A Review of Binderless Polycrystalline Diamonds: Focus on the High-Pressure–High-Temperature Sintering Process
Abstract
1. Introduction
2. The Different Forms of Pure Carbon
2.1. Natural
2.2. Synthetic
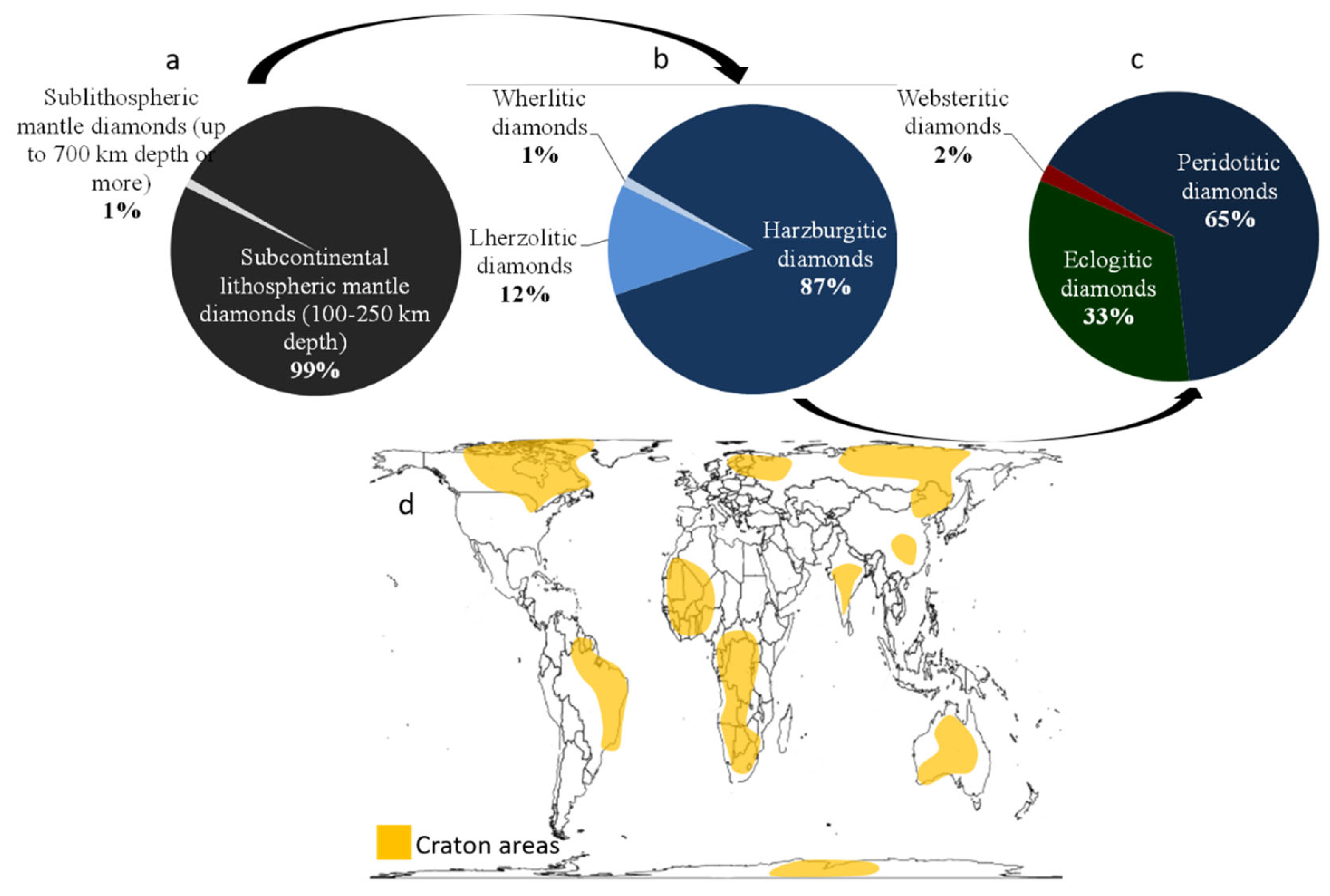
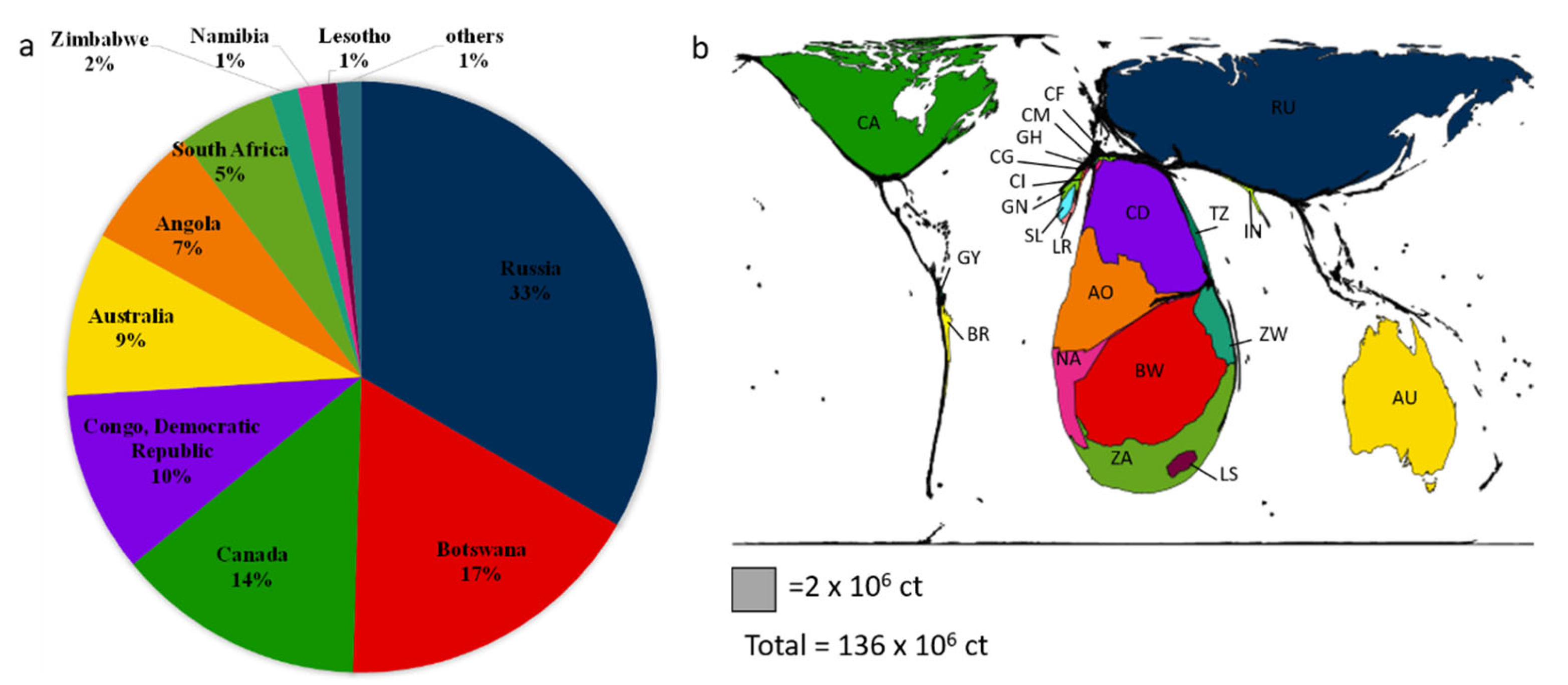
3. Natural Diamonds: Geological Settings and Classification
3.1. Natural Diamonds: Some Geological Settings
3.2. Diamond Classification
3.2.1. The 4-C Classification
3.2.2. The Type Classification
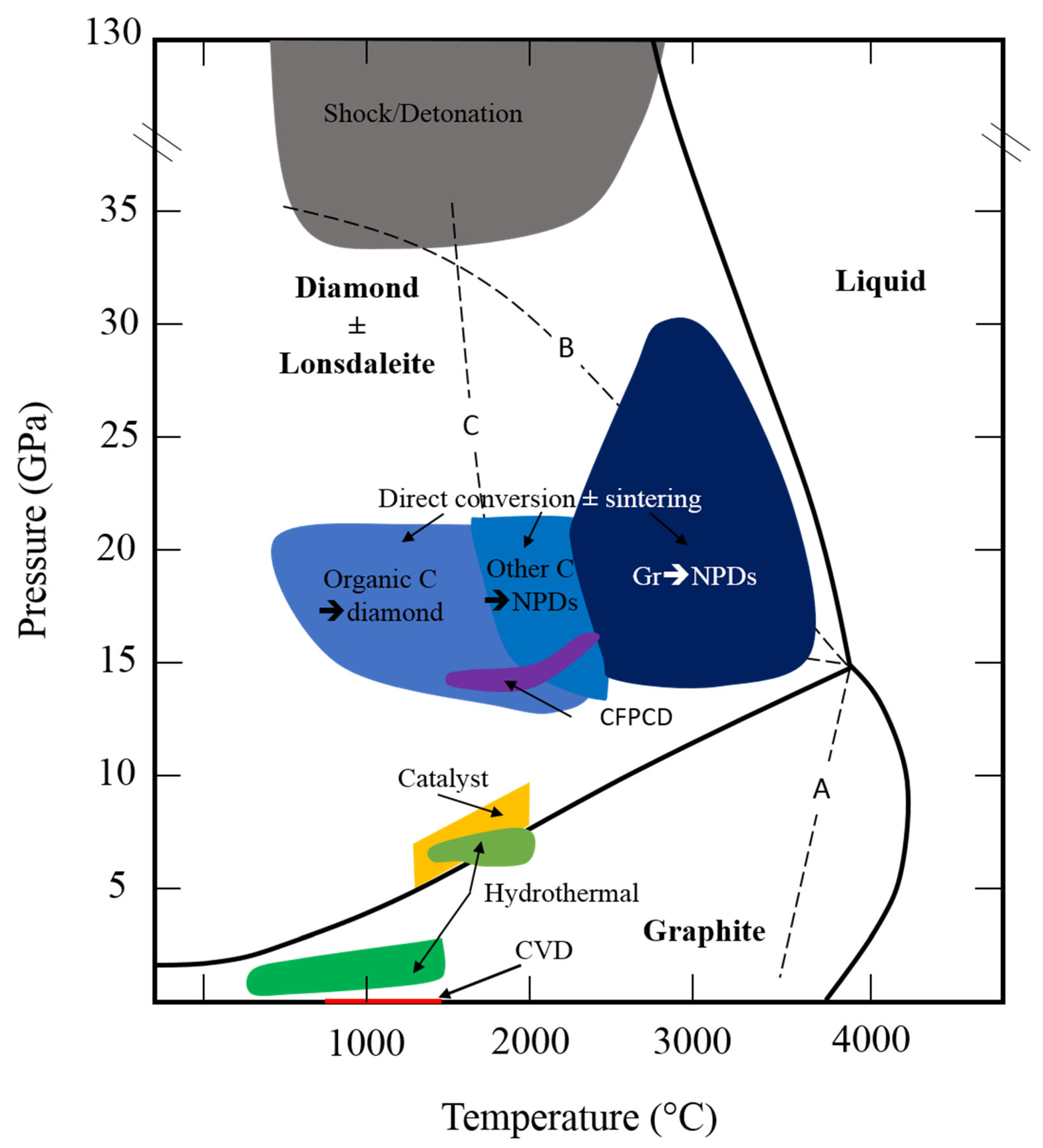
4. HP–HT C-Phase Diagram and the Graphite–Diamond Equilibrium Line
5. On the Way to Catalyst-Free/Binderless Synthetic Diamonds
5.1. Low Pressure Processes: CVD Diamonds
5.2. Medium Pressure Processes: SPS and Hydrothermal Diamonds
5.2.1. SPS Diamonds
5.2.2. Hydrothermal Diamonds
5.3. High-Pressure Processes: Towards Large Binderless Diamond Objects
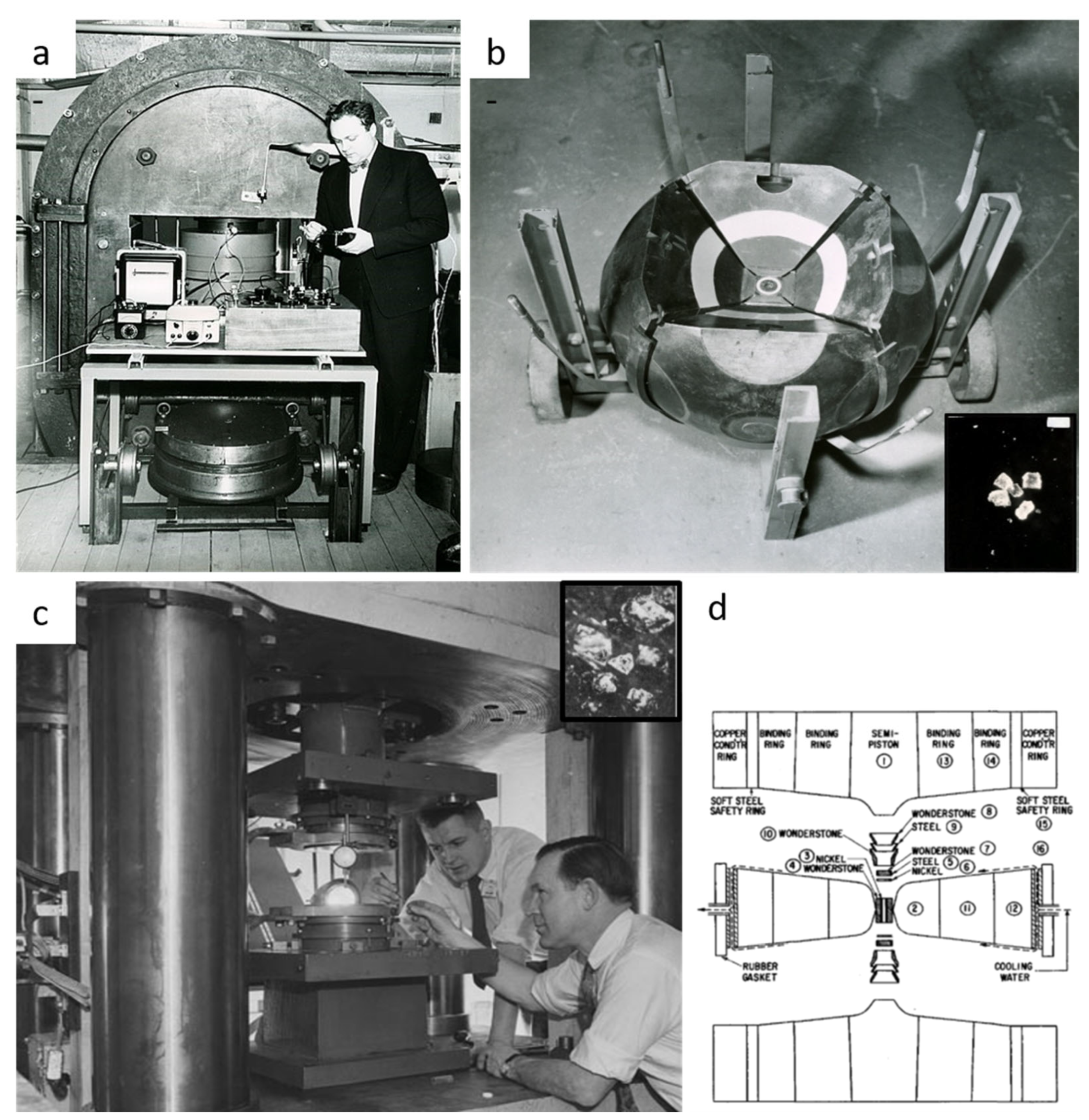
5.3.1. Origin in the 1960s
5.3.2. A Brief Description of Catalyst Synthesis of Diamonds and PDC
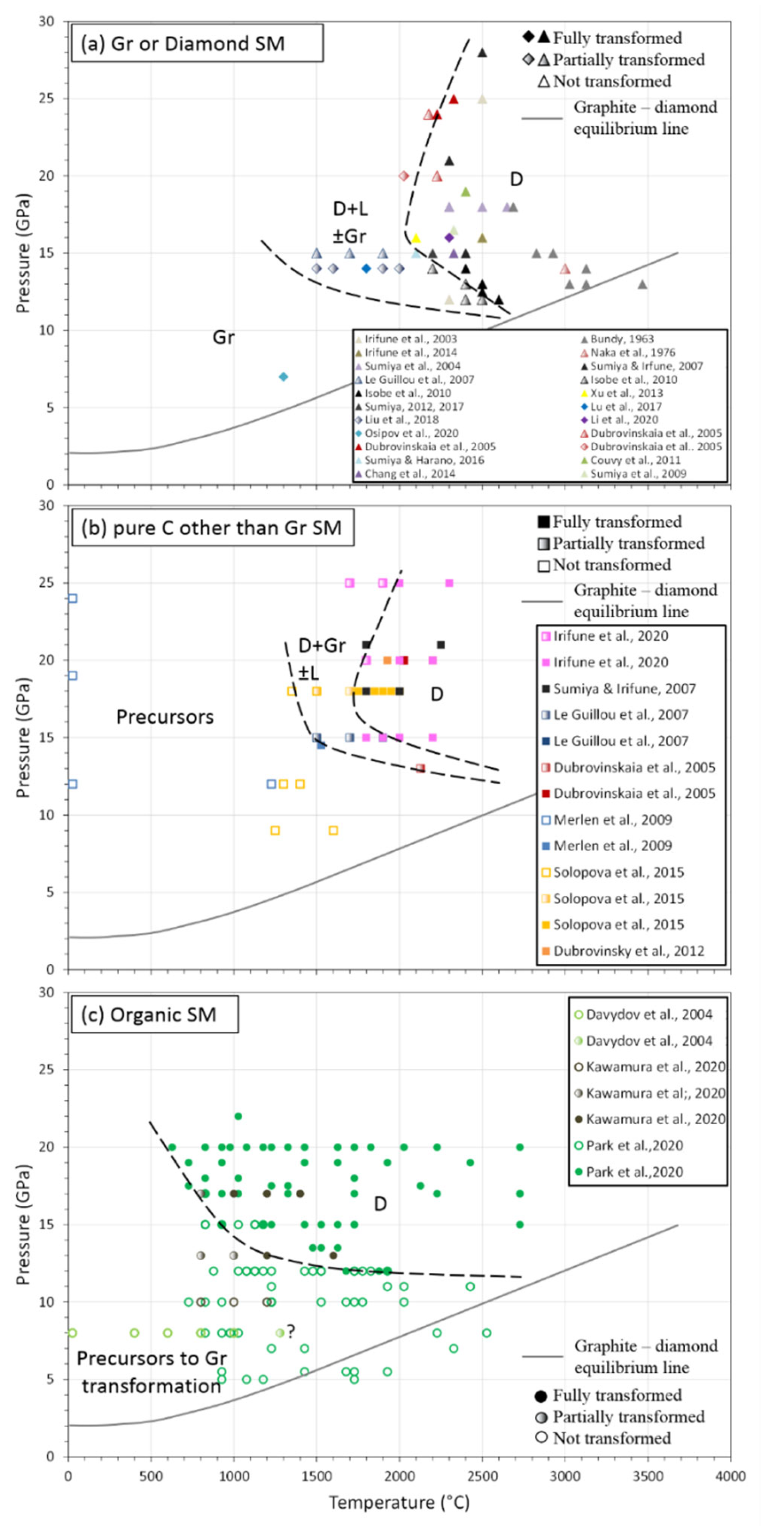
5.3.3. Development in the Last Two Decades
- Binderless Diamond from Graphite: NPDs (Nano-Polycrystalline Diamonds)
- Sintering from Diamonds Precursors
- Sintering from Other Pure Carbon Forms
- Sintering from Carbonaceous Organic Compounds
5.4. Ultra High Procesess: Shock and Detonation Diamonds
5.5. Other Unconventional Expeirmental Set-Ups
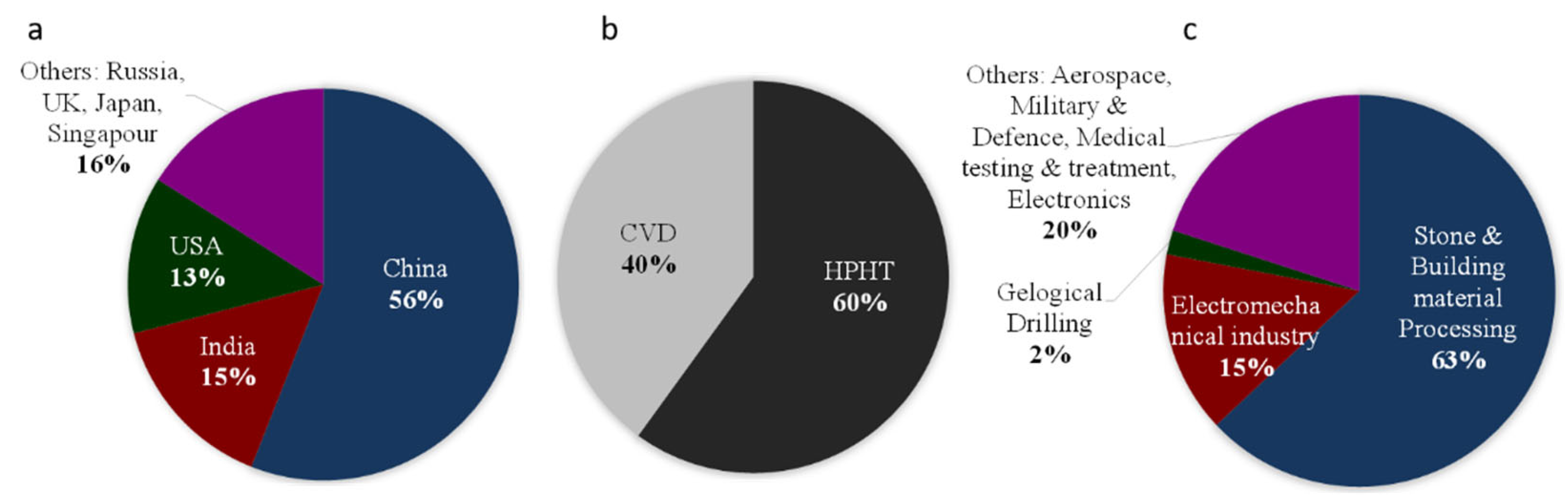
6. Diamonds Properties and Uses
6.1. General Properties
6.2. Specific Properties of Binderless Diamonds
- Mechanical properties
- Thermal properties
- Machining and Cutting properties
- Optical properties
- Uses
7. Summary and Outlook
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fischer, R.A.; Cottrell, E.; Hauri, E.; Lee, K.K.M.; Le Voyer, M. The carbon content of Earth and its core. Proc. Natl. Acad. Sci. USA 2020, 117, 8743–8749. [Google Scholar] [CrossRef]
- Parsons, C.A. Experiments of the Artificial Production of Diamond. Phil. Trans. Roy. Soc. 1918, A220, 67–106. [Google Scholar] [CrossRef][Green Version]
- Bridgman, P.W. An experimental contribution to the problem of diamond synthesis. J. Chem. Phys. 1947, 15, 92. [Google Scholar] [CrossRef]
- Hall, H.T. Ultra-high pressure, high-temperature apparatus: The “Belt”. Rev. Sci. Instrum. 1960, 31, 125–131. [Google Scholar] [CrossRef]
- Irifune, T.; Kurio, A.; Sakamoto, S.; Inoue, T.; Sumiya, H. Ultrahard polycrystalline diamond from graphite. Nature 2003, 421, 599–601. [Google Scholar] [CrossRef] [PubMed]
- Wyckoff, R.W. Crystal Structures 1, 2nd ed.; Interscience Publishers: New York, NK, USA, 1963; pp. 7–83. [Google Scholar]
- Frondel, C.; Marvin, U.B. Lonsdaleite, a Hexagonal Polymorph of Diamond. Nature 1967, 214, 587–589. [Google Scholar] [CrossRef]
- Ross, A.; Steele, A.; Fries, M.D.; Kater, L.; Downes, H.; Jones, A.P.; Smith, C.L.; Zolensky, M.E.; Shaddad, M.H. MicroRaman spectroscopy of diamond and graphite in Almahata Sitta and comparison with other ureilites. Meteorit. Planet. Sci. 2011, 46, 364–378. [Google Scholar] [CrossRef]
- Godard, G.; Frezzotti, M.L.; Palmeri, R.; Smith, D.C. Origin of high-pressure disordered metastable phases (Lonsdaleite and amorphized quartz) in metamorphic rocks: Geodynamic shock or crystal-scale overpressure? In Ultrahigh-Pressure Metamorphism; 25 years after the discovery of coesite and diamond; Elsevier: Amsterdam, The Netherlands, 2011; pp. 125–148. [Google Scholar] [CrossRef]
- Utsumi, W.; Yagi, T.; Taniguchi, T.; Shinomura, O. In situ X-ray observation of the graphite-diamond transition using synchrotron. In Proceedings of the 3rd NIRIM International Symposium on Advanced Materials, Tsukuba, Japan, 4–8 March 1996; pp. 257–261. [Google Scholar] [CrossRef]
- Pan, Z.; Sun, H.; Zhang, Y.; Chen, C. Harder than diamond: Superior indentation of wurtzite BN and Lonsdaleite. Phys. Rev. Lett. 2009, 102, 055503. [Google Scholar] [CrossRef]
- Sundqvist, B. Carbon under pressure. Physic Rep. 2021, 909, 1–73. [Google Scholar] [CrossRef]
- McIntyre, R.A. Common nano-materials and their use in real world application. Sci. Prog. 2012, 95, 1–22. [Google Scholar] [CrossRef]
- Murayama, H.; Tomonoh, S.; Alford, J.M.; Karpuk, M.E. Fullerene production in tons and more: From science to industry. Fuller. Nanotub. Carbon Nanostructures 2004, 12, 1–9. [Google Scholar] [CrossRef]
- Pagni, J. Amroy aims to become nano-leader. European Plastic News, 5 March 2010. [Google Scholar]
- Zhu, Z.; Lu, L.; Yao, X.; Zhang, W.; Liu, W. Carbon nanotube tape stays sticky in extreme temperatures. In Nanowerk Newsletter; American Chemical Society: Washington, DC, USA, 2019. [Google Scholar]
- Noyce, S.G.; Doherty, J.L.; Cheng, Z.; Han, H.; Bowen, S.; Franklin, A.D. Electronic Stability of Carbon Nanotube Transistors Under Long-Term Bias Stress. Nano Lett. 2019, 19, 1460–1466. [Google Scholar] [CrossRef] [PubMed]
- Belkin, A.; Hubler, A.; Bezryadin, A. Self-Assembled Wiggling Nano-Structures and the Principle of Maximum Entropy Production. Sci. Rep. 2015, 5, 8323. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.W.; Tan, K.H.; Ong, Y.T.; Mohamed, A.R.; Sharif, H.S.Z.; Tan, S.H. Energy and environmental applications of carbon nanotubes. Environ. Chem. Lett. 2012, 10, 265–273. [Google Scholar] [CrossRef]
- Tucker, A. Jack Andraka, the Teen Prodigy of Pancreatic Cancer. Smithsonian Magazine, 1 December 2012. [Google Scholar]
- Robertson, J. Diamond-like amorphous carbon. Mater. Sci. Eng. R Rep. 2002, 37, 129–281. [Google Scholar] [CrossRef]
- Grill, A.; Meyerson, B.S.; Patel, V.V. Diamondlike carbon films by RF plasma-assisted chemical vapor deposition from acetylene. IBM J. Res. Dev. 1990, 34, 849–857. [Google Scholar] [CrossRef]
- Polo, M.C.; Andújar, J.L.; Hart, A.; Robertson, J.; Milne, W.I. Preparation of tetrahedral amorphous carbon films by filtered cathodic vacuum arc deposition. Diam. Relat. Mater. 2000, 9, 663–667. [Google Scholar] [CrossRef]
- Ho, M.P.; Lau, A.K.-T. Amorphous carbon nanocomposites. Fill. Reinf. Adv. Nanocompos. 2015, 12, 309–328. [Google Scholar] [CrossRef]
- Narayan, J.; Bhaumik, A. Q-carbon discovery and formation of single-crystal diamond nano- and microneedles and thin films. Mater. Res. Lett. 2016, 4, 118–126. [Google Scholar] [CrossRef][Green Version]
- Narayan, J.; Bhaumik, A.; Gupta, S.; Haque, A.; Sachan, R. Progress in Q-carbon and related materials with extraordinary properties. Mater. Res. Lett. 2018, 6, 353–364. [Google Scholar] [CrossRef]
- Shirey, S.B.; Cartigny, P.; Frost, D.J.; Keshav, S.; Nestola, F.; Nimis, P.; Graham Pearson, D.; Sobolev, N.V.; Walter, M.J. Diamonds and the geology of mantle carbon. Rev. Mineral. Geochem. 2013, 75, 355–421. [Google Scholar] [CrossRef]
- Shirey, S.B.; Shigley, J.E. Recent advances in the understanding the geology of diamonds. Gems Gemol. 2013, 49, 188–232. [Google Scholar] [CrossRef]
- Hough, R.M.; Gilmour, I.; Pillinger, C.T.; Arden, J.W.; Gilkes, K.W.R.; Yuan, J.; Milledge, H.J. Diamond and silicon carbide in impact melt rock from the Ries impact crater. Nature 1995, 378, 41–44. [Google Scholar] [CrossRef]
- Ohfuji, H.; Irifune, T.; Litasov, K.D.; Yamashita, T.; Isobe, F.; Afanasiev, V.P.; Pokhilenko, N.P. Natural occurrence of pure nano-polycrystalline diamond from impact crater. Sci. Rep. 2015, 5, 14702. [Google Scholar] [CrossRef] [PubMed]
- Nabiei, F.; Badro, J.; Dennenwaldt, T.; Oveisi, E.; Cantoni, M.; Hébert, C.; El Goresy, A.; Barrat, J.-A.; Gillet, P. A large planetary body inferred from diamond inclusions in a ureilite meteorite. Nat. Commun. 2018, 9, 1327. [Google Scholar] [CrossRef] [PubMed]
- Lodders, K.; Amari, S. Presolar grains from meteorites: Remnants from the early times of the solar system. Chem. Erde 2005, 65, 93–166. [Google Scholar] [CrossRef]
- Stachel, T.; Luth, R.W. Diamond formation—Where, when and how? Lithos 2015, 220–223, 200–220. [Google Scholar] [CrossRef]
- McCall, G.J.H. The carbonado diamond conundrum. Earth-Sci. Rev. 2009, 93, 85–91. [Google Scholar] [CrossRef]
- Robinson, D.N. The characteristics of natural diamond and their interpretation. Miner. Sci. 1978, 10, 55–72. [Google Scholar]
- Frantsesson, E.V.; Kaminskiy, F.V. Carbonado, a diamond variety of nonkimberlitic origin. Akademiya Nauk SSSR Doklady. Earth Sci. Sect. 1974, 219, 117–119. [Google Scholar]
- Kagi, H.; Takahashi, K.; Hidaka, H.; Masuda, A. Chemical properties of Central American carbonado and its implications. Geochim. Cosm. Acta 1994, 58, 2629–2638. [Google Scholar] [CrossRef]
- Mitchell, R.H. Kinberlites and Lamproites: Primary sources of diamond. Geosci. Can. 1991, 18, 1–16. [Google Scholar]
- Marshall, T.R.; Baxter-Brown, R. Basic principles of alluvial diamond exploration. J. Geochem. Explor. 1995, 53, 277–292. [Google Scholar] [CrossRef]
- King, S.D. Archean cratons and mantle dynamics. Earth Planet. Sci. Lett. 2005, 234, 1–14. [Google Scholar] [CrossRef]
- US Geological Survey. Mineral Commodity Summaries 2020: US Geological Survey; US Geological Survey: Reston, VA, USA, 2020; 200p. [CrossRef]
- Gastner, M.T.; Seguy, V.; More, P. Fast flow-based algorithm for creating density-equalizing map projections. Proc. Natl. Acad. Sci. USA 2018, 115, 2156–2164. [Google Scholar] [CrossRef] [PubMed]
- Yelpaala, K.; Ali, S.H. Multiple scales of diamond mining in Akwatia, Ghana: Addressing environmental and human development impact. Resour. Policy 2005, 30, 145–155. [Google Scholar] [CrossRef]
- Lujala, P.; Gleditsch, N.P.; Gilmore, E. A diamond curse? Civil war and a lootable resource. J. Confl. Resolut. 2005, 49, 538–562. Available online: https://www.jstor.org/stable/30045130 (accessed on 1 December 2021). [CrossRef]
- Le Billon, P. Diamond wars? Conflict diamonds and geographies of resource wars. Ann. Assoc. Am. Geogr. 2008, 98, 345–372. [Google Scholar] [CrossRef]
- Breeding, C.M.; Shigley, J.E. The “type” classification system of diamonds and its importance in gemology. Gems Gemol. 2009, 45, 96–111. [Google Scholar] [CrossRef]
- Roozeboom, B. Die heterogenen Gleichgewichte. Braunschweig 1901, 1, 180. [Google Scholar]
- Tamman, G. Crystal forms which are stable only at high pressures. Z. Phys. Chem. 1909, 69, 569. [Google Scholar]
- Rossini, F.D.; Jessup, R.S. Heat and free energy of formation of carbon dioxide and the transition between graphite and diamond. J. Res. Natl. Bur. Stand. US 1938, 21, 491. [Google Scholar] [CrossRef]
- Leipunskii, O.I. Synthetic diamonds. Usp. Khim. 1939, 8, 1519. [Google Scholar]
- Lijeblad, R. Equilibrium pressure for the different temperatures between graphite and diamonds. Ark. Kemi. 1955, 8, 423. [Google Scholar]
- Berman, R.; Simon, F. The graphite-diamond equilibrium. Z. Electrochem. 1955, 59, 333. [Google Scholar] [CrossRef]
- Liander, H.; Lundblad, E. Some observations on the synthesis of diamonds. Ark. Kemi. 1960, 16, 139–149. [Google Scholar]
- Lundblad, E.G. High-pressure synthesis of diamond in Sweden in 1953. AIP Conf. Proc. 1994, 309, 503–506. [Google Scholar] [CrossRef]
- Bundy, F.P.; Hall, H.T.; Strong, H.M.; Wentorf, R.H., Jr. Man-made diamonds. Nature 1955, 176, 51–55. [Google Scholar] [CrossRef]
- Bundy, F.P. Direct conversion of graphite to diamond in static pressure apparatus. J. Chem. Phys. 1963, 38, 631–643. [Google Scholar] [CrossRef]
- Bundy, F.P. The P, T phase and reaction diagram for elemental carbon, 1979. J. Geophys. Res. 1980, 85, 6930–6936. [Google Scholar] [CrossRef]
- Lazicki, A.; McGonegle, D.; Rygg, J.R.; Braun, D.G.; Swift, D.C.; Gorman, M.G.; Smith, R.F.; Heighway, P.G.; Higginbotham, A.; Suggit, M.J.; et al. Metastability of diamond ramp-compressed to 2 terapascals. Nature 2021, 589, 532–535. [Google Scholar] [CrossRef] [PubMed]
- Benedict, L.X.; Driver, K.P.; Hamel, S.; Militzer, B.; Qi, T.; Correra, A.A.; Saul, A.; Schwegler, E. A Multiphase equation of state for carbon addressing high pressures and temperatures. Phys. Rev. B 2014, 89, 224109. [Google Scholar] [CrossRef]
- Scientific American. How Mr. Hannay Made his Diamonds; Munn & Company: New York, NY, USA, 1880; p. 83. [Google Scholar]
- Moissan, H. Le Four Electrique; G. Steinheil: Paris, France, 1897. [Google Scholar]
- DeCarli, P.S.; Jamieson, J.C. Formation of diamond by explosive shock. Science 1961, 133, 1821–1822. [Google Scholar] [CrossRef] [PubMed]
- Angus, J.C.; Will, H.A.; Stanko, W.S. Growth of diamond seed crystals by vapor deposition. J. Appl. Phys. 1968, 39, 2915–2922. [Google Scholar] [CrossRef]
- Available online: https://statista.com (accessed on 1 December 2021).
- Available online: https://www.alliedmarketresearch.com (accessed on 1 December 2021).
- Expertmarketresearch. Global Industrial Diamond Market Outlook. 2020. Available online: https://www.expertmarketresearch.com/reports/industrial-diamond-market (accessed on 1 December 2021).
- German, R.M. Sintering Theory and Practice; Wiley-VCH: Weinheim, Germany, 1996; p. 568. [Google Scholar]
- German, R.M. Sintering: From empirical observations to scientific principles. In Chapter Ten—Sintering with External Pressure; Butterworth-Heinemann: Oxford, UK, 2014; pp. 305–354. [Google Scholar] [CrossRef]
- Antou, G.; Guyot, P.; Pradeilles, N.; Vandenhende, M.; Maître, A. Identification of densification mechanisms of pressure-assisted sintering: Application to hot pressing and spark plasma sintering of alumina. J. Mater. Sci. 2015, 50, 2327–2336. [Google Scholar] [CrossRef]
- Eversole, W.G. Synthesis of Diamond. US Patents 3,030,187, 3,030,188, 23 July 1958. [Google Scholar]
- Angus, J.C. Diamond synthesis by chemical vapor deposition: The Early years. Diam. Relat. Mater. 2014, 49, 77–86. [Google Scholar] [CrossRef]
- Angus, J.C.; Hayman, C. Low-pressure, metastable growth of diamond and “diamondlike” phases. Science 1988, 241, 913–921. [Google Scholar] [CrossRef]
- Schwander, M.; Partes, K. A review of diamond synthesis by CVD processes. Diam. Relat. Mater. 2011, 20, 1287–1301. [Google Scholar] [CrossRef]
- Tallaire, A. Ingénierie des Défauts Multidimensionnels dans le Diamant Synthétisé par Dépôt Chimique en Phase Vapeur Assisté par Plasma. Ph.D. Thesis, Matériaux, Université Paris 13—Sorbonne Paris Cité, Paris, France, 2017. [Google Scholar]
- Munir, Z.A.; Anselmi-Tamburini, U.; Ohyanagi, M. The effect of electric field and pressure on the synthesis and consolidation of materials: A review of the Spark Plasma Sintering method. J. Mater. Sci. 2006, 41, 763–777. [Google Scholar] [CrossRef]
- Kumar, D.B.; Babu, S.; Aravind Jerrni, K.M.; Joseph, N.; Jiss, A. Review of spark plasma sintering process. IOP Conf. Ser. Mater. Sci. Eng. 2020, 993, 012004. [Google Scholar] [CrossRef]
- Chaim, R. Densification mechanisms in Spark Plasma Sintering of nanocrystalline ceramics. Mater. Sci. Eng. A 2007, 443, 25–32. [Google Scholar] [CrossRef]
- Zhang, F.; Shen, J.; Sun, J.; Zhu, Y.Q.; Wang, G.; McCartney, G. Conversion of carbon nanotubes to diamond by spark plasma sintering. Carbon 2005, 43, 1254–1258. [Google Scholar] [CrossRef]
- Zhang, F.; Shen, J.; Sun, J.; McCartney, G. Direct synthesis of diamond from low purity carbon nanotubes. Carbon 2006, 44, 3113–3148. [Google Scholar] [CrossRef]
- Zhang, F.; Mihoc, C.; Ahmed, F.; Lathe, C.; Burkel, E. Thermal stability of carbon nanotubes, fullerene and graphite under spark plasma sintering. Chem. Phys. Lett. 2011, 510, 109–114. [Google Scholar] [CrossRef]
- Zhang, F.; Adam, M.; Ahmed, F.; Otterstein, E.; Burkel, E. Pulsed electric filed induces diamond synthesis from carbon nanotubes with solvent catalysts. Diam. Relat. Mater. 2011, 20, 853–858. [Google Scholar] [CrossRef]
- Zhang, F.; Ahmed, F.; Holzhüter, G.; Burkel, E. Growth of diamond from fullerene C60 by spark plasma sintering. J. Cryst. Growth 2012, 340, 1–5. [Google Scholar] [CrossRef]
- Zhang, F.; Ahmed, F.; Bednarcik, J.; Burkel, E. Diamond synthesis through the generation of plasma during spark plasma sintering. Phys. Status Solidi A 2012, 209, 2241–2246. [Google Scholar] [CrossRef]
- Zhang, F.; Fan, K.; Yu, J.; Saba, F.; Sun, J. Pulsed direct current field-induced thermal stability and phase transformation of nanodiamonds to carbon onions. RCS Adv. 2019, 9, 14360. [Google Scholar] [CrossRef]
- Zhang, F.; Burkel, E. Synhtesis of diamond using spark plasma sintering. In Sintering of Ceramics—New Emerging Techniques; Arunachalam, L., Ed.; InTech: London, UK, 2012; ISBN 978-953-51-0017-1. [Google Scholar] [CrossRef]
- Nickel, K.G.; Kraft, T.; Gogotsi, Y.G. Hydrothermal synthesis of diamond. In Handbook of Ceramics and Hard Materials; Wiley: New York, NY, USA, 2000; Chapter 3. [Google Scholar] [CrossRef]
- Szymanski, A.; Abgarowicz, E.; Bakon, A.; Niedbalska, A.; Salacinski, R.; Sentek, J. Diamond formed at low pressures and temperatures through liquid-phase hydrothermal synthesis. Diam. Relat. Mater. 1995, 4, 234–235. [Google Scholar] [CrossRef]
- Roy, R.; Ravichandran, D.; Ravindranathan, P.; Badzian, A. Evidence for hydrothermal growth of diamond in the C-H-O and C-H-O halogen system. J. Mater. Res. 1996, 11, 1164–1168. [Google Scholar] [CrossRef]
- Zhao, X.-Z.; Roy, R.; Cherian, K.A.; Badzian, A. Hydrothermal growth of diamond in metal-C-H2O systems. Nature 1997, 385, 513–515. [Google Scholar] [CrossRef]
- Szymanski, A. On the possibility of bulk large diamond single crystal synthesis with hydrothermal process. J. Miner. Soc. Korea 1997, 10, 18–32. [Google Scholar]
- Gogotsi, Y.G.; Kraft, T.; Nickel, K.; Zvanut, M.E. Hydrothermal behavior of diamond. Diam. Relat. Mater. 1998, 7, 1459–1465. [Google Scholar] [CrossRef]
- Yokosawa, K.; Korablov, S.; Tohji, K.; Yamasaki, N. The possibility of diamond sintering by hydrothermal hot pressing. AIP Conf. Proc. 2006, 833, 100–103. [Google Scholar] [CrossRef]
- Korablov, S.; Yokosawa, K.; Korablov, D.; Tohji, K.; Yamasaki, N. Hydrothermal formation of diamond from chlorinated organic compounds. Mater. Lett. 2006, 60, 3041–3044. [Google Scholar] [CrossRef]
- Korablov, S.; Yokosawa, K.; Saski, T.; Korablov, D.; Kawasaki, A.; Ioku, K.; Ishida, E.H.; Yamasaki, N. Synthesis of diamond from a chlorinated organic substance under hydrothermal conditions. J. Mater. Sci. 2007, 42, 7939–7949. [Google Scholar] [CrossRef]
- Roy, R.; Ravichandran, D.; Badzian, A.; Breval, E. Attempted hydrothermal synthesis of diamond by hydrolysis of β-SiC powder. Diam. Relat. Mater. 1996, 5, 973–976. [Google Scholar] [CrossRef]
- Gogotsi, Y.G.; Kofstad, P.; Yoshimura, M.; Nickel, K.G. Formation of sp3-bonded carbon upon hydrothermal treatment of SiC. Diam. Relat. Mater. 1996, 5, 151–162. [Google Scholar] [CrossRef]
- Gogotsi, Y.; Weiz, S.; Ersoy, D.A.; McNallan, M.J. Conversion of silicon carbide to crystalline diamond-structured carbon at ambient pressure. Nature 2001, 411, 283–287. [Google Scholar] [CrossRef]
- Basavalingu, B.; Byrappa, K.; Madhusudan, P. Hydrothermal synthesis of nanosized crystals of diamond under sub-natural conditions. J. Geol. Soc. India 2007, 69, 665–670. [Google Scholar]
- Zhang, W.; Fan, B.; Zhang, Y.; Fan, J. Hydrothermal synthesis of well crystallized C8 and diamond monocrystals and pH-controlled C8-Diamond phase transition. CrystEngComm 2017, 19, 1248–1252. [Google Scholar] [CrossRef]
- Néri, A.; Guignard, J.; Monnereau, M.; Bystricky, M.; Perillat, J.-P.; Andrault, A.; King, A.; Guignot, N.; Tenailleau, C.; Duployer, B.; et al. Reevaluation of metal interconnectivity in a partially molten silicate matrix using 3D microtomography. Phys. Earth Planet. Inter. 2020, 308, 106571. [Google Scholar] [CrossRef]
- Balima, F.; Belin, F.; Michau, D.; Viraphong, O.; Poulon-Quintin, A.; Chung, U.-C.; Dourfaye, A.; Largeteau, A. High pressure pulsed electric current activated equipment (HP-SPS) for material processing. Mater. Des. 2018, 139, 541–548. [Google Scholar] [CrossRef]
- Irifune, T.; Isobe, F.; Shinmei, T. A novel large-volume Kawai-type apparatus and its application to the synthesis of sintered bodies of nano-polycrystalline diamond. Phys. Earth Planet. Inter. 2014, 228, 255–261. [Google Scholar] [CrossRef]
- Zhan, G.D.; Moellendick, T.E.; Li, B.; Gooneratne, C. New ultra-strong and catalyst-free PDC cutting element technology. In Proceedings of the International Petroleum Techology Conference, Dhahran, Saudi Arabia, 13 January 2020. [Google Scholar] [CrossRef]
- Akaishi, M.; Yamaoka, S. Crystallization of diamond from C-H-O fluid under high-pressure and high-temperature conditions. J. Cryst. Growth 2000, 209, 999–1003. [Google Scholar] [CrossRef]
- Akaishi, M.; Shaji Kumar, M.D.; Kanda, H.; Yamaoka, S. Formation process of diamond from supercritical H2O-CO2 fluid under high pressure and high temperature conditions. Diam. Relat. Mater. 2000, 9, 1945–1950. [Google Scholar] [CrossRef]
- Akaishi, M.; Shaji Kumar, M.D.; Akaishi, M.; Yamaoka, S. Formation of diamond from supercritical H2O-CO2 fluid at high pressure and high temperature. J. Cryst. Growth 2000, 213, 203–206. [Google Scholar] [CrossRef]
- Sokol, A.G.; Pal’yanov, Y.N.; Pal’yanova, G.A.; Khokhryakov, A.F.; Borzdov, Y.M. Diamond and graphite crystallization from C-O-H fluids under high pressure and high temperature conditions. Diam. Relat. Mater. 2001, 10, 2131–2136. [Google Scholar] [CrossRef]
- Sokol, A.G.; Pal’yanov, Y.N.; Pal’yanova, G.A.; Tomilenko, A.A. Diamond crystallization in fluid and carbonate-fluid systems under mantle P-T conditions: 1. Fluid composition. Geochem. Int. 2004, 42, 830–838. [Google Scholar]
- Sokol, A.G.; Pal’yanov, Y.N. Diamond crystallization in fluid and carbonate-fluid systems under mantle P-T conditions: 2. An analytical review of experimental data. Geochem. Int. 2004, 42, 1018–1032. [Google Scholar]
- Naka, S.; Horii, K.; Takeda, Y.; Hanawa, T. Direct conversion of graphite to diamond under static pressure. Nature 1976, 259, 38–39. [Google Scholar] [CrossRef]
- Strong, H.M.; Hanneman, R.E. Crystallization of diamond and graphite. J. Chem. Phys. 1967, 46, 3668–3676. [Google Scholar] [CrossRef]
- Strong, H.M.; Chrenko, R.M. Further studies on diamond growth rates and physical properties of laboratory-made diamond. J. Chem. Phys. 1971, 75, 1838–1843. [Google Scholar] [CrossRef]
- Kennedy, C.S.; Kennedy, G.C. The equilibrium boundary between graphite and diamond. J. Geophys. Res. 1976, 81, 2467–2470. [Google Scholar] [CrossRef]
- Wakatsuki, M. New catalysts for synthesis of diamond. Jpn. J. Appl. Phys. 1966, 5, 337. [Google Scholar] [CrossRef]
- Kanda, H.; Akaishi, M.; Yamaoka, S. New catalysts for diamond growth under high pressure and high temperature. Appl. Phys. Lett. 1994, 65, 784–786. [Google Scholar] [CrossRef]
- Polyanov, Y.N.; Kupriyanov, I.N.; Borzdov, Y.M.; Surovtsev, N.V. Germanium: A new catalyst for diamond synthesis and a new optical active impurity in diamond. Sci. Rep. 2015, 5, 14789. [Google Scholar] [CrossRef]
- Polyanov, Y.N.; Kupriyanov, I.N.; Khokhryakov, A.; Borzdov, Y.M. High pressure crystallization and properties of diamond from magnesium-based catalyst. CrystEngComm 2017, 19, 4459–4475. [Google Scholar] [CrossRef]
- Katzman, H.; Libby, W.F. Sintered diamond compacts with a cobalt binder. Science 1971, 171, 1132–1134. [Google Scholar] [CrossRef]
- Liu, C.; Kou, Z.; He, D.; Chen, Y.; Wang, K.; Hui, B.; Zhang, R.; Wang, Y. Effect of removing internal residual metallic phases on wear resistance of polycrystalline diamond compacts. Int. J. Refract. Met. Hard Mater. 2012, 31, 187–191. [Google Scholar] [CrossRef]
- Westraadt, J.E.; Sigalas, I.; Neethling, J.H. Characterization of thermally degraded polycrystalline diamond. Int. J. Refract. Met. Hard Mater. 2015, 48, 286–292. [Google Scholar] [CrossRef]
- Gu, J.; Huang, K. Role of cobalt of polycrystalline diamond compact (PDC) in drilling process. Diam. Relat. Mater. 2016, 66, 98–101. [Google Scholar] [CrossRef]
- Sumiya, H.; Irifune, T.; Kurio, A.; Sakamoto, S.; Inoue, T. Microstructure features of polycrystalline diamond synthesized directly from graphite under static high pressure. J. Mater. Sci. 2004, 39, 445–450. [Google Scholar] [CrossRef]
- Sumiya, H.; Irifune, T. Hardness and deformation microstructures of nano-polycrystalline diamonds synthesized from various carbons under high pressure and high temperature. J. Mater. Res. 2007, 22, 2345–2351. [Google Scholar] [CrossRef]
- Sumiya, H.; Irifune, T. Microstructure and Mechanical Properties of High-Hardness nanopolycrystalline diamonds. SEI Tech. Rev. 2008, 66, 85–91. [Google Scholar]
- Le Guillou, C.; Brunet, F.; Irifune, T.; Ohfuji, H.; Rouzaud, J.-L. Nanodiamond nucleation below 2273 K at 15 GPa from carbons with different structural organizations. Carbon 2007, 45, 636–648. [Google Scholar] [CrossRef]
- Isobe, F.; Irifune, T.; Shinmei, T.; Suga, S.; Nishiyama, N.; Sumiya, H. Lowering P, T Boundary for Synthesis of pure nano-polycrystalline diamond. J. Phys. Conf. Ser. 2010, 215, 012136. [Google Scholar] [CrossRef]
- Xu, C.; He, D.; Wang, H.; Guan, J.; Liu, C.; Peng, F.; Wang, W.; Kou, Z.; He, K.; Yan, X.; et al. Nano-polycrystalline diamond formation under ultra-high pressure. Int. J. Refract. Met. Hard Mater. 2013, 36, 232–237. [Google Scholar] [CrossRef]
- Sumiya, H. Novel development of high-pressure synthetic diamonds “Ultra-hard Nano-polycrystalline Diamonds”. SEI Tech. Rev. 2012, 74, 15–23. [Google Scholar]
- Sumiya, H. Novel superhard nanopolycrystalline materials synthesized by direct conversion sintering under high pressure and high temperature. MRS Bull. 2017, 42, 729–733. [Google Scholar] [CrossRef]
- Sumiya, H.; Harano, K. Innovative Ultra-hard materials: Binderless nano-polycrystalline Diamond and nano-polycrystalline Cubic Boron Nitride. SEI Tech. Rev. 2016, 81, 21–26. [Google Scholar]
- Couvy, H.; Lahiri, D.; Chen, J.; Agarwal, A.; Sen, G. Nanohardness and Young’s modulus of nanocrystalline diamond. Scr. Mater. 2011, 64, 10419–11022. [Google Scholar] [CrossRef]
- Chang, Y.-Y.; Jacobsen, S.D.; Kimura, M.; Irifune, T.; Ohno, I. Elastic properties of transparent nano-polycrystalline diamond measured by GHz-ultrasonic interferometry and resonant sphere methods. Phys. Earth Planet. Inter. 2014, 228, 47–55. [Google Scholar] [CrossRef]
- Sumiya, H.; Harano, K.; Arimoto, K.; Kagi, H.; Odake, S.; Irifune, T. Optical characterisitcs of nano-polycrystalline diamond synthesized directly from graphite under high pressure and high temperature. Jpn. J. Appl. Phys. 2009, 48, 120206. [Google Scholar] [CrossRef]
- Lu, J.; Kou, Z.; Liu, T.; Yan, X.; Liu, F.; Ding, W.; Zhang, Q.; Zhang, L.; Liu, J.; He, D. Submicron binderless polycrystalline diamond sintering under ultra-high pressure. Diam. Relat. Mater. 2017, 77, 41–45. [Google Scholar] [CrossRef]
- Liu, J.; Zhan, G.; Wang, Q.; Yan, X.; Liu, F.; Wang, P.; Lei, L.; Peng, F.; Kou, Z.; He, D. Superstrong micro-grained polycrystalline diamond compact through work hardening under high pressure. Appl. Phys. Lett. 2018, 112, 061901. [Google Scholar] [CrossRef]
- Li, Q.; Zhan, G.; Li, D.; He, D.; Moellendick, T.E.; Gooneratne, C.P.; Alalsayednassir, A.G. Ultrastrong catalyst-free polycrystalline diamond. Sci. Rep. 2020, 10, 22020. [Google Scholar] [CrossRef]
- Osipov, V.Y.; Shakhov, F.M.; Bogdanov, K.V.; Takai, K.; Hayashi, T.; Treussart, F.; Baldycheva, A.; Hogan, B.T.; Jentgens, C. High-quality green-emitting nanodiamonds fabricated by HPHT sintering of polycrystalline shockwave diamonds. Nanoscale Res. Lett. 2020, 15, 209. [Google Scholar] [CrossRef]
- Dubrovinskaia, N.; Dubrovinsky, L.; Crichton, W.; Langenhorst, F.; Richter, A. Aggregated diamond nanorods, the densest and least compressible form of carbon. Appl. Phys. Lett. 2005, 87, 083106. [Google Scholar] [CrossRef]
- Dubrovinskaia, N.; Dubrovinsky, L.; Langenhorst, F.; Jacobsen, S.; Liebske, C. Nanocrystalline diamond synthesized from C60. Diam. Relat. Mater. 2005, 14, 16–22. [Google Scholar] [CrossRef]
- Irifune, T.; Ueda, C.; Ohshita, S.; Ohfuji, H.; Kunimoto, T.; Shinmei, T. Synthesis of nano-polycrystalline diamond from glassy carbon at pressures up to 25 GPa. High Press. Res. 2020, 40, 96–106. [Google Scholar] [CrossRef]
- Merlen, A.; Toulemonde, P.; Le Floch, S.; Montagnac, G.; Hammouda, T.; Marty, O.; San Miguel, A. High pressure-high temperature synthesis of diamond from single-wall pristine and iodine doped nanotube bundles. Carbon 2009, 47, 1643–1651. [Google Scholar] [CrossRef]
- Solopova, N.A.; Dubrovinskaia, N.; Dubrovinsky, L. Synthesis of nanocrystalline diamond from glassy carbon balls. J. Cryst. Growth 2015, 412, 54–59. [Google Scholar] [CrossRef]
- Dubrovinsky, L.; Dubrovinskaia, N.; Prakapenka, V.B.; Abakumov, A.M. Implementation of micro-ball nanodiamond anvils for high-pressure studies above 6 Mbar. Nat. Commun. 2012, 3, 1163. [Google Scholar] [CrossRef]
- Davydov, V.A.; Rakhmanina, A.V.; Agafonov, V.; Narymbetov, B.; Boudou, J.-P.; Szware, H. Conversion of polycyclic aromatic hydrocarbons to graphite and diamond at high pressures. Carbon 2004, 42, 261–269. [Google Scholar] [CrossRef]
- Kawamura, H.; Ohfuji, H. Nano-polycrystalline diamond synthesized through the decomposition of stearic acid. High Press. Res. 2020, 40, 162–174. [Google Scholar] [CrossRef]
- Park, S.; Abate, I.I.; Liu, J.; Wang, C.; Dahl, J.E.P.; Carlson, R.M.K.; Yang, L.; Prakapenka, V.B.; Greenberg, E.; Devereaux, T.P.; et al. Facile diamond synthesis from lower diamondoids. Sci. Adv. 2020, 6, eaay9405. [Google Scholar] [CrossRef]
- Irifune, T. A brief history of nano-polycrystalline diamond. High Press. Res. 2020, 40, 1–2. [Google Scholar] [CrossRef]
- Voronov, O.; Tompa, G.; Sadangi, R.; Kear, B.; Wilson, C.; Yan, P. Superhard Nanophase Cutter Materials for Rock Drilling Applications; Diamond Materials Inc.: Piscataway, NJ, USA, 2000. [Google Scholar] [CrossRef]
- Duanwei, H.; Chao, X.; Haikuo, W. Method for Preapring High-Performance Polycrystalline Diamond by Utilizing Hinge-Type Cubic Press. Patent CN103331129A, 2013. [Google Scholar]
- Shakhov, F.M.; Abyzov, A.M.; Takai, K. Boron doped diamond synthesized from detonation nanodiamond in C-O-H fluid at high pressure and high temperature. J. Solid State Chem. 2017, 256, 72–92. [Google Scholar] [CrossRef]
- Fischbach, D.B. The kinetics and mechanism of graphitization. In The Chemistry and Physics of Carbon 7; Walker, P.L., Ed.; Marcel Dekker: New York, NY, USA, 1971; pp. 1–154. [Google Scholar]
- Bonijoly, M.; Oberlin, M.; Oberlin, A. A possible mechanism for natural graphite formation. Int. J. Coal Geol. 1982, 1, 283–312. [Google Scholar] [CrossRef]
- Wentorf, R.H., Jr. The behavior of some carbonaceous materials at very high pressures and high temperatures. J. Phys. Chemistry 1965, 9, 3063–3069. [Google Scholar] [CrossRef]
- Ekimov, E.A.; Kudryavtsev, O.S.; Mordinova, E.; Lebededv, O.I.; Vlasov, I.I. High-pressure synthesis of nanodiamonds from adamantane: Myth or Reality. Chem. Nanomater. 2018, 4, 269–273. [Google Scholar] [CrossRef]
- Bergmann, O.R.; Bailey, N.F.; Coverly, H.B. Polishing performance of polycrystalline diamond produced by explosive shock wave synthesis. Metallography 1982, 15, 121–139. [Google Scholar] [CrossRef]
- Schrand, A.M.; Ciftan Hens, S.A.; Shenderova, O.A. Nanodiamond particles: Properties and perspectives for bioapplications. Crit. Rev. Solid State Mater. Sci. 2009, 34, 18–74. [Google Scholar] [CrossRef]
- Erksine, D.J.; Nellis, W.J. Shock-induced martensitic phase transformation of orientated graphite to diamond. Nature 1991, 349, 317–319. [Google Scholar] [CrossRef]
- Kurdyumov, A.V.; Britun, V.F.; Yarosh, V.V.; Danilenko, A.I.; Zelyavski, V.B. The influence of the shock compression conditions on the graphite transformation into lonsdaleite and diamond. J. Superhard Mater. 2012, 34, 19–27. [Google Scholar] [CrossRef]
- Kraus, D.; Ravasio, A.; Gauthier, M.; Gericke, D.O.; Vorberger, J.; Frydrych, S.; Helfrich, J.; Fletcher, L.B.; Schaumen, G.; Nagler, B.; et al. Nanosecond formation of diamond and lonsdaleite by shock compression of graphite. Nat. Commun. 2016, 7, 10970. [Google Scholar] [CrossRef]
- Volt, T.J.; Gupta, Y.M. Graphite to diamond transformation under shock compression: Role of orientational order. J. Appl. Phys. 2019, 125, 245902. [Google Scholar] [CrossRef]
- Epanchintsev, O.G.; Zubchenko, A.S.; Korneyev, A.E.; Simonov, V.A. Highly-efficient shock-wave diamond synthesis from fullerenes. J. Phys. Chem. Solids 1997, 58, 1785–1788. [Google Scholar] [CrossRef]
- Donnet, J.B.; Fousson, E.; Wang, T.K.; Samirant, M.; Baras, C.; Pontier Johnson, M. Dynamic synthesis of diamonds. Diam. Relat. Mater. 2000, 9, 887–892. [Google Scholar] [CrossRef]
- Kenkmann, T.; Hornemann, U.; Stöffler, D. Experimental shock synthesis of diamonds in a graphite gneiss. Meteorit. Planet. Sci. 2005, 40, 1299–1310. [Google Scholar] [CrossRef]
- Titov, V.M.; Anisichkin, V.F.; Mal’kov, I.Y. Synthesis of ultradispersed diamond in detonation waves. Fizika Goreniya I Vzeryva 1989, 25, 117–126. [Google Scholar] [CrossRef]
- Pichot, V.; Comet, M.; Fousson, E.; Baras, C.; Senger, A.; Le Normand, F.; Spitzer, D. An efficient purification method for detonation nanodiamonds. Diam. Relat. Mater. 2008, 17, 13–22. [Google Scholar] [CrossRef]
- Pichot, V.; Risse, B.; Schnell, F.; Mory, J.; Sptizer, D. Understanding ultrafine nanodiamond formation using nanostructured explosives. Sci. Rep. 2013, 3, 2159. [Google Scholar] [CrossRef] [PubMed]
- Pichot, V.; Muller, O.; Seve, A.; Yvon, A.; Merlat, L.; Sptizer, D. Optical properties of functionalized nanodiamonds. Sci. Rep. 2017, 7, 14086. [Google Scholar] [CrossRef]
- Batsanov, S.S.; Osavchuk, A.N.; Naumov, S.P.; Efimov, A.E.; Mendis, B.G.; Apperley, D.C.; Batsanov, A.S. Synthesis and properties of hydrogen-free detonation diamond. Propellants Explos. Pyrotech. 2015, 40, 39–45. [Google Scholar] [CrossRef]
- Akira, B.S.; Takamatsu, M.; Akashi, T. Shock compression synthesis of diamonds. Adv. Mater. 1994, 6, 346–354. [Google Scholar] [CrossRef]
- Zousman, B.; Levinson, O. Pure nanodiamonds produced by laser-assisted technique. In Nanodiamond; Williams, O., Ed.; RSC Nanoscience & Nanotechnology: London, UK, 2014; Chapter 5. [Google Scholar]
- Khachatryan, A.; Aloyan, S.G.; May, P.W.; Sargsyan, R.; Khachatryan, V.A.; Baghdasaryan, V.S. Graphite-to-diamond transformation induced by ultrasound cavitation. Diam. Relat. Mater. 2008, 17, 931–936. [Google Scholar] [CrossRef]
- Narayan, J.; Bhaumik, A.; Sachan, R.; Haque, A.; Gupta, S.; Pant, P. Direct conversion of carbon nanofibers and nanotubes into diamonds nanofibers and the subsequent growth of large-size diamonds. Nanoscale 2019, 11, 2238–2248. [Google Scholar] [CrossRef]
- Gao, Y.; Ma, Y.; An, Q.; Levitas, V.; Zhang, Y.; Feng, B.; Chaudhuri, J.; Goddard III, W.A. Shear driven formation of nano-diamonds at sub-gigapascals and 300 K. Carbon 2019, 146, 364–368. [Google Scholar] [CrossRef]
- Fromentin, S. Resistivity of Carbon, Diamond. In The Physics Factbook; Elert, G., Ed.; 2004; Available online: https://hypertextbook.com (accessed on 1 December 2021).
- Bormashov, V.S.; Tarelkin, S.A.; Buga, S.G.; Kuznetsov, M.S.; Terentiev, S.A.; Semenov, A.N.; Blank, V.D. Electrical properties of the high quality boron-doped single crystal diamonds grown by the temperature gradient method. Diam. Relat. Mater. 2013, 35, 19–23. [Google Scholar] [CrossRef]
- Ekimov, E.A.; Sidorov, V.A.; Bauer, E.D.; Mel’nik, N.N.; Curro, N.J.; Thompson, J.D.; Stishov, S.M. Superconductivity in diamond. Nature 2004, 428, 542–545. [Google Scholar] [CrossRef] [PubMed]
- Burgemeister, E.A. Thermal conductivity of natural diamond between 320 and 450 K. Phys. B+C 1978, 93, 165–179. [Google Scholar] [CrossRef]
- Olson, J.R.; Pohl, R.O.; Vandersande, J.W.; Zoltan, A.; Anthony, T.R.; Banholzer, W.F. Thermal conductivity of diamond between 170 and 1200 K and the isotope effect. Phys. Rev. B 1993, 47, 14850. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.; Kuo, P.K.; Thomas, R.L.; Anthony, T.R.; Banholzer, W.F. Thermal conductivity of isotopically modified single crystal diamond. Phys. Rev. Lett. 1993, 70, 3764. [Google Scholar] [CrossRef] [PubMed]
- Zaitseez, A.M. Optical properties of diamond. In A Data Handbook; Springer: Berlin/Heidelberg, Germany, 2001. [Google Scholar]
- Gicquel, A.; Hassouni, K.; Silva, F.; Achard, J. CVD diamond film: From growth to applications. Curr. Appl. Phys. 2001, 1, 479–496. [Google Scholar] [CrossRef]
- Smith, R.L.; Sandland, G.E. An accurate method of determining the hardness of metals, with particular reference to those of a high degree of hardness. Proc. Inst. Mech. Eng. 1922, 102, 623–641. [Google Scholar] [CrossRef]
- Knoop, F.; Peters, C.G.; Emerson, W.B. A sensitive pyramidal-diamond tool for indentation measurements. J. Res. Natl. Bur. Stand. 1939, 23, 39–61. [Google Scholar] [CrossRef]
- Fischer-Cripps, A.C. Nanoindentation, 3rd ed.; Springer: Berlin/Heidelberg, Germany, 2004. [Google Scholar]
- Migliori, A.; Sarrao, J.L.; Vissher, W.M.; Bell, T.M.; Lei, M.; Fisk, Z.; Leisure, R.G. Resonant ultraousnd spectroscopic techniques for measurement of the elastic moduli of solids. Physica B 1993, 183, 1–24. [Google Scholar] [CrossRef]
- Ben Ghorbal, G.; Tricoteaux, A.; Thuault, A.; Louis, G.; Chicot, D. Comparison of conventional Knoop and Vickers hardness of ceramic materials. J. Eur. Ceram. Soc. 2017, 37, 2531–2535. [Google Scholar] [CrossRef]
- Migliori, A.; Ledbetter, H.; Leisure, R.G.; Pantea, C.; Betts, J.B. Diamond’s elastic stiffness from 332 K to 10 K. J. Appl. Phys. 2008, 104, 053512. [Google Scholar] [CrossRef]
- Wang, S.-F.; Hsu, Y.-F.; Pu, J.-C.; Sung, J.C.; Hwa, L.G. Determination of acoustic wave velocities and elastic properties for diamond and other hard materials. Mater. Chem. Phys. 2004, 85, 432–437. [Google Scholar] [CrossRef]
- Tanigaki, K.; Ogi, H.; Sumiya, H.; Kusakabe, K.; Nakamura, N.; Hirao, M.; Ledbetter, H. Observation of higher stiffness in nanopolycrystalline diamond than monocrystal diamond. Nat. Commun. 2013, 4, 2343. [Google Scholar] [CrossRef] [PubMed]
- Gasc, J.; Wang, Y.; Yu, T.; Benea, I.C.; Rosczyk, B.R.; Shinmei, T.; Irifune, T. High-pressure, high-temperature plastic deformation of sintered diamonds. Diam. Relat. Mater. 2015, 59, 95–103. [Google Scholar] [CrossRef]
- Wang, Y.; Shi, F.; Ohfuji, H.; Gasc, J.; Nishiyama, N.; Yu, T.; Officer, T.; Shinmey, T.; Irifune, T. Strength and plastic deformation of polycrystalline diamond composite. High Press. Res. 2019, 40, 35–53. [Google Scholar] [CrossRef]
- Wang, Y.; Shi, F.; Gasc, J.; Ohfuji, H.; Wen, B.; Yu, T.; Officer, T.; Nishiyama, N.; Shinmei, T.; Irifune, T. Plastic deformation and strengthening mechanisms of nanopolycrystalline diamonds. ACS Nano 2021, 15, 8283–8294. [Google Scholar] [CrossRef]
- Ischii, R.; Fukuta, R.; Ischikawa, F.; Matsushita, M.; Ohfuji, H.; Shinmei, T.; Irifune, T.; Funato, M.; Kawakami, Y. Deep-ultraviolet near band-edge emissions from nano-polycrystalline diamond. High Press. Res. 2019, 40, 140–147. [Google Scholar] [CrossRef]
- Dzielgielewski, P.; Mathon, O.; Kantor, I.; Pascarelli, S.; Shinmei, T.; Irifune, T.; Antowicz, J. High pressure atomic structure of Zr-Cu metallic glass via EXAFS spectroscopy and molecular dynamics simulations. High Press. Res. 2019, 40, 54–64. [Google Scholar] [CrossRef]
- Rosa, A.D.; Mathon, O.; Torchio, R.; Jacobs, J.; Pasternak, S.; Irifune, T.; Pascarelli, S. Nano-polycrystalline diamond anvils: Key devices for XAS at extreme conditions: Their use, scientific impact, present status and future needs. High Press. Res. 2019, 40, 65–81. [Google Scholar] [CrossRef]
- Elbers, M.; Sternemann, C.; Julius, K.; Paulus, M.; Surmeier, G.; König, N.; Nase, J.; Bolle, J.; Wagner, R.; Irifune, T.; et al. Pressure stability of the first hydration shell of yttrium in aqueous YCl3 solution. High Press. Res. 2020, 40, 194–204. [Google Scholar] [CrossRef]
- Kousa, M.; Iwasaki, S.; Ishimatsu, N.; Kawamura, N.; Nomura, R.; Kakizawa, S.; Mizumaki, M.; Sumiya, H.; Irifune, T. Element-selective elastic properties of Fe65Ni35 Invar alloy and Fe72Pt28 alloy studied by extended X-ray absorption fine structure. High Press. Res. 2019, 40, 130–139. [Google Scholar] [CrossRef]
- Nataf, L.; Baudelet, F.; Polian, A.; Jonane, I.; Anspoks, A.; Kuzmin, A.; Irifune, T. Recent progress in high pressure X-ray absorption spectroscopy at the ODE beamline. High Press. Res. 2020, 40, 82–87. [Google Scholar] [CrossRef]
- Komatsu, K.; Klotz, S.; Nakano, S.; Machida, S.; Hattori, T.; Sano-Furukawa, A.; Yamashita, K.; Irifune, T. Developments of nano-polycrystalline diamond anvil cells for neutron diffraction experiments. High Press. Res. 2020, 40, 184–193. [Google Scholar] [CrossRef]
- Yamashita, K.; Komatsu, K.; Klotz, S.; Fernandez-Diaz, M.T.; Irifune, T.; Sugiyama, K.; Kawamata, T.; Kagi, H. A nano-polycrystalline diamond anvil cell with bulk metallic glass cylinder for single-crystal neutron diffraction. High Press. Res. 2019, 40, 88–95. [Google Scholar] [CrossRef]
- Kuramochi, K.; Ishimatsu, N.; Sakai, T.; Kawamura, N.; Irifune, T. An applicaton of NPD to double-stage diamond anvil cells: XAS spectra of rhenium metal under high pressures above 300 GPa. High Press. Res. 2019, 40, 119–129. [Google Scholar] [CrossRef]
- Sakai, T.; Yagi, T.; Takeda, R.; Hamatani, T.; Nakamoto, Y.; Kadobayashi, H.; Mimori, H.; Kawaguchi, S.I.; Hirao, N.; Ohishi, Y.; et al. Conical support for double stage diamond anvil apparatus. High Press. Appar. 2019, 40, 12–21. [Google Scholar] [CrossRef]
- Yagi, T.; Sakai, T.; Kadobayashi, H.; Irifune, T. Review: High pressure generation techniques beyond the limit of conventional diamond anvils. High Press. Res. 2019, 40, 148–161. [Google Scholar] [CrossRef]
- Kono, Y.; Kenney-Benson, C.; Shen, G. Opposed type double stage cell for Mbar pressure experiment with large sample volume. High Press. Res. 2020, 40, 175–183. [Google Scholar] [CrossRef]
- Barreda-Argüeso, J.A.; Gonzalez, J.; Valiente, R.; Irifune, T.; Rodriguez, F. Exploiting optical properties of nanopolycrystalline diamond in high pressure experiments. High Press. Res. 2019, 40, 107–118. [Google Scholar] [CrossRef]
- Guan, S.; Peng, F.; Liang, H.; Fan, C.; Tan, L.; Wang, Z.; Zhang, Y.; Zhang, Y.H.; He, D. Fragmentation and stress diversification in diamond powder under high pressure. J. Appl. Phys. 2018, 124, 215902. [Google Scholar] [CrossRef]
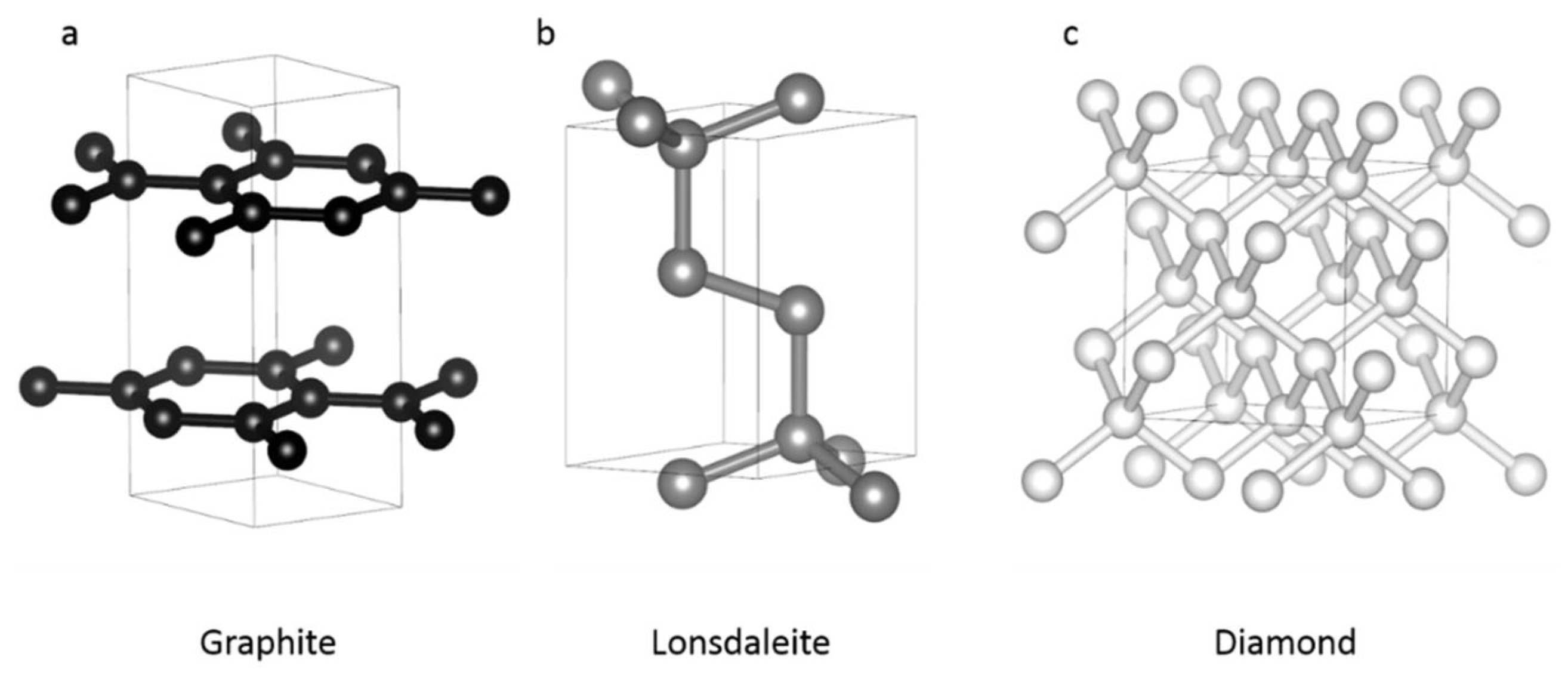

| Occurrence | Lattice | Space Group | Unit Cell (Å) | Density (g/cm3) | Hybridization | |
|---|---|---|---|---|---|---|
| Graphite | Ambient conditions | Hexagonal | (194) P63/mmc | a = b = 2.456 c = 6.696 1 | 2.26 | sp2 |
| Lonsdaleite | HPHT | Hexagonal | (194) P63/mmc | a = b = 2.51 c = 4.12 2 | 4 3.30–5 3.52 | sp3 |
| Diamond | HPHT | Cubic | (227) | a = 3.56679 3 | 3.51 | sp3 |
| Study | Apparatus | Starting Material (SM) | Pressure (GPa) | Temperature (°C) | Time (Min) | Yield (%) | Remarks | ||
|---|---|---|---|---|---|---|---|---|---|
| Phase | Type | Grains Size (μm) | |||||||
| Bundy, 1963 | Belt-type press | Gr, SC, Gr, aC | R | - | 11.8–15 | up to 3900 | ~10−7 | - | 1st direct transition reported |
| Naka et al., 1976 | - | Gr | R | - | 14 | up to 3500 | 4 | 60 | 10–20 μm diamond grain recovered |
| Iriune et al., 2003 | Kawai type MAA | Gr | R | - | 12–25 | 2300–2500 | - | up to 100 | 10–200 nm grain size in NPD |
| Davydov et al., 2004 | Toroïd Press | PAH | P | - | 8 | 1500 | 1 | 60 | 5–40 μm monocrystals, 100% yield for T > 1350 °C, and graphitization step |
| Sumiya et al., 2004 | Kawai type MAA | Gr | R | - | 18 | 2300–2700 | 0.2–170 | 100 | Microstructure: homogenous fine structure + lamellar structure |
| Dubrovinskaia et al., 2005 | Kawai type MAA | C60 | P | - | 20 | 2200 | - | 100 | Diamonds nanorods aggregated 5–20 nm Ø, >1 mm length, compact body of 1.8 mm Ø and 3 mm high |
| Dubrovinskaia et al., 2005 | Kawai type MAA | C60 | P | - | 13–20 | 27–2127 | <90 | up to 100 | 5–12 nm grain size of diamond, lonsdaleite often present |
| aC | 20 | 2027 | |||||||
| Gr | 20–25 | 2177–2327 | |||||||
| D | 20 | 2027 | |||||||
| Le Guillou et al., 20,007 | Kawai type MAA | QAS | - | - | 15 | 1500 | 15–60 | up to 100 | Transforamtion path highly dependent on the nature of the precursors, especially their crystallinity. Lonsdaleite can be present |
| CB | 30–100 × 10−3 | 1500–1700–1900 | |||||||
| HTCB | 30–100 × 10−3 | 1500–1700 | |||||||
| pGr | 0.5–1 | 1500–1700–1900 | |||||||
| HOPG | >1 | 1700 | |||||||
| Sumiya and Irifune, 2007, 2008 | Kawai type MAA | Gr | R | - | 15–28 | 2300–2500 | 0.2–10 | 100 | Lower hardness for other C precursors than graphite |
| aC | P | - | 18–21 | 1800–2000 | 10–20 | ||||
| GC | P | - | 18–21 | 2000–22,500 | 6–20 | ||||
| C60 | P | - | 18 | 1800–2000 | 30–35 | ||||
| Merlen et al., 2009 | DAC, PE, MAA | SWCNT +/− I | P | - | 12–25 | 25–1527 | 60–120 | up to 100 | Graphitization step |
| Isobe et al., 2010 | Kawai type MAA | Gr | R | - | 12–14 | 2200–2600 | 5–30 | up to 100 | Minimum condition for NPD. Higher pressure means lower T to complete transformation. |
| Couvy et al., 2011 | Kawai type MAA | Gr | R | - | 19 | 2400 | 0.5 | 100 | - |
| Dubrovinsky et al., 2012 | Kawai type MAA | GC | B | 20–50 | 20 | 1927 | <15 | 100 | Nano polycrystalline (50 nm) balls of 15–40 microns, presence of NaCl or Na2CO3 to make microballs. |
| He et al., 2013 | Hinge-type cubic press | μD | P | - | 8–20 | 1400–2500 | 1–30 | no need | - |
| Xu et al., 2013 | Hinge-type cubic press | Gr +/− milled | P | 5–20 | 16 | 2500 | 1–10 | 100 | Reaction at lower T with ball-milled Gr |
| Chang et al., 2014 | Kawai type MAA | Gr | R | - | 15 | 2327 | 20 | 100 | NPD shaped in anvil, cyliner or sphere |
| Solopova et al., 2015 | Kawai type MAA | GC | B | 20–50 | 18 | 1850–2000 | 1–5 | up to 100 | Nano polycrystalline balls of 15–40 microns, presence of NaCl or Na2CO3 to make microballs |
| Lu et al., 2017 | Hinge-type cubic press | μD | P | 0.5 | 14 | 2000 | 1 | no need | Cylinder of 3 mm Ø & high at the end. Some backtransformation in graphite. Final grain size of 170 nm |
| Liu et al., 2018 | Hinge-type cubic press | μD | P | 8–12 | 14 | 1000–2000 | - | no need | - |
| Zhan et al., 2020 | Hinge-type cubic press | μD | P | 8–12 | 14 | up to 1900 | - | no need | Cylinder 11 mm Ø, 6 mm high. Not transparent at all. There may be pores and/or cracks |
| Li et al., 2020 | Hinge-type cubic press | μD | P | 8–12 | 16 | 2300 | 10 | no need | Same size body as above, 10-micron final grain size |
| Osipov et al., 2020 | Toroïd press | nD | P | 25 × 10−3 | 7 | 1300 | 0.2 | no need | Purification + sintering using ethanol at HPHT |
| Kawamura et al., 2020 | Kawai type MAA | C18H36O2 | P | - | 10–13-17 | 600–1600 | 5–180 | up to 100 | Graphitization step, 10 nm grain at the end, control fO2 in some exp and remove water during exp |
| Park et al., 2020 | DAC | C10H16,C14H20,C18H24 | P | 0.5–2 × 10−3 | 5–22 | 627–2727 | ~10−7 | up to 100 | Lower transition than with pure C source. Direct transformation, no graphitization step |
| Irifune et al., 2020 | Kawai type MAA | GC | R | - | 15–25 | 1700–2300 | 20 | up to 100 | Grain size dependence on P,T. P,T limits to keep nanograin |
| Mechanical Properties | |
|---|---|
| Poisson’s ratio | 0.1 |
| Young’s modulus (GPa) | 1050 |
| Fracture toughness K1C (MPa.m0.5) | |
| Natural single crystal | 5 |
| Synthetic polycrystalline (NPDs) | 8.5 |
| Fracture strength (GPa) | |
| Natural single crystal | 2.5–3.0 |
| Synthetic polycrystalline (NPDs) | 0.2 to 1.1 |
| Transverse rupture strength (GPa) | |
| Natural single crystal | 1.0–2.0 |
| Synthetic polycrystalline (binder) | 2.5 up to 400 °C, 0.5 at 500 °C |
| Synthetic polycrystalline (NPDs) | 3.0 up to 1000 °C |
| Flank wear width/cutting length (μm/m) | |
| Natural single crystal | 11.25 |
| Synthetic polycrystalline (binder) | 0.50 |
| Synthetic polycrystalline (NPDs) | 0.16 |
| Knoop hardness (GPa) | |
| Natural single crystal | 70 to 120 according to orientation |
| Synthetic polycrystalline (binder) | 50–80 according to binder content |
| Synthetic polycrystalline (binderless) | |
| From graphite or diamond | 125–145 |
| From other C-precursors | 80–100 |
| Friction coefficient (μ) | 0.05 to 0.15 according to orientation |
| Optical properties | |
| Color (see type classification) | colorless to various |
| Transparency | (X-ray) UV to IR |
| Absorption | 2.5–6 μm |
| Fluorescence | UV |
| Refractive index | 2.417 (0.044 dispersion) |
| Electrical properties | |
| Conductivity (Ω.m) | |
| Most natural diamonds | Insulator, 1011 to 1018 |
| Type IIb (boron-doped, natural and synthetic) | Semiconductor, superconductor at 4 K |
| Electronic gap | 5.5 ev |
| Dielectric constant | 5.58 at 35 GHz |
| Thermal properties | |
| Expansion coefficient (10−6 × K−1) | 1.0 at 600 °C, 4.4 at 1300 °C |
| Conductivity (W/(m.K)) | |
| Natural single crystal | 2200 |
| Synthetic (12C enriched) | up to 3200 |
| Stability at 1 bar | |
| 700 °C in air | |
| up to 1600 °C in Ar |
| Techniques | Pressure (GPa) | Temperature (°C) | Synthesis | Yield | Sintering | Size of Sintered Objects |
|---|---|---|---|---|---|---|
| CVD | 10−8–10−9 | 800–1500 | ✓ | very high | ✓ | few mm3 |
| SPS | 0.1 | 1500–1600 | ✓ | low | ✕ | - |
| Hydrothermal | ||||||
| LP-LT(MT) | 0.1–2 | 300–1200 | ✓ | very low | ✕ | - |
| HP–HT(MT) | 3–8 | 1000–2000 | ✓ | low | ✕ | - |
| HPHT | ✓ | |||||
| Catalyst | 4–5 | 1400–1500 | ✓ | high | ✓ | cm3 |
| Binderless | 15–25 | 2000–2500 | ✓ | very high | ✓ | cm3 |
| Shock | >30 | >2000 | ✓ | very high | ✕ | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guignard, J.; Prakasam, M.; Largeteau, A. A Review of Binderless Polycrystalline Diamonds: Focus on the High-Pressure–High-Temperature Sintering Process. Materials 2022, 15, 2198. https://doi.org/10.3390/ma15062198
Guignard J, Prakasam M, Largeteau A. A Review of Binderless Polycrystalline Diamonds: Focus on the High-Pressure–High-Temperature Sintering Process. Materials. 2022; 15(6):2198. https://doi.org/10.3390/ma15062198
Chicago/Turabian StyleGuignard, Jérémy, Mythili Prakasam, and Alain Largeteau. 2022. "A Review of Binderless Polycrystalline Diamonds: Focus on the High-Pressure–High-Temperature Sintering Process" Materials 15, no. 6: 2198. https://doi.org/10.3390/ma15062198
APA StyleGuignard, J., Prakasam, M., & Largeteau, A. (2022). A Review of Binderless Polycrystalline Diamonds: Focus on the High-Pressure–High-Temperature Sintering Process. Materials, 15(6), 2198. https://doi.org/10.3390/ma15062198








