Preparation of Hydrogarnet/Poly(Lactic Acid) Composite Adsorbents for Humic Substance Removal
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of the Samples
2.2. Characterization of the Samples
2.3. Analysis of the Water Remediation Ability of the Samples
3. Results
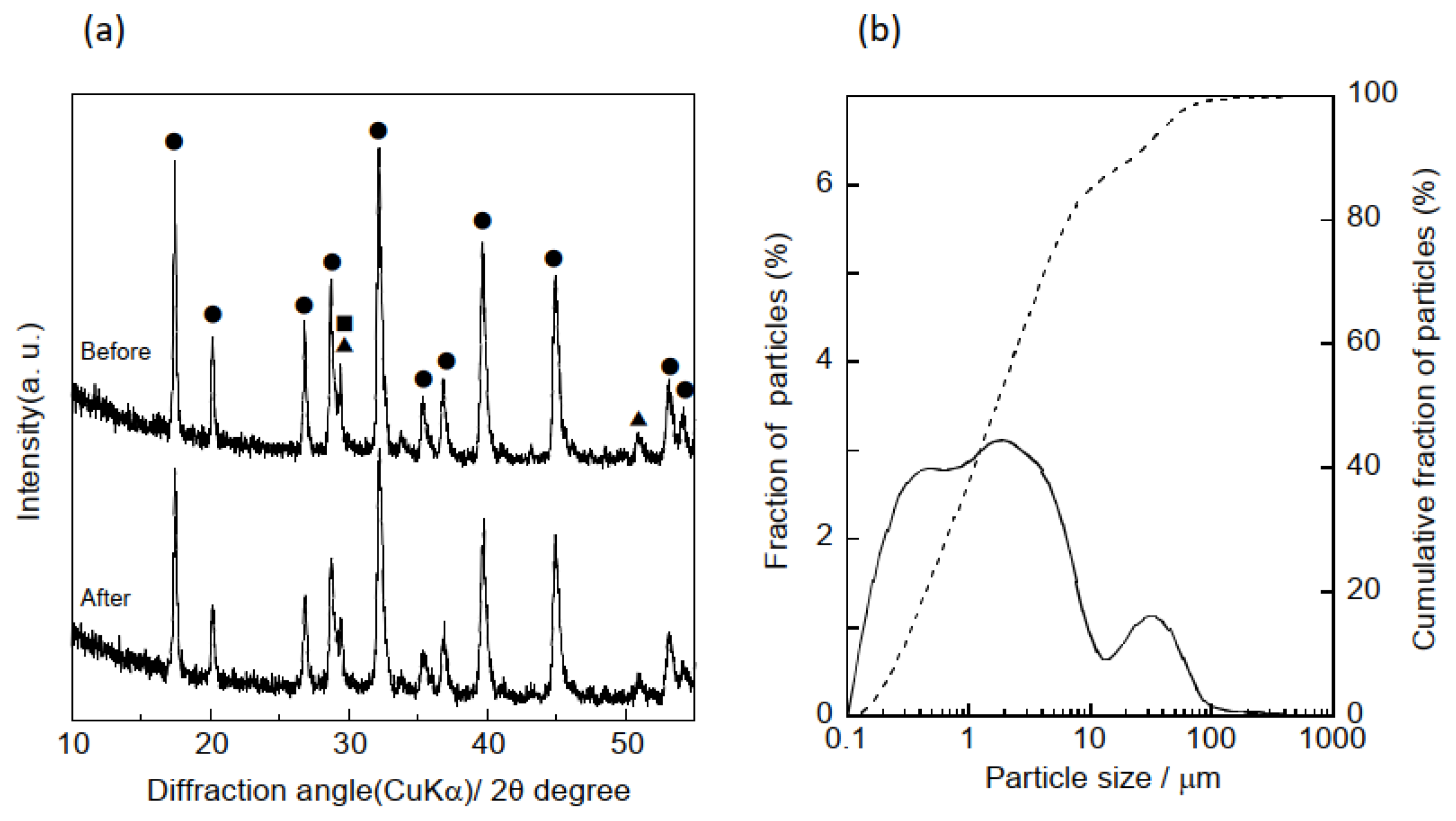
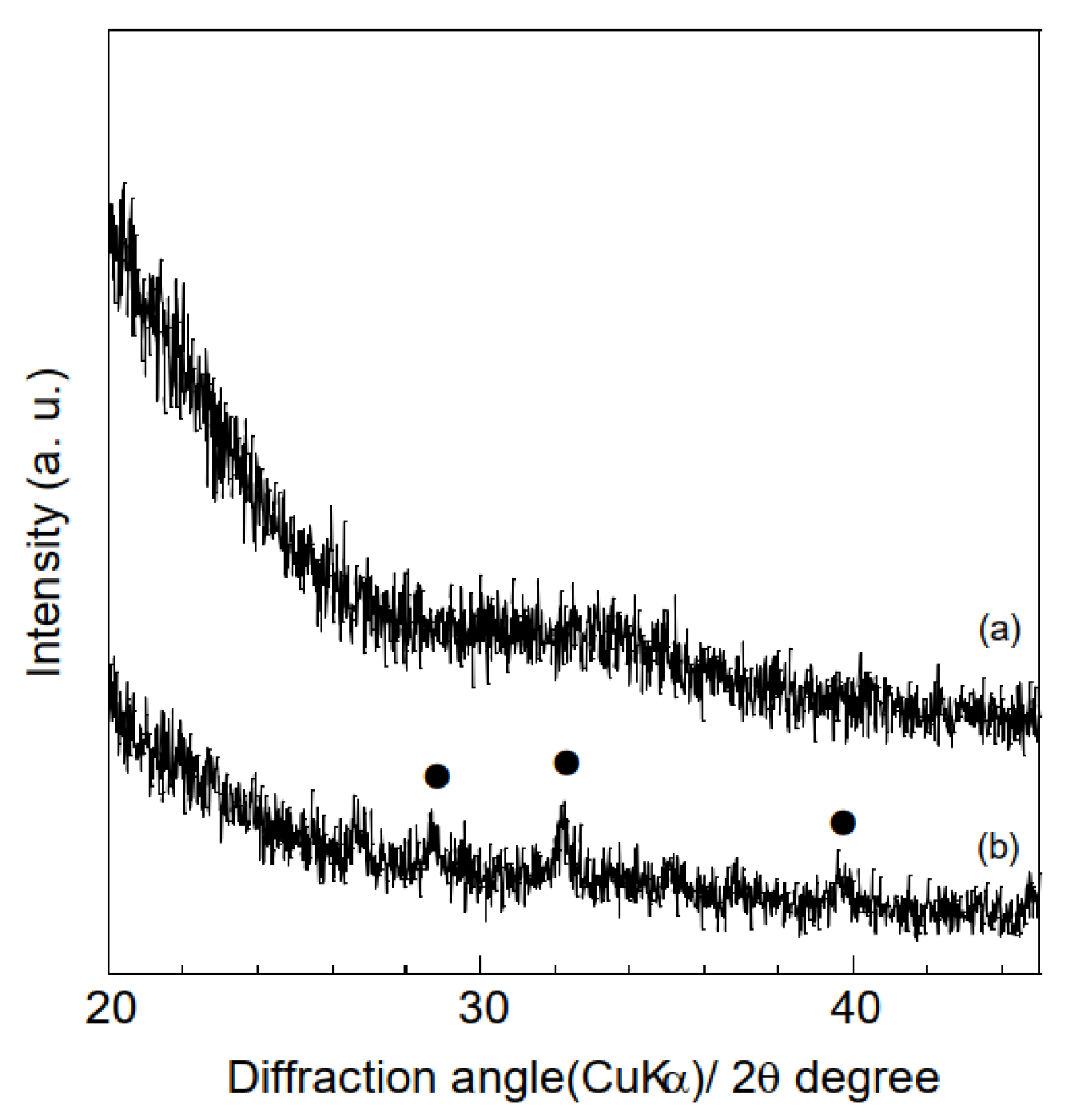
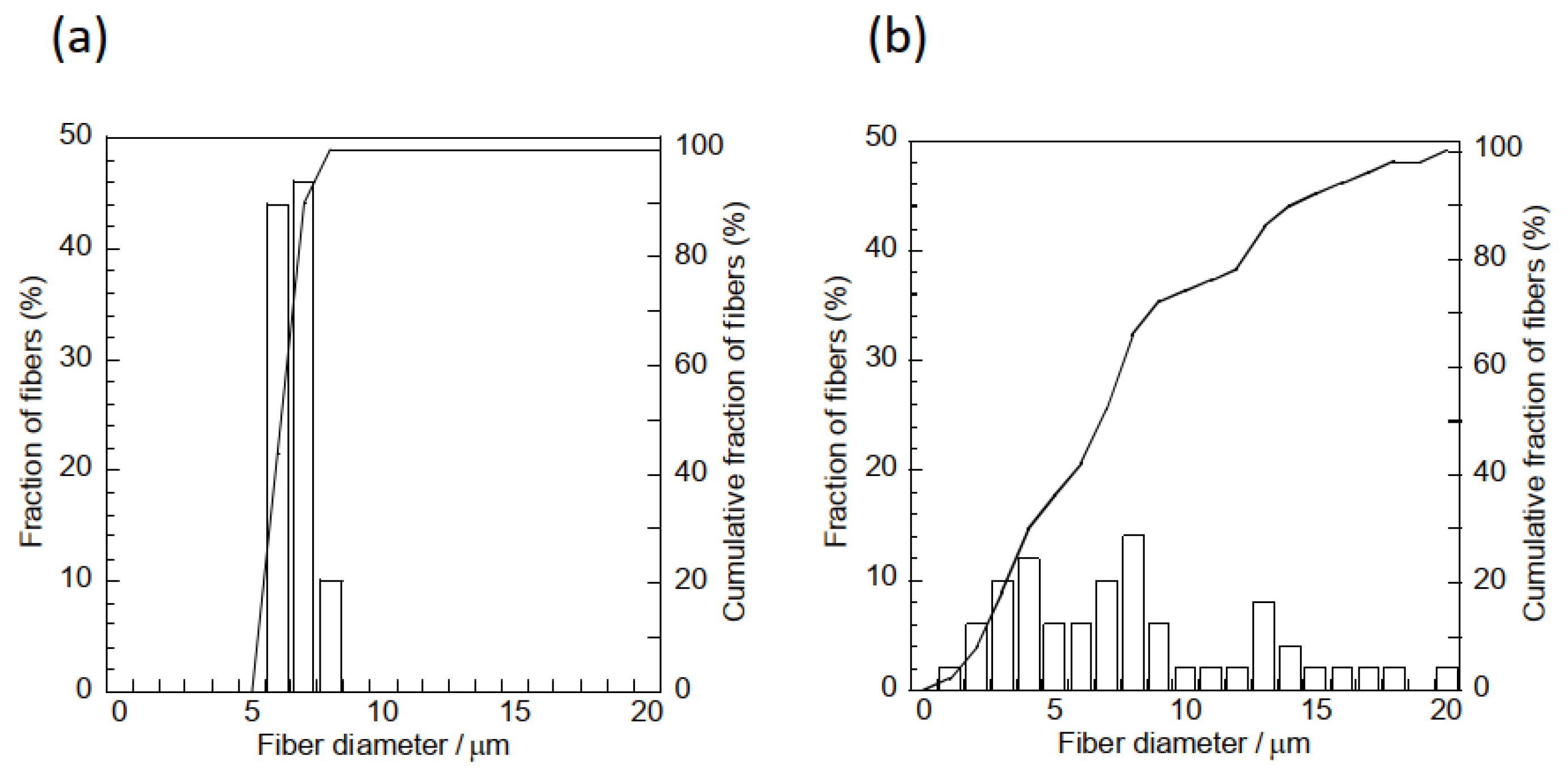
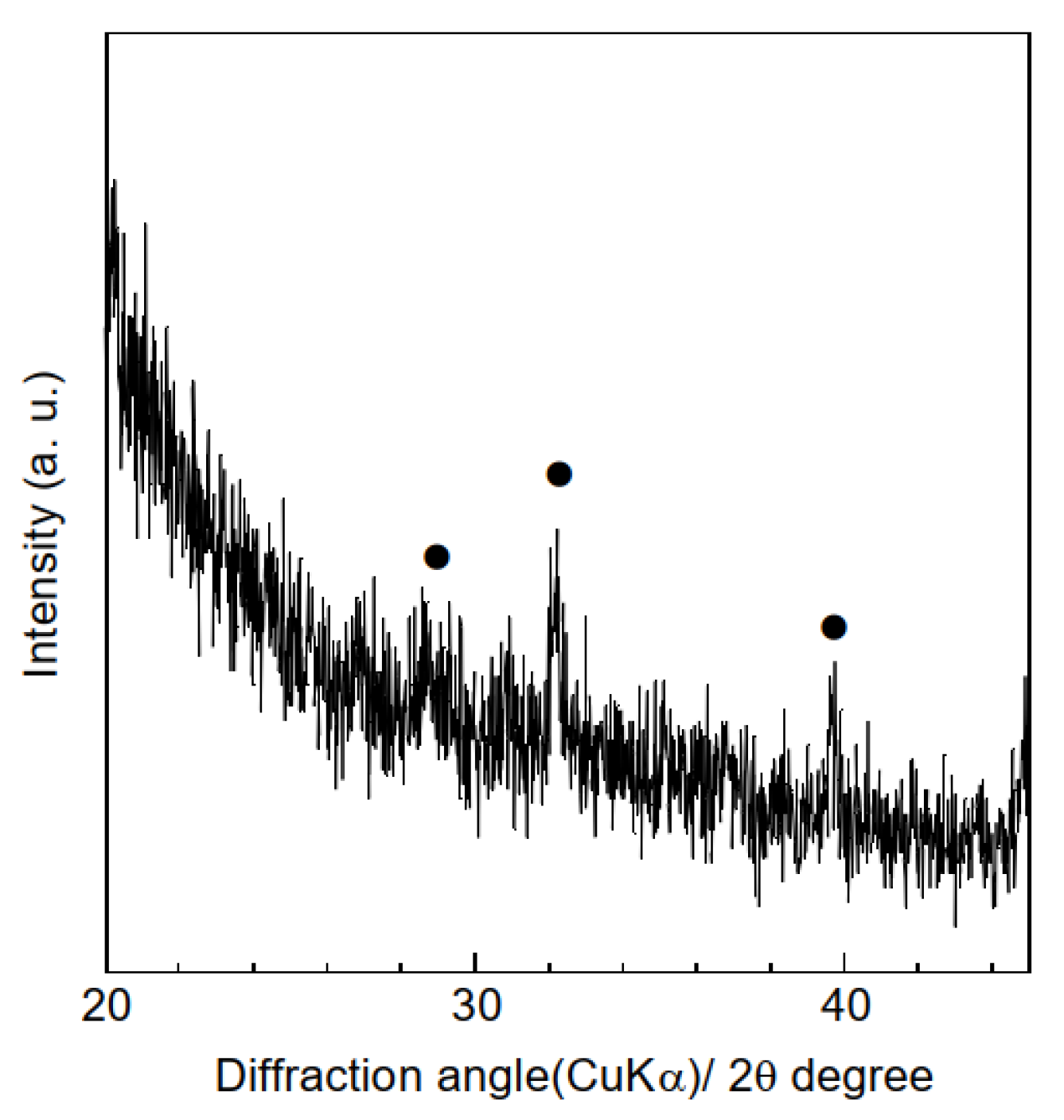
3.1. Characterization of Samples
3.2. Humic Acid Adsorption Proerty of the Samples
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Giasuddin, A.B.M.; Kanel, S.R.; Choi, H. Adsorption of humic acid onto nanoscale zerovalent iron and its effect on arsenic removal. Environ. Sci. Technol. 2007, 41, 2022–2027. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Jiang, Y.; An, X.; Liu, H.; Hu, C.; Qu, J. Transformation of humic acid and halogenated byproduct formation in UV-chlorine processes. Water Res. 2016, 102, 421–427. [Google Scholar] [CrossRef]
- Chen, W.; Ran, X.; Jiang, X.; Min, H.; Li, D.; Zou, L.; Fan, J.; Li, G. Synthesis of TiO2 and TiO2-Pt and their application in photocatalytic degradation of humic acid. Water Environ. Res. 2014, 86, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Uyguner, C.S.; Suphandag, S.A.; Kerc, A.; Bekbolet, M. Evaluation of adsorption and coagulation characteristics of humic acids preceded by alternative advanced oxidation techniques. Desalination 2007, 210, 183–193. [Google Scholar] [CrossRef]
- Terkiatburana, T.; Wang, S.; Tadé, M.O. Competition and complexation of heavy metal ions and humic acid on zeolitic MCM-22 and activated carbon. Chem. Eng. J. 2008, 139, 437–444. [Google Scholar] [CrossRef]
- Capasso, S.; Colella, C.; Coppola, E.; Iovino, P.; Salverstrini, S. Removal of humic substances from water by means of calcium-ion-enriched natural zeolite. Water Environ. Res. 2007, 79, 305–309. [Google Scholar] [CrossRef]
- Maeda, H.; Kurosaki, Y.; Nakayama, M.; Ishida, E.H.; Kasuga, T. Adsorption behaviour of hydrogarnet for humic acid. R. Soc. Open Sci. 2018, 5, 172023. [Google Scholar] [CrossRef] [PubMed]
- Cheng, P.; Liu, Y.; Yang, L.; Wang, X.; Chi, Y.; Yuan, H.; Wang, S.; Ren, Y.-X. Adsorption and recovery of phosphate from aqueous solution by katoite: Performance and mechanism. Colloids Surf. A Physichochem. Eng. Asp. 2022, 655, 130285. [Google Scholar] [CrossRef]
- Satilmis, B.; Isik, T.; Demir, M.M.; Uyar, T. Amidoxime functionalized polymers of intrinsic mictoporositu (PIM-1) electrospun ultrafine fibers for rapid removal of uranyl ions from water. Appl. Surf. Sci. 2019, 467, 648–657. [Google Scholar] [CrossRef]
- Pan, Y.; Zhang, L.; Li, Z.; Ma, L.; Zhang, Y.; Wang, J.; Meng, J. Hierarchical porous membrane via electorspinning PIM-1 for micropollutants removal. Appl. Surf. Sci. 2018, 443, 441–451. [Google Scholar] [CrossRef]
- Liang, Y.; Lin, Z.; Qiu, Y.; Zhang, X. Fabrication and characterization of LATP/PAN composite fiber-based lithium-ion battery separators. Electrochim. Acta 2011, 56, 6474–6480. [Google Scholar] [CrossRef]
- Fujihara, K.; Kotaki, M.; Ramakrishna, S. Guided bone regeneration membrane made of polycaprolactone/calcium carbonate composite nano-fibers. Biomaterials 2005, 26, 4139–4147. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H.; Kurosaki, Y.; Nakamura, T.; Nakayama, M.; Ishida, E.H.; Kasuga, T. Control of chemical composition of hydrogrossular prepared by hydrothermal reaction. Mater. Lett. 2014, 131, 132–134. [Google Scholar] [CrossRef]
- Tsuji, H.; Nakano, M.; Katsura, S.; Mizuno, A. Electrospinning of poly(lactic acid) stereocomplex nanofibers. Biomacromolecules 2006, 7, 3316–3320. [Google Scholar] [CrossRef]
- Rucka, K.; Solipiwko-Pieścik, A.; Wolska, M. Effectiveness of humic substance removal during the coagulation process. SN Appl. Sci. 2019, 1, 535. [Google Scholar] [CrossRef]
- Zhou, H.; Nabiyouni, M.; Lin, B.; Bhaduri, S.B. Fabrication of novel poly(lactic acid)/amorphous magnesium phosphate bionanocomposite fibers for tissue engineering applications via electrospinning. Mater. Sci. Eng. C 2013, 33, 2302–2310. [Google Scholar] [CrossRef]
- Okuyama, T.; Maeda, H.; Ishida, E.H. Preparation of porous poly(L-lactic acid)/tobermorite composite membranes via electrospinning and heat treatment. J. Mater. Sci. 2012, 47, 643–648. [Google Scholar] [CrossRef]
- Wang, S.-Q.; He, J.-H.; Xu, L. Non-ionic surfactants for enhancing electrospinability and for the preparation of electrospun nanofibers. Polym. Int. 2008, 57, 1079–1082. [Google Scholar] [CrossRef]
- He, J.-H.; Wan, Y.-Q.; Yu, J.-Y. Effect of concentration on electrospun polyacrylonitrile (PAN) nanofibers. Fibers Polym. 2008, 9, 140–142. [Google Scholar] [CrossRef]
- Huang, C.; Thomas, N.L. Fabricating porous poly(lactic acid) fibres via electrospinning. Eur. Polym. J. 2018, 99, 464–476. [Google Scholar] [CrossRef]
- Jeun, J.-P.; Kim, Y.-H.; Lim, Y.-M.; Choi, J.-H.; Jung, C.-H.; Kang, P.-H.; Nho, Y.-C. Electrospinning of Poly(L-lactide-co-D, L-lactide). J. Indust. Eng. Chem. 2007, 13, 592–596. [Google Scholar]
- Karanfil, T.; Kitis, M.; Kilduff, J.E.; Wigton, A. Role of granular activated carbon surface chemistry on the adsorption of organic compounds. 2. Natural organic matter. Environ. Sci. Technol. 1999, 33, 3225–3233. [Google Scholar] [CrossRef]
- Kang, S.; Xing, B. Humic acid fractionation upon sequential adsorption onto goethite. Langmuir 2008, 24, 2525–2531. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H.; Minowa, S.; Urushihara, D.; Asaka, T. Controlling the chemical composition of garnet with hydroxy groups for humic acid removal. Ceram. Int. 2022, in press. [Google Scholar] [CrossRef]
- Hyung, H.; Kim, H.H. Natural organic matter (NOM) adsorption to muli-walled carbon nanotubes: Effect of NOM characteristics and water quality parameters. Environ. Sci. Technol. 2008, 42, 4416–4421. [Google Scholar] [CrossRef]





| Polymer Sample | Composite Sample Consisting of Non-Porous Fibers | Composite Sample Consisting of Porous Fibers |
|---|---|---|
| No adsorption | 1.2 ± 0.9 mg/g | 4.6 ± 2.4 mg/g |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Minowa, S.; Maeda, H. Preparation of Hydrogarnet/Poly(Lactic Acid) Composite Adsorbents for Humic Substance Removal. Materials 2023, 16, 336. https://doi.org/10.3390/ma16010336
Minowa S, Maeda H. Preparation of Hydrogarnet/Poly(Lactic Acid) Composite Adsorbents for Humic Substance Removal. Materials. 2023; 16(1):336. https://doi.org/10.3390/ma16010336
Chicago/Turabian StyleMinowa, Shogo, and Hirotaka Maeda. 2023. "Preparation of Hydrogarnet/Poly(Lactic Acid) Composite Adsorbents for Humic Substance Removal" Materials 16, no. 1: 336. https://doi.org/10.3390/ma16010336
APA StyleMinowa, S., & Maeda, H. (2023). Preparation of Hydrogarnet/Poly(Lactic Acid) Composite Adsorbents for Humic Substance Removal. Materials, 16(1), 336. https://doi.org/10.3390/ma16010336







