Abstract
The development of magnesium calcium phosphate bone cements (MCPCs) has garnered substantial attention. MCPCs are bioactive and biodegradable and have appropriate mechanical and antimicrobial properties for use in reconstructive surgery. In this study, the cement powders based on a (Ca + Mg)/P = 2 system doped with Zn2+ at 0.5 and 1.0 wt.% were obtained and investigated. After mixing with a cement liquid, the structural and phase composition, morphology, chemical structure, setting time, compressive strength, degradation behavior, solubility, antibacterial activities, and in vitro behavior of the cement materials were examined. A high compressive strength of 48 ± 5 MPa (mean ± SD) was achieved for the cement made from Zn2+ 1.0 wt.%-substituted powders. Zn2+ introduction led to antibacterial activity against Staphylococcus aureus and Escherichia coli strains, with an inhibition zone diameter of up to 8 mm. Biological assays confirmed that the developed cement is cytocompatible and promising as a potential bone substitute in reconstructive surgery.
1. Introduction
The tendency of growing life expectancy is observed all over the world. The number of age-related diseases and surgical operations has grown accordingly [1]. Among them, there are osteoporosis [2], injuries, and traumas of various types. The latter tends to increase in frequency in all age groups [3]. In addition to osteosarcoma, which is the most prevalent primary bone tumor mainly affecting children and young adults [4], often needs surgical interventions. Currently, surgery is commonly accompanied by antibiotic therapy [5] to reduce the risks of infections and inflammation. On the other hand, antibiotics may pose additional risks, related to the growing prevalence of drug-resistant bacteria [6] or other side effects [7]. Hence, alternative strategies concerning the application of antimicrobial materials, e.g., bone cement, are of great interest.
Bone cement was originally named “materials based on polymethyl methacrylate” and are currently used in clinical practice [8]. Nevertheless, these materials have several disadvantages, such as an exothermic setting reaction (which can lead to damage to adjacent tissues) and the need for revision surgery [9]. Therefore, a search for alternative materials is underway. In recent years, several resorbable calcium phosphate-based bone cements (CPCs) [10], magnesium phosphate-based bone cements (MPCs) [11], and MCPCs were proposed [12,13,14].
The antibacterial activity of such materials may be achieved in several ways. The first one is the impregnation of a cement matrix with antimicrobial agents. Although this solution is widespread and allows for the implementation of the concept of local drug delivery [15,16,17], several issues are still unresolved, such as the control of drug release kinetics and the above-mentioned growing antibiotic resistance of bacteria. Thus, an alternative approach devoid of these shortcomings and involving the introduction of various ions into the cement matrix is attractive.
The most common ion introduced into cement materials to achieve antimicrobial activity is silver. It has been demonstrated that the presence of silver ions can eliminate chronic osteomyelitis [18], and the introduction of Ag+ cations lead to greater mechanical strength and antibacterial effects against both gram-positive and gram-negative bacteria [19]. Nonetheless, it should be noted that silver is not an element that is present in the body in appreciable amounts and, therefore, there may be health risks associated with its toxicity [20]. At present, this question is not fully clarified. An alternative—zinc—is present in various organs and tissues and is involved in various metabolic processes [21]. The Zn2+ ion plays a critical role in bone growth by promoting osteoblast differentiation and inhibiting osteoclast differentiation [22]. Zinc in various compounds can exert antibacterial actions [23,24]. In addition, its presence can lead to further improvement of the mechanical characteristics of a material [25].
A newberyite-based MCPC described in our previous paper shows a neutral pH level (7–8), a setting time of 10–13 min, mechanical strength up to 22 ± 3 MPa, cytocompatibility, and acceptable matrix surface properties [13]. Improved mechanical properties, as well as cytocompatibility and antibacterial activity, have been achieved for newberyite struvite-based bone cements that are doped with silver ions [19]. On the other hand, regarding the controversial effects of silver, the development of Zn2+-doped MCPCs is expected to improve the antibacterial properties and osteogenic ability of MCPCs because a release of Zn2+ may enhance osteogenic differentiation of certain cells for accelerated bone regeneration. Based on the above, the present work is devoted to the development of the new Zn2+-doped MCPC and its setting processes, thermal behavior, and mechanical properties, as well as the characterization of dissolution in model liquids, antibacterial properties, and cytocompatibility in relation to the MG-63 cell line.
2. Materials and Methods
2.1. Synthesis of Cement Materials
The synthesis of cement powders is based on a (Ca + Mg)/P = 2.0 system with 40 mol.%. Mg substitution was carried out by the method of precipitation from an aqueous solution of salts according to the following equation described in our previous paper [13]:
2.4Ca(NO3)2 + 1.6Mg(NO3)2 + 4(NH4)2HPO4 + 8NH4OH = Ca2.4Mg1.6O(PO4)2 + 16NH4NO3 + 6H2O
Zn2+ ions were introduced into the synthesis mixture as solutions of a nitrate [Zn(NO3)2] in the amount of 0.5 and 1.0 wt.%. After solution mixing, the slurries were evaporated to maintain the predicted concentrations.
The cement powders were heat treated at 1150 °C followed by grinding in a planetary ball mill with zirconia balls in a dimethyl ketone medium for 20 min. Cement powders were obtained, and major phases that formed during the synthesis of magnesium-substituted calcium phosphate were MgO, Mg3Ca3(PO4)4, and Ca2.589Mg0.411(PO4)2, which were consistent with a CaO–P2O5–MgO phase diagram [26].
Cement materials were prepared by mixing a cement powder with a cement liquid in a 2:1 ratio under sterile conditions on a glass slide using a spatula. The cement liquid was synthesized on the basis of ammonium phosphate (NH4)3PO4 and magnesium hydrogen phosphate MgHPO4 at a controlled pH value of 4.6. Cement materials with the following names were obtained: MCPC, 0.5%Zn-MCPC, and 1.0%Zn-MCPC. For material characterization and mechanical tests, cement samples were prepared in cylindrical form with a diameter of 0.8 mm; after setting, the samples were hardened in a 100% humidity atmosphere for 24 h.
2.2. Characterization of the Materials
Chemical composition was investigated by atomic emission spectrometry with inductively coupled plasma (AES-ICP, Vista Pro, ICP Expert 4.0 Software). The uncertainty of the ICP results was 2%.
The granulometric composition of cement powders was examined by means of a laser particle analyzer FRITSCH Analysis 22. The phase composition of the materials was determined by X-ray phase analysis (XRD) (on a Shimadzu XRD-6000, Kyoto, Japan) using CuKα radiation in the 2θ range from 10° to 70° with a step of 0.02° using the ICDD database, PDF2. The phases were matched with the following cards: magnesium oxide (MgO: ICDD 77-2364), a magnesium-substituted whitlockite phase (Ca2.589Mg0.411(PO4)2: ICDD 87-1582), stanfieldite (Mg3Ca3(PO4)4: ICDD 73-1182), newberyite (MgHPO4·3H2O: ICDD 75-1714), and struvite (MgNH4PO4·6H2O: ICDD 77-2303). The uncertainty of the XRD results was 5%. The particle size of the cement powders, as well as the microstructure of set cement materials, were investigated by scanning electron microscopy (SEM) (Tescan Vega II, Brno, Czech Republic). An analysis of the SEM data was carried out using ImageJ software 1.52u based on the secant method for at least 50 line segments.
Fourier-transform infrared absorption (FTIR) spectra of the samples were obtained using the KBr technique on a Nicolet Avatar-330 FTIR spectrometer (Thermo Fisher Scientific, Nicolet, Waltham, MA, USA) from 4000 to 400 cm−1 to evaluate the functional groups of the samples.
The pH value of the solutions and temperature during the mixing of the cement paste was measured with the help of a Testo pH meter (Testo, Titisee-Neustadt, Germany).
The setting time of cement materials was determined by immersion of a 1.0 mm (400 g) Vicant needle in a sample according to ISO 9917 (2007) [27]. The pH values of the extracts of set cement samples were determined after 1, 7, and 28 days in model fluids—the Kokubo Simulated Body Fluid (SBF) [28] and Dulbecco’s phosphate-buffered saline without calcium and magnesium (DPBS) (Thermo Scientific, Waltham, MA, USA)—at a ratio of 0.2 g/mL at room temperature.
2.3. Mechanical Testing
An Instron 5581 uniaxial testing machine was employed to measure the compressive strength of the cement samples at a crosshead speed of 1 mm/min according to ASTM D695-91. Five samples of each composition were analyzed to determine compressive strength, and the results are reported as mean ± standard deviation (SD).
2.4. Dissolution Assays
To understand the degradation behavior and solubility of MCPC and 1.0%Zn-MCPC, samples in the form of 28-day set disks (6 mm in diameter and 2 mm in height) were soaked in the model fluids the SBF and DPBS at 37.0 °C at a w/v ratio of 0.2 g/mL and a surface area-to-volume ratio of 0.1 cm−1 for various periods in a closed system [29]. At predetermined time points (1, 3, 7, 14, 21, and 28 days), two samples were taken out of the solutions, frozen in a ULT Freezer MDF-60U50 (SANYO Electric Co., Ltd., Osaka, Osaka, Japan) at −70 °C, and lyophilized in LS-1000 Prointeh-bio before weight loss calculations, and one sample was dried at 60 °C for studying the effect of holding time on phase composition and microstructure upon dissolution.
2.5. Testing of Antibacterial Properties
Antibacterial activities of MCPC, 0.5%Zn-MCPC, and 1.0%Zn-MCPC crystals were assessed against the gram-positive bacterium Staphylococcus aureus (strain ATCC 6538) and the gram-negative bacterium Escherichia coli (strain XL1-Blue) in accordance with ISO 20645 [30]. Tested cement samples were applied to the surface of the agar. The results of antibacterial activity were recorded after 24 h. The size of a zone of growth inhibition of the microorganisms was determined by measuring the distance from the edge of a tested cement sample to the border of the growth of a microorganism around the tested sample, as well as the absence or presence of bacterial growth in the zone of contact of the sample with the agar medium.
2.6. Testing of Cytocompatibility
In vitro cytocompatibility of cement samples was evaluated by the MTT assay on a human osteosarcoma MG-63 cell line on the 1st and 3rd days of cell growth (Russian Collection of Cell Cultures, Institute of Cytology, Russian Academy of Sciences, St. Petersburg, Russia). Before the start of the in vitro assays, the cement samples were sterilized with γ-radiation at a dose of 18 kGy. Cement disk samples 5.0 mm in diameter and 3.0 mm high were placed into 96-well plates for cultivation (Corning Costar, New York, NY, USA) in triplicate at one 96-well plate per incubation period and were covered with a complete growth medium (CGM), which consisted of DMEM (PanEco, Moscow, Russia), 10% of fetal bovine serum (Cytiva, Traun, Austria), 60 mg/mL glutamine (PanEco, Moscow, Russia), 20 mM HEPES buffer (PanEco, Moscow, Russia), and 50 μg/mL gentamycin (PanEco, Moscow, Russia). Before that, in the 96-well plates, MG-63 cells were seeded at a density of 15,000 cells per well in 200 μL of the CGM. All the procedures were performed under sterile conditions at 37 °C in an atmosphere of humidified air that contained 5% CO2. Subsequently, the optical density (OD) of the formazan solution (the end product of the MTT reaction) was evaluated on a Multiscan FC spectrophotometer (Thermoscientific, Waltham, MA, USA) at a wavelength of 540 nm, and the calculation of the population of viable cells (PVC) in relation to the control (in %) was carried out according to the following formula:
where OD is the value of the optical density of the formazan solution.
PVC = ODexp/ODcontr × 100 (%),
A sample of the cement was assumed to be cytocompatible at PVC ≥ 70%. The details of this method are described elsewhere [31].
3. Results
3.1. Characterization of the Cement Powders
Based on the AES-ICP results, Zn2+ concentrations in the cement powders were close to the calculated ones (Table 1).

Table 1.
Phase composition of the cement powders and their particle size distribution.
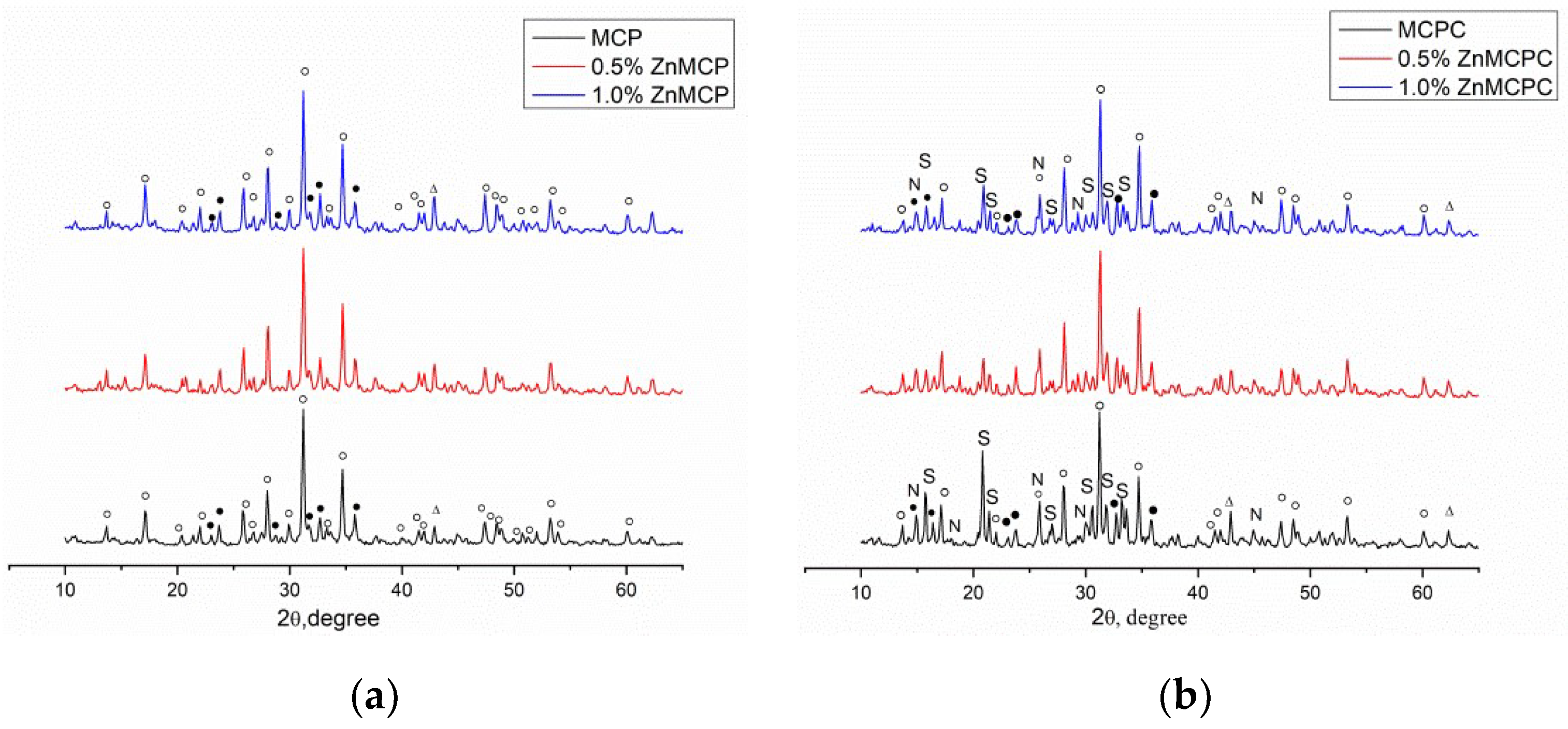
According to the results of the XRD analysis, the main phases of Zn-free magnesium calcium phosphate were magnesium-substituted whitlockite [Ca2.589Mg0.411(PO4)2] in the amount of 42 wt.% and a stanfieldite phase of monoclinic configuration with unit cell parameters of a = 22.8449(2) Å, b = 9.9999(1) Å, and c = 17.0882(4) Å at 29 wt.%; the rest of the magnesium did not enter these phases and crystallized as MgO at 29 wt.% (Table 1). Subsequently, this cement powder (MCPC) was used as a control sample. The Zn2+-substituted materials showed the formation of whitlockite and stanfieldite phases and magnesium oxide, similar to the undoped 40 mol.% Mg cement powder (Figure 1). With the increase in the Zn2+ content of the cement, the quantity of the MgO phase noticeably increased.
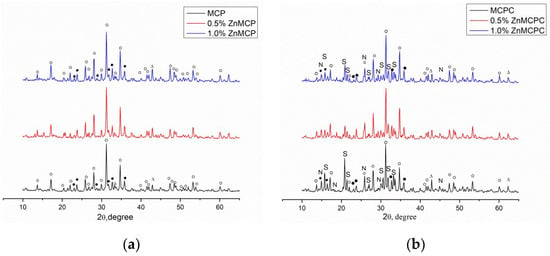
Figure 1.
Diffraction patterns of (a) cement powders and (b) set cement. ○: whitlockite Ca2.589Mg0.411(PO4)2, ●: stanfieldite Mg3Ca3(PO4)4, S: struvite MgNH4PO4·6H2O, N: newberyite MgHPO4·3H2O, ∆: MgO.
The introduction of Zn2+ into the material led to nonsignificant changes (an increase) in unit cell parameters of the stanfieldite phase (a = 22.8274(1) Å, b = 10.0012(1) Å, c = 17.0847(4) Å). We can hypothesize that the substitution of Mg2+ ions with Zn2+ (the ionic radius of Zn2+ is 0.83 Å vs. 0.65 Å in Mg2+ [32]) followed by a rise of the MgO amount in the materials.
The particle size distribution after grinding the cement powders is presented in Table 1. The average particle size of the MCPC is about 20 μm. It was found that under the same grinding conditions, the addition of Zn2+ leads to an increase in particle size up to 36 μm at 1.0 wt.% doping (Figure 2).

Figure 2.
SEM images of the cement powders. (a) MCPC and (b) 1.0%Zn-MCPC in the SE mode.
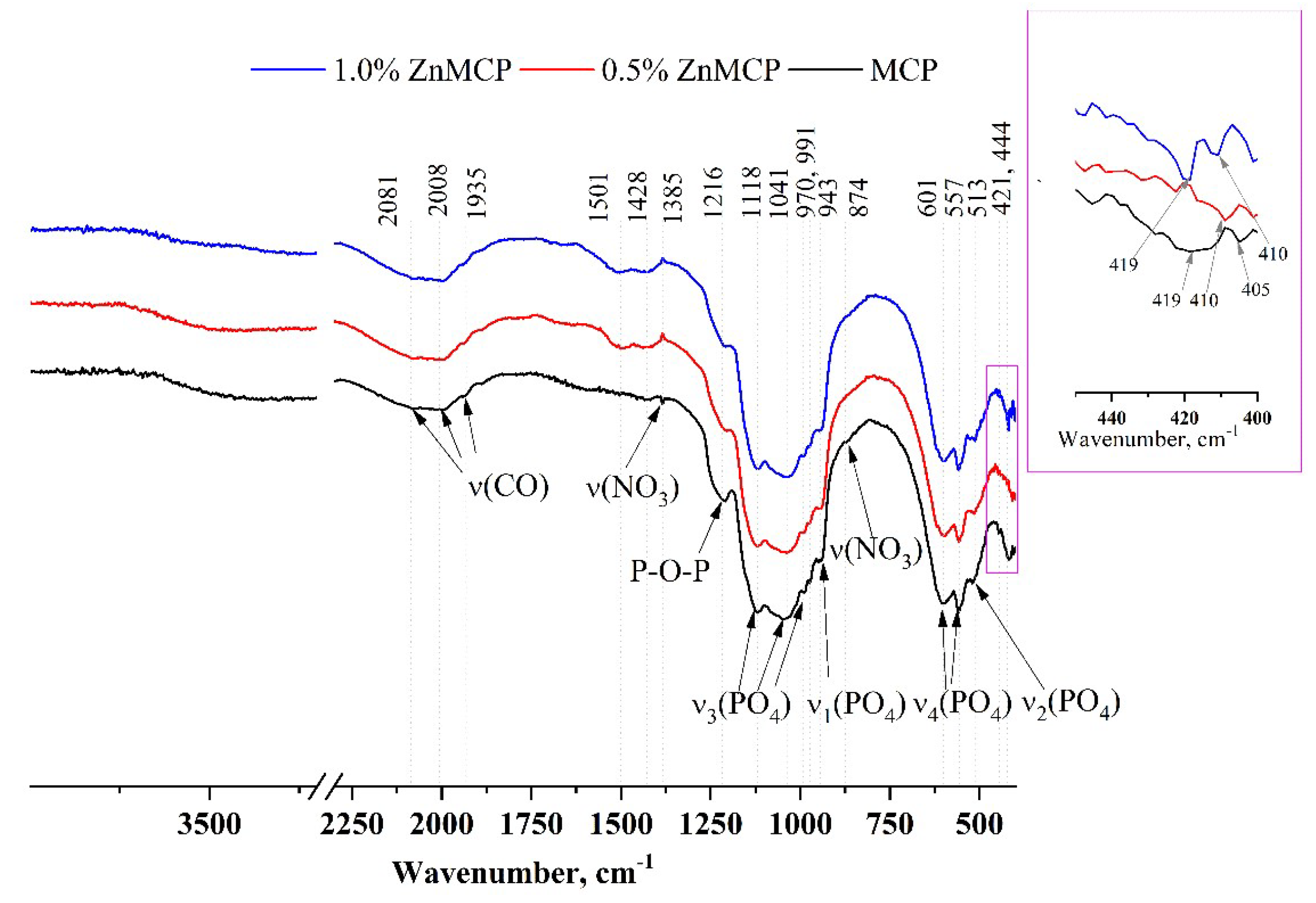
Infrared (IR) spectra of the obtained powders are given in Figure 3. A broad phosphate band in the region of 1200–900 cm−1 and well-resolved bands of ν3(PO4) at 1118, 1034, 991, and 970 cm−1 and ν1(PO4) at 943 cm−1 indicate that the structure of phosphate phases formed successfully. ν4(PO4) bands at 601 and 557 cm−1, as well as ν2(PO4) at 513 cm−1, are also clearly visible in all the materials. A broad peak formed by three bands at 1935, 2008, and 2081 cm−1 is related to the C=O vibration from residual acetone after sample milling. In the absence of dopant ions, this region has a wide band at 419 cm−1 and a low-intensity band at 405 cm−1. In the presence of zinc, a narrower band is observed for the 0.5 wt.% substitution at 410 cm−1, and for the 1.0 wt.% substitution, a doublet is seen at 419 and 410 cm−1.
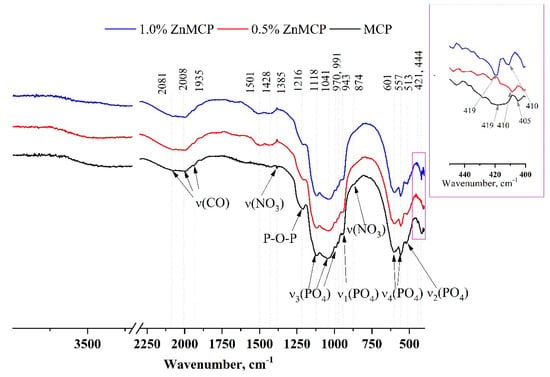
Figure 3.
IR spectra of the powders after calcination and milling in acetone.
3.2. Characterization of the Cement Materials
According to the XRD analysis (Figure 1b), after mixing the cement powders with the cement liquid, the formation of struvite (MgNH4PO4∙6H2O) orthogonal syngony with unit cell parameters of a = 6.9614(2) Å, b = 6.1480(3) Å, and c = 11.2507(2) Å was observed; the preservation of a small amount of initial stanfieldite, magnesium-substituted whitlockite, and MgO were also detected (Figure 4). In contrast, cement materials doped with 0.5 wt.% Zn2+ showed the formation of both struvite (a = 6.9500(1) Å, b = 6.1426(2) Å, and c = 11.2166(1) Å) and newberyite phases orthogonal syngony with unit cell parameters of a = 10.2287(2) Å, b = 10.7026(3) Å, and c = 10.0355(2) Å (MgHPO4∙3H2O) (Table 2). It should be noted that the introduction of the zinc cations in the amount of 0.5 wt.% resulted in a slight increase in the amount in the newberyite phase. Cement material based on the powders doped with 1.0 wt.% Zn formed the struvite phase (a = 6.9497(3) Å, b = 6.1342(4) Å, abd c = 11.2075(2) Å) and newberyite phase (a = 10.2186(1) Å, b = 10.6936(1) Å, c = 10.0312(4) Å).
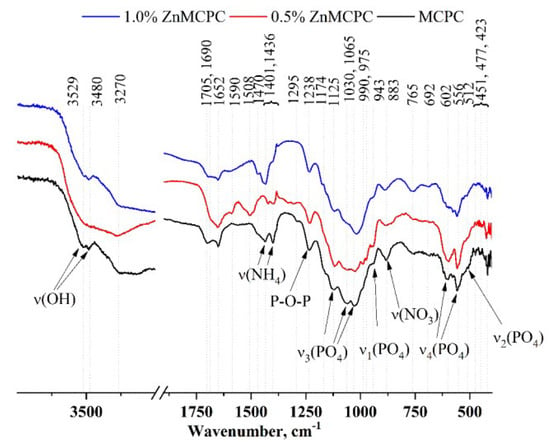
Figure 4.
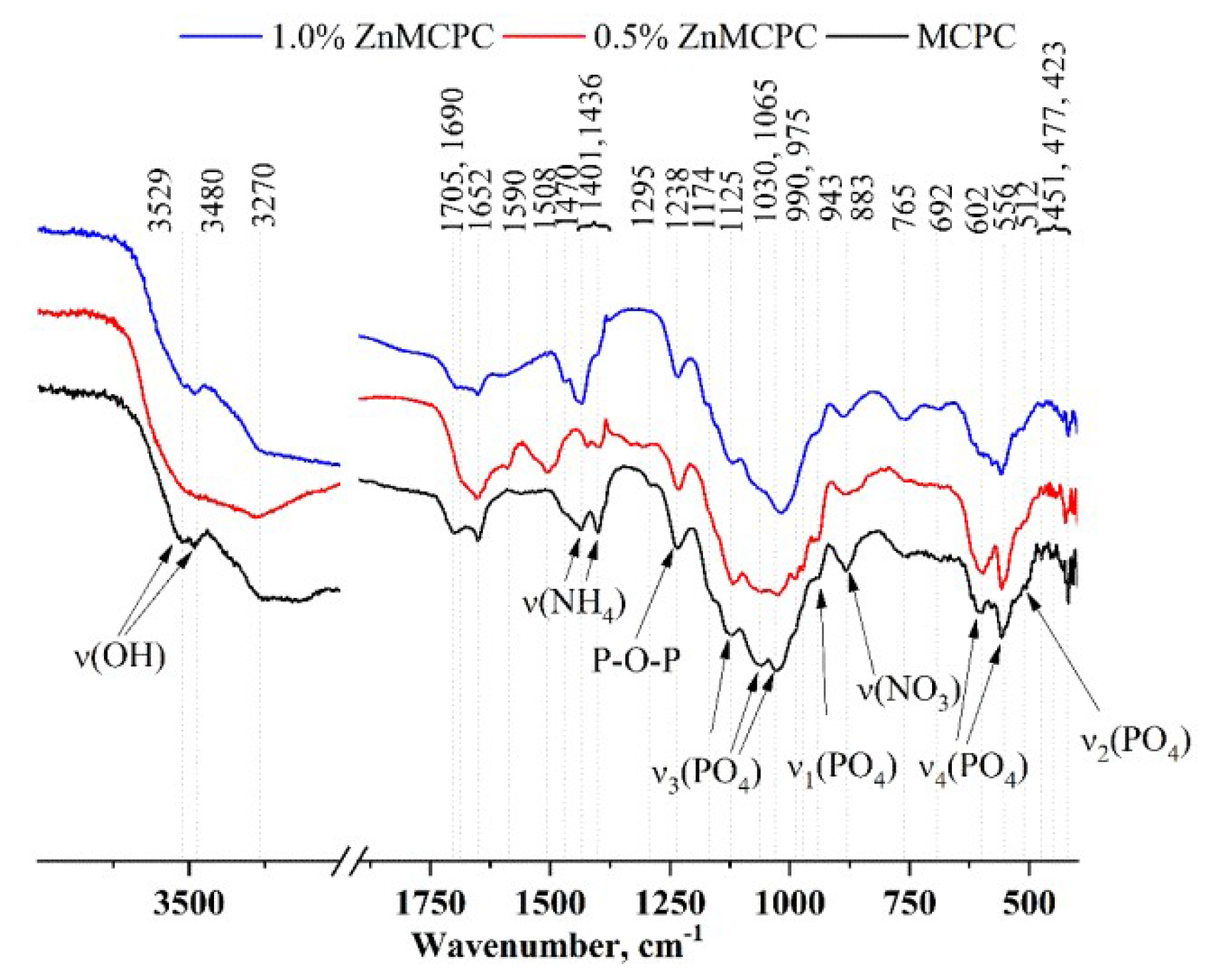
IR spectra of the cement samples after setting and hardening.

Table 2.
Phase composition of the cement materials.
The IR spectra (Figure 4) contain bands related to PO4 vibrations at 1125, 1030, 1065, 990, and 975 cm−1 [ν3(PO4)], 943 cm−1 [ν1(PO4)], 602 and 556 cm−1 [ν4(PO4)], and 512 cm−1 [ν2(PO4)]. It should be pointed out that with the increase in the amount of the dopant ion, the intensity and resolution of these bands noticeably diminished.
Ammonium salt was present in the setting cement liquid, which gave rise to a new phase: struvite (NH4MgPO4∙6H2O). Its presence was confirmed by the XRD analysis. The IR spectra also show absorption related to the NH4 group vibrations in the region 1400–1710 cm−1, as well as in the region of 3600–3200 cm−1. In the latter, H-O-H vibrations of water contribute and are present both in the newly formed crystal hydrates (struvite and brushite) and in free form.
The preparation of a cement material is based on chemical reactions between compounds of the cement powder and cement liquid. When phosphate salts (NH4)3PO4 and MgHPO4 come into contact with the cement liquid, their molecules dissociate and, within a few seconds, the cement liquid is saturated with ions in accordance with the following reactions:
MgHPO4(aq) → HPO42−(aq) + Mg2+(aq)
HPO42−(aq) → PO43−(aq) + H+(aq)
(NH4)3PO4(aq) → PO43−(aq) + 3NH4+(aq)
Futher, MgO recrystallization into struvite occurs (Krokhicheva et al., 2023), and dissolved ions interact with the original phases of the cement powder. Due to the presence of Ca2+ at 60 mol.%, we assume the emergence of amorphous crystalline hydrate phase CaHPO4∙2H2O (brushite) via the following reaction:
CaxMg(3−x)(PO4)2 + (NH4)2HPO4 + (15 − x)H2O →
2MgNH4PO4∙6H2O + (1 − x)MgHPO4·3H2O + xCaHPO4·2H2O
2MgNH4PO4∙6H2O + (1 − x)MgHPO4·3H2O + xCaHPO4·2H2O
The microstructure of the set cement samples was investigated, and it indicated the formation of new cementing phases on the surface (Figure 5). The MCPC sample is characterized by a dense poreless structure of the struvite phase. The introduction of Zn2+ ions led to a struvite crystal shape modification. The crystalline phase has geometric features, such as a honeycomb appearance with an average crystalline size of approximately 5–8 μm for 0.5%Zn-MCPC. On the surface of the Zn2+-doped cement samples, the emergence of the new cementing crystalline hydrate phase newberyite was registered. On the surface of the sample’s cracks, we noticed the newberyite phase in the BSE mode.

Figure 5.
SEM images of the set cement containing 40 mol.% of Mg in the BSE mode (crack).
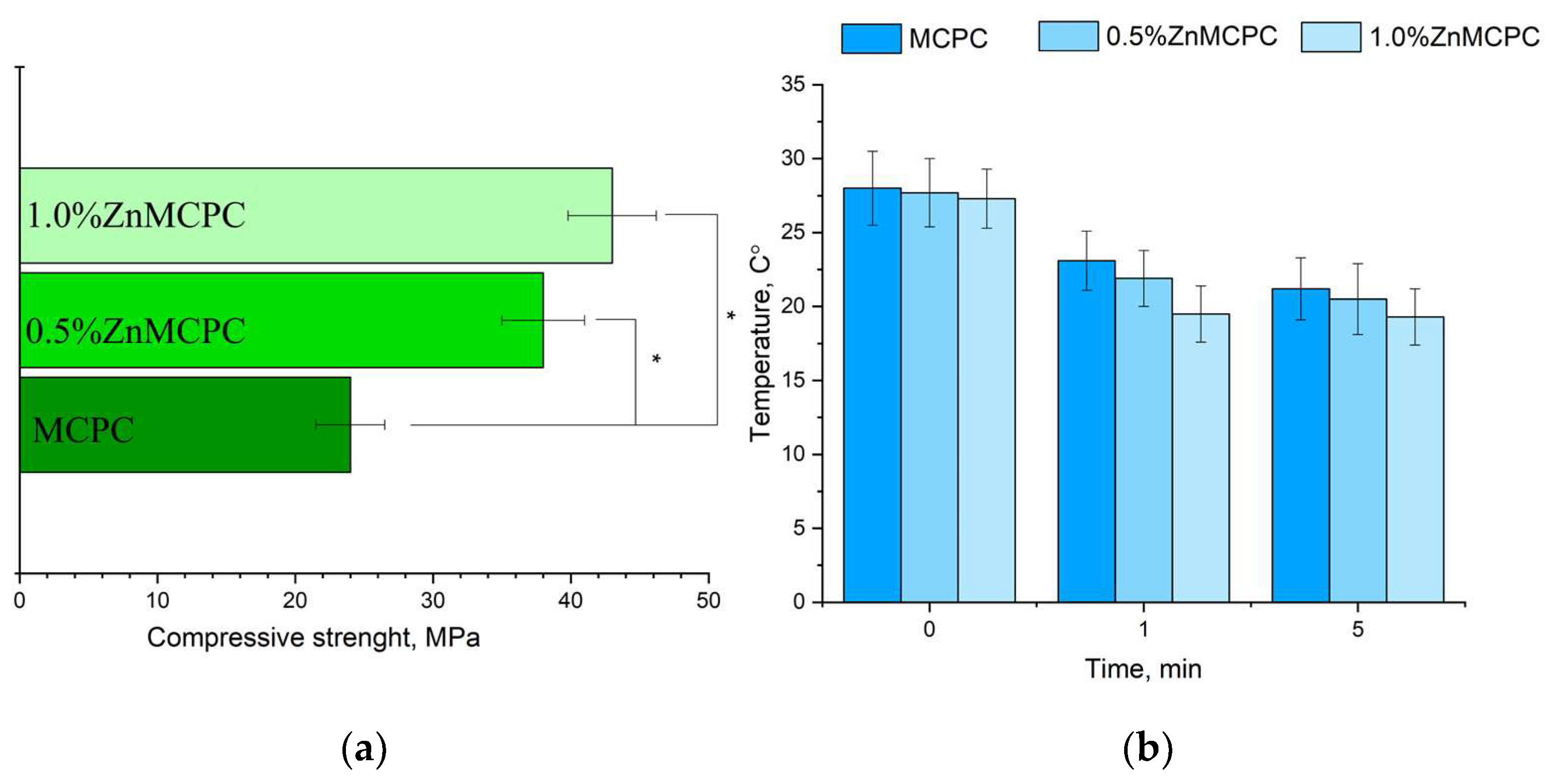
The MCPC samples are characterized by a compressive strength of about 24 ± 3 MPa. It was found that the introduction of Zn2+ cations enhanced the strength. The greatest strength, 48 ± 5 MPa, was observed for 1.0%Zn MCPC samples (Figure 6a). During the mixing of the cement paste, no noticeable thermal reaction was detectable (Figure 6b). Measurement of the pH value of the solutions based on distilled water containing a cement material showed a pH level close to neutral during exposure to the solution for more than 1 day. The pH value of the samples with 40 mol.% of Mg after 24 days of holding in distilled water was 7.2.
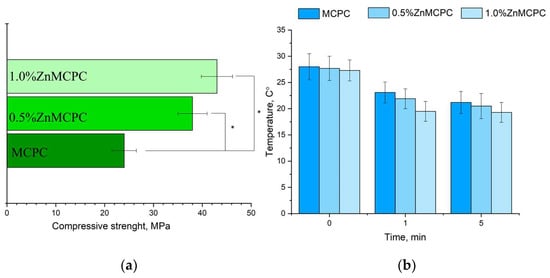
Figure 6.
Results on the cement materials after mechanical testing for compressive strength (a); temperature measure during the setting process of cement materials (b). * Indicates significant differences compared between the Zn-doped samples and the control pure MCPC composition at p < 0.05 levels.
The setting time for the cement materials was approximately 4–7 min regardless of composition. A setting time shorter than 15 min is adequate for surgery [33]. Additionally, the temperature was measured during the setting process of the cement material (Figure 6b). It was shown that the maximum temperature was 28 °C, which was observed at the moment of mixing cement powder with cement liquid. The next process of cement setting during 5 min is shown by the tendency of decreasing temperature for all cement compositions.
3.3. In Vitro Dissolution Assays
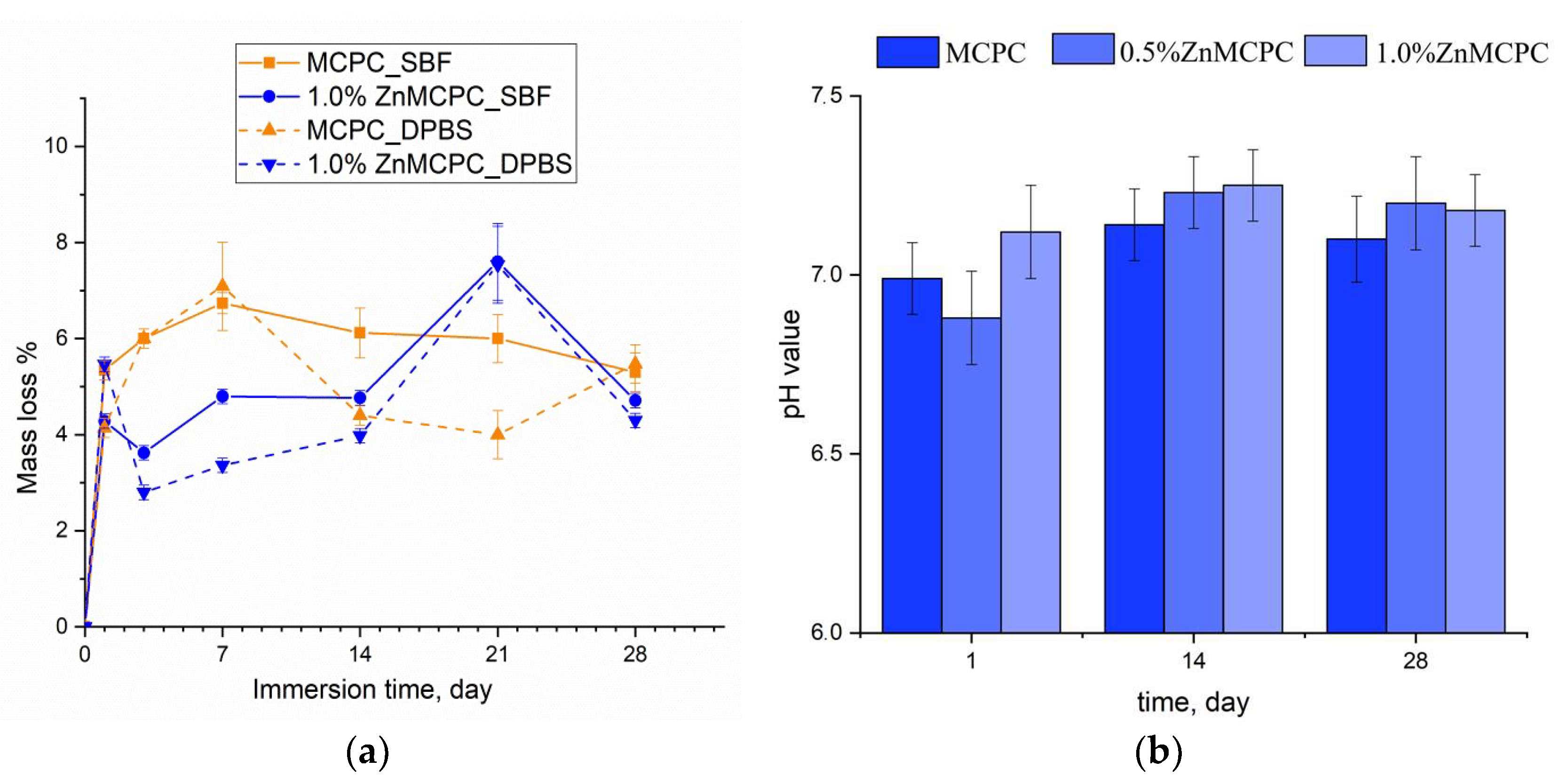
Degradation kinetics are determined by weight loss and pH value, and the profile is plotted as a function of time. The solubility assay of the cement materials revealed that on the first day of the experiment, the mass loss of the samples did not exceed 6%, regardless of the type of mortar. At the initial stage of the dissolution process, the unreacted cement liquid was removed, and subsequent features of the behavior of the cement materials in the model liquids were suggestive of processes taking place on the surface of the material, where interactions proceeded between the dissolved Ca2+, Mg2+, Na+, OH−, HPO42−, and PO43− ions. The greatest mass loss in the MCPC sample was 7% and happened on the 7th day of the experiment in the model liquids; on the 14th day, the solubility in DPBS was less than the SBF; however, by the 28th day, the mass loss reached 5%. The introduction of zinc into the MCPC affected the nature of the solubility of the samples, and mass losses in the model liquids diminished; however, by the 21st day, the loss reached 7%, and by the end of the experiment, the solubility of the samples declined.
Measurement of the pH value in the SBF solution of the cement materials at various time points showed a neutral level of about 6.8–7.3, which was typical for struvite-based MCPCs [34] (Figure 7b).
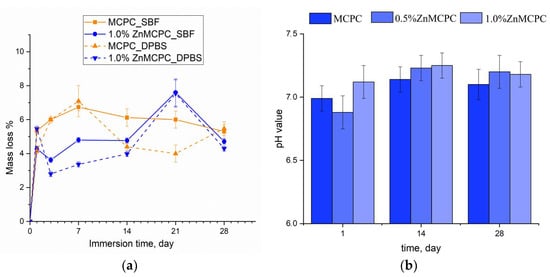
Figure 7.
The dissolution assay of mass loss of the cement samples in the SBF and DPBS (a). The pH value in the SBF (b) throughout the experiment.
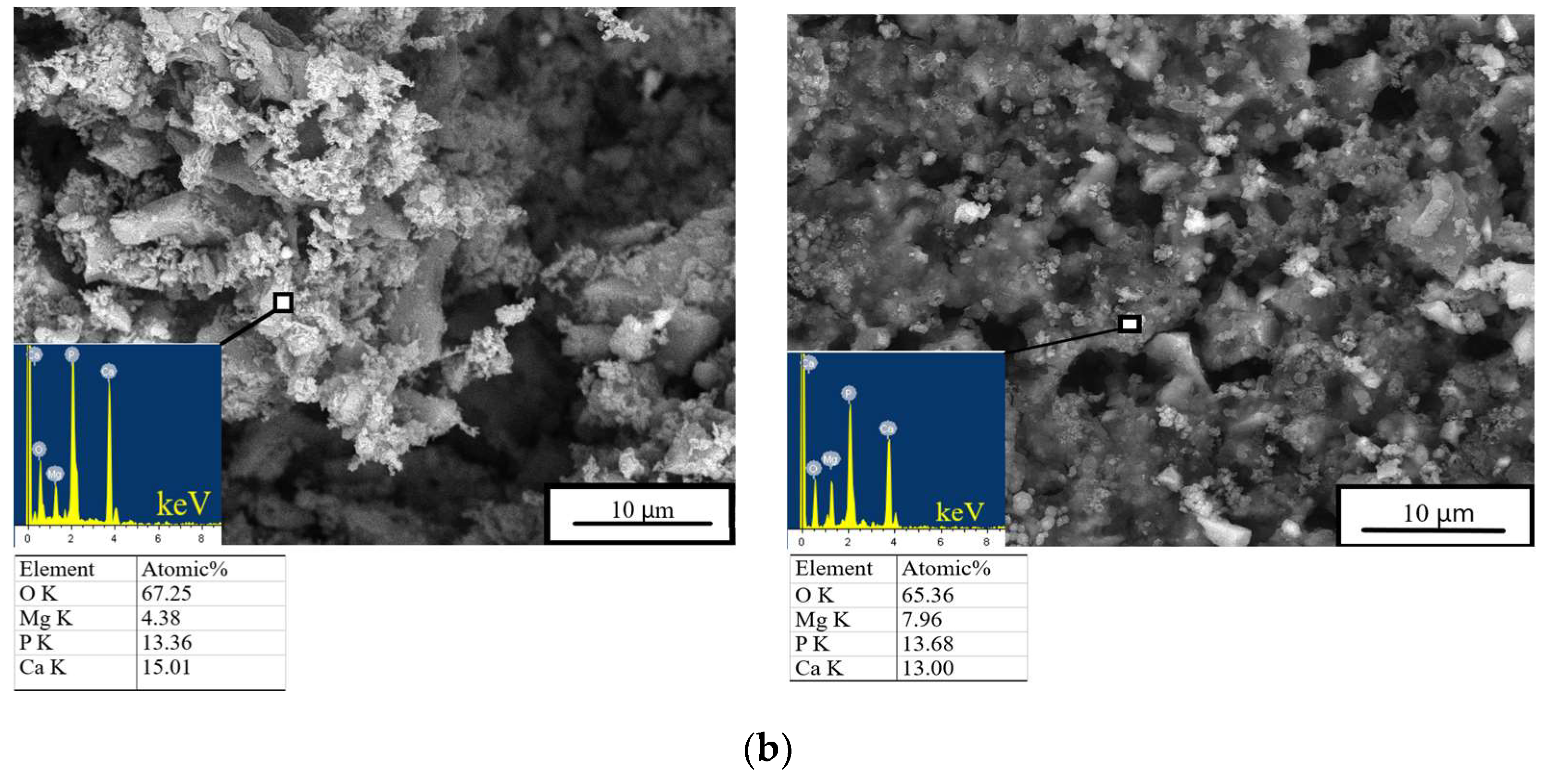
After 28 days of soaking, SEM data revealed the formation of a porous and loose surface with flower-like structures (radial–radiant aggregates) of the newly formed bobierrite phase [Mg3(PO4)2∙8H2O], confirmed by energy-dispersive X-ray (EDX) analysis as a magnesium phosphate phase in the SBF (Figure 8). A structure similar to the above-mentioned radial–radiant structure and composed of whisker-like particles is present in macroporous MPC obtained from amorphous magnesium phosphate cement powders after soaking in the SBF for 7 days [35].
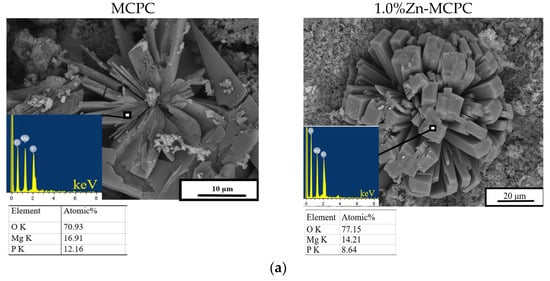
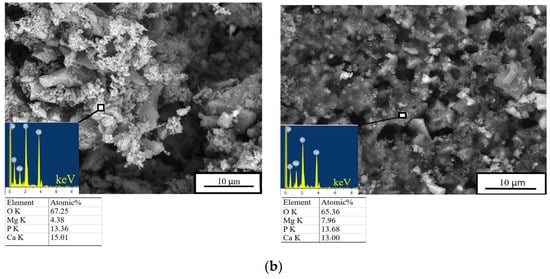
Figure 8.
In vitro dissolution assays. Morphology (EDX data) of the cement materials’ surface after 28 days of soaking in the (a) SBF or (b) DPBS.
The bobierrite phase was probably produced by reaction 6 (see below). MgO is not soluble in water in the absence of acidic components but can create a favorable basic environment for bobierrite synthesis via the following reaction:
3Mg2+(aq) + 2PO43−(aq) + 8H2O = Mg3(PO4)2∙8H2O
According to XRD data, by the 14th day of the experiment, the newberyite that was registered in the Zn2+-doped materials was fully dissolved in the SBF. The emergence of the new bobierrite phase was detected on the surface of the MCPC and Zn2+-doped samples after 28 days of soaking.
3.4. Antibacterial Properties
The antibacterial assays were designed to assess possible antibacterial activities of the doped cement, namely, the ability to inhibit bacterial attachment to a sample’s surface and bacteriostatic action, i.e., the capacity to inhibit bacterial growth. The results are presented in Table 3

Table 3.
Inhibition zones of bacterial growth and an assessment of the efficacy of the antibacterial effect.
According to the contact method results, the pure MCPC sample exerted antibacterial activity toward S. aureus because, owing to its slow dissolution, this material was transformed into magnesium hydroxide, which released hydroxyl ions when it dissociated [36]. By contrast, the struvite-based MCPC does not inhibit the growth of another gram-positive bacterium, S. sanguinis. In our test, the Zn2+-doped samples produced inhibition zones for both S. aureus and E. coli. As the amount of Zn2+ increased, the inhibition areas enlarged, as reported earlier when examining CPC materials [37].
3.5. Cytocompatibility of the Bone Cement Samples
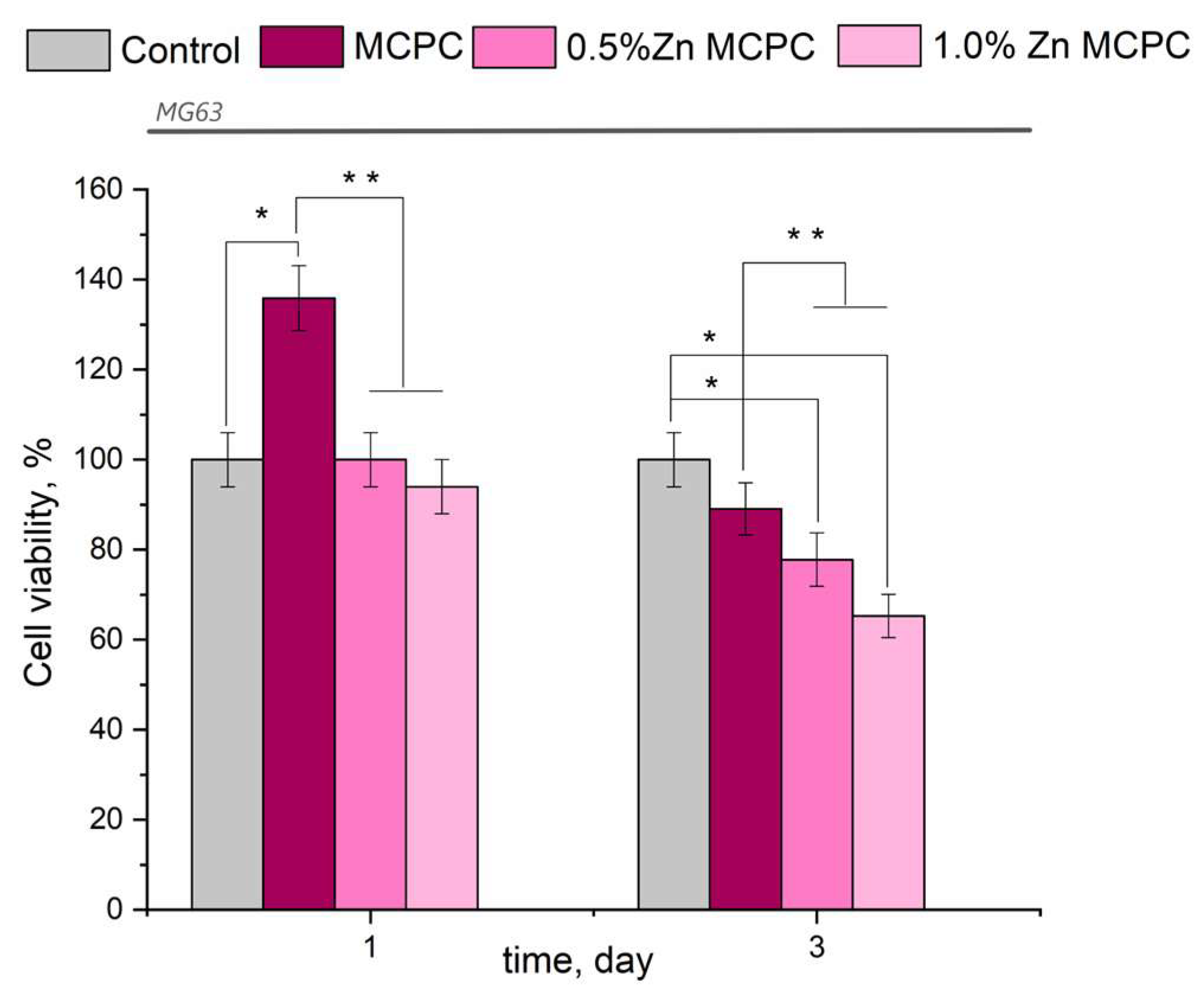
The results of this assay indicated the absence of acute toxicity of the new bone cement to the cultured cells in question (Figure 9). The findings made on the 3rd day were consistent with the cytocompatibility of the newly developed formulations having bactericidal properties because, in their presence, the value of fatty acids in the tested osteosarcoma cells was 77–87%, with the exception of the sample containing 1.0 wt.% of Zn (fatty acid value: 65.3%). The observed behavior may be attributed to the arising amorphous magnesium phosphate phases, which yielded rapid proliferation and differentiation of osteoblast-like cells in contrast to the crystalline phase.
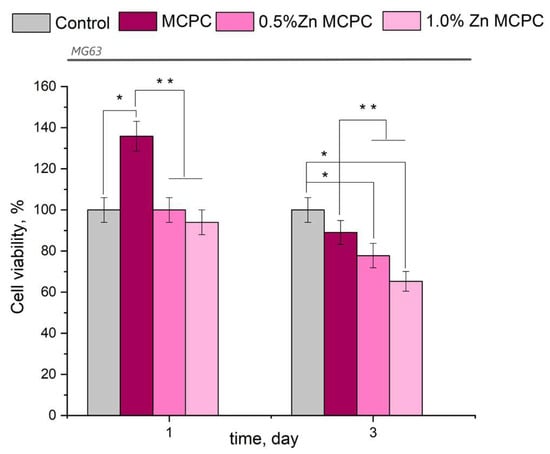
Figure 9.
Viability of MG-63 cells during the cultivation on the bone cement samples or control samples (polystyrene). Data represent means ± SD (n = 2). * Indicates a significant difference compared to the control group at p < 0.05 levels. ** Denotes a statistically significant difference comparison among the groups (p < 0.05).
From the results of the in vitro assays, it can be concluded that the proposed bone cement is nontoxic and cytocompatible.
4. Discussion
The aim of the present study was to determine the effects of Zn2+ ions introduced to MCPCs. According to the XRD analysis, it was revealed that initial powders are characterized by the presence of whitlockite, stanfieldite, and MgO phases. The greater amount of introduced Zn2+ raised the concentration of MgO because there was a large quantity of Mg2+ displaced by Zn2+ ions in the stanfieldite lattice. After mixing the initial cement powder with a cement liquid, the main cementing phase (struvite) crystallized. One of the key findings in this study was that during cementing, the thermal reaction did not proceed at all in the tested compositions of the cement.
It is well-known that MgO is one of the main phases in MPC, which reacts by recrystallizing during its interaction with a cement liquid [38]. Several factors affect the rate of struvite formation during the mixing of magnesium calcium phosphate with a cement liquid based on (NH4)2HPO4 [39]. The hydration of MPC is basically an exothermal acid base neutralization reaction. On the one hand, there is the reactivity of the initial MgO because its specific surface plays a key part in the kinetics of the reaction. On the other hand, the type of phosphate acid applied and its concentration determine the solution’s initial pH, which evolves in the course of the chemical reactions. There is a hypothesis that the successive dissolution of MgO rapidly increases the pH value of the system [40]. When the pH value is greater than 7, NH4MgPO4·6H2O starts to form needle-like crystals and generates a network. The solubility product constant Ksp of struvite is 2.5 × 10−13 at 25 °C, whereas the Ksp of other products is far higher than this; therefore, struvite is the main hydration product of MPC.
Based on the XRD data unit cell parameters, the cell volumes were calculated for MCPC and Zn2+-doped MCPC struvite and newberyite phases. The amount of struvite volume cell of the MCPC is 0.485 nm3, 0.5 wt% Zn is 0.479 nm3, and 1.0 wt% Zn is 0.478 nm3. It could be concluded that doping with Zn2+ leads to a decrease in cell volume of struvite phase. These data were obtained for the first time for MCPCs. In addition, newberyite cell volumes were determined for Zn2+-doped materials; it was found that the introduction of 1.0 wt% Zn2+ led to a decrease in cell volume from 1.096 nm3 to 1.099 nm3 for 0.5 wt% Zn2+. This relation could be explained by assuming cell distortion due to the differences in ionic radii of Zn2+ and Mg2+.
It was noteworthy that the main difference between Zn2+-doped and Zn2+-free MCPCs is the appearance of the newberyite phase during the interaction with the cement liquid, according to XRD data. Boistelle et al. [41] have noted that the pH value and the degree of supersaturation of a substance in a solution play a key role in phase crystallization. They investigated the reaction between MgSO4 and NH4H2PO4 in equimolar amounts and demonstrated that struvite is metastable at specific pH levels (pH < 6.2) and supersaturations. Struvite precipitates first and then dissolves, generating newberyite (MgHPO4·3H2O), which is stable at a low pH value.
As shown in our work, the introduction of Zn2+ into the MCPC cement increases mechanical strength by almost twofold. In Ref. [42], researchers demonstrated the effect of the introduction of ZnO nanoparticles at up to 0.2 wt.% on the mechanical strength of Portland cement (PO 42.5). It was found that with an increase in the concentration of zinc oxide from 0 to 0.2 wt.%, the strength went up and amounted to ~62 and 78 MPa, respectively, by the end of the experiment. By contrast, on the 7th day of the experiment, the strength of zinc-doped cement was ~50 MPa. The same tendency of increasing strength with the introduction of Zn ions into the matrix was documented in Ref. [43], where the strength of cement not doped with zinc was ~15.4 MPa on day 7, whereas, for cement doped with zinc (1.4 wt.% of Zn), it was 17.5 MPa. In the present work, the synthesized MCPC doped with 1 wt.% of Zn2+ had a strength of 48 ± 5 MPa, which is comparable to structural Portland cement and almost two times stronger than CPC. Such high strength of the MCPC may be due to the nascent newberyite phase [44]. A similar effect—the strengthening of magnesium phosphate cement by doping with 1 wt.% of ZnO resulting in a mechanical strength of 49.2 MPa—is explained by the authors of Ref. [45]. The incorporated ZnO not only acted as stress concentration points absorbing a large amount of energy and promoting shear strain of the MPC matrix under the influence of external stress but also served as an effective barrier to dislocation movement, and efficiently impeded crack extension.
As already mentioned in the introduction, the present authors have evaluated MCPCs doped with silver ions [19] but, given the high cost of silver as a metal, the present authors decided to try an alternative: a study on doping with zinc. Zinc is known to have an antibacterial effect. Ref. [43] suggests that the addition of 1.4 wt.% of Zn2+ into brushite cement gives antibacterial properties against E. coli, E. faecium, and P. aeruginosa. In the work, the inhibition zone of E. coli for the MCPC was 14 mm, whereas, for the MCPC doped with 1.0 wt.% of Zn2+, it was 2 mm. This phenomenon is most likely due to the fact that the solubility of calcium phosphate cement is higher, and consequently, the release of zinc ions is more intense. It should be emphasized that gram-negative bacteria are more susceptible to zinc ions than gram-positive ones due to a thicker cell wall. No antibacterial effect against E. coli was observed in the case of the MCPC, but for both Zn2+-doped materials under study, sufficient antibacterial activity was achieved. Thus, doping with Zn2+ contributed to the antibacterial properties of the newly developed MCPCs. In Ref. [37], the average release of zinc ions from CPC at 0.6 wt.% of Zn was 0.307001 mg/L, and at 1.2 wt.% of Zn, it was 0.107001 mg/L. On the other hand, the results of antibacterial assays showed an inhibitory effect against pathogenic E. coli for CPC containing 0.6 wt.% of Zn2+. In our work, we demonstrated high antibacterial activity against the bacterium S. aureus ATCC 6538 at a lower yield of zinc ions, less than 0.1 mg/L. In Ref. [46], the researchers were unable to detect any bactericidal effect of CPC doped with zinc against gram-positive bacterium S. aureus ATCC 25923, whereas in our work, a strong inhibitory effect was registered for all compositions of the cement.
It is known that Zn2+ is a biocompatible ion and is present in many tissues in the human body [21,47]. For instance, the introduction of zinc into brushite cement at 1.4 wt.% improves the viability of NCTC L929 cells by approximately 10% relative to pure cement; this finding can be explained by the positive influence of Zn2+ ions on the proliferation of fibroblasts. A probable reason is the fact that zinc plays an important role in various biological processes and participates in a number of metabolic functions necessary for cell growth and development [43]. The introduction of 5 wt.% of zinc into apatite cement is reported to result in significantly higher proliferation rates of human osteoblasts compared to pure apatite [48], and it is proven that the release of zinc does not have toxic effects on the environment. In our work, all MCPCs were found to be nontoxic to the MG-63 cell line. The observed slight decrease in cell viability can be ascribed to the formation of amorphous phases in the cement and higher solubility on the 1st day of the experiment (6% mass losses). In Ref. [49], it is demonstrated that in biphasic mixtures of amorphous β-tricalcium magnesium phosphate, the viability of human mesenchymal stem cells decreased on the 7th day of the experiment. Those authors explain this outcome is due to rapid proliferation and the confluence of cells, i.e., the fastest cell proliferation at the beginning of the experiment. Further proliferation is slower.
Therefore, the developed cement materials are potentially useful in reconstructive surgery. Additionally, according to our analysis of the literature, there is no information on the doping of MCPCs with zinc cations; therefore, this study is novel in the field of cement materials. Here, in a CaO–P2O5–MgO system at (Ca + Mg)/p = 2.0 and 40 mol.% Mg substitution, we created MCPCs that possess improved physicochemical, mechanical, and biological properties because of a combination of different concentrations of Zn.
5. Conclusions
The present investigation shows the development of MCPCs with antibacterial properties due to the introduction of Zn2+ into cement powders based on MgO, whitlockite, and stanfieldite phases. With the help of XRD and FTIR data, we documented the formation of a newberyite phase during the setting and hardening; this phase led to major alterations in the cement’s properties. During the setting process, a thermal reaction was not observed. We observed greater mass loss during the soaking, enhanced cell growth in the MTT assay, as well as antibacterial activity. The increase in Zn2+ concentration from 0.5% to 1.0% caused a pronounced improvement of mechanical properties of the cement and enlarged the inhibition zone in a disk diffusion test against E. coli and S. aureus and did not lead to noticeable toxicity toward MG-63 cells.
Author Contributions
Investigation, P.A.K., D.R.K., O.S.A., A.S.B., A.V.L., E.M.M., I.V.M., V.A.K., I.K.S. and S.A.A.; data curation—M.A.G.; writing—original draft, P.A.K., M.A.G., A.S.F. and D.R.K.; writing—review and editing, S.M.B., V.S.K., N.S.S.; Supervision, S.M.B.; project administration, V.S.K. and N.S.S. All authors have read and agreed to the published version of the manuscript.
Funding
The research was funded by the Russian Science Foundation, grant number 23-63-10056.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
The authors would like to sincerely thank Nikolai A. Shevchuk for reviewing the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Shinde, A.; Deore, G.; Navsariwala, K.P.; Tabassum, H.; Wani, M. We are all aging, and here’s why. Aging Med. 2022, 5, 211–231. [Google Scholar] [CrossRef] [PubMed]
- Chakhtoura, M.; Dagher, H.; Sharara, S.; Ajjour, S.; Chamoun, N.; Cauley, J.; Mahfoud, Z.; Boudreau, R.; El Hajj Fuleihan, G. Systematic review of major osteoporotic fracture to hip fracture incidence rate ratios worldwide: Implications for Fracture Risk Assessment Tool (FRAX)-derived estimates. J. Bone Miner. Res. 2021, 36, 1942–1956. [Google Scholar] [CrossRef] [PubMed]
- Segura-Palleres, I.; Sobrero, F.; Roccia, F.; de Oliveira Gorla, L.F.; Pereira-Filho, V.A.; Gallafassi, D.; Faverani, L.P.; Romeo, I.; Bojino, A.; Copelli, C.; et al. Characteristics and age-related injury patterns of maxillofacial fractures in children and adolescents: A multicentric and prospective study. Dent. Traumatol. 2022, 38, 213–222. [Google Scholar] [CrossRef] [PubMed]
- Moukengue, B.; Lallier, M.; Marchandet, L.; Baud’huin, M.; Verrecchia, F.; Ory, B.; Lamoureux, F. Origin and Therapies of Osteosarcoma. Cancers 2022, 14, 3503. [Google Scholar] [CrossRef]
- Salgado-Peralvo, A.O.; Mateos-Moreno, M.V.; Velasco-Ortega, E.; Peña-Cardelles, J.F.; Kewalramani, N. Preventive antibiotic therapy in bone augmentation procedures in oral implantology: A systematic review. J. Stomatol. Oral Maxillofac. Surg. 2022, 123, 74–80. [Google Scholar] [CrossRef]
- Bassetti, S.; Tschudin-Sutter, S.; Egli, A.; Osthoff, M. Optimizing antibiotic therapies to reduce the risk of bacterial resistance. Eur. J. Intern. Med. 2022, 99, 7–12. [Google Scholar] [CrossRef]
- Ke Ren, A.D. Drug Delivery Strategies for Treating Osteoporosis. Orthop. Muscular Syst. 2014, s2, 8–11. [Google Scholar] [CrossRef]
- Al-Husinat, L.; Jouryyeh, B.; Al Sharie, S.; Al Modanat, Z.; Jurieh, A.; Al Hseinat, L.; Varrassi, G. Bone Cement and Its Anesthetic Complications: A Narrative Review. J. Clin. Med. 2023, 12, 2105. [Google Scholar] [CrossRef]
- Choryfek, P. Vertebroplasty and kyphoplasty—advantages and disadvantages used bone cement of PMMA. J. Achiev. Mater. Manuf. Eng. 2019, 92, 36–49. [Google Scholar] [CrossRef]
- Vezenkova, A.; Locs, J. Sudoku of porous, injectable calcium phosphate cements—Path to osteoinductivity. Bioact. Mater. 2022, 17, 109–124. [Google Scholar] [CrossRef]
- Kaiser, F.; Schröter, L.; Stein, S.; Krüger, B.; Weichhold, J.; Stahlhut, P.; Ignatius, A.; Gbureck, U. Accelerated bone regeneration through rational design of magnesium phosphate cements. Acta Biomater. 2022, 145, 358–371. [Google Scholar] [CrossRef]
- Wu, J.; Liu, F.; Wang, Z.; Liu, Y.; Zhao, X.; Fang, C.; Leung, F.; Yeung, K.W.K.; Wong, T.M. The Development of a Magnesium-Releasing and Long-Term Mechanically Stable Calcium Phosphate Bone Cement Possessing Osteogenic and Immunomodulation Effects for Promoting Bone Fracture Regeneration. Front. Bioeng. Biotechnol. 2022, 9, 803723. [Google Scholar] [CrossRef]
- Goldberg, M.A.; Krohicheva, P.A.; Fomin, A.S.; Khairutdinova, D.R.; Antonova, O.S.; Baikin, A.S.; Smirnov, V.V.; Fomina, A.A.; Leonov, A.V.; Mikheev, I.V.; et al. Insitu magnesium calcium phosphate cements formation: From one pot powders precursors synthesis to in vitro investigations. Bioact. Mater. 2020, 5, 644–658. [Google Scholar] [CrossRef]
- Liu, J.; Liao, J.; Li, Y.; Yang, Z.; Ying, Q.; Xie, Y.; Zhou, A. Bioactive tetracalcium phosphate/magnesium phosphate composite bone cement for bone repair. J. Biomater. Appl. 2019, 34, 239–249. [Google Scholar] [CrossRef]
- Tseng, T.H.; Chang, C.H.; Chen, C.L.; Chiang, H.; Hsieh, H.Y.; Wang, J.H.; Young, T.H. A simple method to improve the antibiotic elution profiles from polymethylmethacrylate bone cement spacers by using rapid absorbable sutures. BMC Musculoskelet. Disord. 2022, 23, 916. [Google Scholar] [CrossRef]
- Anis, H.K.; Sodhi, N.; Faour, M.; Klika, A.K.; Mont, M.A.; Barsoum, W.K.; Higuera, C.A.; Molloy, R.M. Effect of Antibiotic-Impregnated Bone Cement in Primary Total Knee Arthroplasty. J. Arthroplasty 2019, 34, 2091–2095. [Google Scholar] [CrossRef]
- Hu, M.H.; Chu, P.Y.; Huang, S.M.; Shih, B.S.; Ko, C.L.; Hu, J.J.; Chen, W.C. Injectability, Processability, Drug Loading, and Antibacterial Activity of Gentamicin-Impregnated Mesoporous Bioactive Glass Composite Calcium Phosphate Bone Cement In Vitro. Biomimetics 2022, 7, 121. [Google Scholar] [CrossRef]
- Köse, N.; Asfuroğlu, Z.M.; Köse, A.; Şahintürk, V.; Gürbüz, M.; Doğan, A. Silver ion-doped calcium phosphate-based bone-graft substitute eliminates chronic osteomyelitis: An experimental study in animals. J. Orthop. Res. 2021, 39, 1390–1401. [Google Scholar] [CrossRef]
- Krokhicheva, P.A.; Goldberg, M.A.; Fomin, A.S.; Khayrutdinova, D.R.; Antonova, O.S.; Baikin, A.S.; Konovalov, A.A.; Leonov, A.V.; Mikheev, I.V.; Merzlyak, E.M.; et al. Enhanced bone repair by silver-doped magnesium calcium phosphate bone cements. Ceram. Int. 2023, 49, 19249–19264. [Google Scholar] [CrossRef]
- Fiore, M.; Sambri, A.; Zucchini, R.; Giannini, C.; Donati, D.M.; De Paolis, M. Silver-coated megaprosthesis in prevention and treatment of peri-prosthetic infections: A systematic review and meta-analysis about efficacy and toxicity in primary and revision surgery. Eur. J. Orthop. Surg. Traumatol. 2021, 31, 201–220. [Google Scholar] [CrossRef]
- Molenda, M.; Kolmas, J. The Role of Zinc in Bone Tissue Health and Regeneration—A Review. Biol. Trace Elem. Res. 2023. online ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Qiao, Y.; Zhang, W.; Tian, P.; Meng, F.; Zhu, H.; Jiang, X.; Liu, X.; Chu, P.K. Stimulation of bone growth following zinc incorporation into biomaterials. Biomaterials 2014, 35, 6882–6897. [Google Scholar] [CrossRef]
- Su, Y.; Wang, K.; Gao, J.; Yang, Y.; Qin, Y.X.; Zheng, Y.; Zhu, D. Enhanced cytocompatibility and antibacterial property of zinc phosphate coating on biodegradable zinc materials. Acta Biomater. 2019, 98, 174–185. [Google Scholar] [CrossRef] [PubMed]
- Ijaz, M.; Zafar, M.; Islam, A.; Afsheen, S.; Iqbal, T. A Review on Antibacterial Properties of Biologically Synthesized Zinc Oxide Nanostructures. J. Inorg. Organomet. Polym. Mater. 2020, 30, 2815–2826. [Google Scholar] [CrossRef]
- Uysal, I.; Yilmaz, B.; Evis, Z. Zn-doped hydroxyapatite in biomedical applications. J. Aust. Ceram. Soc. 2021, 57, 869–897. [Google Scholar] [CrossRef]
- Hill, W.L.; Faust, G.T.; Reynolds, D.S. The Binary System P2O5-2CaO.P2O5. Part I. Am. J. Sci. 1944, 242, 457–477. [Google Scholar] [CrossRef]
- ISO 9917-1:2007; Dentistry—Water-Based Cements—Part 1: Powder/Liquid Acid-Base Cements. International Organization for Standardization: Cham, Switzerland, 2007.
- Kokubo, T.; Takadama, H. How useful is SBF in predicting in vivo bone bioactivity? Biomaterials 2006, 27, 2907–2915. [Google Scholar] [CrossRef]
- Devi, K.B.; Tripathy, B.; Roy, A.; Lee, B.; Kumta, P.N.; Nandi, S.K.; Roy, M. In Vitro Biodegradation and in Vivo Biocompatibility of Forsterite Bio-Ceramics: Effects of Strontium Substitution. ACS Biomater. Sci. Eng. 2019, 5, 530–543. [Google Scholar] [CrossRef]
- GOST ISO 20645-2014; Textile Products. Determination of Antibacterial Activity. Diffusive Test in Agar Plate. International Organization for Standardization: Cham, Switzerland, 2014.
- Mosmann, T. Rapid Colorimetric Assay for Cellular Growth and Survival: Application to Proliferation and Cytotoxicity Assays Tim. J. lmmunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Shannon, R.D. Revised Effective Ionic Radii and Systematic Studies of Interatomie Distances in Halides and Chaleogenides. Acta Crystallogr. 1976, A32, 751–767. [Google Scholar] [CrossRef]
- Lee, G.H.; Makkar, P.; Paul, K.; Lee, B.T. Incorporation of BMP-2 loaded collagen conjugated BCP granules in calcium phosphate cement based injectable bone substitutes for improved bone regeneration. Mater. Sci. Eng. C 2017, 77, 713–724. [Google Scholar] [CrossRef]
- Mestres, G.; Ginebra, M.P. Novel magnesium phosphate cements with high early strength and antibacterial properties. Acta Biomater. 2011, 7, 1853–1861. [Google Scholar] [CrossRef]
- Babaie, E.; Lin, B.; Bhaduri, S.B. A new method to produce macroporous Mg-phosphate bone growth substitutes. Mater. Sci. Eng. C 2017, 75, 602–609. [Google Scholar] [CrossRef]
- Wagh, A.S.; Jeong, S.Y. Chemically Bonded Phosphate Ceramics: I, A Dissolution Model of Formation. J. Am. Ceram. Soc. 2003, 86, 1838–1844. [Google Scholar] [CrossRef]
- Graziani, V.; Fosca, M.; Egorov, A.A.; Zobkov, Y.V.; Fedotov, A.Y.; Baranchikov, A.E.; Ortenzi, M.; Caminiti, R.; Komlev, V.S.; Rau, J.V. Zinc-releasing calcium phosphate cements for bone substitute materials. Ceram. Int. 2016, 42, 17310–17316. [Google Scholar] [CrossRef]
- Haque, M.A.; Chen, B. Research progresses on magnesium phosphate cement: A review. Constr. Build. Mater. 2019, 211, 885–898. [Google Scholar] [CrossRef]
- Moseke, C.; Saratsis, V.; Gbureck, U. Injectability and mechanical properties of magnesium phosphate cements. J. Mater. Sci. Mater. Med. 2011, 22, 2591–2598. [Google Scholar] [CrossRef]
- Tansel, B.; Lunn, G.; Monje, O. Struvite formation and decomposition characteristics for ammonia and phosphorus recovery: A review of magnesium-ammonia-phosphate interactions. Chemosphere 2018, 194, 504–514. [Google Scholar] [CrossRef]
- Boistelle, R.; Abbona, F.; Lundager Madsen, H.E. On the transformation of struvite into newberyite in aqueous systems. Phys. Chem. Miner. 1983, 9, 216–222. [Google Scholar] [CrossRef]
- Liu, J.; Jin, H.; Gu, C.; Yang, Y. Effects of zinc oxide nanoparticles on early-age hydration and the mechanical properties of cement paste. Constr. Build. Mater. 2019, 217, 352–362. [Google Scholar] [CrossRef]
- Fadeeva, I.V.; Goldberg, M.A.; Preobrazhensky, I.I.; Mamin, G.V.; Davidova, G.A.; Agafonova, N.V.; Fosca, M.; Russo, F.; Barinov, S.M.; Cavalu, S.; et al. Improved cytocompatibility and antibacterial properties of zinc-substituted brushite bone cement based on β-tricalcium phosphate. J. Mater. Sci. Mater. Med. 2021, 32, 99. [Google Scholar] [CrossRef] [PubMed]
- Klammert, U.; Reuther, T.; Blank, M.; Reske, I.; Barralet, J.E.; Grover, L.M.; Kübler, A.C.; Gbureck, U. Phase composition, mechanical performance and in vitro biocompatibility of hydraulic setting calcium magnesium phosphate cement. Acta Biomater. 2010, 6, 1529–1535. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Shi, F.; Zhao, D.; Yan, Y. Effect of ZnO-doped magnesium phosphate cements on osteogenic differentiation of mBMSCs in vitro. J. Appl. Biomater. Funct. Mater. 2022, 20, 22808000221136369. [Google Scholar] [CrossRef] [PubMed]
- Marques, C.F.; Olhero, S.; Abrantes, J.C.C.; Marote, A.; Ferreira, S.; Vieira, S.I.; Ferreira, J.M.F. Biocompatibility and antimicrobial activity of biphasic calcium phosphate powders doped with metal ions for regenerative medicine. Ceram. Int. 2017, 43, 15719–15728. [Google Scholar] [CrossRef]
- Cockerill, I.; Su, Y.; Sinha, S.; Qin, Y.X.; Zheng, Y.; Young, M.L.; Zhu, D. Porous zinc scaffolds for bone tissue engineering applications: A novel additive manufacturing and casting approach. Mater. Sci. Eng. C 2020, 110, 110738. [Google Scholar] [CrossRef]
- Ishikawa, K.; Miyamoto, Y.; Yuasa, T.; Ito, A.; Nagayama, M.; Suzuki, K. Fabrication of Zn containing apatite cement and its initial evaluation using human osteoblastic cells. Biomaterials 2002, 23, 423–428. [Google Scholar] [CrossRef]
- Singh, S.S.; Roy, A.; Lee, B.; Banerjee, I.; Kumta, P.N. Synthesis, characterization, and in-vitro cytocompatibility of amorphous β-tri-calcium magnesium phosphate ceramics. Mater. Sci. Eng. C 2016, 67, 636–645. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).