Assessing Microplastics and Nanoparticles in the Surface Seawater of Venice Lagoon—Part I: Methodology of Research
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling Procedure
2.2. Processing of MPs and Nanoparticles
2.2.1. Mitigation of Contamination
2.2.2. Wet Sieving
2.2.3. Digestion with H2O2 and Density Separation
2.2.4. Optical Microscopy
- ▪
- The particles have no observable organic or cellular structures;
- ▪
- In the case of fibers, the diameter should be consistent along their length;
- ▪
- Particles should present clear and homogeneous colors;
- ▪
- The further high magnification should be used in the case of transparent or white particles.
2.2.5. Scanning Electron Microscopy/X-ray Energy Dispersive Spectrometry
2.2.6. Filtrate Treatment
2.2.7. Mass Estimation
2.2.8. Statistical Analysis
3. Results
3.1. Physical-Chemical Parameters
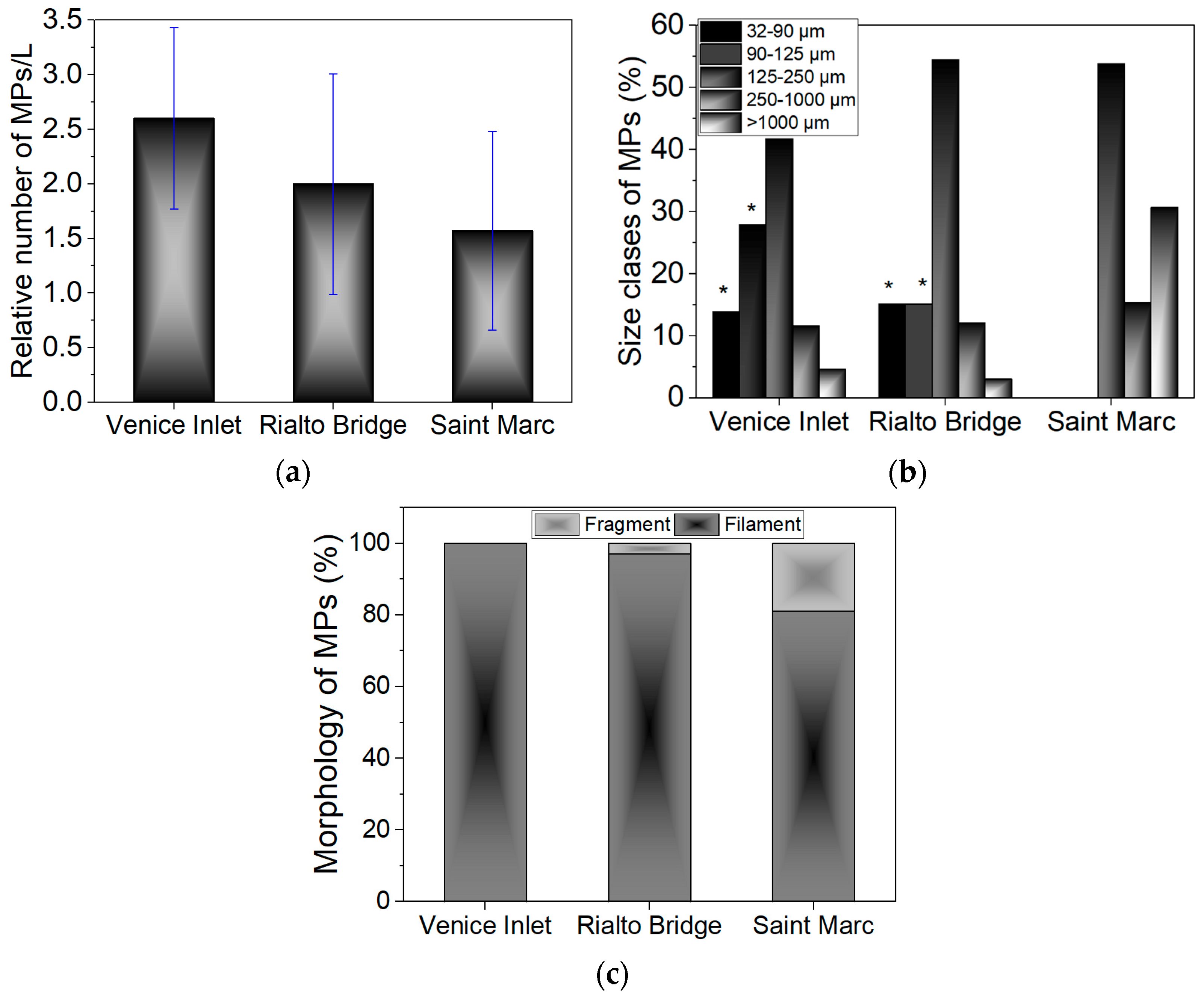
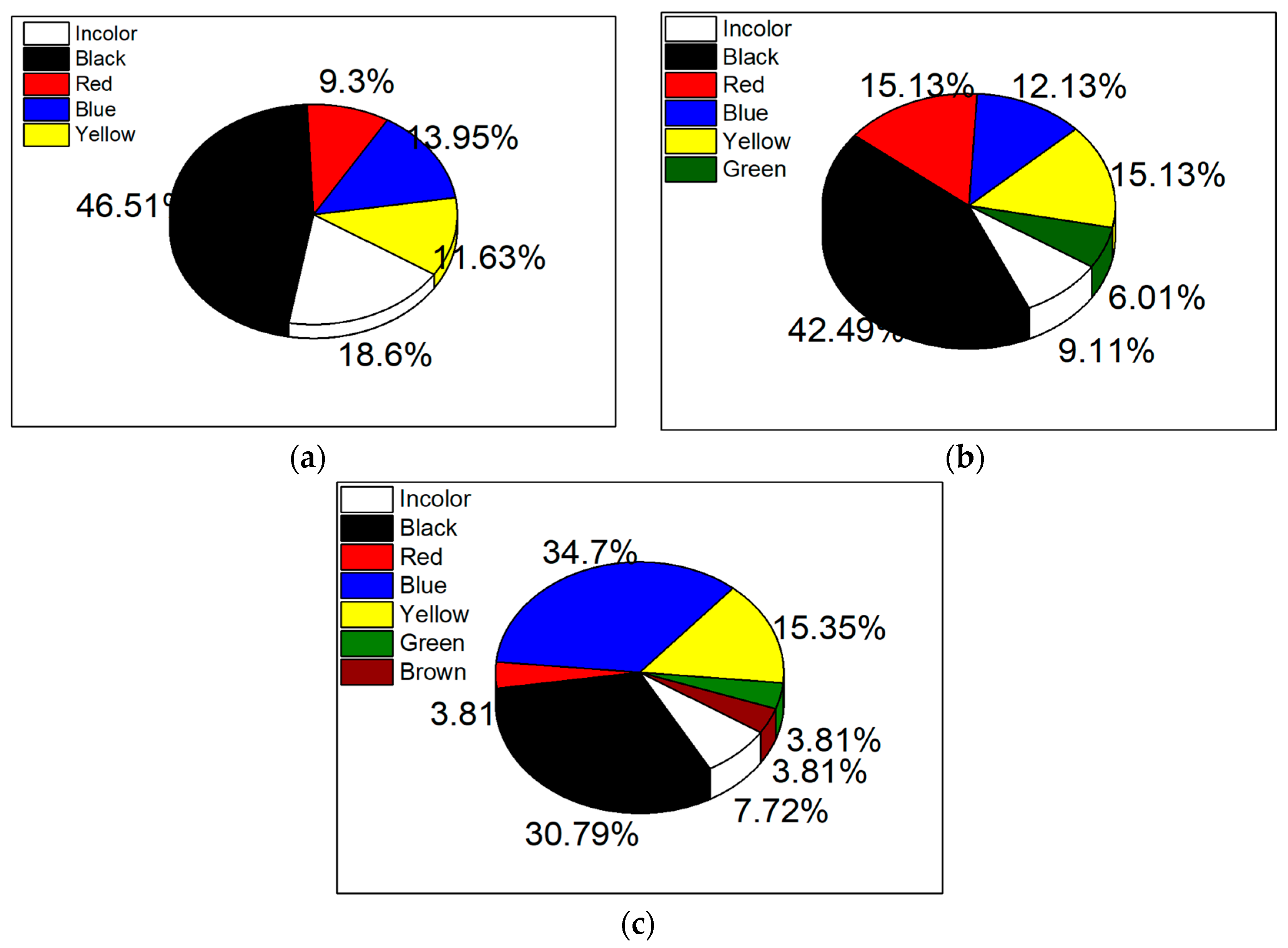
3.2. Microplastic Evaluation
3.2.1. Optical Microscopy
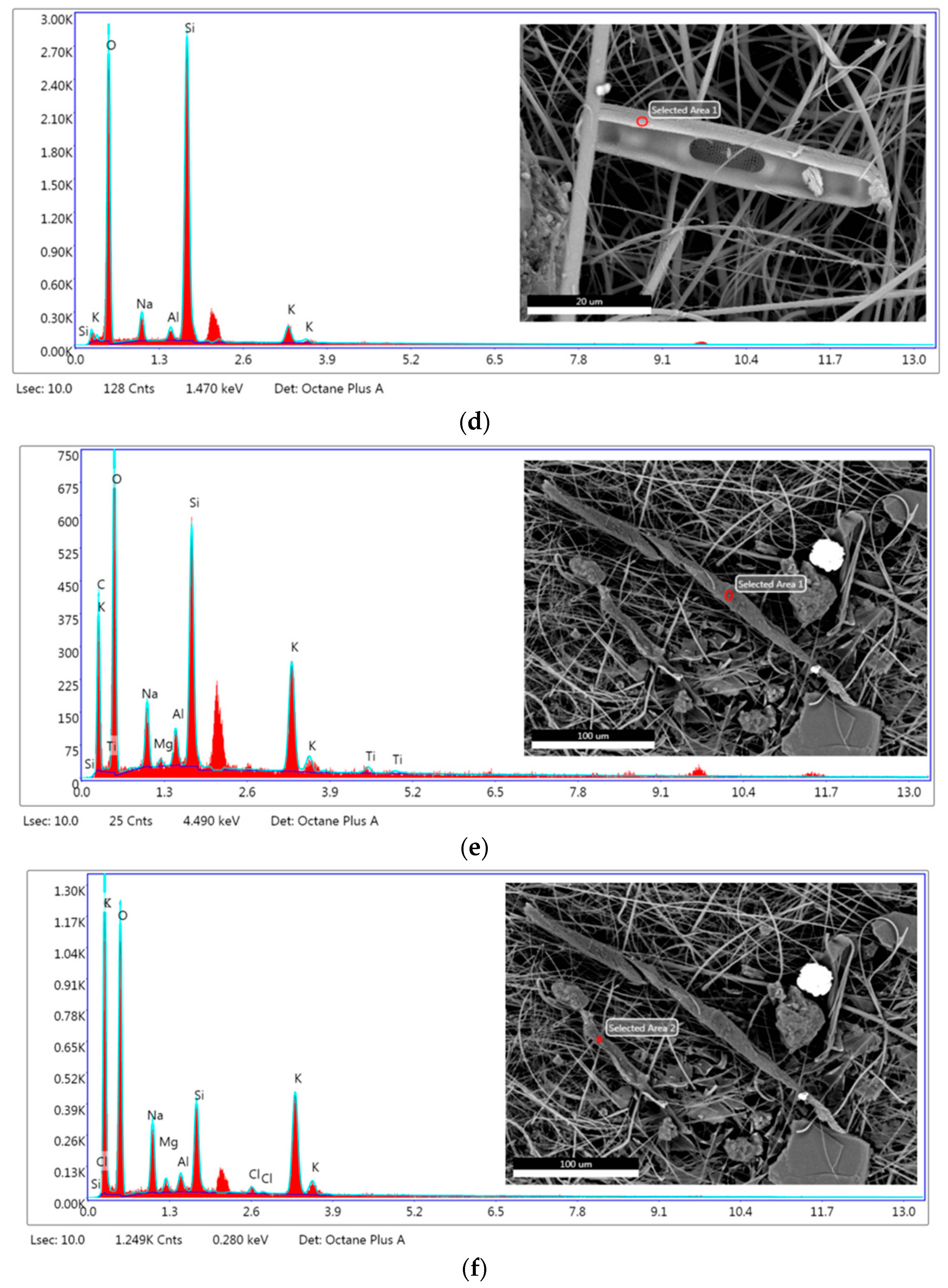
3.2.2. SEM/EDX Analysis
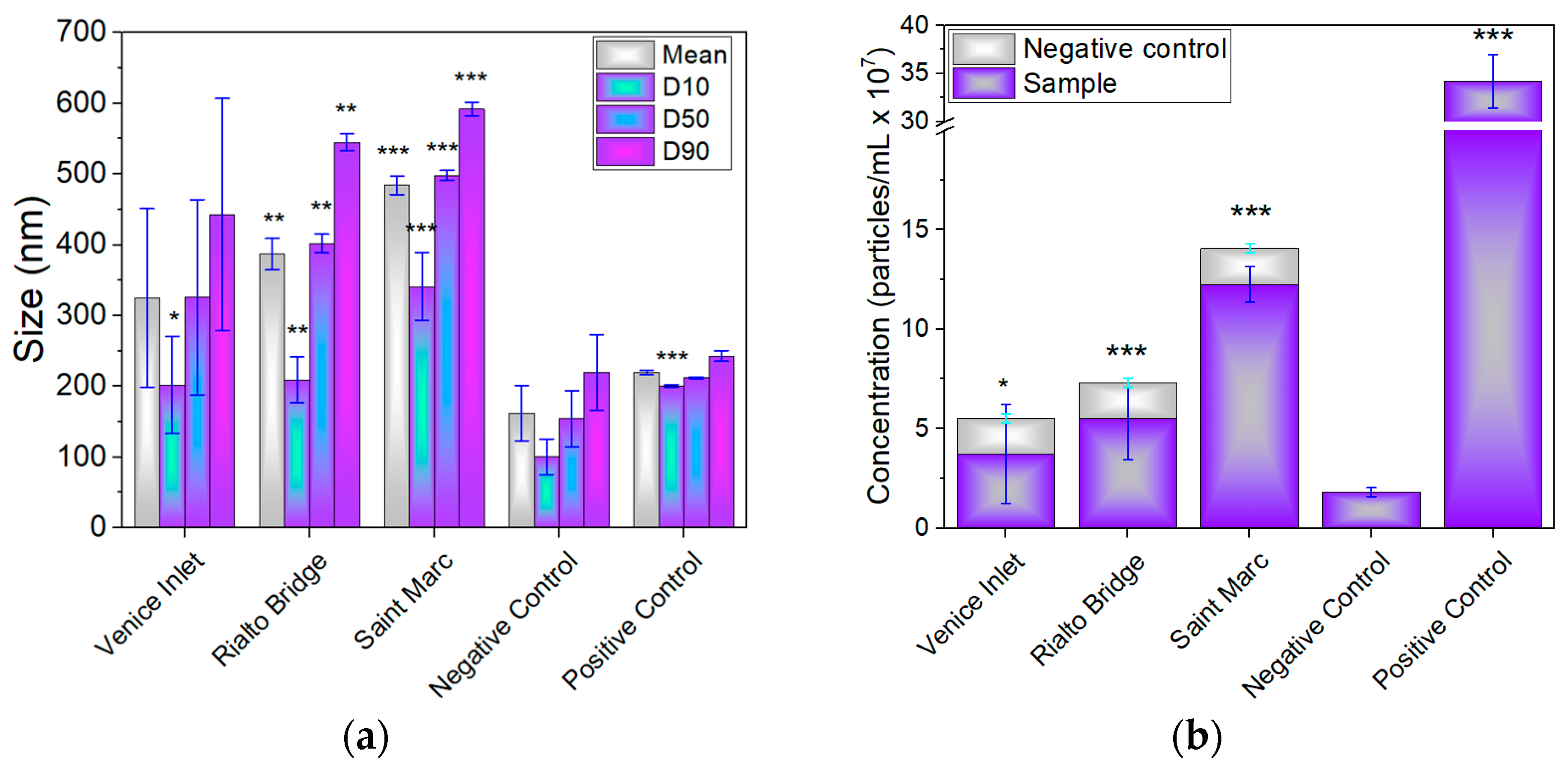
3.3. Estimation of Size and Concentration of Nanoparticles
3.4. Estimation of MP and Nanoparticle Concentration
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kaandorp, M.L.A.; Lobelle, D.; Kehl, C.; Dijkstra, H.A.; van Sebille, E. Global mass of buoyant marine plastics dominated by large long-lived debris. Nat. Geosci. 2023, 16, 689–694. [Google Scholar] [CrossRef]
- Agamuthu, P.; Mehran, S.B.; Norkhairah, A.; Norkhairiyah, A. Marine debris: A review of impacts and global initiatives. Waste Manag. Res. 2019, 37, 987–1002. [Google Scholar] [CrossRef] [PubMed]
- Orthodoxou, D.L.; Loizidou, X.I.; Baldwin, C.; Kocareis, C.; Karonias, A.; Ates, M.A. Seasonal and geographic variations of marine litter: A comprehensive study from the island of Cyprus. Mar. Pollut. Bull. 2022, 177, 113495. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.-T.T.; Ha, N.-H.; Bui, T.-K.L.; Nguyen, K.L.P.; Tran, D.-P.T.; Nguyen, H.Q.; El-Arini, A.; Schuyler, Q.; Nguyen, T.T.L. Baseline Marine Litter Surveys along Vietnam Coasts Using Citizen Science Approach. Sustainability 2022, 14, 4919. [Google Scholar] [CrossRef]
- Hee, J.; Schloegel, K.; Lechthaler, S.; Plaster, J.; Bitter, K.; Blank, L.M.; Quicker, P. Comparative Analysis of the Behaviour of Marine Litter in Thermochemical Waste Treatment Processes. Processes 2021, 9, 13. [Google Scholar] [CrossRef]
- Reinold, S.; Herrera, A.; Stile, N.; Saliu, F.; Hernandez-Gonzalez, C.; Martinez, I.; Ortega, Z.; Marrero, M.D.; Lasagni, M.; Gomez, M. An annual study on plastic accumulation in surface water and sediment cores from the coastline of Tenerife (Canary Island, Spain). Mar. Pollut. Bull. 2021, 173, 113072. [Google Scholar] [CrossRef]
- Li, F.; Zhai, X.; Yao, M.; Bai, X. An inevitable but underestimated photoaging behavior of plastic waste in the aquatic environment: Critical role of nitrate. Environ. Pollut. 2022, 314, 120307. [Google Scholar] [CrossRef]
- Luo, H.W.; Tu, C.L.; He, D.Q.; Zhang, A.P.; Sun, J.Q.; Li, J.; Xu, J.; Pan, X.L. Interactions between microplastics and contaminants: A review focusing on the effect of aging process. Sci. Total Environ. 2023, 899, 165615. [Google Scholar] [CrossRef] [PubMed]
- Syberg, K.; Nielsen, M.B.; Oturai, N.B.; Clausen, L.P.W.; Ramos, T.M.; Hansen, S.F. Circular economy and reduction of micro(nano)plastics contamination. J. Hazard. Mater. Adv. 2022, 5, 100044. [Google Scholar] [CrossRef]
- Winkler, A.; Fumagalli, F.; Cella, C.; Gilliland, D.; Tremolada, P.; Valsesia, A. Detection and formation mechanisms of secondary nanoplastic released from drinking water bottles. Water Res. 2022, 222, 118848. [Google Scholar] [CrossRef] [PubMed]
- Winkler, A.; Santo, N.; Ortenzi, M.A.; Bolzoni, E.; Bacchetta, R.; Tremolada, P. Does mechanical stress cause microplastic release from plastic water bottles? Water Res. 2019, 166, 115082. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.H.; Wong, K.K.; Li, W.; Zhao, H.R.; Wang, T.M.; Stanescu, S.; Boult, S.; van Dongen, B.; Mativenga, P.; Li, L. Characteristics of nano-plastics in bottled drinking water. J. Hazard. Mater. 2022, 424, 127404. [Google Scholar] [CrossRef] [PubMed]
- Rapa, M.; Darie-Nita, R.N.; Matei, E.; Predescu, A.-M.; Berbecaru, A.-C.; Predescu, C. Insights into Anthropogenic Micro- and Nanoplastic Accumulation in Drinking Water Sources and Their Potential Effects on Human Health. Polymers 2023, 15, 2425. [Google Scholar] [CrossRef] [PubMed]
- Yaranal, N.A.; Subbiah, S.; Mohanty, K. Identification, extraction of microplastics from edible salts and its removal from contaminated seawater. Environ. Technol. Innov. 2021, 21, 101253. [Google Scholar] [CrossRef]
- Yap, V.H.S.; Chase, Z.; Wright, J.T.; Hurd, C.L.; Lavers, J.L.; Lenz, M. A comparison with natural particles reveals a small specific effect of PVC microplastics on mussel performance. Mar. Pollut. Bull. 2020, 160, 111703. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Armendariz, C.; Alejandro-Vega, S.; Paz-Montelongo, S.; Gutierrez-Fernandez, A.J.; Carrascosa-Iruzubieta, C.J.; de la Torre, A. Microplastics as Emerging Food Contaminants: A Challenge for Food Safety. Int. J. Environ. Res. Public Health 2022, 19, 1174. [Google Scholar] [CrossRef] [PubMed]
- Cerasa, M.; Teodori, S.; Pietrelli, L. Searching Nanoplastics: From Sampling to Sample Processing. Polymers 2021, 13, 3658. [Google Scholar] [CrossRef] [PubMed]
- Song, F.H.; Li, T.T.; Hur, J.; Wu, F.C.; Meng, X.G. Eco-Colloidal Layer of Micro/Nanoplastics Increases Complexity and Uncertainty of Their Biotoxicity in Aquatic Environments. Environ. Sci. Technol. 2022, 56, 10547–10549. [Google Scholar] [CrossRef]
- Purwiyanto, A.I.S.; Suteja, Y.; Trisno; Ningrum, P.S.; Putri, W.A.E.; Rozirwan; Agustriani, F.; Fauziyah; Cordova, M.R.; Koropitan, A.F. Concentration and adsorption of Pb and Cu in microplastics: Case study in aquatic environment. Mar. Pollut. Bull. 2020, 158, 111380. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.Y.; Yang, W.W.; Cheng, P.; Zhang, S.Q.; Zhang, S.W.; Jiao, W.T.; Sun, Y.H. Adsorption characteristics of cadmium onto microplastics from aqueous solutions. Chemosphere 2019, 235, 1073–1080. [Google Scholar] [CrossRef]
- Davranche, M.; Lory, C.; Le Juge, C.; Blancho, F.; Dia, A.; Grassl, B.; El Hadri, H.; Pascal, P.Y.; Gigault, J. Nanoplastics on the coast exposed to the North Atlantic Gyre: Evidence and traceability. Nanoimpact 2020, 20, 100262. [Google Scholar] [CrossRef]
- Davranche, M.; Veclin, C.; Pierson-Wickmann, A.-C.; El Hadri, H.; Grassl, B.; Rowenczyk, L.; Dia, A.; Ter Halle, A.; Blancho, F.; Reynaud, S.; et al. Are nanoplastics able to bind significant amount of metals? The lead example. Environ. Pollut. 2019, 249, 940–948. [Google Scholar] [CrossRef]
- Camacho, M.; Herrera, A.; Gómez, M.; Acosta-Dacal, A.; Martínez, I.; Henríquez-Hernández, L.A.; Luzardo, O.P. Organic pollutants in marine plastic debris from Canary Islands beaches. Sci. Total Environ. 2019, 662, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Shaw, E.J.; Turner, A. Recycled electronic plastic and marine litter. Sci. Total Environ. 2019, 694, 133644. [Google Scholar] [CrossRef]
- Onrubia, J.A.T.; Djaoudi, K.; Borgogno, F.; Canuto, S.; Angeletti, B.; Besio, G.; Capello, M.; Cutroneo, L.; Stocchino, A.; Mounier, S.; et al. Quantification of Microplastics in North-Western Mediterranean Harbors: Seasonality and Biofilm-Related Metallic Contaminants. J. Mar. Sci. Eng. 2021, 9, 337. [Google Scholar] [CrossRef]
- Lee, C.H.; Fang, J.K.H. Effects of temperature and particle concentration on aggregation of nanoplastics in freshwater and seawater. Sci. Total Environ. 2022, 817, 152562. [Google Scholar] [CrossRef] [PubMed]
- Lv, L.L.; He, L.; Jiang, S.Q.; Chen, J.J.; Zhou, C.X.; Qu, J.H.; Lu, Y.Q.; Hong, P.Z.; Sun, S.L.; Li, C.Y. In situ surface-enhanced Raman spectroscopy for detecting microplastics and nanoplastics in aquatic environments. Sci. Total Environ. 2020, 728, 138449. [Google Scholar] [CrossRef]
- Veclin, C.; Desmet, C.; Pradel, A.; Valsesia, A.; Ponti, J.; El Hadri, H.; Maupas, T.; Pellerin, V.; Gigault, J.; Grassl, B.; et al. Effect of the Surface Hydrophobicity-Morphology-Functionality of Nanoplastics on Their Homoaggregation in Seawater. ACS ES&T Water 2022, 2, 88–95. [Google Scholar] [CrossRef]
- Materic, D.; Holzinger, R.; Niemann, H. Nanoplastics and ultrafine microplastic in the Dutch Wadden Sea—The hidden plastics debris? Sci. Total Environ. 2022, 846, 157371. [Google Scholar] [CrossRef] [PubMed]
- Râpă, M.; Vujanovic, A.; Cecchi, T.; Poletto, D. Detection and Characterization of Micro-and Nanoplastic Pollutants: A Case Study of the Adriatic Sea. In International Conference on Microplastic Pollution in the Mediterranean Sea; Springer International Publishing: Cham, Switzerland, 2023; pp. 29–36. [Google Scholar]
- Pelamatti, T.; Fonseca-Ponce, I.A.; Rios-Mendoza, L.M.; Stewart, J.D.; Marín-Enríquez, E.; Marmolejo-Rodriguez, A.J.; Hoyos-Padilla, E.M.; Galván-Magana, F.; González-Armas, R. Seasonal variation in the abundance of marine plastic debris in Banderas Bay, Mexico. Mar. Pollut. Bull. 2019, 145, 604–610. [Google Scholar] [CrossRef]
- De-la-Torre, G.E.; Dioses-Salinas, D.C.; Pizarro-Ortega, C.I.; López, A.D.F.; Severini, M.D.F.; Rimondino, G.N.; Malanca, F.E.; Dobaradaran, S.; Aragaw, T.A.; Mghili, B.; et al. Plastic and paint debris in marine protected areas of Peru. Sci. Total Environ. 2023, 901, 165788. [Google Scholar] [CrossRef] [PubMed]
- Velmurugan, P.M.; Vijayaprabhakaran, K.; Devika, P.T. Baseline study on identification, characterization, distribution and abundance of microplastics in surface water from Ennore to Kovalam along the east coast of India. Phys. Chem. Earth 2023, 130, 103391. [Google Scholar] [CrossRef]
- Jiang, Y.; Zhao, Y.N.; Wang, X.; Yang, F.; Chen, M.; Wang, J. Characterization of microplastics in the surface seawater of the South Yellow Sea as affected by season. Sci. Total Environ. 2020, 724, 138375. [Google Scholar] [CrossRef] [PubMed]
- Baini, M.; Fossi, M.C.; Galli, M.; Caliani, I.; Campani, T.; Finoia, M.G.; Panti, C. Abundance and characterization of microplastics in the coastal waters of Tuscany (Italy): The application of the MSFD monitoring protocol in the Mediterranean Sea. Mar. Pollut. Bull. 2018, 133, 543–552. [Google Scholar] [CrossRef] [PubMed]
- Ece, E.; Haciosmanoglu, N.; Inci, F. Microfluidics as a Ray of Hope for Microplastic Pollution. Biosensors 2023, 13, 332. [Google Scholar] [CrossRef] [PubMed]
- Phan, S.; Padilla-Gamino, J.L.; Luscombe, C.K. The effect of weathering environments on microplastic chemical identification with Raman and IR spectroscopy: Part I. polyethylene and polypropylene. Polym. Test. 2022, 116, 107752. [Google Scholar] [CrossRef]
- Miserli, K.; Lykos, C.; Kalampounias, A.G.; Konstantinou, I. Screening of Microplastics in Aquaculture Systems (Fish, Mussel, and Water Samples) by FTIR, Scanning Electron Microscopy-Energy Dispersive Spectroscopy and Micro-Raman Spectroscopies. Appl. Sci. 2023, 13, 9705. [Google Scholar] [CrossRef]
- Moses, S.R.; Roscher, L.; Primpke, S.; Hufnagl, B.; Löeder, M.G.J.; Gerdts, G.; Laforsch, C. Comparison of two rapid automated analysis tools for large FTIR microplastic datasets. Anal. Bioanal. Chem. 2023, 415, 2975–2987. [Google Scholar] [CrossRef]
- Elsayed, A.A.; Erfan, M.; Sabry, Y.M.; Dris, R.; Gasperi, J.; Barbier, J.-S.; Marty, F.; Bouanis, F.; Luo, S.; Nguyen, B.T.T.; et al. A microfluidic chip enables fast analysis of water microplastics by optical spectroscopy. Sci. Rep. 2021, 11, 10533. [Google Scholar] [CrossRef] [PubMed]
- Materic, D.; Kasper-Giebl, A.; Kau, D.; Anten, M.; Greilinger, M.; Ludewig, E.; van Sebille, E.; Rockmann, T.; Holzinger, R. Micro- and Nanoplastics in Alpine Snow: A New Method for Chemical Identification and (Semi)Quantification in the Nanogram Range. Environ. Sci. Technol. 2020, 54, 2353–2359. [Google Scholar] [CrossRef] [PubMed]
- Hanif, M.A.; Ibrahim, N.; Dahalan, F.A.; Ali, U.F.M.; Hasan, M.; Jalil, A.A. Microplastics and nanoplastics: Recent literature studies and patents on their removal from aqueous environment. Sci. Total Environ. 2022, 810, 152115. [Google Scholar] [CrossRef] [PubMed]
- Tallec, K.; Blard, O.; Gonzalez-Fernandez, C.; Brotons, G.; Berchel, M.; Soudant, P.; Huvet, A.; Paul-Pont, I. Surface functionalization determines behavior of nanoplastic solutions in model aquatic environments. Chemosphere 2019, 225, 639–646. [Google Scholar] [CrossRef]
- Yang, T.; Luo, J.; Nowack, B. Characterization of Nanoplastics, Fibrils, and Microplastics Released during Washing and Abrasion of Polyester Textiles. Environ. Sci. Technol. 2021, 55, 15873–15881. [Google Scholar] [CrossRef]
- Martin, L.M.A.; Sheng, J.; Zimba, P.V.; Zhu, L.; Fadare, O.O.; Haley, C.; Wang, M.C.; Phillips, T.D.; Conkle, J.; Xu, W. Testing an Iron Oxide Nanoparticle-Based Method for Magnetic Separation of Nanoplastics and Microplastics from Water. Nanomaterials 2022, 12, 2348. [Google Scholar] [CrossRef]
- Liu, Y.; Lin, L.Q.; Yang, B.; Huang, M.H.; Huang, X.X.; Chen, X.X.; Dai, Z.Q.; Sun, S.L.; Yang, Y.Q.; Li, C.Y. Separation and Identification of Nanoplastics via a Two-Phase System Combined with Surface-Enhanced Raman Spectroscopy. ACS Sustain. Chem. Eng. 2024, 12, 1595–1604. [Google Scholar] [CrossRef]
- Zhou, X.-X.; He, S.; Gao, Y.; Li, Z.-C.; Chi, H.-Y.; Li, C.-J.; Wang, D.-J.; Yan, B. Protein Corona-Mediated Extraction for Quantitative Analysis of Nanoplastics in Environmental Waters by Pyrolysis Gas Chromatography/Mass Spectrometry. Anal. Chem. 2021, 93, 6698–6705. [Google Scholar] [CrossRef] [PubMed]
- Dereszewska, A.; Krasowska, K.; Popek, M. Microplastics in Harbour Seawaters: A Case Study in the Port of Gdynia, Baltic Sea. Sustainability 2023, 15, 6678. [Google Scholar] [CrossRef]
- Liu, B.J.; Lu, Y.; Deng, H.Q.; Huang, H.F.; Wei, N.; Jiang, Y.L.; Jiang, Y.X.; Liu, L.Q.Q.; Sun, K.F.; Zheng, H. Occurrence of microplastics in the seawater and atmosphere of the South China Sea: Pollution patterns and interrelationship. Sci. Total Environ. 2023, 889, 164173. [Google Scholar] [CrossRef] [PubMed]
- Kwon, O.Y.; Kang, J.H.; Hong, S.H.; Shim, W.J. Spatial distribution of microplastic in the surface waters along the coast of Korea. Mar. Pollut. Bull. 2020, 155, 110729. [Google Scholar] [CrossRef] [PubMed]
- Kershaw, P.J. (Ed.) Sources, Fate and Effects of Microplastics in the Marine Environment: A Global Assessment; Rep. Stud. GESAMP No. 90; IMO/FAO/UNESCO-IOC/UNIDO/WMO/IAEA/UN/UNEP/UNDP Joint Group of Experts on the Scientific Aspects of Marine Environmental Protection: London, UK, 2015; 96p. [Google Scholar]
- Kershaw, P.J. (Ed.) Sources, Fate and Effects of Microplastics in the Marine Environment: Part Two of a Global Assessment; Rep. Stud. GESAMP No. 93; IMO/FAO/UNESCO-IOC/UNIDO/WMO/IAEA/UN/UNEP/UNDP Joint Group of Experts on the Scientific Aspects of Marine Environmental Protection: London, UK, 2016; 220p. [Google Scholar]
- Guidance on Monitoring of Marine Litter in European Seas; JRC Scientific And Policy Reports; Publications Office of the European Union: Luxembourg, 2013. [CrossRef]
- Masura, J.; Baker, J.E.; Foster, G.D.; Arthur, C.; Herring, C. Laboratory Methods for the Analysis of Microplastics in the Marine Environment: Recommendations for Quantifying Synthetic Particles in Waters and Sediments; NOAA Technical Memorandum NOS-OR&R-48; National Oceanic and Atmospheric Administration: Washington, DC, USA, 2015. [Google Scholar]
- Lares, M.; Ncibi, M.C.; Sillanpää, M. Intercomparison study on commonly used methods to determine microplastics in wastewater and sludge samples. Environ. Sci. Pollut. Res. 2019, 26, 12109–12122. [Google Scholar] [CrossRef]
- Van Mourik, L.M.; Crum, S.; Martinez-Frances, E.; van Bavel, B.; Leslie, H.A.; de Boer, J.; Cofino, W.P. Results of WEPAL-QUASIMEME/NORMANs first global interlaboratory study onmicroplastics reveal urgent need for harmonization (vol 772, 145071, 2021). Sci. Total Environ. 2022, 820, 153237. [Google Scholar] [CrossRef] [PubMed]
- Isobe, A.; Buenaventura, N.T.; Chastain, S.; Chavanich, S.; Cózar, A.; DeLorenzo, M.; Hagmann, P.; Hinata, H.; Kozlovskii, N.; Lusher, A.L.; et al. An interlaboratory comparison exercise for the determination of microplastics in standard sample bottles. Mar. Pollut. Bull. 2019, 146, 831–837. [Google Scholar] [CrossRef]
- Martínez-Francés, E.; van Bavel, B.; Hurley, R.; Nizzetto, L.; Pakhomova, S.; Buenaventura, N.T.; Singdahl-Larsen, C.; Magni, M.L.T.; Johansen, J.E.; Lusher, A. Innovative reference materials for method validation in microplastic analysis including interlaboratory comparison exercises. Anal. Bioanal. Chem. 2023, 415, 2907–2919. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.D.; Santschi, P.H. Composition and cycling of colloids in marine environments. Rev. Geophys. 1997, 35, 17–40. [Google Scholar] [CrossRef]
- Vaillancourt, R.D.; Balch, W.M. Size distribution of marine submicron particles determined by flow field-flow fractionation. Limnol. Oceanogr. 2000, 45, 485–492. [Google Scholar] [CrossRef]
- ISO 3310-1:2016; Test Sieves. Technical Requirements and Testing. Part 1: Test Sieves of Metal wire Cloth. ISO: Geneva, Switzerland, 2016.
- Hurley, R.R.; Lusher, A.L.; Olsen, M.; Nizzetto, L. Validation of a Method for Extracting Microplastics from Complex, Organic-Rich, Environmental Matrices. Environ. Sci. Technol. 2018, 52, 7409–7417. [Google Scholar] [CrossRef] [PubMed]
- Poblete-Chavez, R.; Cortes-Pizarro, E.; Rojas-Araya, R. Treatment of seawater for rotifer culture uses applying adsorption and advanced oxidation processes. Lat. Am. J. Aquat. Res. 2016, 44, 779–791. [Google Scholar] [CrossRef]
- Wang, Z.M.; Wagner, J.; Ghosal, S.; Bedi, G.; Wall, S. SEM/EDS and opticalmicroscopy analyses of microplastics in ocean trawl and fish guts. Sci. Total Environ. 2017, 603, 616–626. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo-Ruz, V.; Gutow, L.; Thompson, R.C.; Thiel, M. Microplastics in the Marine Environment: A Review of the Methods Used for Identification and Quantification. Environ. Sci. Technol. 2012, 46, 3060–3075. [Google Scholar] [CrossRef] [PubMed]
- Cutroneo, L.; Reboa, A.; Besio, G.; Borgogno, F.; Canesi, L.; Canuto, S.; Dara, M.; Enrile, F.; Forioso, I.; Greco, G.; et al. Microplastics in seawater: Sampling strategies, laboratory methodologies, and identification techniques applied to port environment. Environ. Sci. Pollut. Res. 2020, 27, 8938–8952. [Google Scholar] [CrossRef] [PubMed]
- Gross, J.; Sayle, S.; Karow, A.R.; Bakowsky, U.; Garidel, P. Nanoparticle tracking analysis of particle size and concentration detection in suspensions of polymer and protein samples: Influence of experimental and data evaluation parameters. Eur. J. Pharm. Biopharm. 2016, 104, 30–41. [Google Scholar] [CrossRef] [PubMed]
- Vianello, A.; Boldrin, A.; Guerriero, P.; Moschino, V.; Rella, R.; Sturaro, A.; Da Ros, L. Microplastic particles in sediments of Lagoon of Venice, Italy: First observations on occurrence, spatial patterns and identification. Estuar. Coast. Shelf Sci. 2013, 130, 54–61. [Google Scholar] [CrossRef]
- Cui, Y.Z.; Liu, M.Y.; Selvam, S.; Ding, Y.C.; Wu, Q.Q.; Pitchaimani, V.S.; Huang, P.; Ke, H.W.; Zheng, H.W.; Liu, F.J.; et al. Microplastics in the surface waters of the South China sea and the western Pacific Ocean: Different size classes reflecting various sources and transport. Chemosphere 2022, 299, 134456. [Google Scholar] [CrossRef] [PubMed]
- Carbery, M.; Herb, F.; Reynes, J.; Pham, C.K.; Fong, W.K.; Lehner, R. How small is the big problem? Small microplastics <300 μm abundant in marine surface waters of the Great Barrier Reef Marine Park. Mar. Pollut. Bull. 2022, 184, 114179. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Tu, C.; Yang, J.; Fu, C.C.; Li, Y.; Waniek, J.J. Trapping of Microplastics in Halocline and Turbidity Layers of the Semi-enclosed Baltic Sea. Front. Mar. Sci. 2021, 8, 761566. [Google Scholar] [CrossRef]
- Sbrana, A.; Valente, T.; Bianchi, J.; Franceschini, S.; Piermarini, R.; Saccomandi, F.; de Lucia, A.G.; Camedda, A.; Matiddi, M.; Silvestri, C. From inshore to offshore: Distribution of microplastics in three Italian seawaters. Environ. Sci. Pollut. Res. 2023, 30, 21277–21287. [Google Scholar] [CrossRef] [PubMed]
- Covernton, G.A.; Pearce, C.M.; Gurney-Smith, H.J.; Chastain, S.G.; Ross, P.S.; Dower, J.F.; Dudas, S.E. Size and shape matter: A preliminary analysis of microplastic sampling technique in seawater studies with implications for ecological risk assessment. Sci. Total Environ. 2019, 667, 124–132. [Google Scholar] [CrossRef]
- Zhou, Z.; Wan, L.; Cai, W.Q.; Tang, J.; Wu, Z.J.; Zhang, K.D. Species-specific microplastic enrichment characteristics of scleractinian corals from reef environment: Insights from an in-situ study at the Xisha Islands. Sci. Total Environ. 2022, 815, 152845. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, A.; Sadeghi, P.; Abadi, Z.T.R. Characterization of microplastics in digestive tract of commercial fish species from the Oman Sea. Mar. Pollut. Bull. 2023, 197, 115769. [Google Scholar] [CrossRef] [PubMed]
- Galafassi, S.; Campanale, C.; Massarelli, C.; Uricchio, V.F.; Volta, P. Do Freshwater Fish Eat Microplastics? A Review with A Focus on Effects on Fish Health and Predictive Traits of MPs Ingestion. Water 2021, 13, 2214. [Google Scholar] [CrossRef]
- Gunaalan, K.; Nielsen, T.G.; Torres, R.R.; Lorenz, C.; Vianello, A.; Andersen, C.A.; Vollertsen, J.; Almeda, R. Is Zooplankton an Entry Point of Microplastics into the Marine Food Web? Environ. Sci. Technol. 2023, 57, 11643–11655. [Google Scholar] [CrossRef] [PubMed]
- Bermudez, J.R.; Swarzenski, P.W. A microplastic size classification scheme aligned with universal plankton survey methods. Methodsx 2021, 8, 101516. [Google Scholar] [CrossRef] [PubMed]
- Alfonso, M.B.; Lindsay, D.J.; Arias, A.H.; Nakano, H.; Jandang, S.; Isobe, A. Zooplankton as a suitable tool for microplastic research. Sci. Total Environ. 2023, 905, 167329. [Google Scholar] [CrossRef] [PubMed]
- Sfriso, A.A.; Tomio, Y.; Juhmani, A.S.; Sfriso, A.; Munari, C.; Mistri, M. Macrophytes: A Temporary Sink for Microplastics in Transitional Water Systems. Water 2021, 13, 3032. [Google Scholar] [CrossRef]
- Sheath, R.G.; Wehr, J.D. 1—Introduction to freshwater algae. In Aquatic Ecology, Freshwater Algae of North America; Ecology and Classification; Academic Press: Cambridge, MA, USA, 2003; pp. 1–9. [Google Scholar] [CrossRef]
- Wu, Y.P.; Beardall, J.; Gao, K.S. Physiological Responses of a Model Marine Diatom to Fast pH Changes: Special Implications of Coastal Water Acidification. PLoS ONE 2015, 10, e0141163. [Google Scholar] [CrossRef] [PubMed]
- Pakhomova, S.; Berezina, A.; Lusher, A.L.; Zhdanov, I.; Silvestrova, K.; Zavialov, P.; van Bavel, B.; Yakushev, E. Microplastic variability in subsurface water from the Arctic to Antarctica. Environ. Pollut. 2022, 298, 118808. [Google Scholar] [CrossRef] [PubMed]
- Faussone, G.C.; Krzan, A.; Grilc, M. Conversion of Marine Litter from Venice Lagoon into Marine Fuels via Thermochemical Route: The Overview of Products, Their Yield, Quality and Environmental Impact. Sustainability 2021, 13, 9481. [Google Scholar] [CrossRef]
- Consoli, P.; Falautano, M.; Sinopoli, M.; Perzia, P.; Canese, S.; Esposito, V.; Battaglia, P.; Romeo, T.; Andaloro, F.; Galgani, F.; et al. Composition and abundance of benthic marine litter in a coastal area of the central Mediterranean Sea. Mar. Pollut. Bull. 2018, 136, 243–247. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Wang, Q.; Chen, H. Challenges in characterization of nanoplastics in the environment. Front. Environ. Sci. Eng. 2022, 16, 11. [Google Scholar] [CrossRef]




| Study Area | Sampling | Analytical Tool | Characteristics of MPs/NPs | Ref. |
|---|---|---|---|---|
| Two sites from Wadden Sea (The Netherlands) | 5 L Niskin bottle—site A, PP bucket—site B. | Thermal desorption—proton transfer reaction—mass spectrometry (TD-PTR-MS) | PS NPs, PET NPs. | [29] |
| Saint Marc basin, Rialto Bridge, Venice-Lido port inlet | Bulk, 5 L jerry cans, sampling conducted on 15th June 2022. | Optical microscopy | 3.6 ± 1.1 MPs/L, 1.6 ± 1.1 MPs/L, 1 ± 0 MPs/L. | [30] |
| Banderas Bay, Mexico | Wooplankton net (0.3 m diameter, 333 μm mesh size), floating plastic monitoring conducted from 2016 to 2018. | µ-ATR-FTIR | 79% of marine litter were MPs (45% PP, 43% PE). The most represented size class was the 1–2 mm. | [31] |
| Marine-protected areas from Peru | Bulk method—15 L bucket. Sampling conducted from January to May of 2022. | ATR-FTIR | 4.19 ± 2.23 MPs/L (1.60–9.37 MPs/L). The particles were largely composed of fibers (91.6%) of blue color (81.8%). MPs mean size of 1260 µm. Size classes: 46.4% in the range of 1000–5000 µm. 26.2% in the range of 500–1000 µm. | [32] |
| 34 sites from India’s east coast | Manta nets, neuston nets, plankton nets, and bongo nets 750 mL collection jar. Sampling conducted in the monsoon period of August 2022. | Optical microscopy, SEM, and FTIR | 43% PE, 42% PP, and 15% PS. Mean: 12 MPs/site. Morphology: 59% fibers, 24% films, 10% fragments, and 7% pellets. Color distribution: 26% white, 16% black, 12% grey 12%, 14% red, 12% blue, 10% yellow, and 10% green. | [33] |
| 16 different stations of the South Yellow Sea | 100 L of surface seawater (0–50 cm) was collected in January, April, and August 2018. | ATR-FTIR | High abundance of 6.5 ± 2.1 items/L. 78% of MPs were <500 μm. ~90% were fibers. | [34] |
| Tuscan coast, Italy | Manta trawl (330 μm mesh size), 24 surface tows. Sampling conducted on November–December 2013 and April–May 2014. | Stereomicroscope FT-IR | 1586 MPs, corresponding to 0.16 ± 0.26 mg/m3. The most abundant size class is 1–2.5 mm; 81% fragments; PE > 66% and PP 28%. | [35] |
| Venice-Lido Port Inlet | Rialto Bridge | Saint Marc Basin | |
|---|---|---|---|
| pH (pH units) | 7.8 ± 0.04 | 7.7 ± 0.02 | 7.9 ± 0 |
| Electrical conductivity (mS/cm) | 49.5 ± 0.47 | 48.7 ± 0.1 | 48.8 ± 0.05 |
| Salinity | 32.5 ± 0.15 | 31.7 ± 0.15 | 31.8 ± 0.2 |
| Chloride ions (g/L) | 20.2 ± 0.95 | 14.4 ± 0.7 | 20.1 ± 1.13 |
| Total dissolved solids (g/L) | 30.8 ± 0.15 | 30.0 ± 0.15 | 30.1 ± 0.15 |
| Element | Weight (%) | |||||
|---|---|---|---|---|---|---|
| Venice-Lido Port Inlet | Grand Canal at the Rialto Bridge | Saint Marc Basin | ||||
| (a) | (b) | (c) | (d) | (e) | (f) | |
| Carbon (C) | 62.83 | 16.45 | 35.42 | 31.01 | 41.85 | |
| Nitrogen (N) | 1.50 | 0.03 | ||||
| Oxygen (O) | 32.51 | 47.78 | 41.20 | 55.84 | 42.32 | 39.28 |
| Sodium (Na) | 1.21 | 6.62 | 4.29 | 5.80 | 5.32 | 5.57 |
| Magnesium (Mg) | 0.04 | 0.44 | 0.63 | |||
| Aluminum (Al) | 0.33 | 2.30 | 1.79 | 1.60 | 1.69 | 0.77 |
| Silicon (Si) | 1.22 | 23.86 | 9.72 | 33.07 | 9.83 | 3.69 |
| Chloride (Cl) | 0.21 | 3.69 | 0.36 | |||
| Potassium (K) | 0.19 | 1.57 | 2.35 | 3.69 | 8.68 | 7.87 |
| Calcium (Ca) | 1.35 | 1.19 | ||||
| Titanium (Ti) | 0.68 | 0.72 | ||||
| Water Sample | Concentration of MPs (mg/m3) | Concentration of Nanoparticles (mg/m3) | Total Concentration (mg/m3) |
|---|---|---|---|
| Venice-Lido port inlet | 67.31 | 0.66 × 10−3 | 67.311 |
| Grand Canal at the Rialto Bridge | 34.93 | 0.167 × 10−2 | 34.397 |
| Saint Marc basin | 256.52 | 0.727 × 10−2 | 256.532 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cecchi, T.; Poletto, D.; Berbecaru, A.C.; Cârstea, E.M.; Râpă, M. Assessing Microplastics and Nanoparticles in the Surface Seawater of Venice Lagoon—Part I: Methodology of Research. Materials 2024, 17, 1759. https://doi.org/10.3390/ma17081759
Cecchi T, Poletto D, Berbecaru AC, Cârstea EM, Râpă M. Assessing Microplastics and Nanoparticles in the Surface Seawater of Venice Lagoon—Part I: Methodology of Research. Materials. 2024; 17(8):1759. https://doi.org/10.3390/ma17081759
Chicago/Turabian StyleCecchi, Teresa, Davide Poletto, Andrei Constantin Berbecaru, Elfrida Mihaela Cârstea, and Maria Râpă. 2024. "Assessing Microplastics and Nanoparticles in the Surface Seawater of Venice Lagoon—Part I: Methodology of Research" Materials 17, no. 8: 1759. https://doi.org/10.3390/ma17081759
APA StyleCecchi, T., Poletto, D., Berbecaru, A. C., Cârstea, E. M., & Râpă, M. (2024). Assessing Microplastics and Nanoparticles in the Surface Seawater of Venice Lagoon—Part I: Methodology of Research. Materials, 17(8), 1759. https://doi.org/10.3390/ma17081759







