez, S.; Vesely, M.; Garrido, P.; Palma, S. Factors Affecting the Compressive Strength of Geopolymers: A Review. Minerals 2021, 11, 1317. [Google Scholar] [CrossRef]
- Khan, R.; Iqbal, S.; Soliyeva, M.; Ali, A.; Elboughdiri, N. Advanced Clay-Based Geopolymer: Influence of Structural and Material Parameters on Its Performance and Applications. RSC Adv. 2025, 15, 12443–12471. [Google Scholar] [CrossRef]
- Hu, X.; Guo, X.; He, M.; Li, S. pH-Dependent Release Characteristics of Antimony and Arsenic from Typical Antimony-Bearing Ores. J. Environ. Sci. 2016, 44, 171–179. [Google Scholar] [CrossRef]
- Wang, H.; Xu, J.; Gomez, M.A.; Shi, Z.; Jia, Y. A Study on the Effects of Anion, Cation, Organic Compounds, and pH on the Release Behaviors of As and Sb from Sediments. Environ. Sci. Pollut. Res. 2021, 28, 45199–45211. [Google Scholar] [CrossRef]
- Padan, E.; Bibi, E.; Ito, M.; Krulwich, T.A. Alkaline pH Homeostasis in Bacteria: New Insights. Biochim. Biophys. Acta (BBA) Biomembr. 2005, 1717, 67–88. [Google Scholar] [CrossRef]
- Brennan, L.E.; Kumawat, L.K.; Piatek, M.E.; Kinross, A.J.; McNaughton, D.A.; Marchetti, L.; Geraghty, C.; Wynne, C.; Tong, H.; Kavanagh, O.N.; et al. Potent Antimicrobial Effect Induced by Disruption of Chloride Homeostasis. Chem 2023, 9, 3138–3158. [Google Scholar] [CrossRef]
- Weckwerth, P.H.; Zapata, R.O.; Vivan, R.R.; Tanomaru Filho, M.; Maliza, A.G.A.; Duarte, M.A.H. In Vitro Alkaline pH Resistance of Enterococcus Faecalis. Braz. Dent. J. 2013, 24, 474–476. [Google Scholar] [CrossRef]
- Adhikary, S.K.; D’Angelo, A.; Viola, V.; Catauro, M.; Perumal, P. Alternative Construction Materials from Industrial Side Streams: Are They Safe? Energy Ecol. Environ. 2023, 9, 206–214. [Google Scholar] [CrossRef]
- D’Angelo, A.; Viola, V.; Fiorentino, M.; Dal Poggetto, G.; Blanco, I. Use of Natural Dyes to Color Metakaolin-Based Geopolymer Materials. Ceram. Int. 2024, 51, 5528–5535. [Google Scholar] [CrossRef]
- Jin, G.; Wang, X.; Mao, H.; Ji, S.; Shi, Q. Preparation and Properties of Metakaolin-Fumed-Silica Geopolymer Modified with Sodium Silicate and Potassium Silicate Activators. Chin. J. Anal. Chem. 2024, 52, 100352. [Google Scholar] [CrossRef]
- Kim, G.; Cho, S.; Im, S.; Yoon, J.; Suh, H.; Kanematsu, M.; Machida, A.; Shobu, T.; Bae, S. Evaluation of the Thermal Stability of Metakaolin-Based Geopolymers According to Si/Al Ratio and Sodium Activator. Cem. Concr. Compos. 2024, 150, 105562. [Google Scholar] [CrossRef]

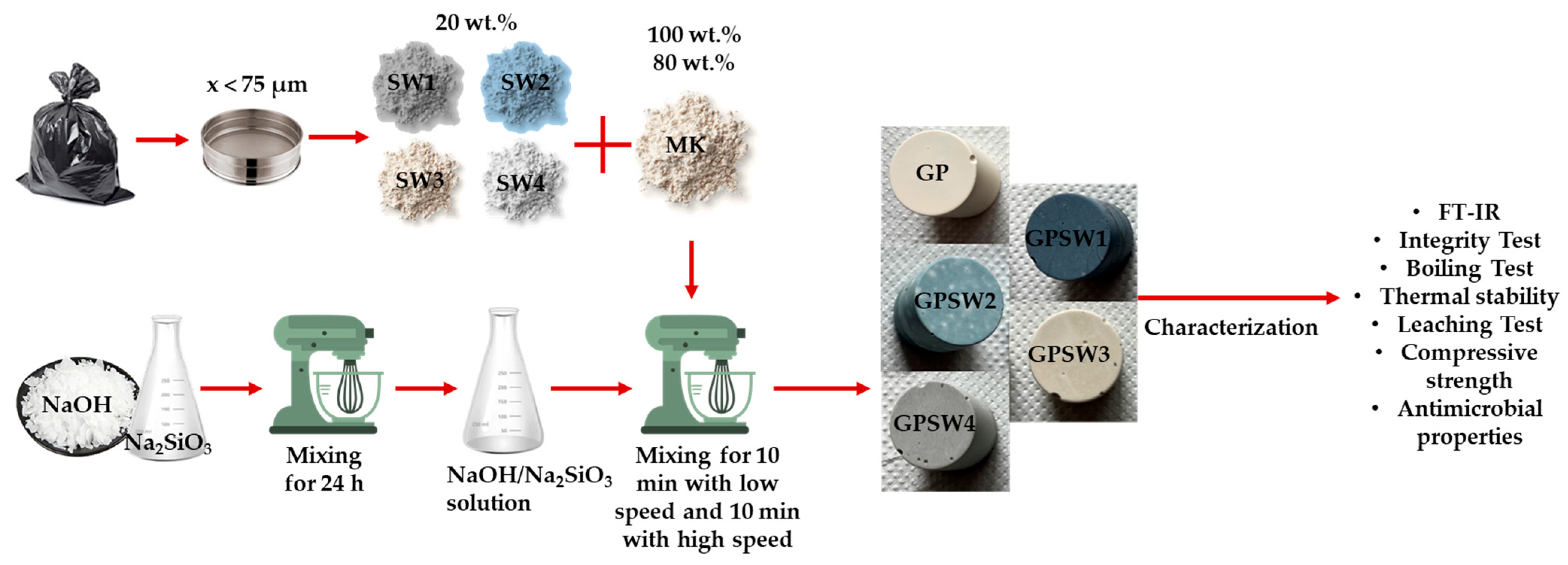



| Type of Waste | Suction Dust | Red Mud a | Electro-Filter Dust | Extraction Sludge b |
|---|---|---|---|---|
| Sample label | SW1 | SW2 | SW3 | SW4 |
| Characteristics | Powdery, grey-black color | Powdery, grey-green color | Powdery, white color | Grainy, beige color |
| Residue (%) at 105 °C | 99.1 ± 0.8 | 58.3 ± 2.0 | 98.6 ± 2.5 | 65.0 ± 3.7 |
| pH | 7.2 | 3.0 | 11.2 | 7.1 |
| Chlorides (mg/L) | <5 | 25,678 | 25,980 | - |
| Sulfates (mg/L) | <5 | 64.4 | 16,510 | - |
| Fluorides (mg/L) | <5 | 0.35 | 3900 | - |
| Nitrates (mg/L) | <5 | 17.5 | - | - |
| Phosphates (mg/L) | <5 | - | - | - |
| Cyanides (mg/L) | - | <50 | - | - |
| Hydrocarbons (mg/L) | <100 | C10–C40 < 2.5 C5–C8 = 294 | C10–C40 < 18 C5–C8 < 1 | C10–C40 < 100 |
| Metal content (mg/kg) | As = 870 Sb = 145,265 Ca = 369 Fe = 219 Ni = 23 Pb = 228 K = 171 Sn = 257,741 Zn = 60 | Al = 114 Cr = 130 Fe = 371 Mn = 371 Ni = 176 Pb = 25 Cu = 52 Zn = 25,426 | As < 2.3 Cd < 2.3 Co = 1.35 Cr = 15.5 Mn = 15.5 Mn = 2.7 Mg = 43.2 Ni = 7.43 Pb = 21.5 Cu = 25.7 Sn = 7.4 Tl = 0.676 V = 1.35 Zn = 7.43 | Sb < 10 As < 5 Cd < 0.6 Cr < 10 Mn = 22 Mg < 0.5 Ni = 2 Pb < 5 Cu < 5 Se < 100 V < 5 Zn = 25 |
| Heavy metal leaching (mg/L) | Sb = 19.3 | Cr = 0.2 Cu = 2.6 Ni = 7.37 Pb = 1.16 Zn = 168 | As = 2.3 | As < 0.01 Cd < 0.001 Cr = 0.01 Cu = 0.01 Mg < 0.0005 Ni = 0.01 Sb < 0.0005 Se < 0.01 Zn = 0.9 V < 0.02 |
| Geopolymer Name | Mass/g | ||||||
|---|---|---|---|---|---|---|---|
| Na2SiO3 | NaOH | MK | SW1 | SW2 | SW3 | SW4 | |
| GP0 | 145.63 | 13.31 | 100.00 | - | - | - | - |
| GPSW1 | 116.50 | 10.65 | 80.00 | 20.00 | - | - | - |
| GPSW2 | 116.50 | 10.65 | 80.00 | - | 20.00 | - | - |
| GPSW3 | 116.50 | 10.65 | 80.00 | - | - | 20.00 | - |
| GPSW4 | 116.50 | 10.65 | 80.00 | - | - | - | 20.00 |
| Data from Integrity Tests | GP | GPSW1 | GPSW2 | GPSW3 | GPSW4 |
|---|---|---|---|---|---|
| IC (mS/m)-7D | 2.2 | 4.4 | 4.5 | 17.5 | 4.6 |
| IC (mS/m)-14D | 2.0 | 2.1 | 3.3 | 12.6 | 4.4 |
| IC (mS/m)-28D | 2.8 | 2.2 | 3.3 | 12.7 | 3.2 |
| pH-7D | 10.9 | 10.9 | 11.0 | 11.0 | 10.9 |
| pH-14D | 11.2 | 11.5 | 11.7 | 11.8 | 11.6 |
| pH-28D | 10.9 | 10.9 | 11.3 | 11.3 | 11.0 |
| Sample | 7D | 28D | ||
|---|---|---|---|---|
| Trange/°C | Mass/% | Trange/°C | Mass/% | |
| GP | 14–275 | 19.9 | 16–277 | 21.0 |
| GPSW1 | 11–258 | 16.9 | 12–290 | 16.6 |
| GPSW2 | 32–263 | 18.4 | 20–253 | 16.3 |
| GPSW3 | 27–283 | 25.1 | 19–276 | 23.3 |
| GPSW4 | 12–285 | 20.0 | 24–278 | 17.5 |
| Parameter | Cr | Ni | Cu | Zn | As | Cd | Pb | Ba | Mo | Sb | Se |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Inert | 0.05 | 0.04 | 0.2 | 0.4 | 0.05 | 0.004 | 0.05 | 2.00 | 0.05 | 0.006 | 0.01 |
| Non-Hazardous | 1.00 | 1.00 | 5.0 | 5.0 | 0.20 | 0.100 | 1.00 | 10.0 | 1.00 | 0.070 | 0.05 |
| Hazardous | 7.00 | 4.00 | 10.0 | 20.0 | 2.50 | 0.500 | 5.00 | 30.0 | 3.00 | 0.500 | 0.70 |
| Sample | 28D | |||||
|---|---|---|---|---|---|---|
| pH | IC/mS/m | Mass Loss at 1000 °C/% | σmax/MPa | Leaching Sb/ppm | IHD E. coli/cm | |
| GP | 10.9 | 2.8 | 21.0 | 22.0 | - | 1.9 |
| GPSW1 | 10.9 | 2.2 | 32.0 | 6.9 | 127.0 | 1.7 |
| 80GP20SW1 | 7.6 | 23.1 | 15.0 | 68.5 | 25.0 | 2.2 |
| GPSW2 | 11.3 | 3.3 | 28.0 | 3.4 | - | 2.0 |
| 80GP20SW2 | 10.0 | 107.6 | 23.0 | 37.8 | - | 2.5 |
| GPSW3 | 11.3 | 12.7 | 29.5 | 8.0 | - | 3.2 |
| 80GP20SW3 | 9.8 | 165.1 | 13.7 | 39.4 | - | 2.5 |
| GPSW4 | 11.0 | 3.2 | 24.8 | 12.6 | 0.128 | 1.9 |
| 80GP20SW4 | 7.9 | 43.2 | 12.7 | 40.4 | - | 1.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Catauro, M.; D’Angelo, A.; Genua, F.; Giovini, M.; Silva Ferraz, J.M.; Vecchio Ciprioti, S. The Behavior of Industrial Wastes as a Replacement for Metakaolin Before Geopolymerization: A Comparative Study. Materials 2025, 18, 4035. https://doi.org/10.3390/ma18174035
Catauro M, D’Angelo A, Genua F, Giovini M, Silva Ferraz JM, Vecchio Ciprioti S. The Behavior of Industrial Wastes as a Replacement for Metakaolin Before Geopolymerization: A Comparative Study. Materials. 2025; 18(17):4035. https://doi.org/10.3390/ma18174035
Chicago/Turabian StyleCatauro, Michelina, Antonio D’Angelo, Francesco Genua, Mattia Giovini, José Miguel Silva Ferraz, and Stefano Vecchio Ciprioti. 2025. "The Behavior of Industrial Wastes as a Replacement for Metakaolin Before Geopolymerization: A Comparative Study" Materials 18, no. 17: 4035. https://doi.org/10.3390/ma18174035
APA StyleCatauro, M., D’Angelo, A., Genua, F., Giovini, M., Silva Ferraz, J. M., & Vecchio Ciprioti, S. (2025). The Behavior of Industrial Wastes as a Replacement for Metakaolin Before Geopolymerization: A Comparative Study. Materials, 18(17), 4035. https://doi.org/10.3390/ma18174035










