Application of ANN for Analysis of Hole Accuracy and Drilling Temperature When Drilling CFRP/Ti Alloy Stacks
Abstract
1. Introduction
2. Materials and Methods
2.1. Workpiece Material and Cutting Tool
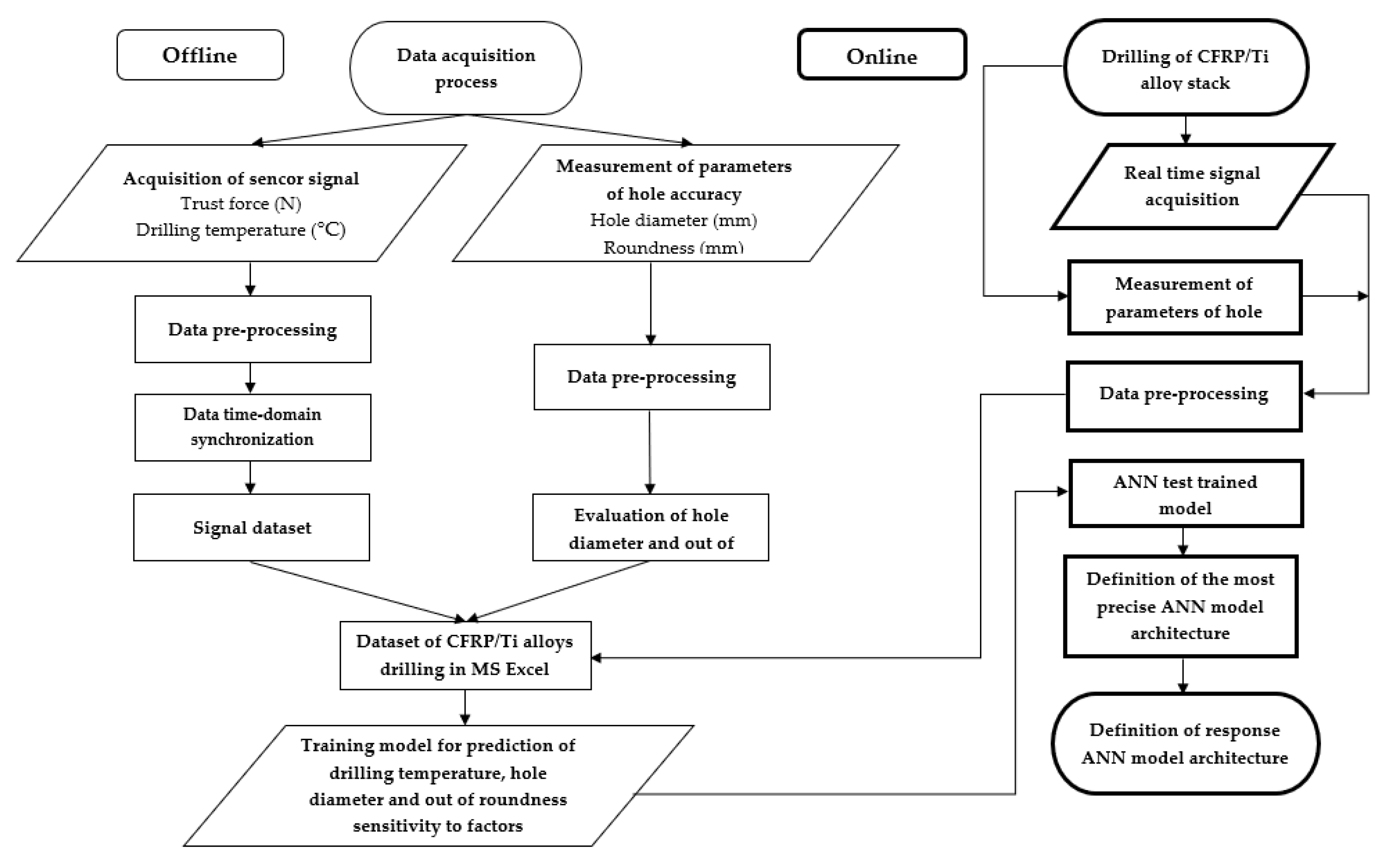
2.2. Proposed Approach for Drilling Temperature, Hole Diameter, and Roundness Prediction
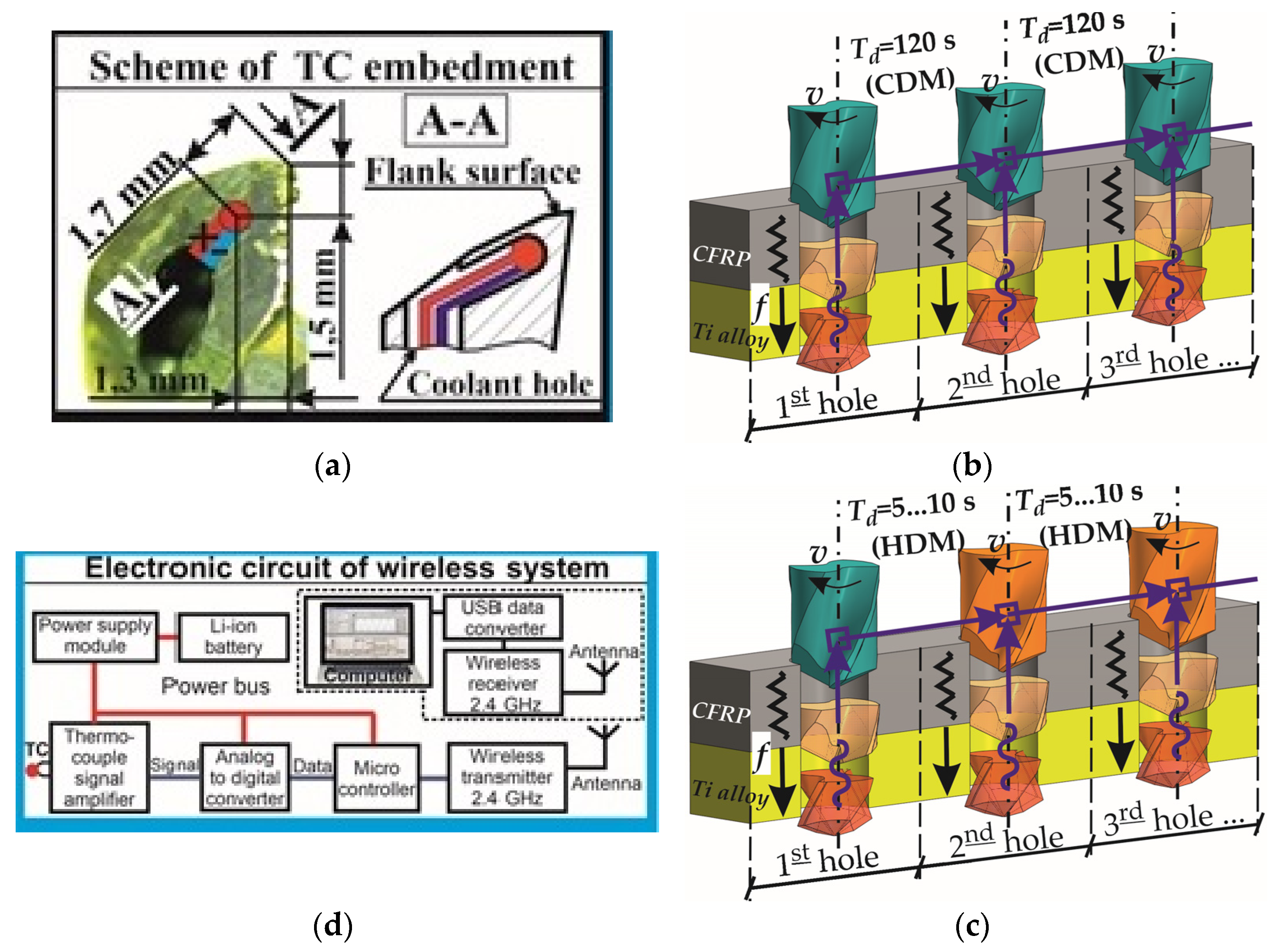
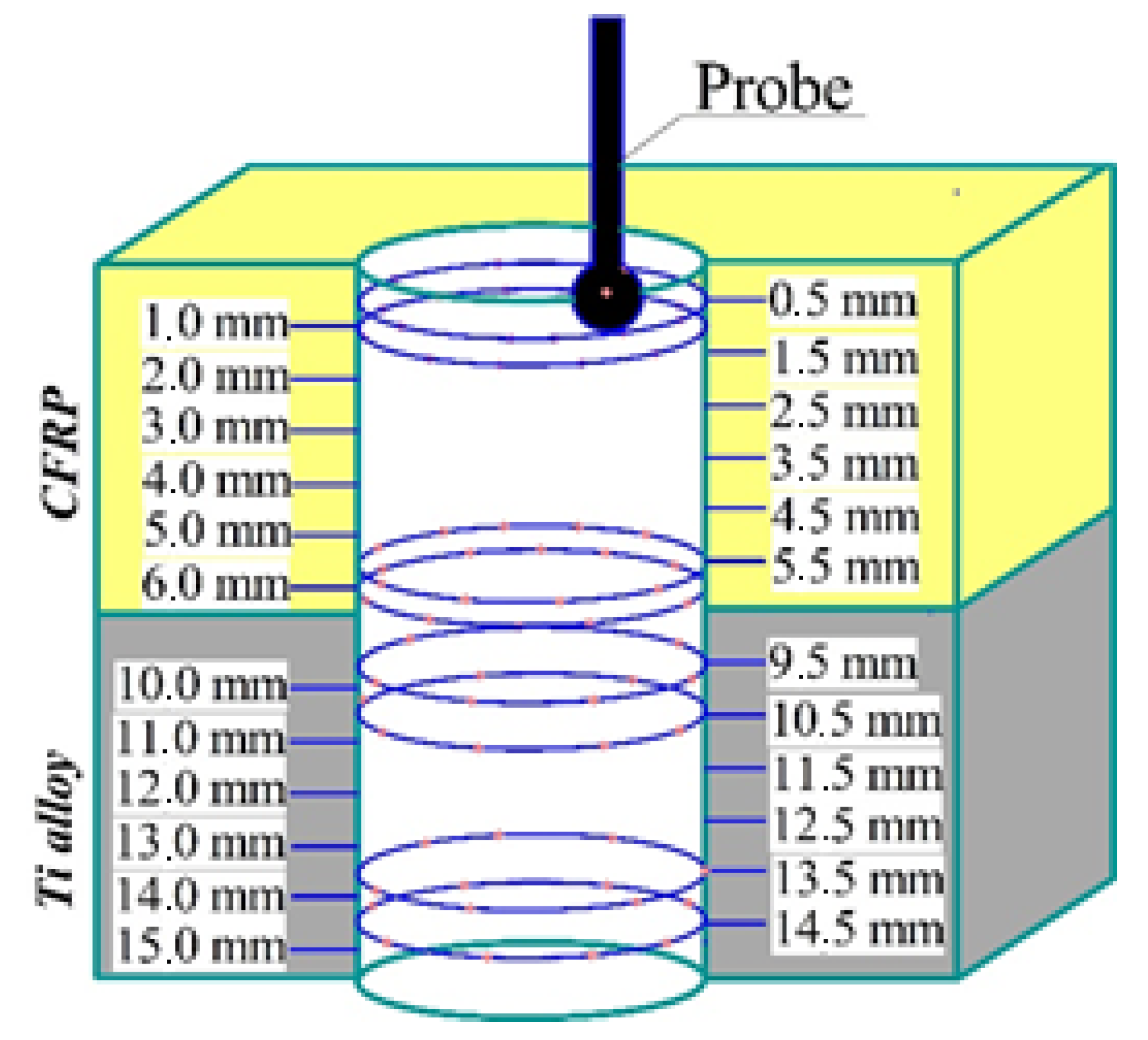
2.3. Experimental Set-Up
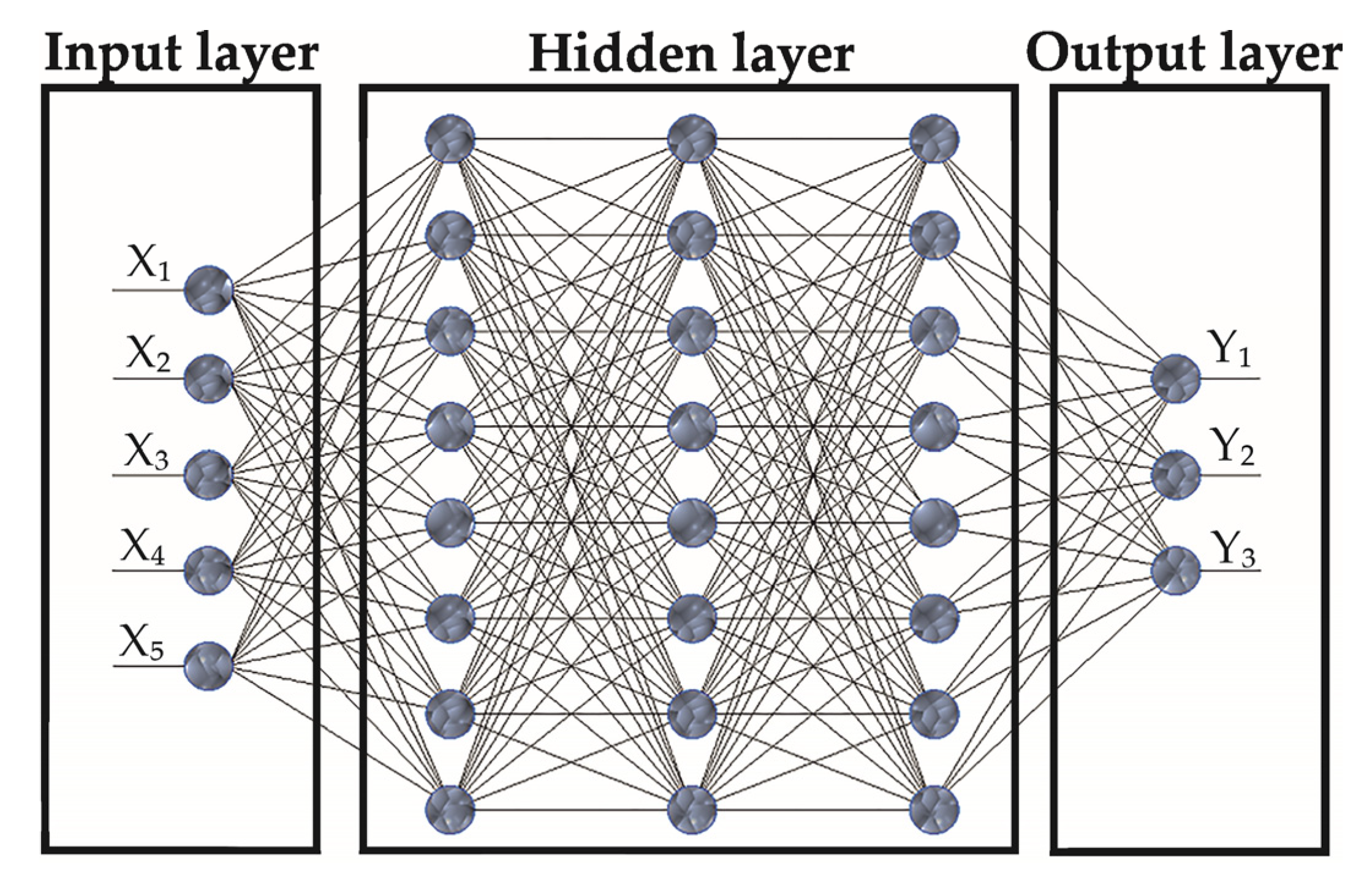
2.4. Methodology of ANN Analysis
- -
- From 50 to 70% to train new neural networks;
- -
- From 15 to 25% to control overfitting of neural networks;
- -
- From 15 to 25% to test trained neural networks.
3. Experimental Results and Discussion
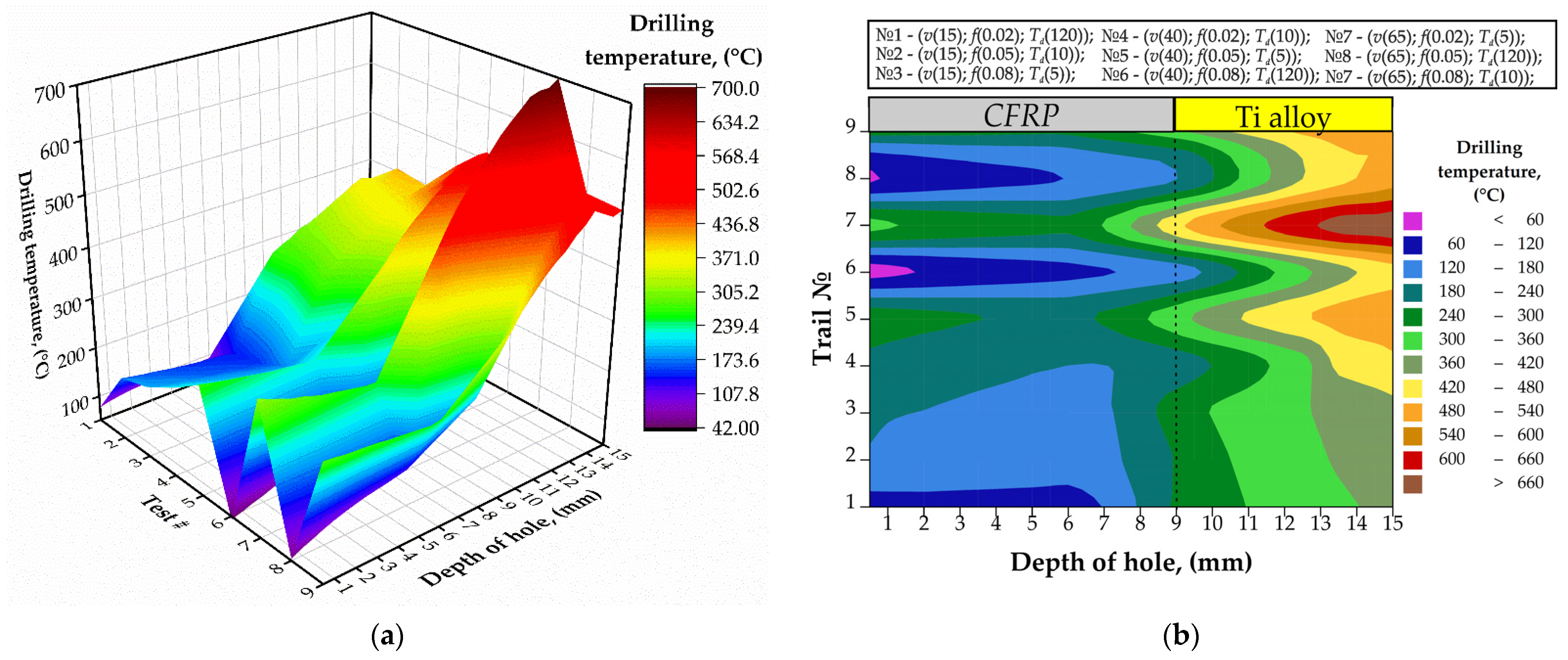
3.1. Drilling Temperature
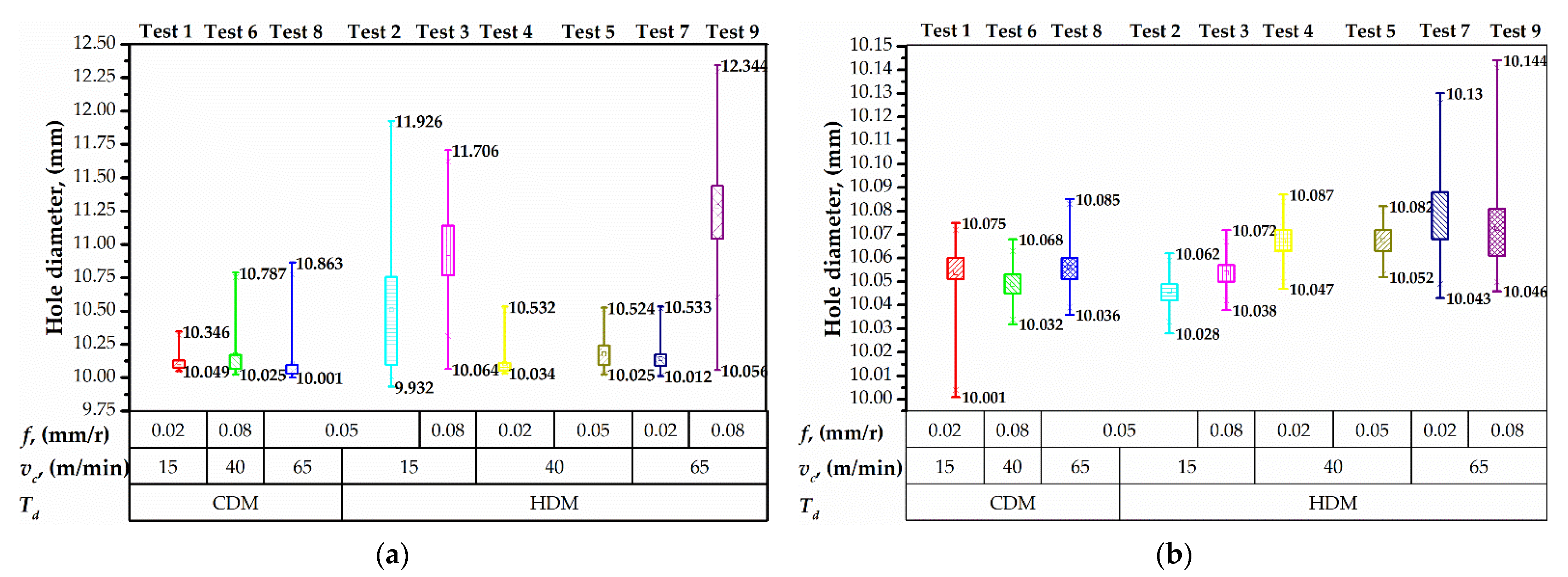
3.2. Hole Accuracy
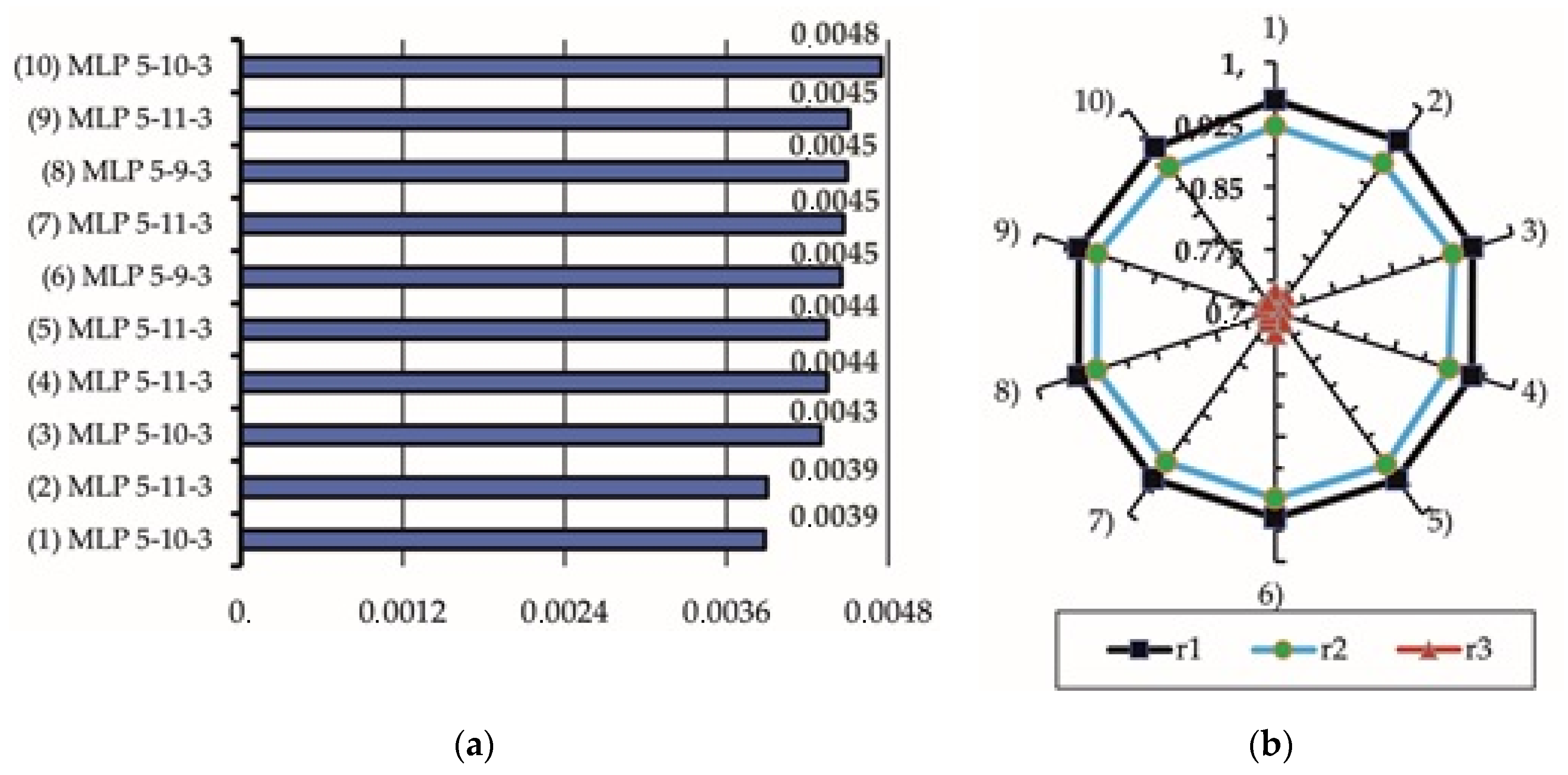
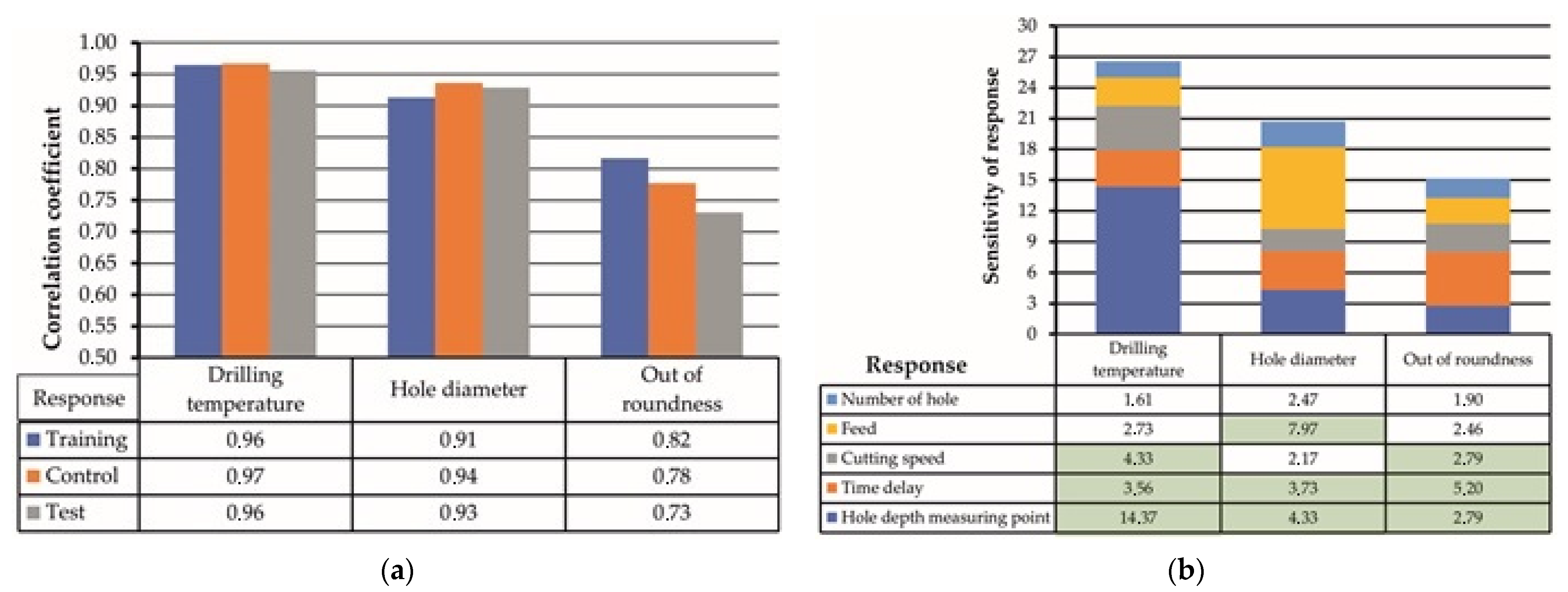
3.3. ANN Analysis of Experimental Results
4. Conclusions
- The drilling temperature is significantly affected by the time delay factor during single-shot drilling of the CFRP/Ti alloy stack. Increasing the drilling delay time between the two adjusted holes from 5 s to 120 s decreased the maximum drilling temperature from 350 °C to 150 °C in CFRP and from 690 °C to 575 °C in the Ti plate. Thus, the drill bit temperature is the key factor in controlling the drilling temperature;
- The hole diameter and out of roundness are affected by the drilling temperature. When drilling with a short time delay and aggressive feed, the drilling-induced heat transfers into the CFRP/Ti workpiece, deteriorating its fracture toughness and chip formation mechanism, respectively, that eventually results in oversizing. The diameter and out of roundness in CFRP grown along with the feed rate from 10.064 to 12.344 mm, and from 0.142 to 0.894, respectively. Low drilling temperature ensures higher hole accuracy;
- The ANN analysis allowed quantifying the sensitivities of the hole quality parameters to the drilling parameters. The drilling temperature was sensitive to cutting speed (4.33 points) and time delay (3.59 points) when drilling CFRP →Ti sequence, with a correlation coefficient of 0.97. The hole diameter was affected mainly by feed (7.97 points) and time delay (3.73 points) with a correlation coefficient of 0.94, while hole out roundness changed under the influence of time delay (5.20 points) and cutting speed (2.79 points) with a correlation coefficient of 0.74. Thus, ANN has a great potential for optimizing cutting parameters and other operational conditions of drilling holes in CFRP/Ti alloy stacks in industrial production;
- The most accurate ANN architecture to accomplish such a task is MLP-5-10-3, with a hyperbolic tangent function to activate hidden neurons, an exponential function to activate output neurons, and a gradient learning algorithm that provides an error of prediction up to 0.00388.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nekrasov, S.; Zhyhylii, D.; Dovhopolov, A.; Karatas, M.A. Research on the manufacture and strength of the innovative joint of FRP machine parts. J. Manuf. Process. 2021, 72, 338–349. [Google Scholar] [CrossRef]
- Osadchiy, I.; Kryvoruchko, D.; Kolesnyk, V.; Hatala, M.; Duplak, J.; Mital, D. Development of Integrated Technology of FRP Gear Manufacturing. Manuf. Technol. 2016, 16, 574–578. [Google Scholar] [CrossRef]
- Xu, J.Y.; Mkaddem, A.; El Mansori, M. Recent advances in drilling hybrid FRP/Ti composite: A state-of-the-art review. Compos. Struct. 2016, 135, 316–338. [Google Scholar] [CrossRef]
- Giasin, K.; Hawxwell, J.; Sinke, J.; Dhakal, H.; Köklü, U.; Brousseau, E. The effect of cutting tool coating on the form and dimensional errors of machined holes in GLARE® fibre metal laminates. Int. J. Adv. Manuf. Technol. 2020, 107, 2817–2832. [Google Scholar] [CrossRef]
- Xu, C.Y.; Wang, Y.W.; Xu, J.Z.; Liu, X.L. Design of internal-chip-removal drill for CFRP drilling and study of influencing factors of drilling quality. Int. J. Adv. Manuf. Technol. 2020, 106, 1657–1669. [Google Scholar] [CrossRef]
- Denysenko, Y.; Ivanov, V.; Luscinski, S.; Zaloga, V. An Integrated Approach for Improving Tool Provisioning Efficiency. Manag. Prod. Eng. Rev. 2020, 11, 4–12. [Google Scholar] [CrossRef]
- Wojciechowski, S. Estimation of Minimum Uncut Chip Thickness during Precision and Micro-Machining Processes of Various Materials—A Critical Review. Materials 2022, 15, 59. [Google Scholar] [CrossRef]
- Jia, Z.-Y.; Zhang, C.; Wang, F.-J.; Fu, R.; Chen, C. Multi-margin drill structure for improving hole quality and dimensional consistency in drilling Ti/CFRP stacks. J. Mater. Process. Technol. 2020, 276, 116405. [Google Scholar] [CrossRef]
- Wang, B.; Zhao, H.; Zhang, F.; Wang, M.; Zheng, Y. Comparison of the geometric accuracy of holes made in CFRP/Ti laminate by drilling and helical milling. Int. J. Adv. Manuf. Technol. 2021, 112, 3343–3350. [Google Scholar] [CrossRef]
- An, Q.; Dang, J.; Li, J.; Wang, C.; Chen, M. Investigation on the cutting responses of CFRP/Ti stacks: With special emphasis on the effects of drilling sequences. Compos. Struct. 2020, 253, 112794. [Google Scholar] [CrossRef]
- Kolesnyk, V.; Peterka, J.; Kuruc, M.; Šimna, V.; Moravčíková, J.; Vopát, T.; Lisovenko, D. Experimental Study of Drilling Temperature, Geometrical Errors and Thermal Expansion of Drill on Hole Accuracy When Drilling CFRP/Ti Alloy Stacks. Materials 2020, 13, 3232. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Ke, Y.L.; Dong, H.Y.; Chen, Z.P.; Gao, K.Y. Hole diameter variation and roundness in dry orbital drilling of CFRP/Ti stacks. Int. J. Adv. Manuf. Technol. 2016, 87, 811–824. [Google Scholar] [CrossRef]
- Qiu, X.-Y.; Yu, Z.; Li, C.-P.; Niu, Q.-L.; Li, S.-J.; Li, P.-N.; Ko, T.J. Influence of main cutting edge structure on hole defects in CFRP/titanium alloy stacks drilling. J. Manuf. Process. 2021, 69, 503–513. [Google Scholar] [CrossRef]
- Wang, Q.; Jia, X. Optimization of cutting parameters for improving exit delamination, surface roughness, and production rate in drilling of CFRP composites. Int. J. Adv. Manuf. Technol. 2021, 117, 3487–3502. [Google Scholar] [CrossRef]
- Yaşar, N.; Korkmaz, M.E.; Gupta, M.K.; Boy, M.; Günay, M. A novel method for improving drilling performance of CFRP/Ti6AL4V stacked materials. Int. J. Adv. Manuf. Technol. 2021, 117, 653–673. [Google Scholar] [CrossRef]
- Nie, P.; Ma, Y.; Liu, S. Research on CFRP inner wall roughness during vibration drilling of CFRP/Ti based on acoustic emission. J. Phys. Conf. Ser. 2021, 1952, 032060. [Google Scholar] [CrossRef]
- Rajkumar, G.M.; Bhardwaj, D.; Kannan, C.; Oyyaravelu, R.; Balan, A.S.S. Effect of chilled air on delamination, induced vibration, burr formation and surface roughness in CFRP drilling: A comparative study. Mater. Res. Express 2019, 6, 035305. [Google Scholar] [CrossRef]
- Liu, S.; Wang, N.; Zhang, K.; Luo, B. A subjective-objective evaluation method of hole surface quality in drilling CFRP-Ti stacks. Proc. Inst. Mech. Eng. Part B J. Eng. Manuf. 2020, 236, 64–76. [Google Scholar] [CrossRef]
- Li, J.; Zou, P.; Qiao, C.; Dong, L. Temperature Field Distribution Model in Drilling of CFRP/Ti Stacks Structure. J. Phys. Conf. Ser. 2020, 1626. [Google Scholar] [CrossRef]
- Voß, R.; Henerichs, M.; Rupp, S.; Kuster, F.; Wegener, K. Evaluation of bore exit quality for fibre reinforced plastics including delamination and uncut fibres. CIRP J. Manuf. Sci. Technol. 2016, 12, 56–66. [Google Scholar] [CrossRef]
- Alonso, U.; Calamaz, M.; Girot, F.; Iriondo, E. Influence of flute number and stepped bit geometry when drilling CFRP/Ti6Al4V stacks. J. Manuf. Process. 2019, 39, 356–370. [Google Scholar] [CrossRef]
- Kuo, C.; Soo, S.; Aspinwall, D.; Carr, C.; Bradley, S.; M’Saoubi, R.; Leahy, W. Development of single step drilling technology for multilayer metallic-composite stacks using uncoated and PVD coated carbide tools. J. Manuf. Process. 2018, 31, 286–300. [Google Scholar] [CrossRef]
- Poór, D.I.; Geier, N.; Pereszlai, C.; Xu, J.Y. A critical review of the drilling of CFRP composites: Burr formation, characterisation and challenges. Compos. Part B Eng. 2021, 223, 109155. [Google Scholar] [CrossRef]
- Melentiev, R.; Priarone, P.C.; Robiglio, M.; Settineri, L. Effects of Tool Geometry and Process Parameters on Delamination in CFRP Drilling: An Overview. Procedia CIRP 2016, 45, 31–34. [Google Scholar] [CrossRef]
- Han, C.; Bin Kim, K.; Lee, S.W.; Jun, M.B.-G.; Jeong, Y.H. Thrust Force-Based Tool Wear Estimation Using Discrete Wavelet Transformation and Artificial Neural Network in CFRP Drilling. Int. J. Precis. Eng. Manuf. 2021, 22, 1527–1536. [Google Scholar] [CrossRef]
- Ivchenko, O.; Zhyhylii, D.; Zaloha, O.; Zaloga, V.; Dehtiarenko, O. Resolution of the Friction Coefficient of Adhesion Under Cutting. In Advanced Manufacturing Processes. InterPartner 2019. Lecture Notes in Mechanical Engineering; Springer: Cham, Switzerland, 2019. [Google Scholar] [CrossRef]
- Wang, Q.; Wang, F.; Zhang, C.; Chen, C. Combined effects of various materials on tool wear in drilling of Ti/CFRP stacks. Proc. Inst. Mech. Eng. Part C J. Mech. Eng. Sci. 2019, 234, 2750–2759. [Google Scholar] [CrossRef]
- Rao, U.S.; Rodrigues, L.L.R. Tjprc An Application of Dissimilar ANN Algorithms to Improve the Simulation Performance of Flank Wear Extrapolation in GFRP Composite Drilling. Int. J. Mech. Prod. Eng. Res. Dev. 2018, 8, 325–336. [Google Scholar] [CrossRef]
- Hou, G.Y.; Qiu, J.P.; Zhang, K.F.; Cao, S.P.; Cheng, H.; Luo, B.; Cheng, Y. Comparative tool wear and hole quality investigation in drilling of aerospace grade T800 CFRP using different external cooling lubricants. Int. J. Adv. Manuf. Technol. 2020, 106, 937–951. [Google Scholar] [CrossRef]
- Kulesh, E.A.; Piliptsou, D.G.; Rogachev, A.V.; Hong, J.X.; Fedosenko, N.N.; Kolesnyk, V. Boron-Carbon Coatings: Structure, Morphology, and Mechanical Properties. J. Eng. Sci. 2020, 7, C1–C9. [Google Scholar] [CrossRef]
- Kim, D.; Swan, S.R.; He, B.; Khominich, V.; Bell, E.; Lee, S.-W.; Kim, T.-G. A study on the machinability of advanced arc PVD AlCrN-coated tungsten carbide tools in drilling of CFRP/titanium alloy stacks. Carbon Lett. 2020, 31, 497–507. [Google Scholar] [CrossRef]
- Liu, L.; Jiang, X.; Ermakov, S.F.; Nikolaev, V.I.; Yarmolenko, M.A.; Piliptsou, D.G.; Shershnev, E.B. Effect of Lubricants with Different Nature and Properties on Friction in a DLC–UHMWP Endoprosthesis Pair. J. Frict. Wear 2021, 42, 146–151. [Google Scholar] [CrossRef]
- Kayihan, M.; Karaguzel, U.; Bakkal, M. Experimental analysis on drilling of Al/Ti/CFRP hybrid composites. Mater. Manuf. Process. 2020, 36, 215–222. [Google Scholar] [CrossRef]
- Álvarez-Alcón, M.; López de Lacalle, L.N.; Fernández-Zacarías, F. Multiple Sensor Monitoring of CFRP Drilling to Define Cutting Parameters Sensitivity on Surface Roughness, Cylindricity and Diameter. Materials 2020, 13, 2796. [Google Scholar] [CrossRef]
- Bañon, F.; Sambruno, A.; Batista, M.; Vidal, S.R.F.; Salguero, J. Study of the one-shot drilling of CFRP/Ti6Al4V stacks with a double tip angle cutting-tool geometry. AIP Conf. Proc. 2019, 2113, 080010. [Google Scholar] [CrossRef]
- Kuntoğlu, M.; Aslan, A.; Pimenov, D.Y.; Giasin, K.; Mikolajczyk, T.; Sharma, S. Modeling of Cutting Parameters and Tool Geometry for Multi-Criteria Optimization of Surface Roughness and Vibration via Response Surface Methodology in Turning of AISI 5140 Steel. Materials 2020, 13, 4242. [Google Scholar] [CrossRef]
- Kim, C.-J. Temperature-Dependent Dynamic Characteristics of Carbon-Fiber-Reinforced Plastic for Different Spectral Loading Patterns. Materials 2020, 13, 5238. [Google Scholar] [CrossRef] [PubMed]
- Zitoune, R.; Cadorin, N.; Collombet, F.; Šíma, M. Temperature and wear analysis in function of the cutting tool coating when drilling of composite structure: In situ measurement by optical fiber. Wear 2017, 376–377, 1849–1858. [Google Scholar] [CrossRef]
- Xu, J.; Mansori, M.E. Experimental Studies on the Cutting Characteristics of Hybrid CFRP/Ti Stacks. Procedia Manuf. 2016, 5, 270–281. [Google Scholar] [CrossRef][Green Version]
- Xu, J.Y.; Li, C.; Chen, M.; Mansori, M.E.; Davim, J.P. On the analysis of temperatures, surface morphologies and tool wear in drilling CFRP/Ti6Al4V stacks under different cutting sequence strategies. Compos. Struct. 2019, 234, 111708. [Google Scholar] [CrossRef]
- Luo, B.; Zhang, K.; Liu, S.; Cheng, H.; Wang, R. Investigation on the interface damage in drilling low-stiffness CFRP/Ti stacks. Chin. J. Aeronaut. 2019, 32, 2211–2221. [Google Scholar] [CrossRef]
- Dahnel, A.N.; Ascroft, H.; Barnes, S. An investigation of hole quality during drilling of carbon fibre reinforced plastic and titanium (Ti6Al4V) using tungsten carbide drills. Mater. Today Proc. 2020, 29, 161–167. [Google Scholar] [CrossRef]
- Tang, L.-Y.; Li, P.-N.; Yu, Z.; Li, C.-P.; Ko, T.J.; Niu, Q.-L.; Li, S.-J.; Qiu, X.-Y. New drilling method for damage reduction of CFRP/Ti stacks drilling. Int. J. Adv. Manuf. Technol. 2021, 115, 595–602. [Google Scholar] [CrossRef]
- Iqbal, A.; Zhao, G.; Zaini, J.; Gupta, M.; Jamil, M.; He, N.; Nauman, M.; Mikolajczyk, T.; Pimenov, D. Between-the-Holes Cryogenic Cooling of the Tool in Hole-Making of Ti-6Al-4V and CFRP. Materials 2021, 14, 795. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Pérez, J.; Cantero, J.L.; Díaz-Álvarez, J.; Miguélez, M.H. Hybrid Composite-Metal Stack Drilling with Different Minimum Quantity Lubrication Levels. Materials 2019, 12, 448. [Google Scholar] [CrossRef]
- Sun, L.; Gao, H.; Wang, B.; Bao, Y.; Wang, M.; Ma, S. Mechanism of reduction of damage during helical milling of titanium/CFRP/aluminium stacks. Int. J. Adv. Manuf. Technol. 2020, 107, 4741–4753. [Google Scholar] [CrossRef]
- Priarone, P.C.; Robiglio, M.; Melentiev, R.; Settineri, L. Diamond Drilling of Carbon Fiber Reinforced Polymers: Influence of Tool Grit Size and Process Parameters on Workpiece Delamination. Procedia CIRP 2017, 66, 181–186. [Google Scholar] [CrossRef]
- Fu, Q.; Wu, S.J.; Li, C.H.; Xu, J.Y.; Wang, D.Z. Delamination and chip breaking mechanism of orthogonal cutting CFRP/Ti6Al4V composite. J. Manuf. Process. 2021, 73, 183–196. [Google Scholar] [CrossRef]
- An, Q.; Zhong, B.; Wang, X.; Zhang, H.; Sun, X.; Chen, M. Effects of drilling strategies for CFRP/Ti stacks on static mechanical property and fatigue behavior of open-hole CFRP laminates. J. Manuf. Process. 2021, 64, 409–420. [Google Scholar] [CrossRef]
- Shu, L.M.; Li, S.H.; Fang, Z.L.; Kizaki, T.; Kimura, K.; Arai, G.; Arai, K.; Sugita, N. Study on dedicated drill bit design for carbon fiber reinforced polymer drilling with improved cutting mechanism. Compos. Part A Appl. Sci. Manuf. 2021, 142, 106259. [Google Scholar] [CrossRef]
- Xu, J.Y.; Ji, M.; Davim, J.P.; Chen, M.; Mansori, M.E.; Krishnaraj, V. Comparative study of minimum quantity lubrication and dry drilling of CFRP/titanium stacks using TiAlN and diamond coated drills. Compos. Struct. 2020, 234, 111727. [Google Scholar] [CrossRef]
- Bolar, G.; Sridhar, A.K.; Ranjan, A. Drilling and helical milling for hole making in multi-material carbon reinforced aluminum laminates. Int. J. Light. Mater. Manuf. 2022, 5, 113–125. [Google Scholar] [CrossRef]
- Voss, R.; Henerichs, M.; Kuster, F. Comparison of conventional drilling and orbital drilling in machining carbon fibre reinforced plastics (CFRP). CIRP Ann. 2016, 65, 137–140. [Google Scholar] [CrossRef]
- Xu, J.Y.; Ji, M.; Chen, M.; Mansori, M.E. Experimental investigation on drilling machinability and hole quality of CFRP/Ti6Al4V stacks under different cooling conditions. Int. J. Adv. Manuf. Technol. 2020, 109, 1527–1539. [Google Scholar] [CrossRef]
- Karpat, Y.; Karagüzel, U.; Bahtiyar, O. A thermo-mechanical model of drill margin-borehole surface interface contact conditions in dry drilling of thick CFRP laminates. Int. J. Mach. Tools Manuf. 2020, 154, 103565. [Google Scholar] [CrossRef]
- Shao, Z.; Jiang, X.; Geng, D.; Liu, Y.; Zhou, Z.; Li, S.; Zhang, D.; Zheng, W. The interface temperature and its influence on surface integrity in ultrasonic-assisted drilling of CFRP/Ti stacks. Compos. Struct. 2021, 266, 113803. [Google Scholar] [CrossRef]
- Xu, J.; Li, C.; Mansori, M.E.; Liu, G.; Chen, M. Study on the Frictional Heat at Tool-Work Interface when Drilling CFRP Composites. Procedia Manuf. 2018, 26, 415–423. [Google Scholar] [CrossRef]
- Leng, S.; Wang, Z.; Min, T.; Dai, Z.Q.; Chen, G. Detection of Tool Wear in Drilling CFRP/TC4 Stacks by Acoustic Emission. J. Vib. Eng. Technol. 2020, 8, 463–470. [Google Scholar] [CrossRef]
- Wang, B.; Yin, W.D.A.; Wang, M.H.; Zheng, Y.H.; Li, X.P.; Ma, Z.B. Edge chipping mechanism and failure time prediction on carbide cemented tool during drilling of CFRP/Ti stack. Int. J. Adv. Manuf. Technol. 2017, 91, 3015–3024. [Google Scholar] [CrossRef]
- Emel’Yanov, S.G.; Yatsun, E.I.; Remnev, A.I.; Shvets, S.V. Chip curling in metal cutting. Russ. Eng. Res. 2011, 31, 679–683. [Google Scholar] [CrossRef]
- Moghaddas, M.A.; Yi, A.Y.; Graff, K.F. Temperature measurement in the ultrasonic-assisted drilling process. Int. J. Adv. Manuf. Technol. 2019, 103, 187–199. [Google Scholar] [CrossRef]
- Wang, H.X.; Zhang, X.H.; Duan, Y.G. Effects of drilling area temperature on drilling of carbon fiber reinforced polymer composites due to temperature-dependent properties. Int. J. Adv. Manuf. Technol. 2018, 96, 2943–2951. [Google Scholar] [CrossRef]
- Peterka, J.; Vozar, M.; Vopat, T.; Pokorny, P.; Boris, P. Using multi-criteria analysis to evaluate the impact of drag-finishing technological parameters on the carbide tool radius. Mater. Today Proc. 2020, 22, 205–211. [Google Scholar]
- Peterka, J.; Pokorný, P. Influence of the Lead Angle from the Vertical Axis Milling on Effective Radius of the Cutter. Key Eng. Mater. 2013, 581, 44–49. [Google Scholar] [CrossRef]
- Tomas, V.; Jozef, P.; Mario, K.; Ivan, B. The Wear Measurement Process of Ball Nose end Mill in the Copy Milling Operations. In Proceedings of the 24th Daaam International Symposium on Intelligent Manufacturing and Automation, Zadar, Croatia, 23–26 October 2013; Volume 69, pp. 1038–1047. [Google Scholar] [CrossRef]
- Borkin, D.; Nemeth, M.; Nemethova, A. Using Autoregressive Integrated Moving Average (ARIMA) for Prediction of Time Series Data. Intell. Syst. Appl. Softw. Eng. 2019, 1046, 470–476. [Google Scholar] [CrossRef]
- Nemeth, M.; Borkin, D.; Michalconok, G. The Comparison of Machine-Learning Methods XGBoost and LightGBM to Predict Energy Development. Comput. Stat. Math. Modeling Methods Intell. Syst. 2019, 1047, 208–215. [Google Scholar] [CrossRef]
- Peterkova, A.; Nemeth, M.; Bohm, A. Computing missing values using neural networks in medical field. In Proceedings of the 22nd IEEE International Conference on Intelligent Engineering Systems (INES), Las Palmas de Gran Canaria, Spain, 21–23 June 2018; pp. 151–154. [Google Scholar]
- Pavlenko, I.; Trojanowska, J.; Ivanov, V.; Liaposhchenko, O. Parameter Identification of Hydro-Mechanical Processes Using Artificial Intelligence Systems. Int. J. Mechatron. Appl. Mech. 2019, 5, 19–26. [Google Scholar]
- Pavlenko, I.; Saga, M.; Kuric, I.; Kotliar, A.; Basova, Y.; Trojanowska, J.; Ivanov, V. Parameter Identification of Cutting Forces in Crankshaft Grinding Using Artificial Neural Networks. Materials 2020, 13, 5357. [Google Scholar] [CrossRef]
- Parmar, J.G.; Dave, K.; Gohil, A.; Trivedi, H. Prediction of end milling process parameters using artificial neural network. Mater. Today Proc. 2020, 38, 3168–3176. [Google Scholar] [CrossRef]
- Abbas, A.T.; Pimenov, D.Y.; Erdakov, I.N.; Taha, M.A.; Soliman, M.S.; Rayes, M.M.E. ANN Surface Roughness Optimization of AZ61 Magnesium Alloy Finish Turning: Minimum Machining Times at Prime Machining Costs. Materials 2018, 11, 808. [Google Scholar] [CrossRef]
- Zhou, G.; Xu, C.; Ma, Y.; Wang, X.-H.; Feng, P.-F.; Zhang, M. Prediction and control of surface roughness for the milling of Al/SiC metal matrix composites based on neural networks. Adv. Manuf. 2020, 8, 486–507. [Google Scholar] [CrossRef]
- Mia, M.; Khan, A.; Dhar, N.R. Study of surface roughness and cutting forces using ANN, RSM, and ANOVA in turning of Ti-6Al-4V under cryogenic jets applied at flank and rake faces of coated WC tool. Int. J. Adv. Manuf. Technol. 2017, 93, 975–991. [Google Scholar] [CrossRef]
- Sada, S.O. Improving the predictive accuracy of artificial neural network (ANN) approach in a mild steel turning operation. Int. J. Adv. Manuf. Technol. 2021, 112, 2389–2398. [Google Scholar] [CrossRef]
- Wu, T.Y.; Lei, K.W. Prediction of surface roughness in milling process using vibration signal analysis and artificial neural network. Int. J. Adv. Manuf. Technol. 2019, 102, 305–314. [Google Scholar] [CrossRef]
- Paturi, U.M.R.; Devarasetti, H.; Narala, S.K.R. Application Of Regression And Artificial Neural Network Analysis In Modelling Of Surface Roughness In Hard Turning Of AISI 52100 Steel. Mater. Today Proc. 2018, 5, 4766–4777. [Google Scholar] [CrossRef]
- Rifai, A.P.; Aoyama, H.; Tho, N.H.; Dawal, S.Z.M.; Masruroh, N.A. Evaluation of turned and milled surfaces roughness using convolutional neural network. Measurement 2020, 161, 107860. [Google Scholar] [CrossRef]
- Savkovic, B.; Kovac, P.; Rodic, D.; Strbac, B.; Klancnik, S. Comparison of artificial neural network, fuzzy logic and genetic algorithm for cutting temperature and surface roughness prediction during the face milling process. Adv. Prod. Eng. Manag. 2020, 15, 137–150. [Google Scholar] [CrossRef]
- Li, B.; Tian, X. An Effective PSO-LSSVM-Based Approach for Surface Roughness Prediction in High-Speed Precision Milling. IEEE Access 2021, 9, 80006–80014. [Google Scholar] [CrossRef]
- Feito, N.; Muñoz-Sánchez, A.; Díaz-Álvarez, A.; Loya, J.A. Analysis of the Machinability of Carbon Fiber Composite Materials in Function of Tool Wear and Cutting Parameters Using the Artificial Neural Network Approach. Materials 2019, 12, 2747. [Google Scholar] [CrossRef]
- Teti, R.; Segreto, T.; Caggiano, A.; Nele, L. Smart Multi-Sensor Monitoring in Drilling of CFRP/CFRP Composite Material Stacks for Aerospace Assembly Applications. Appl. Sci. 2020, 10, 758. [Google Scholar] [CrossRef]
- Cui, J.; Liu, W.; Zhang, Y.; Gao, C.; Lu, Z.; Li, M.; Wang, F. A novel method for predicting delamination of carbon fiber reinforced plastic (CFRP) based on multi-sensor data. Mech. Syst. Signal Process. 2021, 157, 107708. [Google Scholar] [CrossRef]
- SAE International. Aerospace Material Specifications. Titanium Alloy, Bars, Wire, and Rings, Titanium 6-2-4-2, Solution and Precipitation Heat Treated. SAE AMS 4975. 2017. Available online: https://www.sae.org/standards/content/ams4975/ (accessed on 20 January 2022).
- Ivanov, V.; Dehtiarov, I.; Pavlenko, I.; Kosov, M.; Hatala, M. Technological Assurance and Features of Fork-Type Parts Machining. Adv. Des. Simul. Manuf. Ii 2019, LNME, 114–125. [Google Scholar] [CrossRef]
- Graupe, D. Principles of Artificial Neural Networks, 3rd ed.; World Scientific Publishing Co. Pte. Ltd.: Singapore, 2013; p. 384. [Google Scholar]
- Hassoun, M.H. Fundamentals Of Artificial Neural Networks; MIT Press: Cambridge, MA, USA, 2003; p. 511. [Google Scholar]
- Quatember, A. Pseudo-Populations: A Basic Concept in Statistical Surveys; Springer: Berlin/Heidelberg, Germany, 2015; p. 150. [Google Scholar]
- Xu, J.; Lin, T.; Li, L.; Ji, M.; Davim, J.P.; Geier, N.; Chen, M. Numerical study of interface damage formation mechanisms in machining CFRP/Ti6Al4V stacks under different cutting sequence strategies. Compos. Struct. 2022, 285, 115236. [Google Scholar] [CrossRef]
- Sorrentino, L.; Turchetta, S.; Colella, L.; Bellini, C. Analysis of Thermal Damage in FRP Drilling. Procedia Eng. 2016, 167, 206–215. [Google Scholar] [CrossRef]




| Ti | Al | C | O | Si | Mn | Fe |
|---|---|---|---|---|---|---|
| 96.42% | 1.92% | 0.21% | 0.19% | 0.17%, | 0.89% | 0.20% |
| Geometric Parameters | Drill Number in Respect to the Test Number | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | |
| D, (mm) | 10.008 | 10.003 | 10.003 | 10.003 | 10.000 | 10.000 | 10.000 | 10.000 | 10.000 |
| Radial runout, (mm) | 0.010 | 0.012 | 0.016 | 0.008 | 0.008 | 0.008 | 0.008 | 0.008 | 0.008 |
| Point angle, (θ°) | 140.52 | 140.61 | 140.35 | 140.30 | 140.84 | 140.60 | 140.60 | 140.60 | 140.60 |
| Axial relief angle, (αa.r°) | 7.54 | 7.42 | 8.18 | 20.14 | 7.58 | 7.54 | 8.26 | 8.52 | 7.50 |
| Chisel edge angle, (ψ°) | 44.21 | 45.33 | 55.17 | 24.62 | 55.62 | 54.40 | 52.27 | 53.59 | 58.33 |
| Helix angle, (ω°) | 30.00 | 30.09 | 29.92 | 29.93 | 29.91 | 29.99 | 29.81 | 29.97 | 30.06 |
| Drilling Performance (Factors) | Levels of Factors | |||
|---|---|---|---|---|
| 1 | 2 | 3 | ||
| A | Cutting speed, vc (m/min) | 15 | 40 | 65 |
| B | Feed, f (mm/r) | 0.02 | 0.05 | 0.08 |
| C | Time delay, Td (s) | 120 | 10 | 5 |
| Test № | Cutting Speed, v (m/min) | Feed, f (mm/r) | Time Delay, Td (s) | Remark to Time Delay |
|---|---|---|---|---|
| 1 | 15 | 0.02 | 120 | CDM |
| 2 | 15 | 0.05 | 10 | HDM |
| 3 | 15 | 0.08 | 5 | HDM |
| 4 | 40 | 0.02 | 10 | HDM |
| 5 | 40 | 0.05 | 5 | HDM |
| 6 | 40 | 0.08 | 120 | CDM |
| 7 | 65 | 0.02 | 5 | HDM |
| 8 | 65 | 0.05 | 120 | CDM |
| 9 | 65 | 0.08 | 10 | HDM |
| A | B | C | D | E | F | G | H | I | |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Test № | Hole № | Cutting Speed, v (m/min) | Feed, f (mm/r) | Time Delay, Tt (s) | Hole Depth Measuring Point, (mm) | Drilling Temperature, t (°C) | Hole Diameter,D (mm) | Hole Out of Roundness, ΔD (mm) |
| 2 | X1 | X2 | X3 | X4 | X5 | Y1 | Y2 | Y3 | |
| 3 | 1 | 1 | 15 | 0.02 | 120 | 0.5 | 58.68 | 10.118 | 0.031 |
| 4 | 1 | 1 | 15 | 0.02 | 120 | 1 | 61.26 | 10.139 | 0.022 |
| 5 | 1 | 1 | 15 | 0.02 | 120 | 1.5 | 63.37 | 10.178 | 0.026 |
| 6 | 1 | 1 | 15 | 0.02 | 120 | 2 | 65.06 | 10.184 | 0.031 |
| 7 | 1 | 1 | 15 | 0.02 | 120 | 2.5 | 67 | 10.202 | 0.053 |
| 8 | 1 | 1 | 15 | 0.02 | 120 | 3 | 67.44 | 10.215 | 0.062 |
| 9 | 1 | 1 | 15 | 0.02 | 120 | 3.5 | 67.68 | 10.239 | 0.067 |
| 10 | 1 | 1 | 15 | 0.02 | 120 | 4 | 67.79 | 10.282 | 0.082 |
| 4745 | 9 | 23 | 65 | 0.08 | 10 | 10.5 | 422.14 | 10.097 | 0.048 |
| 4746 | 9 | 23 | 65 | 0.08 | 10 | 11 | 442.94 | 10.079 | 0.052 |
| 4747 | 9 | 23 | 65 | 0.08 | 10 | 11.5 | 460.33 | 10.064 | 0.055 |
| 4748 | 9 | 23 | 65 | 0.08 | 10 | 12 | 474.01 | 10.065 | 0.055 |
| 4749 | 9 | 23 | 65 | 0.08 | 10 | 12.5 | 494.37 | 10.063 | 0.063 |
| 4750 | 9 | 23 | 65 | 0.08 | 10 | 13 | 508.78 | 10.065 | 0.061 |
| 4751 | 9 | 23 | 65 | 0.08 | 10 | 13.5 | 529.73 | 10.057 | 0.063 |
| 4752 | 9 | 23 | 65 | 0.08 | 10 | 14 | 533.29 | 10.055 | 0.065 |
| 4753 | 9 | 23 | 65 | 0.08 | 10 | 14.5 | 504.25 | 10.056 | 0.07 |
| 4754 | 9 | 23 | 65 | 0.08 | 10 | 15 | 510.09 | 10.057 | 0.077 |
| № | Architecture | Function to Activate Hidden Neurons | Function to Activate Output Neurons | Learning Algorithm | Sensitivity | ||||
|---|---|---|---|---|---|---|---|---|---|
| X1 | X2 | X3 | X4 | X5 | |||||
| 1 | MLP 5-10-3 | Hyperbolic tangent | Exponential | Gradient | 1.422 | 3.534 | 2.276 | 4.802 | 16.83 |
| 2 | MLP 5-11-3 | Logistic sigmoid | Logistic sigmoid | BFGS | 1.979 | 7.209 | 2.74 | 8.326 | 14.37 |
| 3 | MLP 5-10-3 | Logistic sigmoid | Exponential | BFGS | 1.663 | 3.164 | 1.694 | 46.87 | 12.62 |
| 4 | MLP 5-11-3 | Hyperbolic tangent | Exponential | BFGS | 1.239 | 2.445 | 1.195 | 6.851 | 14.32 |
| 5 | MLP 5-11-3 | Identity | Logistic sigmoid | BFGS | 1.243 | 2.941 | 1.401 | 29.51 | 13.15 |
| 6 | MLP 5-9-3 | Logistic sigmoid | Exponential | BFGS | 2.223 | 1.965 | 1.254 | 7.033 | 14.04 |
| 7 | MLP 5-11-3 | Hyperbolic tangent | Logistic sigmoid | BFGS | 1.521 | 4.18 | 1.792 | 4.207 | 12.28 |
| 8 | MLP 5-9-3 | Logistic sigmoid | Exponential | Gradient | 1.225 | 2.845 | 1.859 | 3.878 | 12.52 |
| 9 | MLP 5-11-3 | Logistic sigmoid | Logistic sigmoid | Gradient | 2.123 | 3.669 | 1.771 | 3.469 | 12.27 |
| 10 | MLP 5-10-3 | Exponential | Logistic sigmoid | BFGS | 1.803 | 3.373 | 2.847 | 13.75 | 10.63 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kolesnyk, V.; Peterka, J.; Alekseev, O.; Neshta, A.; Xu, J.; Lysenko, B.; Sahul, M.; Martinovič, J.; Hrbal, J. Application of ANN for Analysis of Hole Accuracy and Drilling Temperature When Drilling CFRP/Ti Alloy Stacks. Materials 2022, 15, 1940. https://doi.org/10.3390/ma15051940
Kolesnyk V, Peterka J, Alekseev O, Neshta A, Xu J, Lysenko B, Sahul M, Martinovič J, Hrbal J. Application of ANN for Analysis of Hole Accuracy and Drilling Temperature When Drilling CFRP/Ti Alloy Stacks. Materials. 2022; 15(5):1940. https://doi.org/10.3390/ma15051940
Chicago/Turabian StyleKolesnyk, Vitalii, Jozef Peterka, Oleksandr Alekseev, Anna Neshta, Jinyang Xu, Bohdan Lysenko, Martin Sahul, Jozef Martinovič, and Jakub Hrbal. 2022. "Application of ANN for Analysis of Hole Accuracy and Drilling Temperature When Drilling CFRP/Ti Alloy Stacks" Materials 15, no. 5: 1940. https://doi.org/10.3390/ma15051940
APA StyleKolesnyk, V., Peterka, J., Alekseev, O., Neshta, A., Xu, J., Lysenko, B., Sahul, M., Martinovič, J., & Hrbal, J. (2022). Application of ANN for Analysis of Hole Accuracy and Drilling Temperature When Drilling CFRP/Ti Alloy Stacks. Materials, 15(5), 1940. https://doi.org/10.3390/ma15051940










