Tribological Characteristics of a-C:H:Si and a-C:H:SiOx Coatings Tested in Simulated Body Fluid and Protein Environment
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
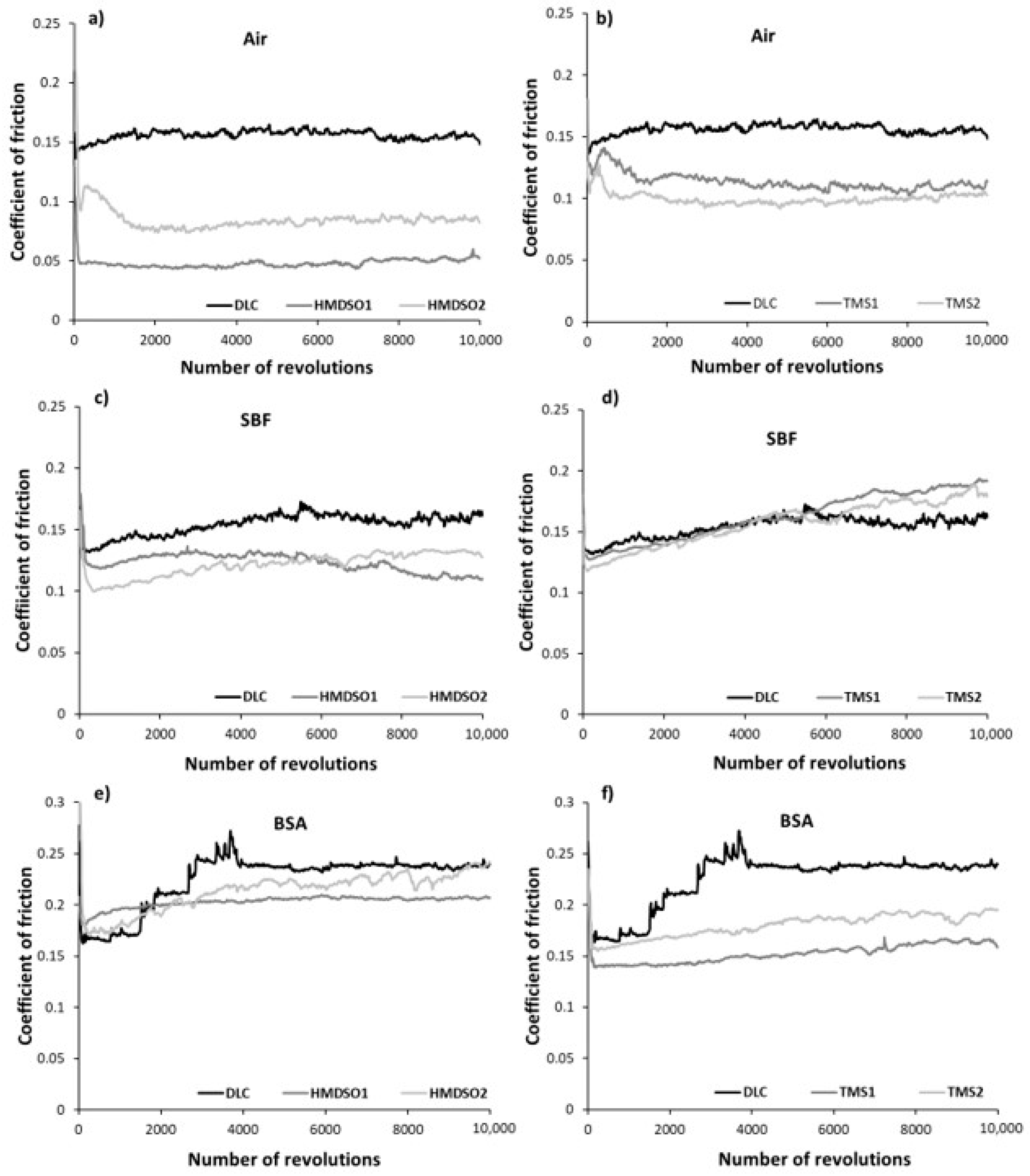
3.1. Frictional Behavior
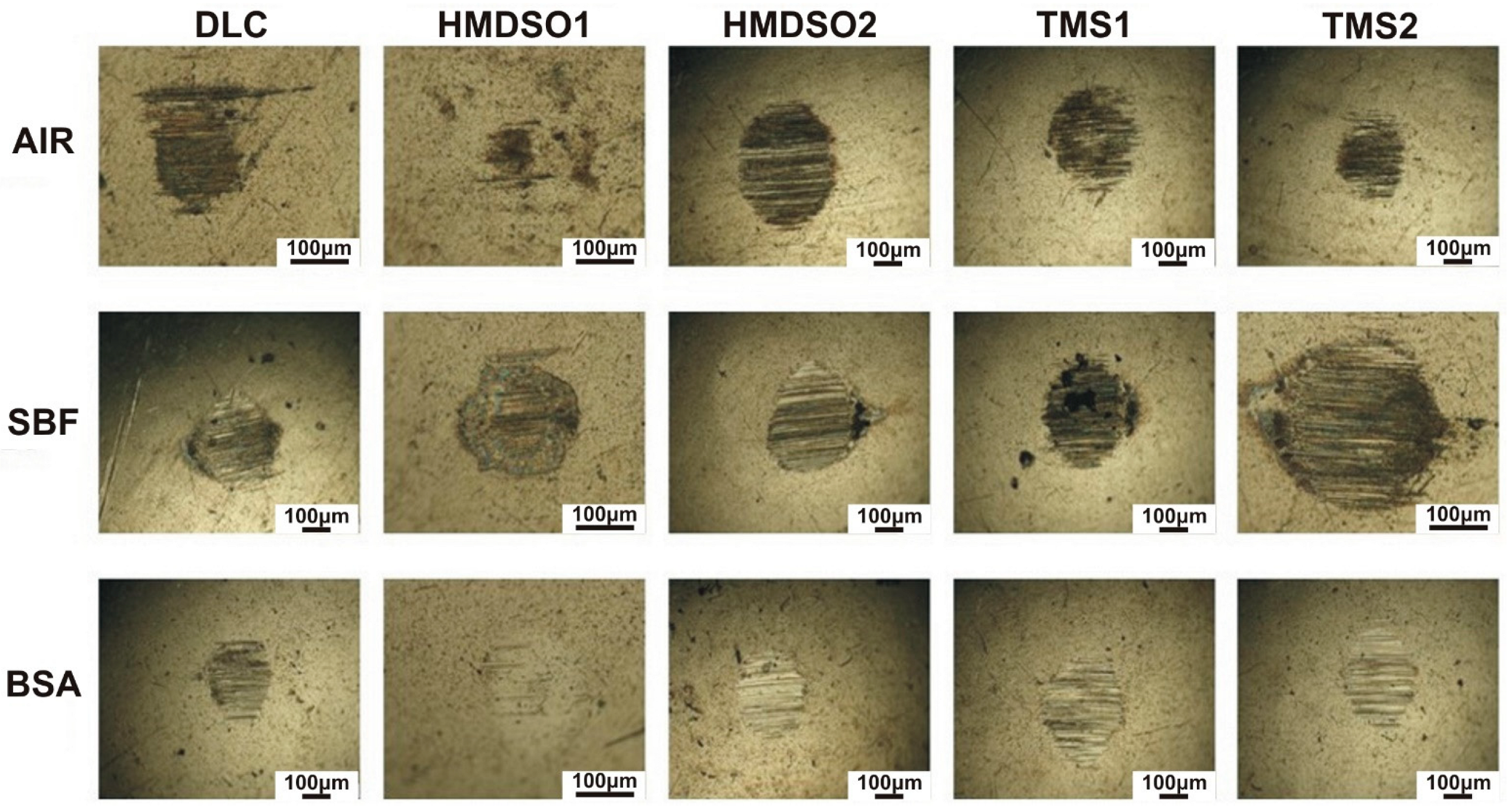
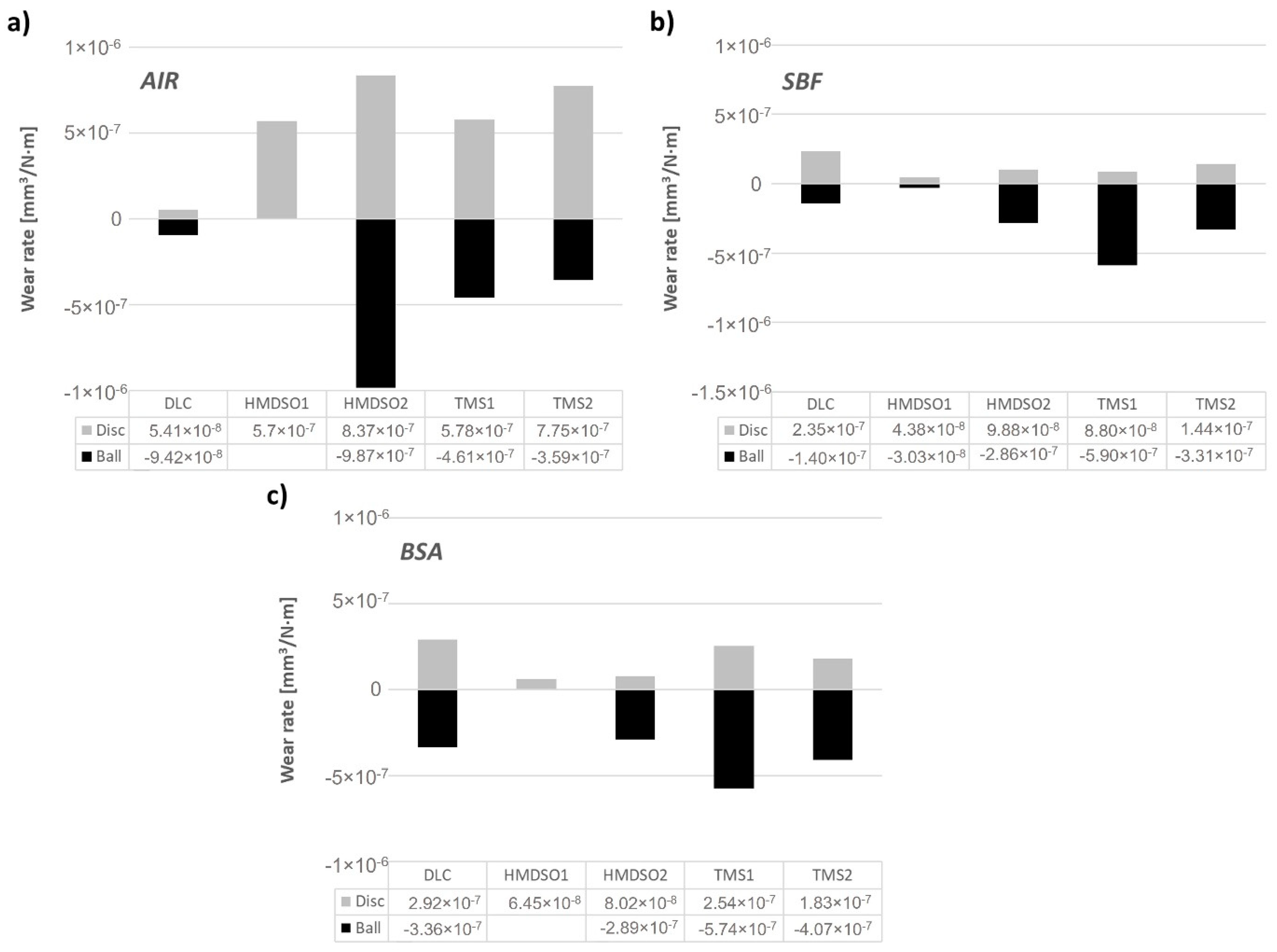
3.2. Wear Rate Analysis
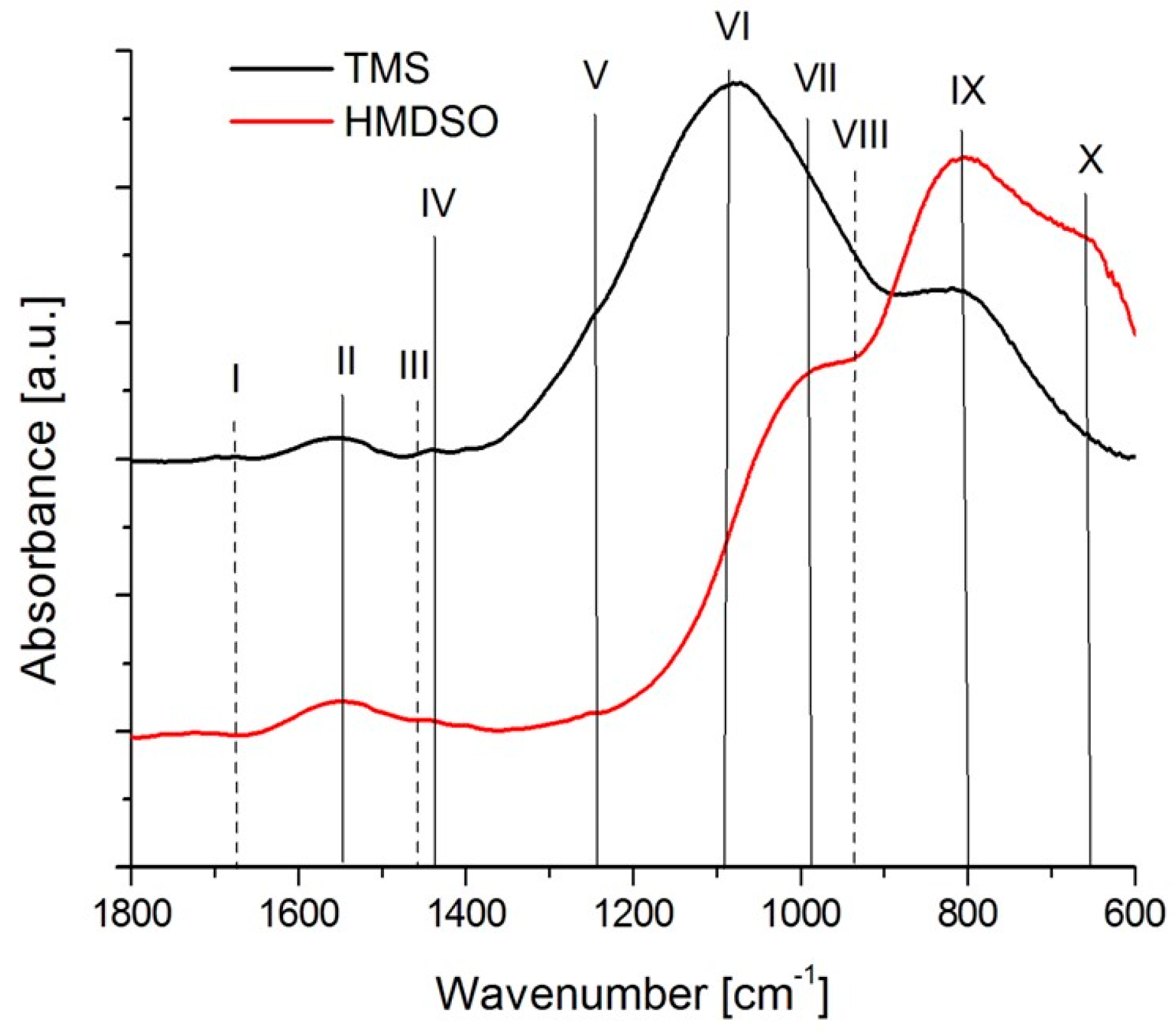
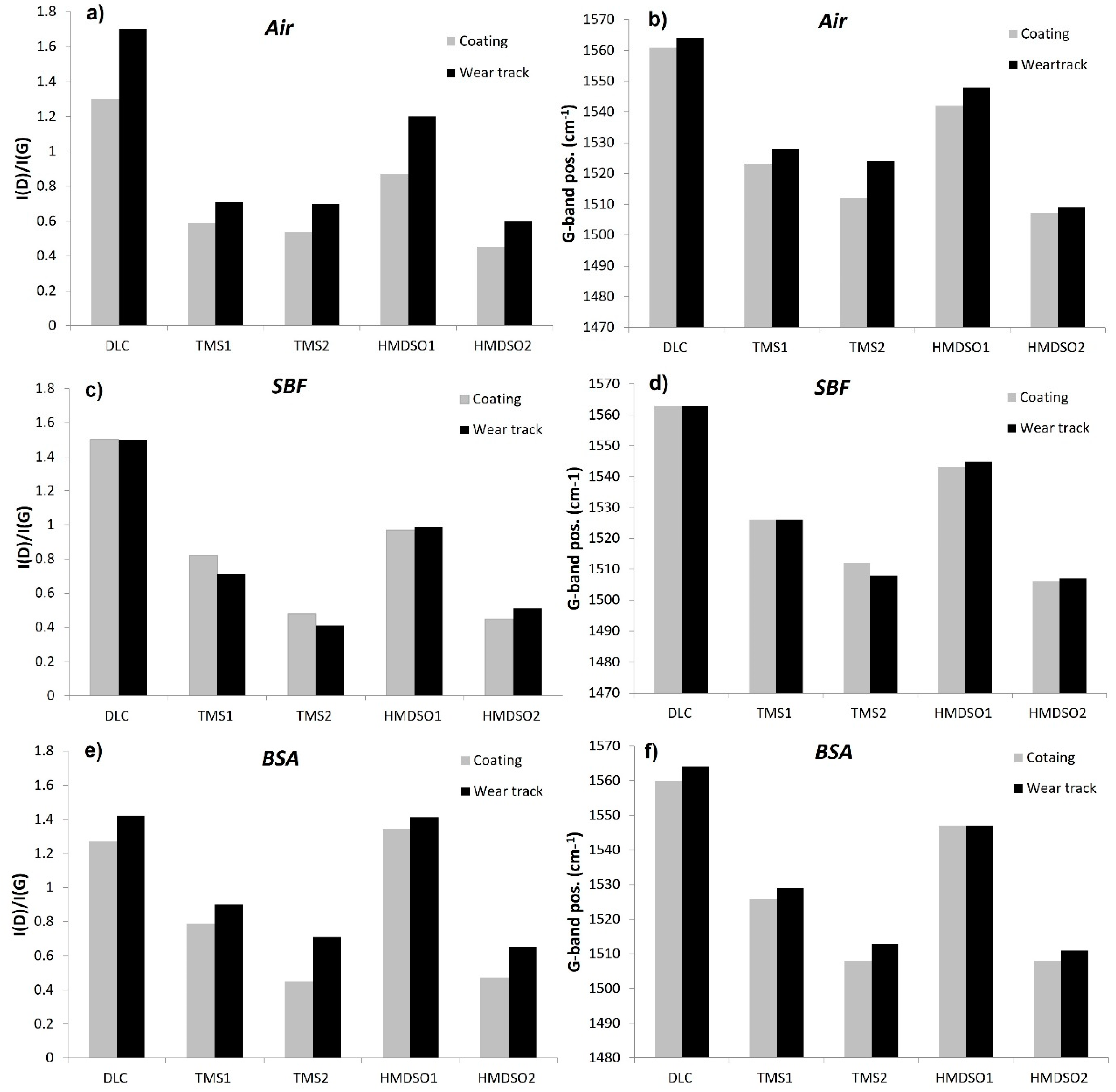
3.3. Chemical Characterization
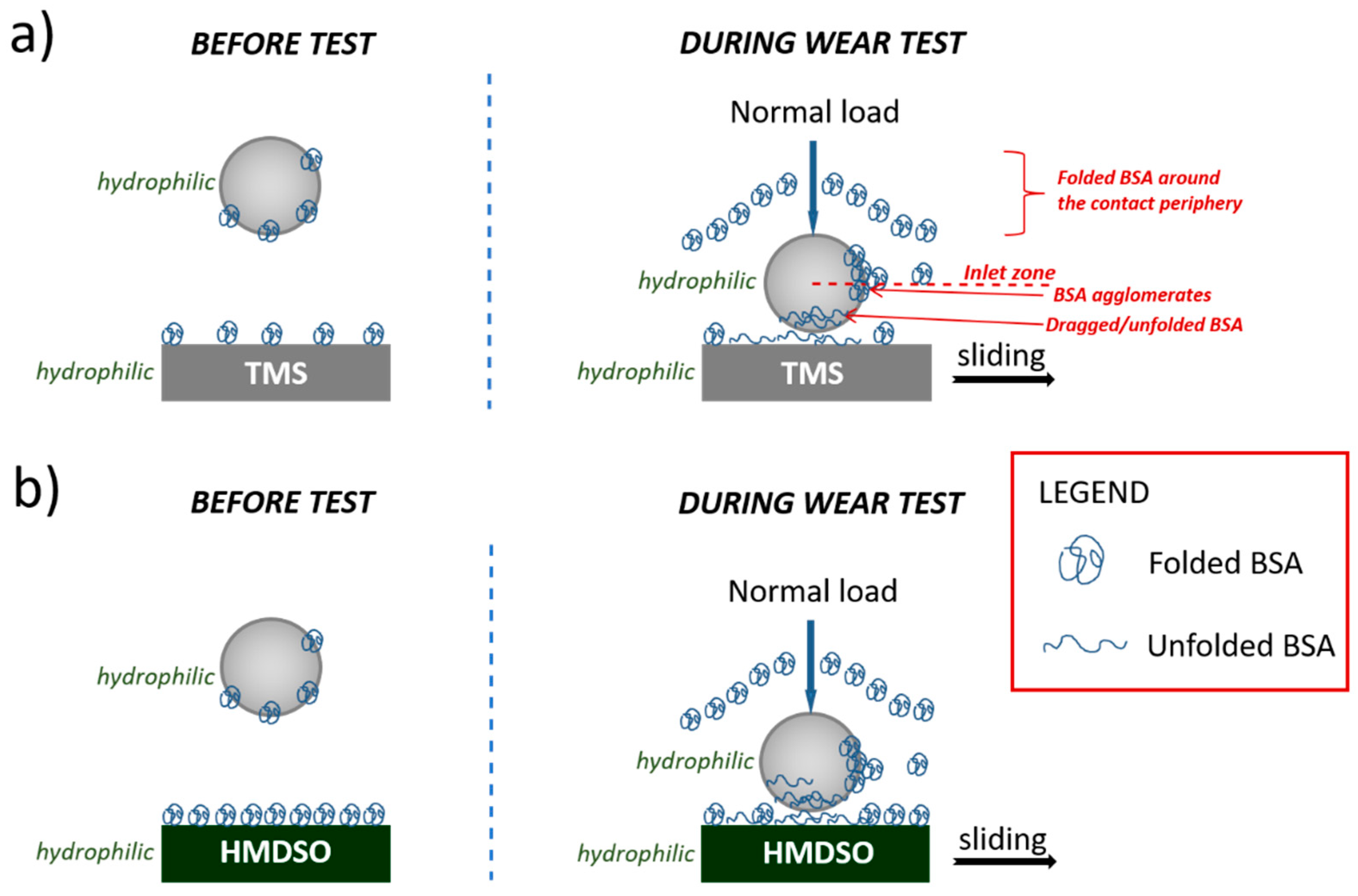
3.4. Model of Interaction between BSA and Silicon-Incorporated DLC Films
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Shtansky, D.V.; Roy, M. Surface engineering for biotribological application. In Surface Engineering for Enhanced Performance against Wear; Roy, M., Ed.; Springer: Vienna, Austria, 2013. [Google Scholar]
- Merola, M.; Affatato, S. Materials for Hip Prostheses: A Review of Wear and Loading Considerations. Materials 2019, 12, 495. [Google Scholar] [CrossRef] [PubMed]
- Bauer, S.; Schmuki, P.; von der Mark, K.; Park, J. Engineering biocompatible implant surfaces: Part I: Materials and surfaces. Prog. Mater. Sci. 2013, 58, 261–326. [Google Scholar] [CrossRef]
- Hesketh, J.; Meng, Q.; Dowson, D.; Neville, A. Biotribocorrosion of metal-on-metal hip replacements: How surface degradation can influence metal ion formation. Tribol. Int. 2013, 65, 128–137. [Google Scholar] [CrossRef]
- Kenney, C.; Dick, S.; Lea, J.; Liu, J.; Ebraheim, N.A. A systematic review of the causes of failure of Revision Total Hip Arthroplasty. J. Orthop. 2019, 16, 393–395. [Google Scholar] [CrossRef]
- Khatod, M.; Cafri, G.; Inacio, M.; Schepps, A.L.; Paxton, E.W.; Bini, S.A. Revision total hip arthoplasty: Factors associated with Re-revision surgery. J. Bone Jt. Surg. Am. 2015, 97, 359–366. [Google Scholar] [CrossRef]
- Ramos, B.; Saito, E.; Trava-Airoldi, V.; Lobo, A.O.; Marciano, F. Diamond-like carbon electrochemical corrosion resistance by addition of nanocrystalline diamond particles for biomedical applications. Surf. Coat. Technol. 2014, 259, 732–736. [Google Scholar] [CrossRef]
- Capote, G.; Ramírez, M.; da Silva, P.; Lugo, D.; Trava-Airoldi, V. Improvement of the properties and the adherence of DLC coatings deposited using a modified pulsed-DC PECVD technique and an additional cathode. Surf. Coat. Technol. 2016, 308, 70–79. [Google Scholar] [CrossRef]
- Gotzmann, G.; Beckmann, J.; Scholz, B.; Herrmann, U.; Wetzel, C. Low-energy electron-beam modification of DLC coatings reduces cell count while maintaining biocompatibility. Surf. Coat. Technol. 2018, 336, 34–38. [Google Scholar] [CrossRef]
- Jamesh, M.I.; Li, P.; Bilek, M.; Boxman, R.; McKenzie, D.R.; Chu, P.K. Evaluation of corrosion resistance and cytocompatibility of graded metal carbon film on Ti and NiTi prepared by hybrid cathodic arc/glow discharge plasma-assisted chemical vapor deposition. Corros. Sci. 2015, 97, 126–138. [Google Scholar] [CrossRef]
- Hang, R.; Ma, S.; Chu, P.K. Corrosion behavior of DLC-coated NiTi alloy in the presence of serum proteins. Diam. Relat. Mater. 2010, 19, 1230–1234. [Google Scholar] [CrossRef]
- Gerke, L.; Schauer, J.-C.; Pohl, M.; Winter, J. Adhesion and elasticity of plasma deposited wear resistant a-C:H coatings on nickel–titanium. Surf. Coat. Technol. 2009, 203, 3214–3218. [Google Scholar] [CrossRef]
- Erdemir, A.; Donnet, C. Tribology of diamond-like carbon films: Recent progress and future prospects. J. Phys. D Appl. Phys. 2006, 39, 311–327. [Google Scholar] [CrossRef]
- Batory, D.; Szymanski, W.; Clapa, M. Mechanical and tribological properties of gradient a-C:H/Ti coatings. Mater. Sci. Pol. 2013, 31, 415–423. [Google Scholar] [CrossRef]
- Bociąga, D.; Sobczyk-Guzenda, A.; Szymanski, W.; Jedrzejczak, A.; Jastrzebska, A.; Olejnik, A.; Jastrzębski, K. properties, chemical analysis and evaluation of antimicrobial response of Si-DLC coatings fabricated on AISI 316 LVM substrate by a multi-target DC-RF magnetron sputtering method for potential biomedical applications. Appl. Surf. Sci. 2017, 417, 23–33. [Google Scholar] [CrossRef]
- Ishihara, M.; Kosaka, T.; Nakamura, T.; Tsugawa, K.; Hasegawa, M.; Kokai, F.; Koga, Y. Antibacterial activity of fluorine incorporated DLC films. Diam. Relat. Mater. 2006, 15, 1011–1014. [Google Scholar] [CrossRef]
- Zhang, T.F.; Wan, Z.X.; Ding, J.C.; Zhang, S.; Wang, Q.M.; Kim, K.H. Microstructure and high-temperature tribological properties of Si-doped hydrogenated diamond-like carbon films. Appl. Surf. Sci. 2018, 435, 963–973. [Google Scholar] [CrossRef]
- Wu, Y.; Zhou, S.; Zhao, W.; Ouyang, L. Comparative corrosion resistance properties between (Cu, Ce)-DLC and Ti co-doped (Cu, Ce)/Ti-DLC films prepared via magnetron sputtering method. Chem. Phys. Lett. 2018, 705, 50–58. [Google Scholar] [CrossRef]
- Batory, D.; Gorzedowski, J.; Rajchel, B.; Szymanski, W.; Kolodziejczyk, L. Silver implanted diamond-like carbon coatings. Vacuum 2014, 110, 78–86. [Google Scholar] [CrossRef]
- Batory, D.; Jedrzejczak, A.; Kaczorowski, W.; Szymanski, W.; Kolodziejczyk, L.; Clapa, M.; Niedzielski, P. Influence of the process parameters on the characteristics of silicon-incorporated a-C:H:SiOx coatings. Surf. Coat. Technol. 2015, 271, 112–118. [Google Scholar] [CrossRef]
- Batory, D.; Jedrzejczak, A.; Szymanski, W.; Niedzielski, P.; Fijalkowski, M.; Louda, P.; Kotela, I.; Hromadka, M.; Musil, J. Mechanical characterization of a-C:H:SiOx coatings synthesized using radio-frequency plasma-assisted chemical vapor deposition metod. Thin Solid Film. 2015, 590, 299–305. [Google Scholar] [CrossRef]
- Batory, D.; Jedrzejczak, A.; Kaczorowski, W.; Kolodziejczyk, L.; Burnat, B. The effect of Si incorporation on the corrosion resistance of a-C:H:SiOx coatings. Diam. Relat. Mater. 2016, 67, 1–7. [Google Scholar] [CrossRef]
- Namus, R.; Rainforth, W. Influence of protein adsorption on tribocorrosion behaviour of CoCrMo biomedical-grade alloys. Tribol. Int. 2020, 150, 106364. [Google Scholar] [CrossRef]
- Wang, Z.; Yan, Y.; Su, Y.; Qiao, L. Effect of proteins on the surface microstructure evolution of a CoCrMo alloy in bio-tribocorrosion processes. Colloids Surf. B 2016, 145, 176–184. [Google Scholar] [CrossRef] [PubMed]
- Dimah, M.K.; Devesa Albeza, F.; Amigó Borrás, V.; Igual Muñoz, A. Study of the biotribocorrosion behaviour of titanium biomedical alloys in simulated body fluids by electrochemical techniques. Wear 2012, 294, 409–418. [Google Scholar] [CrossRef]
- Hang, R.; Qi, Y. A study of biotribological behavior of DLC coatings and its influence to human serum albumin. Diam. Relat. Mater. 2010, 19, 62–66. [Google Scholar] [CrossRef]
- Alemón, B.; Flores, M.; Ramírez, W.; Huegel, J.C.; Broitman, E. Tribocorrosion behavior and ions release of CoCrMo alloy coated with aTiAlVCN/CNx multilayer in simulated body fluid plus bovine serum albumin. Tribol. Int. 2015, 81, 159–168. [Google Scholar] [CrossRef]
- Escudeiro, A.; Polcar, T.; Cavaleiro, A. Adsorption of bovine serum albumin on Zr co-sputtered a-C(:H) films: Implication wear behavior. J. Mech. Behav. Biomed. Mater. 2014, 39, 316–327. [Google Scholar] [CrossRef][Green Version]
- Scholes, C. A frictional study of total hip joint replacements. Phys. Med. Biol. 2000, 45, 3721. [Google Scholar] [CrossRef]
- Ahlroos, T.; Saikko, V. Wear of prosthetic joint materials in various lubricants. Wear 1997, 211, 113–119. [Google Scholar] [CrossRef]
- Carapeto, A.P.; Serro, A.P.; Nunes, B.M.F.; Martins, M.C.L.; Todorovic, S.; Duarte, M.T.; André, V.; Colaço, R.; Saramago, B. Characterization of two DLC coatings for joint prosthesis: The role of albumin on the tribological behavior. Surf. Coat. Technol. 2010, 204, 3451–3458. [Google Scholar] [CrossRef]
- Jedrzejczak, A.; Kolodziejczyk, L.; Szymanski, W.; Piwonski, I.; Cichomski, M.; Kisielewska, A.; Dudek, M.; Batory, D. Friction and wear of a-C:H:SiOx coatings in combination with AISI 316L and ZrO2 counterbodies. Tribol. Int. 2017, 112, 155–162. [Google Scholar] [CrossRef]
- Jedrzejczak, A.; Batory, D.; Dominik, M.; Smietana, M.; Cichomski, M.; Szymanski, W.; Bystrzycka, E.; Prowizor, M.; Kozlowski, W.; Dudek, M. Carbon coatings with high concentrations of silicon deposited by RF PECVD method at relatively high self-bias. Surf. Coat. Technol. 2017, 329, 212–217. [Google Scholar] [CrossRef]
- Sonnenfeld, A.; Tun, T.M.; Zajíčková, L.; Kozlov, K.V.; Wagner, H.-E.; Behnke, J.F.; Hippler, R. Deposition process based on organosilicon precursors in dielectric barrier discharges at atmospheric pressure—A comparison. Plasmas Polym. 2001, 6, 237–266. [Google Scholar] [CrossRef]
- Hauert, R.; Müller, U. An overview on tailored tribological and biological behavior of diamond-like carbon. Diam. Relat. Mater. 2003, 12, 171–177. [Google Scholar] [CrossRef]
- Roy, R.K.; Choi, H.-W.; Park, S.-J.; Lee, K.-R. Surface energy of the plasma treated Si incorporated diamond-like carbon films. Diam. Relat. Mater. 2007, 16, 1732–1738. [Google Scholar] [CrossRef]
- Wu, B.; Deng, Q.; Leng, Y.; Wang, C.; Huang, N. Characterization of adsorption and lubrication of synovial fluid proteins and HA on DLC joint bearings surface. Surf. Coat. Technol. 2017, 320, 320–332. [Google Scholar] [CrossRef]
- Lambert, J.B. Introduction to Organic Spectroscopy; Macmillan Publ.: New York, NY, USA, 1987; Chapter 7. [Google Scholar]
- Vorsina, I.A.; Grigorieva, T.F.; Barinova, A.P.; Lyakhov, N.Z. Mechanochemical interaction of silicon dioxide with organic acids. Russ. J. Bioorg. Chem. 2013, 39, 765–770. [Google Scholar]
- Das, L.; Guleria, A.; Adhikari, S. Aqueous phase one-pot green synthesis of SnSe nanosheets in a protein matrix: Negligible cytotoxicity and room temperature emission in the visible region. RSC Adv. 2015, 75, 61390–61397. [Google Scholar] [CrossRef]
- Lee Smith, A. Analysis of Silicones; Wiley: New York, NY, USA, 1974; Chapter 10. [Google Scholar]
- Arkles, B. Infrared Analysis of Organosilicon Compounds: Spectra-Structure Correlations, Reprinted from Silicon Compounds: Silanes & Silicones; Gelest. Inc.: Morrisville, PA, USA, 2013. [Google Scholar]
- Siliprandi, R.A.; Zanini, S.; Grimoldi, E.; Fumagalli, F.; Barni, R.; Riccardi, C. Atmospheric pressure plasma discharge for polysiloxane thin films deposition and comparison with low pressure process. Plasma Chem. Plasma Process. 2011, 31, 353–372. [Google Scholar] [CrossRef]
- Jing, S.-Y.; Lee, H.-J.; Choi, C.K. Bond Structure on Si-O-C Composite films with a low dielectric constant deposited by using inductively coupled plasma chemical vapor deposition. J. Korean Phys. Soc. 2002, 41, 769–773. [Google Scholar]
- Casiraghi, C.; Piazza, F.; Ferrari, A.C.; Grambole, D.; Robertson, J. Boding in hydrogenated diamond-like carbon by Raman spectroscopy. Diam. Relat. Mater. 2005, 14, 1098–1102. [Google Scholar] [CrossRef]
- Papakonstantinou, P.; Zhao, J.F.; Lemoine, P.; McAdams, E.T.; McLaughlin, J.A. The effects of Si incorporation on the electrochemical and nanomechanical properties of DLC thin films. Diam. Relat. Mater. 2002, 11, 1074–1080. [Google Scholar] [CrossRef]
- Camargo, S.; Neto, A.B.; Santos, R.; Freire, F.; Carius, R.; Finger, F. Improved high-temperature stability of Si-incorporated a-C:H films. Diam. Relat. Mater. 1998, 7, 1155–1162. [Google Scholar] [CrossRef]
- Wu, W.-J.; Hon, M.-H. Thermal stability of diamond-like carbon films with added silicon. Surf. Coat. Technol. 1999, 111, 134–140. [Google Scholar] [CrossRef]
- Dwyer-Joyce, R.S. Predicting the abrasive wear of ball bearings by lubricant debris. Wear 1999, 233–235, 692–701. [Google Scholar] [CrossRef]
- Myant, C.; Cann, P. On the matter of synovial fluid lubrication: Implications for Metal-on-Metal hip tribology. J. Mech. Behav. Biomed. Mater. 2014, 34, 338–348. [Google Scholar]



| Coating | Pressure [Pa] | Gas Mixture and Flow Ratio | Bias | Deposition Time | Si Content [at.%] | O Content [at.%] |
|---|---|---|---|---|---|---|
| DLC | 20 | CH4 | −800 | 37 | - | - |
| TMS1 | CH4/TMS-18/4 | 17 | 5.0 | - | ||
| TMS2 | CH4/TMS-17/8 | 15 | 10.0 | - | ||
| HMDSO1 | CH4/HMDSO-18/3 | 28 | 0.45 | 9.3 | ||
| HMDSO2 | CH4/HMDSO-16/12 | 21 | 5.3 | 10.3 |
| No. | Wavenumber [cm−1] | Vibrating Mode [cm−1] | Ref. | TMS | HMDSO |
|---|---|---|---|---|---|
| I | 1680 | C=O (stretch) | [38] | +(weak) | - |
| II | 1550 | C=O (stretch) | [39] | + | + |
| III | 1460 | CH3 (deformation) | [38] | + | + |
| IV | 1440 | C-OH/Si-OH (bend) | [38,40,41] | + | - |
| V | 1250 | Si-CH3 (bend) | [41,42] | + | + |
| VI | 1080 | Si-O-C/Si-CH2-Si (stretch) | [43,44] | +(strong) | +(weak) |
| VII | 998 | Si-O(stretch) | [41,42,43] | + | +(strong) |
| VIII | 940 | Si-OH/Si-O (stretch) | [41,42] | + | + |
| IX | 798 | Si-C (stretch) | [41,42] | +(strong) | + |
| X | 660 | Si-O (bend) | [38,40,43] | + | - |
| No. | Wavenumber [cm−1] | Vibrating Mode [cm−1] | Ref. | TMS SBF | TMS BSA | HMDSO SBF | HMDSO BSA |
|---|---|---|---|---|---|---|---|
| 1 | 1750 | C=O (stretch) | [38] | + | - | - | - |
| 2 | 1680 | C=O (stretch) | [38] | + | + | - | - |
| 3 | 1659 | C=O, C-N (stretch) (amid I) | [38,40] | - | + | - | +(strong) |
| 4 | 1630 | C=C (stretch) | [38] | + | - | - | - |
| 5 | 1550 | C=O (stretch) | [39] | +(strong) | + | + | + |
| 6 | 1540 | N-H, C-N (deformation) (amid II) | [38,40] | - | + | - | +(strong) |
| 7 | 1460 | CH3 (deformation) | [38] | + | + | + | + |
| 8 | 1440 | C-OH/Si-OH (Bend) | [38,41,42] | + | + | + | + |
| 9 | 1390 | C-N (stretch) (amide III band) | [38,41] | - | + | - | +(strong) |
| 10 | 1370 | SiOCOCH3 | [41,42] | +(weak) | +(weak) | - | - |
| 11 | 1305 | C=O (stretch) | [38] | +(strong) | - | - | - |
| 12 | 1299 | C-N (stretch) (amide III) | [38,40] | - | - | - | + |
| 13 | 1250 | Si-CH3 (bend) | [41,42] | + | + | + | + |
| 14 | 1240 | C-N (stretch) | [38,40] | - | + | - | + |
| 15 | 1213 | C-O-C (stretch) | [38] | + | - | - | - |
| 16 | 1170 | C-C-N (stretch) | [38] | - | + | - | + |
| 17 | 1080 | Si-O-C/R1-Si-O-Si-R2 | [43,44] | + | + | + | + |
| 18 | 1057 | (stretch) | [41,42,43] | +(strong) | + | + | + |
| 19 | 1038 | Si-O/Si-CH2-Si (stretch) | [41,42,43] | +(strong) | + | - | - |
| 20 | 1000 | Si-O-Si (stretch) | [41,42,43] | + | + | + | + |
| 21 | 920 | Si-O(stretch) | [38] | + | - | - | - |
| 22 | 857 | CH=CH (deformation) | [44] | + | + | + | + |
| 23 | 802 | Si-O-C (stretch) | [41,42] | + | + | + | + |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jedrzejczak, A.; Szymanski, W.; Kolodziejczyk, L.; Sobczyk-Guzenda, A.; Kaczorowski, W.; Grabarczyk, J.; Niedzielski, P.; Kolodziejczyk, A.; Batory, D. Tribological Characteristics of a-C:H:Si and a-C:H:SiOx Coatings Tested in Simulated Body Fluid and Protein Environment. Materials 2022, 15, 2082. https://doi.org/10.3390/ma15062082
Jedrzejczak A, Szymanski W, Kolodziejczyk L, Sobczyk-Guzenda A, Kaczorowski W, Grabarczyk J, Niedzielski P, Kolodziejczyk A, Batory D. Tribological Characteristics of a-C:H:Si and a-C:H:SiOx Coatings Tested in Simulated Body Fluid and Protein Environment. Materials. 2022; 15(6):2082. https://doi.org/10.3390/ma15062082
Chicago/Turabian StyleJedrzejczak, Anna, Witold Szymanski, Lukasz Kolodziejczyk, Anna Sobczyk-Guzenda, Witold Kaczorowski, Jacek Grabarczyk, Piotr Niedzielski, Agnieszka Kolodziejczyk, and Damian Batory. 2022. "Tribological Characteristics of a-C:H:Si and a-C:H:SiOx Coatings Tested in Simulated Body Fluid and Protein Environment" Materials 15, no. 6: 2082. https://doi.org/10.3390/ma15062082
APA StyleJedrzejczak, A., Szymanski, W., Kolodziejczyk, L., Sobczyk-Guzenda, A., Kaczorowski, W., Grabarczyk, J., Niedzielski, P., Kolodziejczyk, A., & Batory, D. (2022). Tribological Characteristics of a-C:H:Si and a-C:H:SiOx Coatings Tested in Simulated Body Fluid and Protein Environment. Materials, 15(6), 2082. https://doi.org/10.3390/ma15062082










