Synthesis Methods and Optical Sensing Applications of Plasmonic Metal Nanoparticles Made from Rhodium, Platinum, Gold, or Silver
Abstract
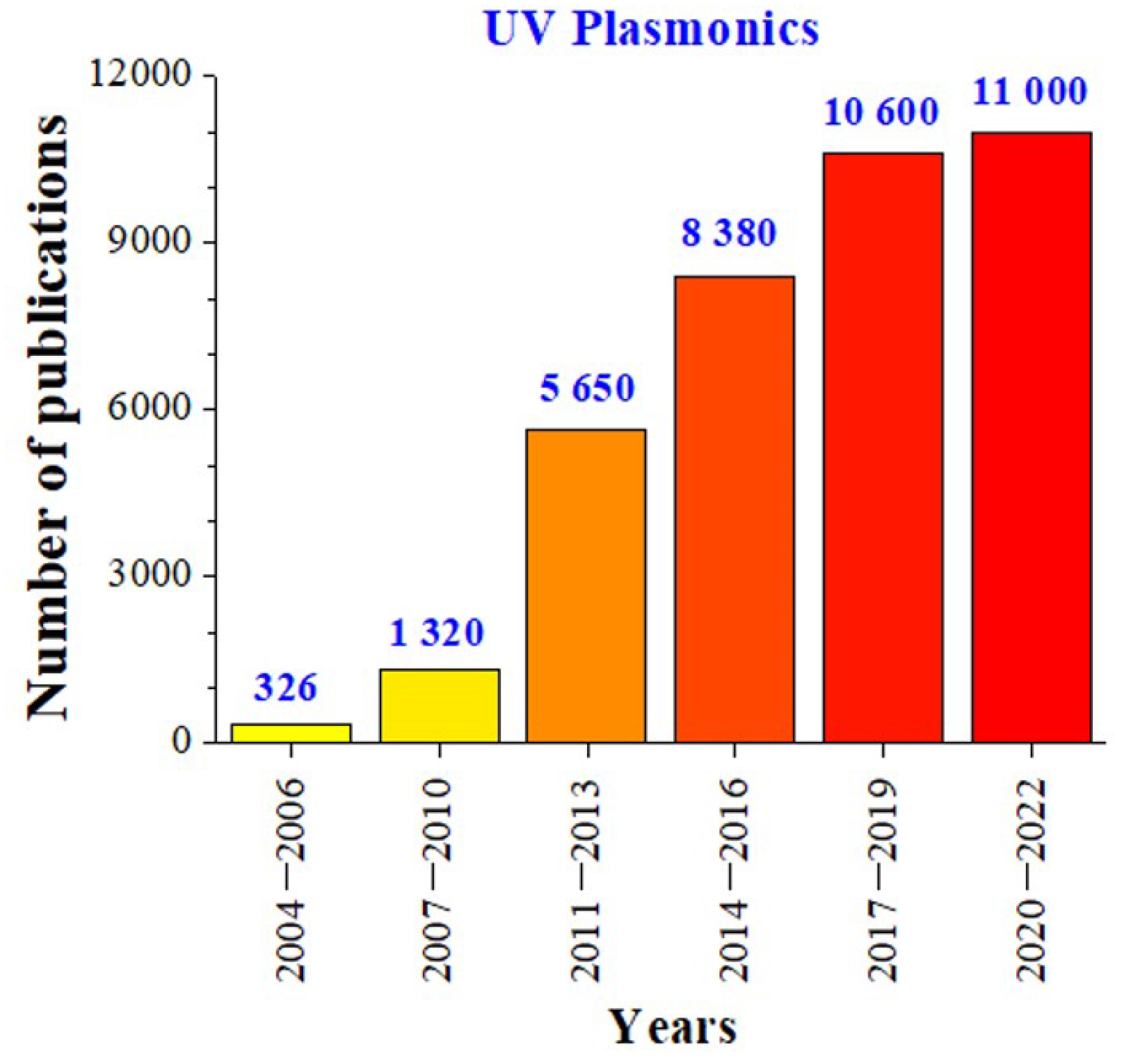
1. Introduction
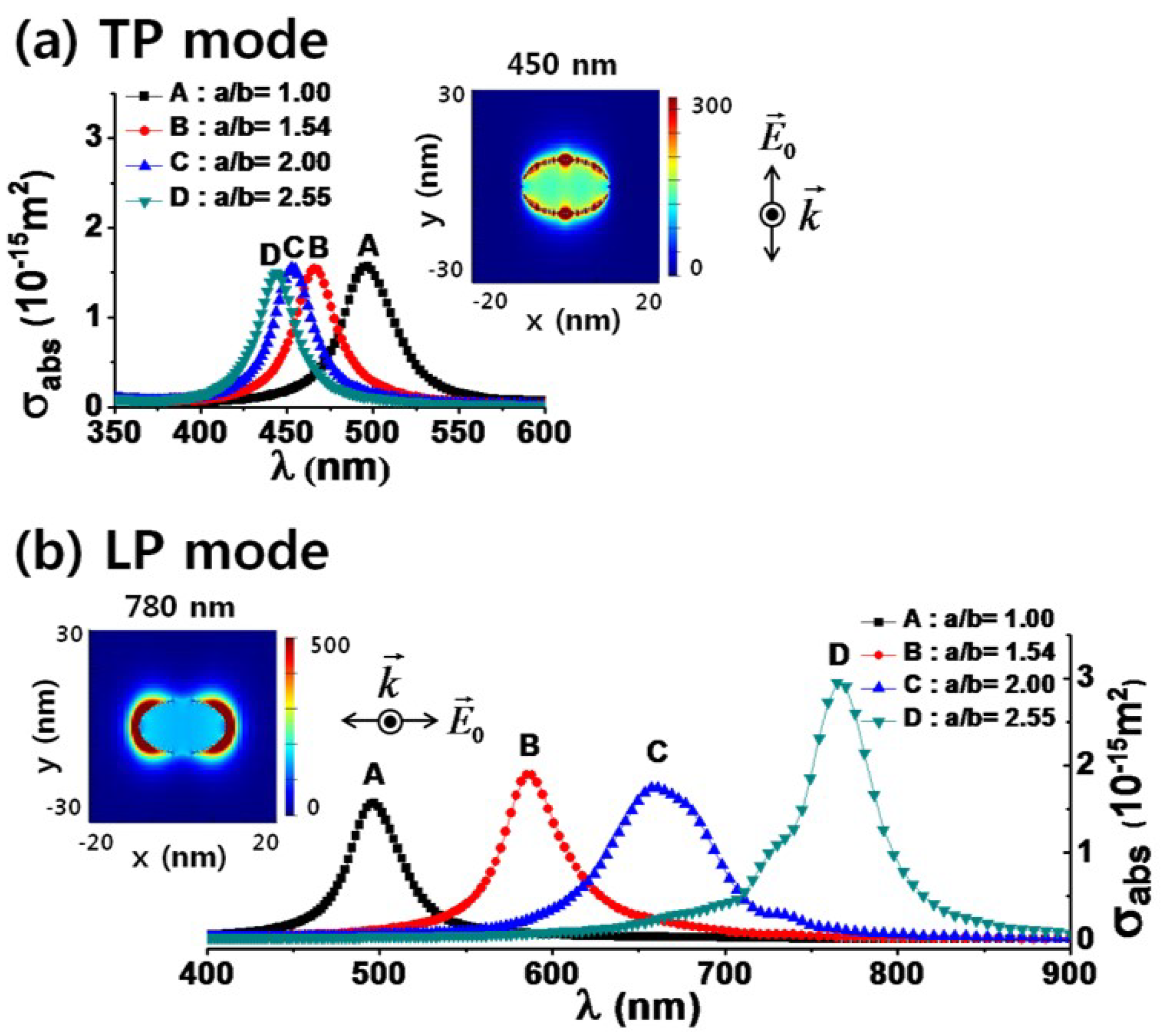
2. Theoretical Background for Plasmon-Enhanced Optical Sensing of Metal Nanoparticles
3. Optical Sensing Applications of Rhodium, Platinum, Gold, and Silver Nanoparticles
3.1. Rhodium- and Platinum-Nanoparticle-Based Optical Sensing
3.1.1. Rhodium Nanoparticle-Based Optical Sensing
3.1.2. Platinum Nanoparticle-Based Optical Sensing
3.2. Gold- and Silver-Nanoparticle-Based Optical Sensing
3.2.1. Gold Nanowire-Based Optical Sensing
3.2.2. Gold Nanoparticle-Based Optical Sensing
3.2.3. Silver Nanoparticle-Based Optical Sensing
3.2.4. Practical Applications of Plasmonic Metal Nanoparticle-Based Optical Sensing
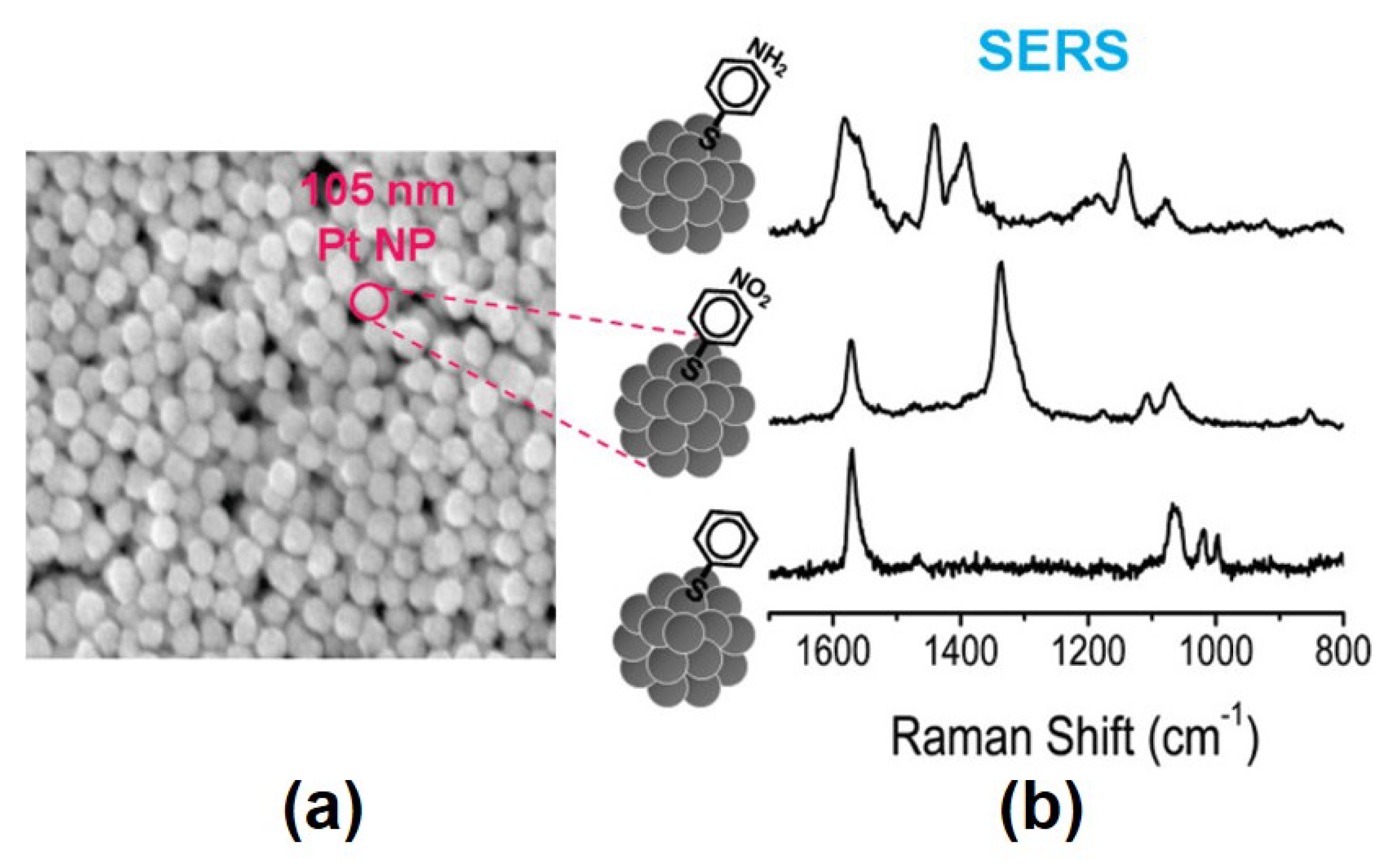
4. Synthesis of Rhodium and Platinum Nanoparticles
4.1. Chemical Synthesis of Rhodium and Platinum Nanoparticles
4.1.1. Chemical Synthesis of Rhodium Nanoparticles
4.1.2. Chemical Synthesis of Platinum Nanoparticles
4.2. Physical Synthesis of Rhodium and Platinum Nanoparticles
4.2.1. Physical Synthesis of Rhodium Nanoparticles
4.2.2. Physical Synthesis of Platinum Nanoparticles
4.3. Green Synthesis of Rhodium and Platinum Nanoparticles
4.3.1. Green Synthesis of Rhodium Nanoparticles
4.3.2. Green Synthesis of Platinum Nanoparticles
5. Synthesis of Gold and Silver Nanoparticles
6. Concluding Remarks
- Compared with semiconductor nanoparticles, plasmonic metal nanoparticles have advantages and limitations [212]. The advantages of metal nanoparticles are their high sensitivity, simple fabrication process, tunable shape, and size. The limitations include high cost, low stability, and poor biocompatibility.
- Plasmonic metal nanoparticles can be used in theranostics for cancer diagnostics and treatment. Various gold-based organic and inorganic nanoparticles have attracted increasing research attention due to the feasibility of surface functionalization, excellent tumor specificity, high drug-loading capacity, and biocompatibility [108,213]. Platinum and rhodium nanoparticles are promising for applications in deep-UV SERS, therapeutics, diagnostics, and biosensing fields [214,215].
- Plasmonic metal nanoparticles can be used for single-molecule and single-cell detection purposes [105,216]. They can be extremely useful in the sensing of pathogens [121,217] and intracellular components [218]. Metal nanoparticle-based SERS sensing is widely used in environmental contaminant monitoring [123]. Another challenging issue is metallic nanoparticle application as nanocarriers for drug delivery systems [219].
- This paper highlights the promising potential of optical sensors over a wide range of tasks. It provides a detailed analysis for optical sensing applications of plasmonic metal nanoparticles, and reveals the prospects for optical sensing applications of metal nanoparticles in the UV and visible ranges. In the near future, optical sensing applications of metal nanoparticles by IR- and far-IR-plasmonics will be reviewed.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Agranovich, V.M.; Mills, D.L. Surface Polaritons: Electromagnetic Waves at Surfaces and Interfaces; North Holland: Amsterdam, The Netherlands, 1982; Chapter 9. [Google Scholar]
- Philip, A.; Kumar, A.R. The performance enhancement of surface plasmon resonance optical sensors using nanomaterials: A review. Coord. Chem. Rev. 2022, 458, 214424. [Google Scholar]
- Hutter, E.; Fendler, J.H. Exploitation of localized surface plasmon resonance. Adv. Mater. 2004, 16, 1685–1706. [Google Scholar]
- Maier, S.A.; Atwater, H.A. Plasmonics: Localization and guiding of electromagnetic energy in metal/dielectric structures. J. Appl. Phys. 2005, 98, 011101. [Google Scholar]
- Petryayeva, E.; Krull, U.J. Localized surface plasmon resonance: Nanostructures, bioassays and biosensing—A review. Anal. Chim. Acta 2011, 706, 8–24. [Google Scholar] [CrossRef]
- Ozbay, E. Plasmonics: Merging photonics and electronics at nanoscale dimensions. Science 2006, 311, 189–193. [Google Scholar]
- Huang, L.; Chen, X.; Mühlenbernd, H.; Zhang, H.; Chen, S.; Bai, B.; Tan, Q.; Jin, G.; Cheah, K.W.; Qiu, C.W.; et al. Three-dimensional optical holography using a plasmonic metasurface. Nat. Commun. 2013, 4, 1–8. [Google Scholar] [CrossRef]
- Kim, M.S.; Ham, W.K.; Kim, W.; Hwangbo, C.K.; Choi, E.H.; Lee, G.J. Optical properties of nucleobase thin films as studied by attenuated total reflection and surface-enhanced Raman spectroscopy. Opt. Mater. 2018, 78, 531–537. [Google Scholar] [CrossRef]
- Kim, J.B.; Zou, Y.; Kim, Y.D.; Kim, J.J.; Hwangbo, C.K. Multiple surface plasmon waves in [prism/Ag/SiO2 helical thin film] Kretschmann configuration. Thin Solid Film. 2011, 520, 1451–1453. [Google Scholar] [CrossRef]
- Kim, J.B.; Peranantham, P.; Shin, Y.S.; Hwangbo, C.K.; Zou, Y. Surface plasmon resonances at a [Ag/SiO2 helical thin-film] interface. J. Kor. Phys. Soc. 2012, 60, 1249–1252. [Google Scholar] [CrossRef]
- Lee, G.J.; Choi, E.H.; Ham, W.K.; Hwangbo, C.K.; Cho, M.J.; Choi, D.H. Circular dichroism, surface-enhanced Raman scattering, and spectroscopic ellipsometry studies of chiral polyfluorene-phenylene films. Opt. Mater. Express 2016, 6, 767–781. [Google Scholar]
- Chau, Y.F.; Yeh, H.H. A comparative study of solid-silver and silver-shell nanodimers on surface plasmon resonances. J. Nanopart. Res. 2011, 13, 637–644. [Google Scholar] [CrossRef]
- Chau, Y.F.; Jiang, J.C.; Chao, C.T.C.; Chiang, H.P.; Lim, C.M. Manipulating near field enhancement and optical spectrum in a pair-array of the cavity resonance based plasmonic nanoantennas. J. Phys. D Appl. Phys. 2016, 49, 475102. [Google Scholar] [CrossRef]
- Moskovits, M.; Tay, L.L.; Yang, J.; Haslett, T. SERS and the single molecule. In Optical Properties of Nanostructured Random Media; Springer: Berlin/Heidelberg, Germany, 2002; pp. 215–227. [Google Scholar]
- Wang, Z.J.; Pan, S.L.; Krauss, T.D.; Du, H.; Rothberg, L.J. The structural basis for giant enhancement enabling single-molecule Raman scattering. Proc. Natl. Acad. Sci. USA 2003, 100, 8638–8643. [Google Scholar] [CrossRef]
- Lakowicz, J.R.; Malicka, J.; Gryczynski, I.; Gryczynski, Z.; Geddes, C.D. Radiative decay engineering: The role of photonic mode density in biotechnology. J. Phys. D: Appl. Phys. 2003, 36, R240. [Google Scholar] [CrossRef]
- Guzatov, D.V.; Klimov, V.V. Radiative decay engineering by triaxial nanoellipsoids. Chem. Phys. Lett. 2005, 412, 341–346. [Google Scholar] [CrossRef]
- Wang, L.; Hasanzadeh Kafshgari, M.; Meunier, M. Optical properties and applications of plasmonic-metal nanoparticles. Adv. Funct. Mater. 2020, 30, 2005400. [Google Scholar] [CrossRef]
- de Aberasturi, D.J.; Serrano-Montes, A.B.; Liz-Marzan, L.M. Modern applications of plasmonic nanoparticles: From energy to health. Adv. Opt. Mater. 2015, 3, 602–617. [Google Scholar] [CrossRef]
- Vo-Dinh, T.; Liu, Y.; Fales, A.M.; Ngo, H.; Wang, H.N.; Register, J.K.; Yuan, H.; Norton, S.J.; Griffin, G.D. SERS nanosensors and nanoreporters: Golden opportunities in biomedical applications. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2015, 7, 17–33. [Google Scholar] [CrossRef]
- Fan, X.; Zheng, W.; Singh, D.J. Light scattering and surface plasmons on small spherical particles. Light Sci. Appl. 2014, 3, e179. [Google Scholar] [CrossRef]
- Stiles, P.L.; Dieringer, J.A.; Shah, N.C.; Van Duyne, R.P. Surface-enhanced Raman spectroscopy. Annu. Rev. Anal. Chem. 2008, 1, 601–626. [Google Scholar] [CrossRef]
- Kelly, K.L.; Coronado, E.; Zhao, L.L.; Schatz, G.C. The optical properties of metal nanoparticles: The influence of size, shape, and dielectric environment. J. Phys. Chem. B 2003, 107, 668–677. [Google Scholar] [CrossRef]
- Qiu, Y.; Kuang, C.; Liu, X.; Tang, L. Single-molecule surface-enhanced Raman spectroscopy. Sensors 2022, 22, 4889. [Google Scholar] [CrossRef]
- Li, Z.Y. Mesoscopic and microscopic strategies for engineering plasmon-enhanced Raman scattering. Adv. Opt. Mater. 2018, 6, 1701097. [Google Scholar] [CrossRef]
- Jeong, Y.; Kook, Y.M.; Lee, K.; Koh, W.G. Metal enhanced fluorescence (MEF) for biosensors: General approaches and a review of recent developments. Biosens. Bioelectron. 2018, 111, 102–116. [Google Scholar] [CrossRef]
- Deng, W.; Xie, F.; Baltar, H.T.; Goldys, E.M. Metal-enhanced fluorescence in the life sciences: Here, now and beyond. Phys. Chem. Chem. Phys. 2013, 15, 15695–15708. [Google Scholar] [CrossRef]
- Lakowicz, J.R.; Geddes, C.D.; Gryczynski, I.; Malicka, J.; Gryczynski, Z.; Aslan, K.; Lukomska, J.; Matveeva, E.; Zhang, J.; Badugu, R.; et al. Advances in surface-enhanced fluorescence. J. Fluoresc. 2004, 14, 425–441. [Google Scholar] [CrossRef]
- Aslan, K.; Gryczynski, I.; Malicka, J.; Matveeva, E.; Lakowicz, J.R.; Geddes, C.D. Metal-enhanced fluorescence: An emerging tool in biotechnology. Curr. Opin. Biotech. 2005, 16, 55–62. [Google Scholar] [CrossRef]
- Aslan, K.; Lakowicz, J.R.; Szmacinski, H.; Geddes, C.D. Metal-enhanced fluorescence solution-based sensing platform. J. Fluoresc. 2004, 14, 677–679. [Google Scholar] [CrossRef]
- Taflove, A.; Hagness, S.C. Computational Electrodynamics: The Finite-Difference Time-Domain Method, 3rd ed.; Artech House: Boston, MA, USA, 2005; Chapter 3. [Google Scholar]
- Oubre, C.; Nordlander, P. Optical properties of metallodielectric nanostructures calculated using the finite difference time domain method. J. Phys. Chem. B 2004, 108, 17740–17747. [Google Scholar] [CrossRef]
- Zyubin, A.Y.; Kon, I.I.; Poltorabatko, D.A.; Samusev, I.G. FDTD simulations for rhodium and platinum nanoparticles for UV plasmonics. Nanomaterials 2023, 13, 897. [Google Scholar] [CrossRef]
- Kim, J.; Lee, G.J.; Park, I.; Lee, Y.P. Finite-difference time-domain numerical simulation study on the optical properties of silver nanocomposites. J. Nanosci. Nanotechnol. 2012, 12, 5527–5531. [Google Scholar] [CrossRef]
- Kim, J.; Lee, G.J.; Park, I.; Lee, Y.P. Influence of ZnO nanorod morphology on optical confinement–Finite-Difference Time-Domain numerical simulation study. J. Nanosci. Nanotechnol. 2011, 11, 7238–7241. [Google Scholar] [CrossRef]
- Tira, C.; Tira, D.; Simon, T.; Astilean, S. Finite-Difference Time-Domain (FDTD) design of gold nanoparticle chains with specific surface plasmon resonance. J. Mol. Struct. 2014, 1072, 137–143. [Google Scholar] [CrossRef]
- Hermann, R.J.; Gordon, M.J. Quantitative comparison of plasmon resonances and field enhancements of near-field optical antennae using FDTD simulations. Opt. Express 2018, 26, 27668. [Google Scholar] [CrossRef]
- Bertó-Roselló, F.; Xifré-Pérez, E.; Ferré-Borrull, J.; Marsal, L.F. 3D-FDTD modelling of optical biosensing based on gold-coated nanoporous anodic alumina. Results Phys. 2018, 11, 1008–1014. [Google Scholar] [CrossRef]
- Gao, Y.; Zhang, J.; Zhang, Z.; Li, Z.; Xiong, Q.; Deng, L.; Zhou, Q.; Meng, L.; Du, Y.; Zuo, T.; et al. Plasmon-enhanced perovskite solar cells with efficiency beyond 21%: The asynchronous synergistic effect of water and gold nanorods. ChemPlusChem 2021, 86, 291–297. [Google Scholar] [CrossRef]
- Feng, L.; Niu, M.; Wen, Z.; Hao, X. Recent advances of plasmonic organic solar cells: Photophysical investigations. Polymers 2018, 10, 123. [Google Scholar] [CrossRef]
- Cheng, L.; Zhu, G.; Liu, G.; Zhu, L. FDTD simulation of the optical properties for gold nanoparticles. Mater. Res. Express 2020, 7, 125009. [Google Scholar] [CrossRef]
- Zeng, S.; Baillargeat, D.; Ho, H.P.; Yong, K.T. Nanomaterials enhanced surface plasmon resonance for biological and chemical sensing applications. Chem. Soc. Rev. 2014, 43, 3426–3452. [Google Scholar] [CrossRef]
- Zhao, J.; Pinchuk, A.O.; McMahon, J.M.; Li, S.; Ausman, L.K.; Atkinson, A.L.; Schatz, G.C. Methods for describing the electromagnetic properties of silver and gold nanoparticles. Accounts Chem. Res. 2008, 41, 1710–1720. [Google Scholar] [CrossRef]
- Huang, Y.; Ma, L.; Li, J.; Zhang, Z. Nanoparticle-on-mirror cavity modes for huge and/or tunable plasmonic field enhancement. Nanotechnology 2017, 28, 105203. [Google Scholar] [CrossRef] [PubMed]
- Rahaman, M.H.; Kemp, B.A. Analytical model of plasmonic resonance from multiple core-shell nanoparticles. Opt. Eng. 2017, 56, 121903. [Google Scholar] [CrossRef]
- Seker, I.; Karatutlu, A.; Gölcök, K.; Karakız, M.; Ortac, B.A. A systematic study on Au-capped Si nanowhiskers for size-dependent improved biosensing applications. Plasmonics 2020, 15, 1739–1745. [Google Scholar] [CrossRef]
- Masharin, M.A.; Berestennikov, A.S.; Barettin, D.; Voroshilov, P.M.; Ladutenko, K.S.; Carlo, A.D.; Makarov, S.V. Giant enhancement of radiative recombination in perovskite light-emitting diodes with plasmonic core-shell nanoparticles. Nanomaterials 2020, 11, 45. [Google Scholar] [CrossRef]
- Xie, Z.; Zhao, F.; Zou, S.; Zhu, F.; Zhang, Z.; Wang, W. TiO2 nanorod arrays decorated with Au nanoparticles as sensitive and recyclable SERS substrates. J. Alloys Compd. 2021, 861, 157999. [Google Scholar] [CrossRef]
- Butt, M.A.; Khonina, S.N.; Kazanskiy, N.L. Plasmonic refractive index sensor based on metal-insulator-metal waveguides with high sensitivity. J. Mod. Opt. 2019, 66, 1038–1043. [Google Scholar] [CrossRef]
- Ovejero, J.G.; Morales, I.; de la Presa, P.; Mille, N.; Carrey, J.; Garcia, M.A.; Hernando, A.; Herrasti, P. Hybrid nanoparticles for magnetic and plasmonic hyperthermia. Phys. Chem. Chem. Phys. 2018, 20, 24065–24073. [Google Scholar] [CrossRef]
- Manrique-Bedoya, S.; Abdul-Moqueet, M.; Lopez, P.; Gray, T.; Disiena, M.; Locker, A.; Kwee, S.; Tang, L.; Hood, R.L.; Feng, Y.; et al. Multiphysics modeling of plasmonic photothermal heating effects in gold nanoparticles and nanoparticle arrays. J. Phys. Chem. C 2020, 124, 17172–17182. [Google Scholar] [CrossRef]
- Elhoussaine, O.; Abdelaziz, E.; Hicham, M.; Abdenbi, B. Plasmonic Nanorods investigating for cancer cell imaging and photothermal therapy. In Proceedings of the 2020 1st International Conference on Innovative Research in Applied Science, Engineering and Technology (IRASET), Meknes, Morocco, 16–19 April 2020; pp. 1–4. [Google Scholar]
- Xu, P.; Lu, W.; Zhang, J.; Zhang, L. Efficient hydrolysis of ammonia borane for hydrogen evolution catalyzed by plasmonic Ag@Pd core-shell nanocubes. ACS Sustain. Chem. Eng. 2020, 8, 12366–12377. [Google Scholar] [CrossRef]
- Sosa, I.O.; Noguez, C.; Barrera, R.G. Optical properties of metal nanoparticles with arbitrary shapes. J. Phys. Chem. B 2003, 107, 6269–6275. [Google Scholar] [CrossRef]
- Makkar, P.; Ghosh, N.N. A review on the use of DFT for the prediction of the properties of nanomaterials. RSC Adv. 2021, 11, 27897–27924. [Google Scholar] [CrossRef] [PubMed]
- Jackson, J.D. Classical Electrodynamics, 3rd ed.; John Wiley & Sons: Hoboken, NJ, USA, 1999; Chapter 6. [Google Scholar]
- Darienzo, R.E.; Chen, O.; Sullivan, M.; Mironava, T.; Tannenbaum, R. Au nanoparticles for SERS: Temperature-controlled nanoparticle morphologies and their Raman enhancing properties. Mater. Chem. Phys. 2020, 240, 122143. [Google Scholar] [CrossRef] [PubMed]
- Qayyum, H.; Amin, S.; Ahmed, W.; Mohamed, T.; Rehman, Z.U.; Hussain, S. Laser-based two-step synthesis of Au-Ag alloy nanoparticles and their application for surface-enhanced Raman spectroscopy (SERS) based detection of rhodamine 6G and urea nitrate. J. Mol. Liq. 2022, 365, 120120. [Google Scholar] [CrossRef]
- Kim, K.; Lee, H.S. Effect of Ag and Au nanoparticles on the SERS of 4-aminobenzenethiol assembled on powdered copper. J. Phys. Chem. B 2005, 109, 18929–18934. [Google Scholar] [CrossRef]
- Aslan, K.; Malyn, S.N.; Geddes, C.D. Angular-dependent metal-enhanced fluorescence from silver colloid-deposited films: Opportunity for angular-ratiometric surface assays. Analyst 2007, 132, 1112–1121. [Google Scholar] [CrossRef]
- Aslan, K.; Malyn, S.N.; Geddes, C.D. Metal-enhanced fluorescence from gold surfaces: Angular dependent emission. J. Fluoresc. 2007, 17, 7–13. [Google Scholar] [CrossRef]
- Cabello, G.; Nwoko, K.C.; Marco, J.F.; Sánchez-Arenillas, M.; Méndez-Torres, A.M.; Feldmann, J.; Yáñez, C.; Smith, T.A. Cu@ Au self-assembled nanoparticles as SERS-active substrates for (bio) molecular sensing. J. Alloys Compd. 2019, 791, 184–192. [Google Scholar] [CrossRef]
- de Barros Santos, E.; Sigoli, F.A.; Mazali, I.O. Metallic Cu nanoparticles dispersed into porous glass: A simple green chemistry approach to prepare SERS substrates. Mater. Lett. 2013, 108, 172–175. [Google Scholar] [CrossRef]
- Zhang, Y.; Aslan, K.; Previte, M.J.; Geddes, C.D. Metal-enhanced fluorescence from copper substrates. Appl. Phys. Lett. 2007, 90, 173116. [Google Scholar] [CrossRef]
- Gutiérrez, Y.; Alcaraz de la Osa, R.; Ortiz, D.; Saiz, J.M.; González, F.; Moreno, F. Plasmonics in the ultraviolet with aluminum, gallium, magnesium and rhodium. Appl. Sci. 2018, 8, 64. [Google Scholar] [CrossRef]
- McMahon, J.M.; Schatz, G.C.; Gray, S.K. Plasmonics in the ultraviolet with the poor metals Al, Ga, In, Sn, Tl, Pb, and Bi. Phys. Chem. Chem. Phys. 2013, 15, 5415–5423. [Google Scholar] [CrossRef]
- Tanabe, I.; Shimizu, M.; Kawabata, R.; Katayama, C.; Fukui, K.I. Far-and deep-ultraviolet surface plasmon resonance using Al film for efficient sensing of organic thin overlayer. Sens. Actuators A-Phys. 2020, 301, 111661. [Google Scholar] [CrossRef]
- Knight, M.W.; King, N.S.; Liu, L.; Everitt, H.O.; Nordlander, P.; Halas, N.J. Aluminum for plasmonics. ACS Nano 2014, 8, 834–840. [Google Scholar] [CrossRef]
- Gutierrez, Y.; Ortiz, D.; Sanz, J.M.; Saiz, J.M.; Gonzalez, F.; Everitt, H.O.; Moreno, F. How an oxide shell affects the ultraviolet plasmonic behavior of Ga, Mg, and Al nanostructures. Opt. Express 2016, 24, 20621–20631. [Google Scholar] [CrossRef]
- Yang, Y.; Wu, P.C.; Kim, T.H.; Brown, A.S.; Everitt, H.O. Gallium nanoparticle plasmonics. APS March Meet. Abstr. 2010, 55, Y14.00005. [Google Scholar]
- Gutierrez, Y.; González, F.; Saiz, J.M.; Alcaraz de la Osa, R.; Albella, P.; Ortiz, D.; Everitt, H.O.; Moreno, F. Metals and dielectrics for UV plasmonics. SPIE Proc. Nanophotonics VIII 2020, 11345, 26–34. [Google Scholar]
- Voet, D.; Gratzer, W.B.; Cox, R.A.; Doty, P. Absorption spectra of nucleotides, polynucleotides, and nucleic acids in the far ultraviolet. Biopolymers 1963, 1, 193–208. [Google Scholar] [CrossRef]
- Cardinal, M.F.; Vander Ende, E.; Hackler, R.A.; McAnally, M.O.; Stair, P.C.; Schatz, G.C.; Van Duyne, R.P. Expanding applications of SERS through versatile nanomaterials engineering. Chem. Soc. Rev. 2017, 46, 3886–3903. [Google Scholar] [CrossRef]
- Lee, H.K.; Lee, Y.H.; Koh, C.S.L.; Phan-Quang, G.C.; Han, X.; Lay, C.L.; Sim, H.Y.F.; Kao, Y.C.; An, Q.; Ling, X.Y. Designing surface-enhanced Raman scattering (SERS) platforms beyond hot-spot engineering: Emerging opportunities in analyte manipulations and hybrid materials. Chem. Soc. Rev. 2019, 48, 731–756. [Google Scholar] [CrossRef]
- Laing, S.; Gracie, K.; Faulds, K. Multiplex in vitro detection using SERS. Chem. Soc. Rev. 2016, 45, 1901–1918. [Google Scholar] [CrossRef] [PubMed]
- Halas, N. Playing with plasmons: Tuning the optical resonant properties of metallic nanoshells. MRS Bull. 2005, 30, 362–367. [Google Scholar] [CrossRef]
- Schlücker, S. Surface-enhanced Raman spectroscopy: Concepts and chemical applications. Phys. Plasmas 2014, 53, 4756–4795. [Google Scholar] [CrossRef] [PubMed]
- Kundu, S.; Chen, Y.; Dai, W.; Ma, L.; Sinyukov, A.M.; Liang, H. Enhanced catalytic and SERS activities of size-selective rhodium nanoparticles on DNA scaffolds. J. Mater. Chem. C 2017, 5, 2577–2590. [Google Scholar] [CrossRef]
- Zettsu, N.; McLellan, J.M.; Wiley, B.; Yin, Y.; Li, Z.Y.; Xia, Y. Synthesis, stability, and surface plasmonic properties of rhodium multipods, and their use as substrates for surface-enhanced Raman scattering. Angew. Chem. 2006, 118, 1310–1314. [Google Scholar] [CrossRef]
- Tian, Z.Q.; Ren, B.; Wu, D.Y. Surface-enhanced Raman scattering: From noble to transition metals and from rough surfaces to ordered nanostructures. J. Phys. Chem. B 2002, 106, 9463–9483. [Google Scholar] [CrossRef]
- Hunyadi Murph, S.E.; Coopersmith, K.J. Fabrication of silver-rhodium nanomaterials for chemical sensing applications. In Nanocomposites VI: Nanoscience and Nanotechnology in Advanced Composites; Springer: Berlin/Heidelberg, Germany, 2019; pp. 95–104. [Google Scholar]
- Sangeetha, K.; Sankar, S.S.; Karthick, K.; Anantharaj, S.; Ede, S.R.; Wilson, S.; Kundu, S. Synthesis of ultra-small Rh nanoparticles congregated over DNA for catalysis and SERS applications. Colloids Surf. B Biointerfaces 2019, 173, 249–257. [Google Scholar] [CrossRef]
- Kumaravel, S.; Karthick, K.; Sankar, S.S.; Karmakar, A.; Kundu, S. Engineered Rh Nano-networks on DNA for SERS Applications. J. Nanotechnol. Nanomater. 2020, 1, 57–64. [Google Scholar]
- Zhang, Y.; Geddes, C.D. Metal-enhanced fluorescence from thermally stable rhodium nanodeposits. J. Mater. Chem. 2010, 20, 8600–8606. [Google Scholar] [CrossRef]
- Rodrigues, M.P.D.S.; Dourado, A.H.; Cutolo, L.D.O.; Parreira, L.S.; Alves, T.V.; Slater, T.J.; Haigh, S.J.; Camargo, P.H.C.; Cordoba de Torresi, S.I. Gold-rhodium nanoflowers for the plasmon-enhanced hydrogen evolution reaction under visible light. ACS Catal. 2021, 11, 13543–13555. [Google Scholar] [CrossRef]
- Akbay, N.; Mahdavi, F.; Lakowicz, J.R.; Ray, K. Metal-enhanced intrinsic fluorescence of nucleic acids using platinum nanostructured substrates. Chem. Phys. Lett. 2012, 548, 45–50. [Google Scholar] [CrossRef]
- Chu, C.S.; Sung, T.W.; Lo, Y.L. Enhanced optical oxygen sensing property based on Pt (II) complex and metal-coated silica nanoparticles embedded in sol–gel matrix. Sens. Actuators B-Chem. 2013, 1852, 287–292. [Google Scholar] [CrossRef]
- Bigall, N.C.; Hartling, T.; Klose, M.; Simon, P.; Eng, L.M.; Eychmuller, A. Monodisperse platinum nanospheres with adjustable diameters from 10 to 100 nm: Synthesis and distinct optical properties. Nano Lett. 2008, 8, 4588–4592. [Google Scholar] [CrossRef] [PubMed]
- Mafune, F.; Kohno, J.Y.; Takeda, Y.; Kondow, T. Formation of stable platinum nanoparticles by laser ablation in water. J. Phys. Chem. B 2003, 107, 4218–4223. [Google Scholar] [CrossRef]
- Cueto, M.; Piedrahita, M.; Caro, C.; Martinez-Haya, B.; Sanz, M.; Oujja, M.; Castillejo, M. Platinum nanoparticles as photoactive substrates for mass spectrometry and spectroscopy sensors. J. Phys. Chem. C 2014, 118, 11432–11439. [Google Scholar] [CrossRef]
- Kim, K.; Lee, H.B.; Choi, J.Y.; Kim, K.L.; Shin, K.S. Surface-enhanced Raman scattering of 4-aminobenzenethiol in nanogaps between a planar Ag substrate and Pt nanoparticles. J. Phys. Chem. C 2011, 115, 13223–13231. [Google Scholar] [CrossRef]
- Kim, K.; Kim, K.L.; Lee, H.B.; Shin, K.S. Surface-enhanced Raman scattering on aggregates of platinum nanoparticles with definite size. J. Phys. Chem. C 2010, 114, 18679–18685. [Google Scholar] [CrossRef]
- Yang, Y.; Huang, Z.; Li, D.; Nogami, M. Solvothermal synthesis of platinum nanoparticles and their SERS properties. In Proceedings of the 5th International Symposium on Advanced Optical Manufacturing and Testing Technologies: Optoelectronic Materials and Devices for Detector, Imager, Display, and Energy Conversion Technology, Dalian, China, 26–29 April 2010; Volume 7658, p. 76580H. [Google Scholar]
- Cui, L.; Wang, A.; Wu, D.Y.; Ren, B.; Tian, Z.Q. Shaping and shelling Pt and Pd nanoparticles for ultraviolet laser excited surface-enhanced Raman scattering. J. Phys. Chem. C 2008, 112, 17618–17624. [Google Scholar] [CrossRef]
- Abdelsalam, M.E.; Mahajan, S.; Bartlett, P.N.; Baumberg, J.J.; Russell, A.E. SERS at structured palladium and platinum surfaces. J. Am. Chem. Soc. 2007, 23, 7399–7406. [Google Scholar] [CrossRef]
- Kämmer, E.; Dorfer, T.; Csaki, A.; Schumacher, W.; Da Costa Filho, P.A.; Tarcea, N.; Fritzsche, W.; Rösch, P.; Schmitt, M.; Popp, J. Evaluation of colloids and activation agents for determination of melamine using UV–SERS. J. Phys. Chem. C 2012, 116, 6083–6091. [Google Scholar] [CrossRef]
- Tran, M.; Whale, A.; Padalkar, S. Exploring the efficacy of platinum and palladium nanostructures for organic molecule detection via Raman spectroscopy. Sensors 2018, 18, 147. [Google Scholar] [CrossRef]
- Fan, Y.; Girard, A.; Waals, M.; Salzemann, C.; Courty, A. Ag@Pt core-shell nanoparticles for plasmonic catalysis. ACS Appl. Nano Mater. 2023, 6, 1193–1202. [Google Scholar] [CrossRef]
- Pang, P.; Teng, X.; Chen, M.; Zhang, Y.; Wang, H.; Yang, C.; Yang, W.; Barrow, C.J. Ultrasensitive enzyme-free electrochemical immunosensor for microcystin-LR using molybdenum disulfide/gold nanoclusters nanocomposites as platform and Au@ Pt core-shell nanoparticles as signal enhancer. Sens. Actuators B Chem. 2018, 266, 400–407. [Google Scholar] [CrossRef]
- Proniewicz, E.; Gralec, B.; Ozaki, Y. Homogeneous Pt nanostructures surface functionalized with phenylboronic acid phosphonic acid derivatives as potential biochemical nanosensors and drugs: SERS and TERS studies. J. Biomed. Mater. Res. Part B 2023, 111. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Ze, H.; Zhang, X.G.; Zhang, Y.J.; Song, J.; Zhang, H.; Zhong, H.L.; Yang, Z.L.; Yang, C.; Li, J.F.; et al. Direct and simultaneous identification of multiple mitochondrial reactive oxygen species in living cells using a SERS borrowing strategy. Angew. Chem. Int. Ed. 2015, 61, e202203511. [Google Scholar]
- Yoon, I.; Kang, T.; Choi, W.; Kim, J.; Yoo, Y.; Joo, S.W.; Park, Q.H.; Ihee, H.; Kim, B. Single Nanowire on a Film as an Efficient SERS-Active Platform. J. Am. Chem. Soc. 2009, 131, 758–762. [Google Scholar] [CrossRef] [PubMed]
- Ranjan, M.; Facsko, S. Anisotropic surface enhanced Raman scattering in nanoparticle and nanowire arrays. Phys. Plasmas 2012, 23, 485307. [Google Scholar] [CrossRef] [PubMed]
- Lim, D.K.; Jeon, K.S.; Hwang, J.H.; Kim, H.; Kwon, S.; Suh, Y.D.; Nam, J.M. Highly uniform and reproducible surface-enhanced Raman scattering from DNA-tailorable nanoparticles with 1-nm interior gap. Nat. Nanotechnol. 2011, 6, 452–460. [Google Scholar] [CrossRef]
- Nie, S.; Emory, S.R. Probing single molecules and single nanoparticles by surface-enhanced Raman scattering. Science 1997, 275, 1102–1106. [Google Scholar] [CrossRef]
- Kneipp, K.; Wang, Y.; Kneipp, H.; Perelman, L.T.; Itzkan, I.; Dasari, R.R.; Feld, M.S. Single molecule detection using surface-enhanced Raman scattering (SERS). Phys. Rev. Lett. 1997, 78, 1667–1670. [Google Scholar] [CrossRef]
- Kneipp, K.; Haka, A.S.; Kneipp, H.; Badizadegan, K.; Yoshizawa, N.; Boone, C.; Shafer-Peltier, K.E.; Motz, J.T.; Dasari, R.R.; Feld, M.S. Surface-enhanced Raman spectroscopy in single living cells using gold nanoparticles. Appl. Spectrosc. 2002, 56, 150–154. [Google Scholar] [CrossRef]
- Qian, X.; Peng, X.H.; Ansari, D.O.; Yin-Goen, Q.; Chen, G.Z.; Shin, D.M.; Yang, L.; Young, A.N.; Wang, M.D.; Nie, S. In vivo tumor targeting and spectroscopic detection with surface-enhanced Raman nanoparticle tags. Nat. Biotechnol. 2008, 26, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Rycenga, M.; Cobley, C.M.; Zeng, J.; Li, W.; Moran, C.H.; Zhang, Q.; Qin, D.; Xia, Y. Controlling the synthesis and assembly of silver nanostructures for plasmonic applications. Chem. Rev. 2011, 111, 3669–3712. [Google Scholar] [CrossRef] [PubMed]
- He, R.X.; Liang, R.; Peng, P.; Zhou, Y.N. Effect of the size of silver nanoparticles on SERS signal enhancement. J. Nanopart. Res. 2017, 19, 267. [Google Scholar] [CrossRef]
- Lee, S.J.; Morrill, A.R.; Moskovits, M. Hot spots in silver nanowire bundles for surface-enhanced Raman spectroscopy. J. Am. Chem. Soc. 2006, 128, 2200–2201. [Google Scholar] [CrossRef]
- Fan, M.; Brolo, A.G. Silver nanoparticles self assembly as SERS substrates with near single molecule detection limit. Phys. Chem. Chem. Phys. 2009, 11, 7381–7389. [Google Scholar] [CrossRef]
- Gopal, J.; Abdelhamid, H.N.; Huang, J.H.; Wu, H.F. Nondestructive detection of the freshness of fruits and vegetables using gold and silver nanoparticle mediated graphene enhanced Raman spectroscopy. Sens. Actuators B-Chem. 2016, 224, 413–424. [Google Scholar] [CrossRef]
- Sheng, S.Z.; Zheng, S.Q.; Feng, X.F.; Zhou, L.R.; Tao, Z.C.; Liu, J.W. Anisotropic nanoparticle arrays guided by ordered nanowire films enhance surface-enhanced Raman scattering. Adv. Opt. Mater. 2022, 10, 2201682. [Google Scholar] [CrossRef]
- Wei, H.; Hao, F.; Huang, Y.; Wang, W.; Nordlander, P.; Xu, H. Polarization dependence of surface-enhanced Raman scattering in gold nanoparticle-nanowire system. Nano Lett. 2008, 8, 2497–2502. [Google Scholar] [CrossRef]
- Anwar, S.; Khawar, M.B.; Ovais, M.; Afzal, A.; Zhang, X. Gold nanocubes based optical detection of HIV-1 DNA via surface-enhanced Raman spectroscopy. J. Pharm. Biomed. Anal. 2023, 226, 115242. [Google Scholar] [CrossRef]
- Sadanandan, S.; Ramkumar, K.; Pillai, N.P.; Anuvinda, P.; Devika, V.; Ramanunni, K.; Sreejaya, M.M. Biorecognition elements appended gold nanoparticle biosensors for the detection of food-borne pathogens-A review. Food Control 2022, 148, 109510. [Google Scholar] [CrossRef]
- Wang, Z.; Sha, H.; Yang, K.; Zhu, Y.; Zhang, J. Self-assembled monolayer silver nanoparticles: Fano resonance and SERS application. Opt. Laser Technol. 2023, 157, 108771. [Google Scholar] [CrossRef]
- Shariat, M. Plasma jet printing of silver nanoparticles on polyester fabric as a surface-enhanced Raman scattering substrate. Optik 2015, 277, 170698. [Google Scholar] [CrossRef]
- Avci, E.; Yilmaz, H.; Sahiner, N.; Tuna, B.G.; Cicekdal, M.B.; Eser, M.; Basak, K.; Altıntoprak, F.; Zengin, I.; Dogan, S.; et al. Label-free surface enhanced raman spectroscopy for cancer detection. Cancers 2022, 14, 5021. [Google Scholar] [CrossRef] [PubMed]
- Craig, A.P.; Franca, A.S.; Irudayaraj, J. Surface-enhanced Raman spectroscopy applied to food safety. Annu. Rev. Food Sci. Technol. 2013, 4, 369–380. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Dutt, S.; Sharma, P.; Sundramoorthy, A.K.; Dubey, A.; Singh, A.; Arya, S. Future of nanotechnology in food industry: Challenges in processing, packaging, and food safety. Glob. Chall. 2023, 7, 2200209. [Google Scholar] [CrossRef] [PubMed]
- Terry, L.R.; Sanders, S.; Potoff, R.H.; Kruel, J.W.; Jain, M.; Guo, H. Applications of surface-enhanced Raman spectroscopy in environmental detection. Anal. Sci. Adv. 2022, 3, 113–145. [Google Scholar] [CrossRef]
- Hail, C.U.; Michel, A.K.U.; Poulikakos, D.; Eghlidi, H. Optical metasurfaces: Evolving from passive to adaptive. Adv. Opt. Mater. 2019, 7, 1801786. [Google Scholar] [CrossRef]
- Meinzer, N.; Barnes, W.L.; Hooper, I.R. Plasmonic meta-atoms and metasurfaces. Nat. Photonics 2014, 8, 889–898. [Google Scholar] [CrossRef]
- Gwo, S.; Chen, H.Y.; Lin, M.H.; Sun, L.; Li, X. Nanomanipulation and controlled self-assembly of metal nanoparticles and nanocrystals for plasmonics. Chem. Soc. Rev. 2016, 45, 5672–5716. [Google Scholar] [CrossRef]
- Gwo, S.; Wang, C.Y.; Chen, H.Y.; Lin, M.H.; Sun, L.; Li, X.; Chen, W.L.; Chang, Y.M.; Ahn, H. Plasmonic metasurfaces for nonlinear optics and quantitative SERS. ACS Photonics 2016, 22, 1371–1384. [Google Scholar] [CrossRef]
- Biswas, A.; Bayer, I.S.; Biris, A.S.; Wang, T.; Dervishi, E.; Faupel, F. Advances in top-down and bottom-up surface nanofabrication: Techniques, applications and future prospects. Adv. Colloid Interface Sci. 2012, 170, 2–27. [Google Scholar] [CrossRef] [PubMed]
- Dhand, C.; Dwivedi, N.; Loh, X.J.; Ying, A.N.J.; Verma, N.K.; Beuerman, R.W.; Lakshminarayanan, R.; Ramakrishna, S. Methods and strategies for the synthesis of diverse nanoparticles and their applications: A comprehensive overview. RSC Adv. 2015, 5, 105003–105037. [Google Scholar] [CrossRef]
- Xue, X.; Zhou, Z.; Peng, B.; Zhu, M.M.; Zhang, Y.J.; Ren, W.; Ye, Z.G.; Chen, X.; Liu, M. Review on nanomaterials synthesized by vapor transport method: Growth and their related applications. RSC Adv. 2015, 5, 79249–79263. [Google Scholar] [CrossRef]
- Yang, P.; Zheng, J.; Xu, Y.; Zhang, Q.; Jiang, L. Colloidal synthesis and applications of plasmonic metal nanoparticles. Adv. Mater. 2016, 28, 10508–10517. [Google Scholar] [CrossRef]
- Das, R.K.; Pachapur, V.L.; Lonappan, L.; Naghdi, M.; Pulicharla, R.; Maiti, S.; Cledon, M.; Dalila, L.M.A.; Sarma, S.J.; Brar, S.K. Biological synthesis of metallic nanoparticles: Plants, animals and microbial aspects. Nanotechnol. Environ. Eng. 2017, 2, 1–21. [Google Scholar] [CrossRef]
- Ghasemi, A.; Rabiee, N.; Ahmadi, S.; Hashemzadeh, S.; Lolasi, F.; Bozorgomid, M.; Kalbasi, A.; Nasseri, B.; Dezfuli, A.S.; Aref, A.R.; et al. Optical assays based on colloidal inorganic nanoparticles. Analyst 2018, 143, 3249–3283. [Google Scholar] [CrossRef]
- Wu, J.; Yang, H. Platinum-based oxygen reduction electrocatalysts. Accounts Chem. Res. 2013, 46, 1848–1857. [Google Scholar] [CrossRef]
- Xu, L.; Liu, D.; Chen, D.; Liu, H.; Yang, J. Size and shape-controlled synthesis of rhodium nanoparticles. Heliyon 2019, 5, e01165. [Google Scholar] [CrossRef]
- Hirai, H.; Nakao, Y.; Toshima, N. Preparation of colloidal rhodium in poly(vinyl alcohol) by reduction with methanol. J. Macromol. Sci. 1978, 12, 1117–1141. [Google Scholar] [CrossRef]
- Lokesh, K.S.; Shivaraj, Y.; Dayananda, B.P.; Chandra, S. Synthesis of phthalocyanine stabilized rhodium nanoparticles and their application in biosensing of cytochrome c. Bioelectrochemistry 2009, 75, 104–109. [Google Scholar] [CrossRef]
- Long, N.V.; Chien, N.D.; Hirata, H.; Matsubara, T.; Ohtaki, M.; Nogami, M. Highly monodisperse cubic and octahedral rhodium nanocrystals: Their evolutions from sharp polyhedrons into branched nanostructures and surface-enhanced Raman scattering. J. Cryst. Growth. 2011, 320, 78–89. [Google Scholar] [CrossRef]
- Park, J.Y.; Zhang, Y.; Grass, M.; Zhang, T.; Somorjai, G.A. Tuning of catalytic CO oxidation by changing composition of Rh-Pt bimetallic nanoparticles. Nano Lett. 2008, 8, 673–677. [Google Scholar] [CrossRef] [PubMed]
- Lim, B.; Jiang, M.; Tao, J.; Camargo, P.H.C.; Zhu, Y.; Xia, Y. Shape-controlled synthesis of Pd nanocrystals in aqueous solutions. Adv. Funct. Mater. 2009, 19, 189–200. [Google Scholar] [CrossRef]
- Choi, S.I.; Lee, S.R.; Ma, C.; Oliy, B.; Luo, M.; Chi, M.; Xia, Y. Facile synthesis of rhodium icosahedra with controlled sizes up to 12 nm. ChemNanoMat 2016, 2, 61–66. [Google Scholar] [CrossRef]
- Zhang, X.; Li, P.; Barreda, Á.; Gutiérrez, Y.; González, F.; Moreno, F.; Everitt, H.O.; Liu, J. Size-tunable rhodium nanostructures for wavelength-tunable ultraviolet plasmonics. Nanoscale Horizons 2016, 1, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Watson, A.M.; Zhang, X.; Alcaraz de La Osa, R.; Sanz, J.M.; Gonzalez, F.; Moreno, F.; Finkelstein, G.; Everitt, H.O. Rhodium nanoparticles for ultraviolet plasmonics. Nano Lett. 2015, 15, 1095–1100. [Google Scholar] [CrossRef]
- Lee, S.R.; Vara, M.; Hood, Z.D.; Zhao, M.; Gilroy, K.D.; Chi, M.; Xia, Y. Rhodium decahedral nanocrystals: Facile synthesis, mechanistic insights, and experimental controls. ChemNanoMat 2018, 4, 66–70. [Google Scholar] [CrossRef]
- Biacchi, A.J.; Schaak, R.E. Ligand-induced fate of embryonic species in the shape-controlled synthesis of rhodium nanoparticles. ACS Nano 2015, 9, 1707–1720. [Google Scholar] [CrossRef]
- Koebel, M.M.; Jones, L.C.; Somorjai, G.A. Preparation of size-tunable, highly monodisperse PVP-protected Pt-nanoparticles by seed-mediated growth. J. Nanopart. Res. 2008, 10, 1063–1069. [Google Scholar] [CrossRef]
- Herricks, T.; Chen, J.; Xia, Y. Polyol synthesis of platinum nanoparticles: Control of morphology with sodium nitrate. Nano Lett. 2004, 4, 2367–2371. [Google Scholar] [CrossRef]
- Long, N.V.; Chien, N.D.; Hayakawa, T.; Hirata, H.; Lakshminarayana, G.; Nogami, M. The synthesis and characterization of platinum nanoparticles: A method of controlling the size and morphology. Nanotechnology 2009, 21, 035605. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Sun, X. One-step preparation of poly (vinyl alcohol)-protected Pt nanoparticles through a heat-treatment method. Mater. Lett. 2007, 61, 2015–2017. [Google Scholar] [CrossRef]
- Maicu, M.; Schmittgens, R.; Hecker, D.; Glöß, D.; Frach, P.; Gerlach, G. Synthesis and deposition of metal nanoparticles by gas condensation process. J. Vac. Sci. Technol. A Vac. Surfaces Films 2014, 32, 02B113. [Google Scholar] [CrossRef]
- Pedone, D.; Moglianetti, M.; Luca, E.D.; Bardi, G.; Pompa, P.P. Platinum nanoparticles in nanobiomedicine. Chem. Soc. Rev. 2017, 46, 4951–4975. [Google Scholar] [CrossRef] [PubMed]
- Fazio, E.; Gökce, B.; De Giacomo, A.; Meneghetti, M.; Compagnini, G.; Tommasini, M.; Waag, F.; Lucotti, A.; Zanchi, C.G.; Ossi, P.M.; et al. Nanoparticles engineering by pulsed laser ablation in liquids: Concepts and applications. Nanomaterials 2020, 10, 2317. [Google Scholar] [CrossRef]
- Zhang, D.; Gokce, B.; Barcikowski, S. Laser synthesis and processing of colloids: Fundamentals and applications. Chem. Rev. 2017, 117, 3990–4103. [Google Scholar]
- Fazio, E.; Neri, F.; Patane, S.; D’urso, L.; Compagnini, G. Optical limiting effects in linear carbon chains. Carbon 2011, 49, 306–310. [Google Scholar] [CrossRef]
- Volpato, G.A.; Arboleda, D.M.; Brandiele, R.; Carraro, F.; Sartori, G.B.; Cardelli, A.; Badocco, D.; Pastore, P.; Agnoli, S.; Durante, C.; et al. Clean rhodium nanoparticles prepared by laser ablation in liquid for high performance electrocatalysis of the hydrogen evolution reaction. Nanoscale Adv. 2019, 1, 4296–4300. [Google Scholar] [CrossRef]
- Zhang, J.; Chaker, M.; Ma, D. Pulsed laser ablation based synthesis of colloidal metal nanoparticles for catalytic applications. J. Colloid Interf. Sci. 2017, 489, 138–149. [Google Scholar] [CrossRef]
- Kishida, M.; Tago, T.; Hatsuta, T.; Wakabayashi, K. Preparation of silica-coated rhodium nanoparticles using water-in-oil microemulsion. Chem. Lett. 2000, 29, 1108–1109. [Google Scholar] [CrossRef]
- Gautam, A.; Guleria, P.; Kumar, V. Platinum nanoparticles: Synthesis strategies and applications. Nanoarchitectonics 2020, 1, 70–86. [Google Scholar]
- Jeyaraj, M.; Gurunathan, S.; Qasim, M.; Kang, M.H.; Kim, J.H. A comprehensive review on the synthesis, characterization, and biomedical application of platinum nanoparticles. Nanomaterials 2019, 9, 1719. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Li, C.; Ren, X. Fine platinum nanoparticles supported on a porous ceramic membrane as efficient catalysts for the removal of benzene. Sci. Rep. 2017, 7, 16589. [Google Scholar] [CrossRef] [PubMed]
- Prasetya, O.D.; Khumaeni, A. Synthesis of colloidal platinum nanoparticles using pulse laser ablation method. AIP Conf. Proc. 2018, 2014, 020050. [Google Scholar]
- Cueto, M.; Sanz, M.; Oujja, M.; Gamez, F.; Martiinez-Haya, B.; Castillejo, M. Platinum nanoparticles prepared by laser ablation in aqueous solutions: Fabrication and application to laser desorption ionization. J. Phys. Chem. C 2011, 115, 22217–22224. [Google Scholar] [CrossRef]
- Mafune, F.; Kondow, T. Selective laser fabrication of small nanoparticles and nano-networks in solution by irradiation of UV pulsed laser onto platinum nanoparticle. Chem. Phys. Lett. 2004, 383, 343–347. [Google Scholar] [CrossRef]
- Dobrzanski, L.A.; Szindler, M.; Pawlyta, M.; Szindler, M.M.; Borylo, P.; Tomiczek, B. Synthesis of Pt nanowires with the participation of physical vapor deposition. Open Phys. 2016, 14, 159–165. [Google Scholar] [CrossRef]
- Hu, X.; Saran, A.; Hou, S.; Wen, T.; Ji, Y.; Liu, W.; Zhang, H.; He, W.; Yin, J.J.; Wu, X. Au@PtAg core/shell nanorods: Tailoring enzyme-like activities via alloying. RSC Adv. 2013, 3, 6095–6105. [Google Scholar] [CrossRef]
- Patil, S.; Chandrasekaran, R. Biogenic nanoparticles: A comprehensive perspective in synthesis, characterization, application and its challenges. J. Genet. Eng. Biotechnol. 2020, 182, 1–23. [Google Scholar] [CrossRef]
- Mughal, B.; Zaidi, S.Z.J.; Zhang, X.; Hassan, S.U. Biogenic nanoparticles: Synthesis, characterisation and applications. Appl. Sci. 2021, 11, 2598. [Google Scholar] [CrossRef]
- Kumari, S.; Tyagi, M.; Jagadevan, S. Mechanistic removal of environmental contaminants using biogenic nanomaterials. Int. J. Environ. Sci. Technol. 2019, 16, 7591–7606. [Google Scholar] [CrossRef]
- Ali, M.A.; Ahmed, T.; Wu, W.; Hossain, A.; Hafeez, R.; Masum, M.M.I.; Wang, Y.; An, Q.; Sun, G.; Li, B. Advancements in plant and microbe-based synthesis of metallic nanoparticles and their antimicrobial activity against plant pathogens. Nanomaterials 2020, 10, 1146. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Xu, H.; Chen, Z.S.; Chen, G. Biosynthesis of nanoparticles by microorganisms and their applications. J. Nanomater. 2011, 2011, 1–16. [Google Scholar] [CrossRef]
- Tamaoki, K.; Saito, N.; Nomura, T.; Konishi, Y. Microbial recovery of rhodium from dilute solutions by the metal ion-reducing bacterium Shewanella algae. Hydrometallurgy 2013, 139, 26–29. [Google Scholar] [CrossRef]
- Srivastava, S.K.; Constanti, M. Room temperature biogenic synthesis of multiple nanoparticles (Ag, Pd, Fe, Rh, Ni, Ru, Pt, Co, and Li) by Pseudomonas aeruginosa SM1. J. Nanopart. Res. 2012, 14, 1–10. [Google Scholar] [CrossRef]
- Dobrucka, R. Synthesis and structural characteristic of platinum nanoparticles using herbal bidens tripartitus extract. J. Inorg. Organomet. Polym. Mater. 2015, 26, 219–225. [Google Scholar] [CrossRef]
- Thirumurugan, A.; Aswitha, P.; Kiruthika, C.; Nagarajan, S.; Nancy, C.A. Green synthesis of platinum nanoparticles using Azadirachta indica–An eco-friendly approach. Mater. Lett. 2016, 170, 175–178. [Google Scholar] [CrossRef]
- Al-Radadi, N.S. Green synthesis of platinum nanoparticles using Saudi’s Dates extract and their usage on the cancer cell treatment. Arab. J. Chem. 2019, 12, 330–349. [Google Scholar] [CrossRef]
- Mavukkandy, M.O.; Chakraborty, S.; Abbasi, T.; Abbasi, S.A. A clean-green synthesis of platinum nanoparticles utilizing a pernicious weed lantana (Lantana Camara). Am. J. Eng. Appl. Sci. 2016, 9, 84–90. [Google Scholar] [CrossRef]
- Velmurugan, P.; Shim, J.; Kim, K.; Oh, B.T. Prunus × yedoensis tree gum mediated synthesis of platinum nanoparticles with antifungal activity against phytopathogens. Mater. Lett. 2016, 174, 61–65. [Google Scholar] [CrossRef]
- Rokade, S.S.; Joshi, K.A.; Mahajan, K.; Tomar, G.; Dubal, D.S.; Parihar, V.S.; Kitture, R.; Jayesh Bellare, J.; Ghosh, S. Novel anticancer platinum and palladium nanoparticles from Barleria prionitis. Glob. J. Nanomed. 2017, 2, 555600. [Google Scholar]
- Vinod, V.T.P.; Saravanan, P.; Sreedhar, B.; Devi, D.K.; Sashidhar, R.B. A facile synthesis and characterization of Ag, Au and Pt nanoparticles using a natural hydrocolloid gum kondagogu (Cochlospermum gossypium). Colloids Surf. B Biointerfaces 2011, 83, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Sheny, D.S.; Philip, D.; Mathew, J. Synthesis of platinum nanoparticles using dried Anacardium occidentale leaf and its catalytic and thermal applications. Spectrochim. Acta A 2013, 114, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Song, J.Y.; Kwon, E.Y.; Kim, B.S. Biological synthesis of platinum nanoparticles using Diopyros kaki leaf extract. Bioproc. Biosyst. Eng. 2010, 33, 159–164. [Google Scholar] [CrossRef] [PubMed]
- Zheng, B.; Kong, T.; Jing, X.; Odoom-Wubah, T.; Li, X.; Sun, D.; Lu, F.; Zheng, Y.; Huang, J.; Li, Q. Plant-mediated synthesis of platinum nanoparticles and its bioreductive mechanism. J. Colloid Interf. Sci. 2013, 396, 138–145. [Google Scholar] [CrossRef]
- Dauthal, P.; Mukhopadhyay, M. Biofabrication, characterization, and possible bio-reduction mechanism of platinum nanoparticles mediated by agro-industrial waste and their catalytic activity. J. Ind. Eng. Chem. 2015, 22, 185–191. [Google Scholar] [CrossRef]
- Lim, S.K.; Chung, K.J.; Kim, C.K.; Shin, D.W.; Kim, Y.H.; Yoon, C.S. Surface-plasmon resonance of Ag nanoparticles in polyimide. J. Appl. Phys. 2005, 98, 084309. [Google Scholar] [CrossRef]
- Kibis, L.S.; Stadnichenko, A.I.; Pajetnov, E.M.; Koscheev, S.V.; Zaykovskii, V.I.; Boronin, A.I. The investigation of oxidized silver nanoparticles prepared by thermal evaporation and radio-frequency sputtering of metallic silver under oxygen. Appl. Surf. Sci. 2010, 257, 404–413. [Google Scholar] [CrossRef]
- Pingali, K.C.; Rockstraw, D.A.; Deng, S. Silver nanoparticles from ultrasonic spray pyrolysis of aqueous silver nitrate. Aerosol Sci. Technol. 2005, 39, 1010–1014. [Google Scholar] [CrossRef]
- Kodu, M.; Berholts, A.; Kahro, T.; Avarmaa, T.; Kasikov, A.; Niilisk, A.; Alles, H.; Jaaniso, R. Highly sensitive NO2 sensors by pulsed laser deposition on graphene. Appl. Phys. Lett. 2016, 109, 113108. [Google Scholar] [CrossRef]
- Henglein, A.; Giersig, M. Formation of colloidal silver nanoparticles: Capping action of citrate. J. Phys. Chem. B 1999, 103, 9533–9539. [Google Scholar] [CrossRef]
- Pillai, Z.S.; Kamat, P.V. What factors control the size and shape of silver nanoparticles in the citrate ion reduction method. J. Phys. Chem. B 2004, 108, 945–951. [Google Scholar] [CrossRef]
- Lee, P.C.; Meisel, D. Adsorption and surface-enhanced Raman of dyes on silver and gold sols. J. Phys. Chem. 1982, 86, 3391–3395. [Google Scholar] [CrossRef]
- Hachem, K.; Ansari, M.J.; Saleh, R.O.; Kzar, H.H.; Al-Gazally, M.E.; Altimari, U.S.; Hussein, S.A.; Mohammed, H.T.; Hammid, A.T.; Kianfar, E. Methods of chemical synthesis in the synthesis of nanomaterial and nanoparticles by the chemical deposition method: A review. BioNanoScience 2022, 12, 1032–1057. [Google Scholar] [CrossRef]
- Chang, C.C.; Wu, H.L.; Kuo, C.H.; Huang, M.H. Hydrothermal synthesis of monodispersed octahedral gold nanocrystals with five different size ranges and their self-assembled structures. Chem. Mater. 2008, 20, 7570–7574. [Google Scholar] [CrossRef]
- Yin, H.; Yamamoto, T.; Wada, Y.; Yanagida, S. Large-scale and size-controlled synthesis of silver nanoparticles under microwave irradiation. Mater. Chem. Phys. 2004, 83, 66–70. [Google Scholar] [CrossRef]
- Eisa, W.H.; Abdel-Moneam, Y.K.; Shaaban, Y.; Abdel-Fattah, A.A.; Abou Zeid, A.M. Gamma-irradiation assisted seeded growth of Ag nanoparticles within PVA matrix. Mater. Chem. Phys. 2011, 128, 109–113. [Google Scholar] [CrossRef]
- Kondeti, V.S.S.K.; Gangal, U.; Yatom, S.; Bruggeman, P.J. Ag+ reduction and silver nanoparticle synthesis at the plasma-liquid interface by an RF driven atmospheric pressure plasma jet: Mechanisms and the effect of surfactant. J. Vac. Sci. Technol. A 2017, 35, 6130. [Google Scholar] [CrossRef]
- Zhang, Y.T.; Guo, Y.; Ma, T.C. Plasma catalytic synthesis of silver nanoparticles. Chin. Phys. Lett. 2011, 28, 105201. [Google Scholar] [CrossRef]
- Lee, H.; Park, S.H.; Jung, S.C.; Yun, J.J.; Kim, S.J.; Kim, D.H. Preparation of nonaggregated silver nanoparticles by the liquid phase plasma reduction method. J. Mater. Res. 2013, 28, 1105–1110. [Google Scholar] [CrossRef]
- Turkevich, J.; Stevenson, P.C.; Hiller, J. A study of the nucleation and growth processes in the synthesis of colloidal gold. Discuss. Faraday Soc. 1951, 11, 55–75. [Google Scholar] [CrossRef]
- Kimling, J.; Maier, M.; Okenve, B.; Kotaidis, V.; Ballot, H.; Plech, A. Turkevich method for gold nanoparticle synthesis revisited. J. Phys. Chem. B 2006, 110, 15700–15707. [Google Scholar] [CrossRef] [PubMed]
- De Souza, C.D.; Nogueira, B.R.; Rostelato, M.E.C. Review of the methodologies used in the synthesis gold nanoparticles by chemical reduction. J. Alloys Compd. 2019, 798, 714–740. [Google Scholar] [CrossRef]
- Lee, G.J.; Kang, M.; Kim, Y.; Choi, E.H.; Cho, M.J.; Choi, D.H. Optical assessment of chiral-achiral polymer blends based on surface plasmon resonance effects of gold nanoparticles. J. Phys. D Appl. Phys. 2019, 53, 095102. [Google Scholar] [CrossRef]
- Dragieva, I.D.; Stoynov, Z.B.; Klabunde, K.J. Synthesis of nanoparticles by borohydride reduction and their applications. Scripta Mater. 2001, 44, 2187–2191. [Google Scholar] [CrossRef]
- Mavani, K.; Shah, M. Synthesis of silver nanoparticles by using sodium borohydride as a reducing agent. Int. J. Eng. Res. Technol. 2013, 2, 1–5. [Google Scholar]
- Seney, C.S.; Gutzman, B.M.; Goddard, R.H. Correlation of size and surface-enhanced Raman scattering activity of optical and spectroscopic properties for silver nanoparticles. J. Phys. Chem. C 2009, 113, 74–80. [Google Scholar] [CrossRef]
- Bhattacharjee, R.R.; Dasgupta, U. Seed-mediated synthesis of silver nanoparticles: Tunable surface plasmon and their facile fabrication. Mater. Today Proc. 2021, 43, 1342–1347. [Google Scholar] [CrossRef]
- Szunerits, S.; Praig, V.G.; Manesse, M.; Boukherroub, R. Gold island films on indium tin oxide for localized surface plasmon sensing. Nanotechnology 2008, 19, 195712. [Google Scholar] [CrossRef]
- Billot, L.; de La Chapelle, M.L.; Grimault, A.S.; Vial, A.; Barchiesi, D.; Bijeon, J.L.; Adam, P.M.; Royer, P. Surface enhanced Raman scattering on gold nanowire arrays: Evidence of strong multipolar surface plasmon resonance enhancement. Chem. Phys. Lett. 2006, 422, 303–307. [Google Scholar] [CrossRef]
- Tan, B.J.Y.; Sow, C.H.; Koh, T.S.; Chin, K.C.; Wee, A.T.S.; Ong, C.K. Fabrication of size-tunable gold nanoparticles array with nanosphere lithography, reactive ion etching, and thermal annealing. J. Phys. Chem. B 2005, 109, 11100–11109. [Google Scholar] [CrossRef] [PubMed]
- Nikolov, A.S.; Nedyalkov, N.N.; Nikov, R.G.; Atanasov, P.A.; Alexandrov, M.T. Characterization of Ag and Au nanoparticles created by nanosecond pulsed laser ablation in double distilled water. Appl. Surf. Sci. 2011, 257, 5278–5282. [Google Scholar] [CrossRef]
- Nguyen, L.N.; Lamichhane, P.; Choi, E.H.; Lee, G.J. Structural and optical sensing properties of nonthermal atmospheric plasma-synthesized polyethylene glycol-functionalized gold nanoparticles. Nanomaterials 2021, 11, 1678. [Google Scholar] [CrossRef]
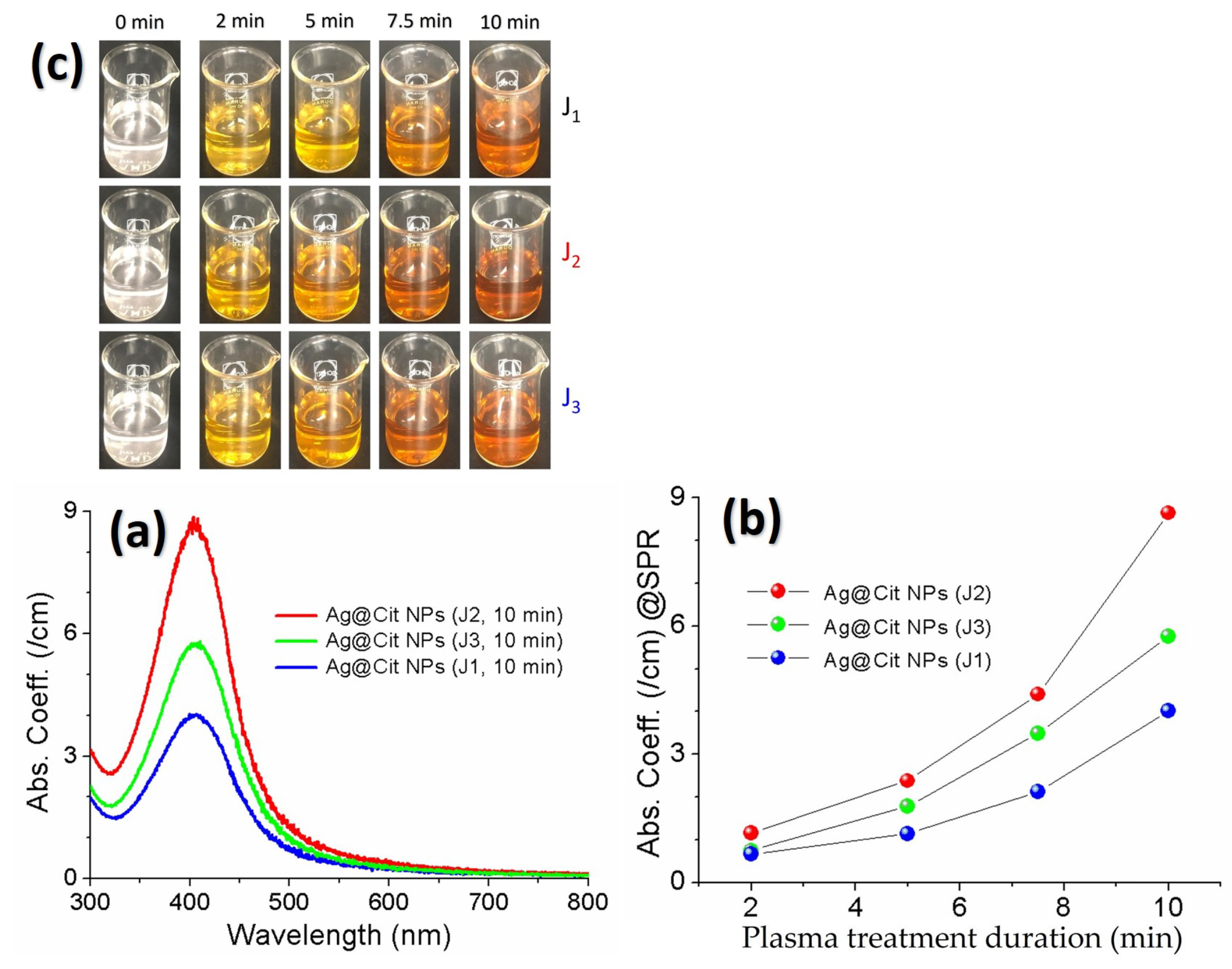
- Acharya, T.R.; Lee, G.J.; Choi, E.H. Influences of plasma plume length on structural, optical and dye degradation properties of citrate-stabilized silver nanoparticles synthesized by plasma-assisted reduction. Nanomaterials 2022, 12, 2367. [Google Scholar] [CrossRef]
- Nilghaz, A.; Mahdi Mousavi, S.; Amiri, A.; Tian, J.; Cao, R.; Wang, X. Surface-enhanced Raman spectroscopy substrates for food safety and quality analysis. J. Agric. Food Chem. 2022, 70, 5463–5476. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.; Zhang, J.; Gao, J.; Zhang, Z.; Zhu, H.; Wang, D. Gold nanoparticles in cancer theranostics. Front. Bioeng. Biotechnol. 2021, 9, 647905. [Google Scholar] [CrossRef] [PubMed]
- King, M.E.; Guzman, M.V.F.; Ross, M.B. Material strategies for function enhancement in plasmonic architectures. Nanoscale 2022, 14, 602–611. [Google Scholar] [CrossRef]
- Azharuddin, M.; Zhu, G.H.; Das, D.; Ozgur, E.; Uzun, L.; Turner, A.P.; Patra, H.K. A repertoire of biomedical applications of noble metal nanoparticles. Chem. Commun. 2019, 55, 6964–6996. [Google Scholar] [CrossRef]
- Mauriz, E.; Lechuga, L.M. Plasmonic biosensors for single-molecule biomedical analysis. Biosensors 2021, 11, 123. [Google Scholar] [CrossRef]
- Alula, M.T.; Krishnan, S.; Hendricks, N.R.; Karamchand, L.; Blackburn, J.M. Identification and quantitation of pathogenic bacteria via in-situ formation of silver nanoparticles on cell walls, and their detection via SERS. Microchim. Acta 2017, 184, 219–227. [Google Scholar] [CrossRef]
- Wang, J.; Fu, J.; Chen, H.; Wang, A.; Ma, Y.; Yan, H.; Li, Y.; Yu, D.; Gao, F.; Li, S. Trimer structures formed by target-triggered AuNPs self-assembly inducing electromagnetic hot spots for SERS-fluorescence dual-signal detection of intracellular miRNAs. Biosens. Bioelectron. 2023, 224, 115051. [Google Scholar] [CrossRef] [PubMed]
- Jahangirian, H.; Lemraski, E.G.; Webster, T.J.; Rafiee-Moghaddam, R.; Abdollahi, Y. A review of drug delivery systems based on nanotechnology and green chemistry: Green nanomedicine. Int. J. Nanomed. 2017, 12, 2957. [Google Scholar] [CrossRef] [PubMed]
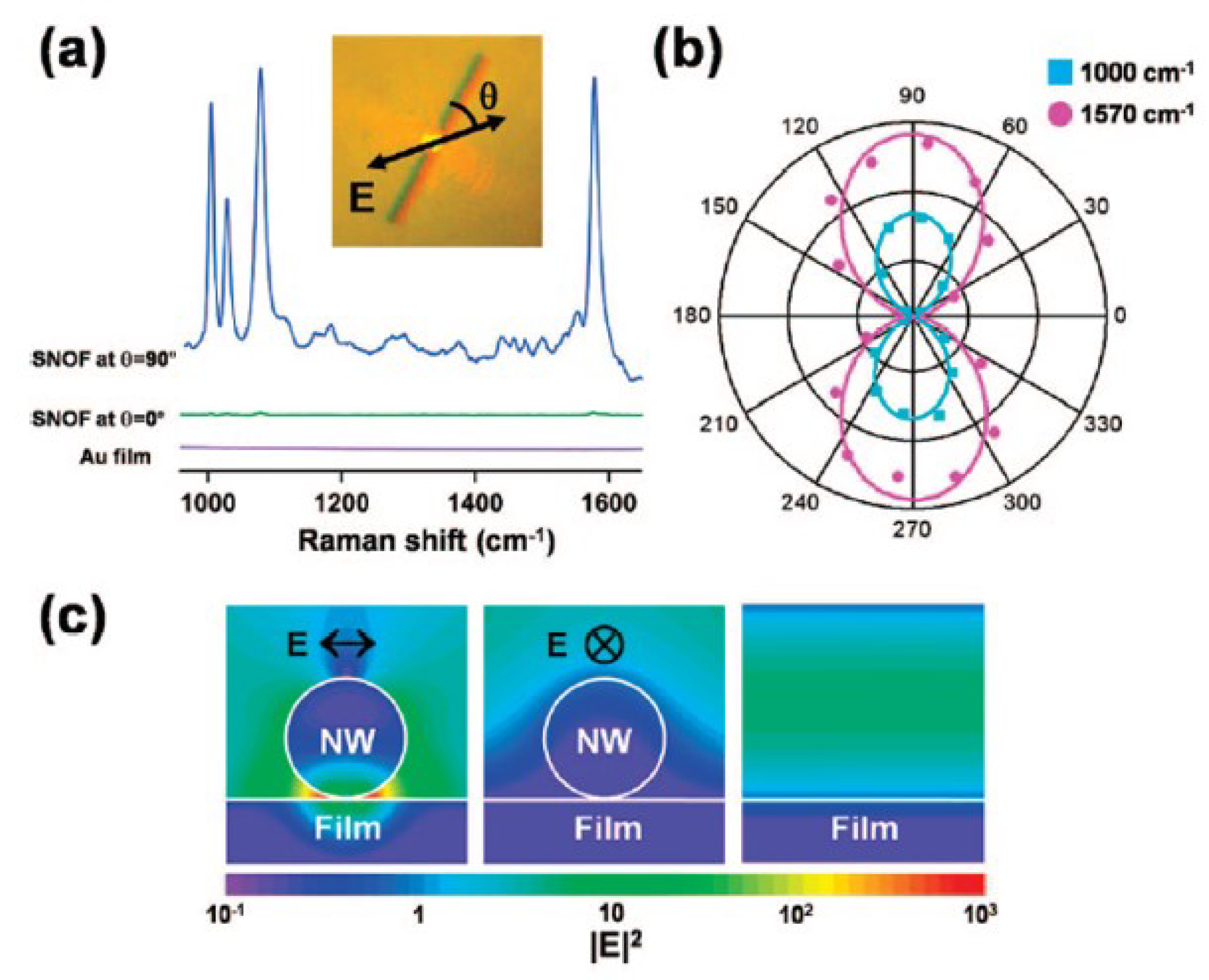





| Synthesis Methods | Optical Sensing Applications | Ref. | |
|---|---|---|---|
| Rh-based nanomaterials | • Chemical reduction of RhCl with alkaline 2,7-DHN on DNA scaffolds under UV-light irradiation. | • SERS sensing by ultra-small, self-assembled RhNPs on DNA scaffold. | [78] |
| • Co-reduction of the metal precursors by polyol at elevated temperature. | • SERS sensing by Ag–Rh nanomaterials. | [81] | |
| • Chemical reduction of RhCl with NaBH on DNA scaffolds. | • SERS sensing by Rh@DNA NPs. | [82] | |
| • Polyol synthesis. (Chemical reduction of NaRhCl by ethylene glycol). | • SERS sensing by RhNP multipods (Tripod and tetrapod RhNPs). | [79] | |
| • Electron-beam physical-vapor-deposition. | • MEF sensing by RhNP substrates. | [84] | |
| Pt-based nanomaterials | • Chemical reduction of HPtCl and physical ablation from bulk Pt. | • SERS sensing by PtNPs. | [90] |
| • Electrodeposition of Pd or Pt through a template of self-assembled polystyrene latex spheres onto a suitable conducting surface. | • SERS sensing by structured Pt and Pd surfaces. | [95] | |
| • Chemical reduction of HPtCl with sodium citrate, sodium borohydride, and L-ascorbic acid. | • Gap-enhanced Raman scattering of 4-ABT positioned in the gaps formed by a flat Ag substrate and 20–150 nm PtNPs. | [91] | |
| • Synthesis of Pt nanocubes, Pt nanospheres, Au core Pt shell (Au@Pt), and Au core Pd shell (Au@Pd) nanoparticles from chemical reduction. | • Shaping and shelling PtNPs and PdNPs for UV–SERS. | [94] | |
| • Chemical reduction of HPtCl using a multistep seed-mediated approach. | • SERS sensing of melamine by PtNPs with different shapes and sizes. | [96] | |
| • Deposition on the n-type Si substrate by galvanic displacement method. | • SERS sensing by Pt- and Pd-nanostructures. | [80] | |
| • Core-shell Au@Pt NPs by seed-mediated chemical method. (Synthesis of Au seeds by the Frens method, and then deposition of the Pt shells on the Au surface by in situ reduction. ) | • Simultaneous identification of multiple mitochondrial ROS in living cells by a SERS-based nanoprobe (core-shell Au@Pt NPs). | [101] | |
| • Synthesis of AuNPs by citrate reduction of HAuCl, and then synthesis of core-shell Au@Pt NPs by reduction of HPtCl with ascorbic acid. | • Ag@Pt NPs as an enzymatic reporter to identify microcystin-leucine arginine antibodies. | [99] | |
| • Chemical reduction of PtCl in ethylene glycol under boiling for 3 h by Lewera polylene method. | • SERS and TERS studies for the selective adsorption of PBA–PA derivatives on the surface of PtNPs. | [100] |
| Synthesis Methods | Ref. | ||
|---|---|---|---|
| Gold-based nanomaterials | • Citrate reduction of gold precursor; Deposition of AuNPs on the ordered tellurium nanowire template. | • Efficient SERS platform by highly ordered gold nanowire arrays. ➀ Anisotropic NP arrays. ➁ Polarization-dependent SERS. | [114] |
| • Oblique incidence physical-vapor-deposition on pre-patterned rippled substrate by low energy ion irradiation. | • Anisotropic SERS of gold nanowire arrays and NP chains. ➀ Comparative study of nanowire arrays and NP chains. ➁ Polarization-dependent SERS. | [103] | |
| • Chemical reduction of gold precursor with NaBH. | • Polarization-dependent SERS in gold nanoparticle-nanowire system. | [115] | |
| • Vapor transport method. | • SERS sensing of benzenethiol () by a single gold nanowire on a gold film. ➀ Nano-gap-enhanced Raman scattering. ➁ Polarization-dependent SERS. | [102] | |
| • Citrate reduction method. | • Single molecule and single living cell detections by AuNP-based SERS. | [106] | |
| • Citrate-stabilized AuNPs. | • In vivo tumor targeting and SERS detection by scFv, EGFR-conjugated AuNPs. | [108] | |
| • CTAB-stabilized Au nanocubes by NaBH reduction of gold precursor. | • SERS sensing of human immunodeficiency virus (HIV) by Au nanocubes. | [116] | |
| • Citrate reduction method. | • SERS sensing of food-borne pathogens by biorecognition element-conjugated AuNPs. | [117] | |
| • Numerical simulation study. | • Influence of size, shape, and dielectric environment on the optical properties of metal NPs. | [23] | |
| Silver-based nanomaterials | • Chemical reduction of silver precursor with ascorbic acid. | • Efficient SERS platform by self-assembled AgNP monolayer. | [118] |
| • Plasma reduction of silver precursor. | • SERS sensing by AgNPs loaded on a polyester fabric by plasma jet printing. | [119] | |
| • Citrate reduction of silver precursor; Deposition of AgNPs on graphene nanosheets. | • SERS sensing of the freshness of fruits and vegetables by AgNPs and AuNPs supported on graphene nanosheets. | [113] | |
| • Citrate reduction method. | • Cancer detection by AgNP-based SERS. | [120] | |
| • Citrate reduction method. | • Single-molecule detection of rhodamine 6G by AgNP-based SERS. | [105] | |
| • Fabrication of Ag nanowires in highly ordered porous aluminum oxide (PAO) template by AC electrodeposition. | • Efficient SERS platform by Ag nanowire bundles. | [111] | |
| • Review for synthesis methods and plasmonic sensing applications. | • Synthesis methods and SERS/SEF sensing of AgNPs. | [109] |
| • Sputtering | |
| • Electron beam evaporation | |
| • Thermal evaporation | |
| • Radiolysis | |
| • Plasma synthesis | |
| • Laser ablation | |
| • Ultrasonication | |
| • UV photolysis | |
| • Laser pyrolysis | |
| • Spray pyrolysis | |
| • Mechanical milling | |
| • Lithography | |
| • Chemical reduction of metal salts | |
| • Polyol synthesis | |
| • Sol-gel synthesis | |
| • Vapor transport method | |
| • Chemical vapor deposition | |
| • Plasma-enhanced chemical vapor deposition | |
| • Electrochemical synthesis | |
| • Phytochemical synthesis | |
| • Microemulsion synthesis | |
| • Plant extract-assisted synthesis | |
| – Leaves, Flowers, Fruits, Roots | |
| • Microorganism-assisted synthesis | |
| – Fungi, Yeast, Bacteria, Viruses, Actinomycetes | |
| • Algae-based synthesis |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Demishkevich, E.; Zyubin, A.; Seteikin, A.; Samusev, I.; Park, I.; Hwangbo, C.K.; Choi, E.H.; Lee, G.J. Synthesis Methods and Optical Sensing Applications of Plasmonic Metal Nanoparticles Made from Rhodium, Platinum, Gold, or Silver. Materials 2023, 16, 3342. https://doi.org/10.3390/ma16093342
Demishkevich E, Zyubin A, Seteikin A, Samusev I, Park I, Hwangbo CK, Choi EH, Lee GJ. Synthesis Methods and Optical Sensing Applications of Plasmonic Metal Nanoparticles Made from Rhodium, Platinum, Gold, or Silver. Materials. 2023; 16(9):3342. https://doi.org/10.3390/ma16093342
Chicago/Turabian StyleDemishkevich, Elizaveta, Andrey Zyubin, Alexey Seteikin, Ilia Samusev, Inkyu Park, Chang Kwon Hwangbo, Eun Ha Choi, and Geon Joon Lee. 2023. "Synthesis Methods and Optical Sensing Applications of Plasmonic Metal Nanoparticles Made from Rhodium, Platinum, Gold, or Silver" Materials 16, no. 9: 3342. https://doi.org/10.3390/ma16093342
APA StyleDemishkevich, E., Zyubin, A., Seteikin, A., Samusev, I., Park, I., Hwangbo, C. K., Choi, E. H., & Lee, G. J. (2023). Synthesis Methods and Optical Sensing Applications of Plasmonic Metal Nanoparticles Made from Rhodium, Platinum, Gold, or Silver. Materials, 16(9), 3342. https://doi.org/10.3390/ma16093342










