Effect of Si and B on the Electrochemical Behavior of FeCoNiCr-Based High-Entropy Amorphous Alloys
Abstract
1. Introduction
2. Experimental
2.1. Materials and Methods
2.2. Potentiodynamic Measurements
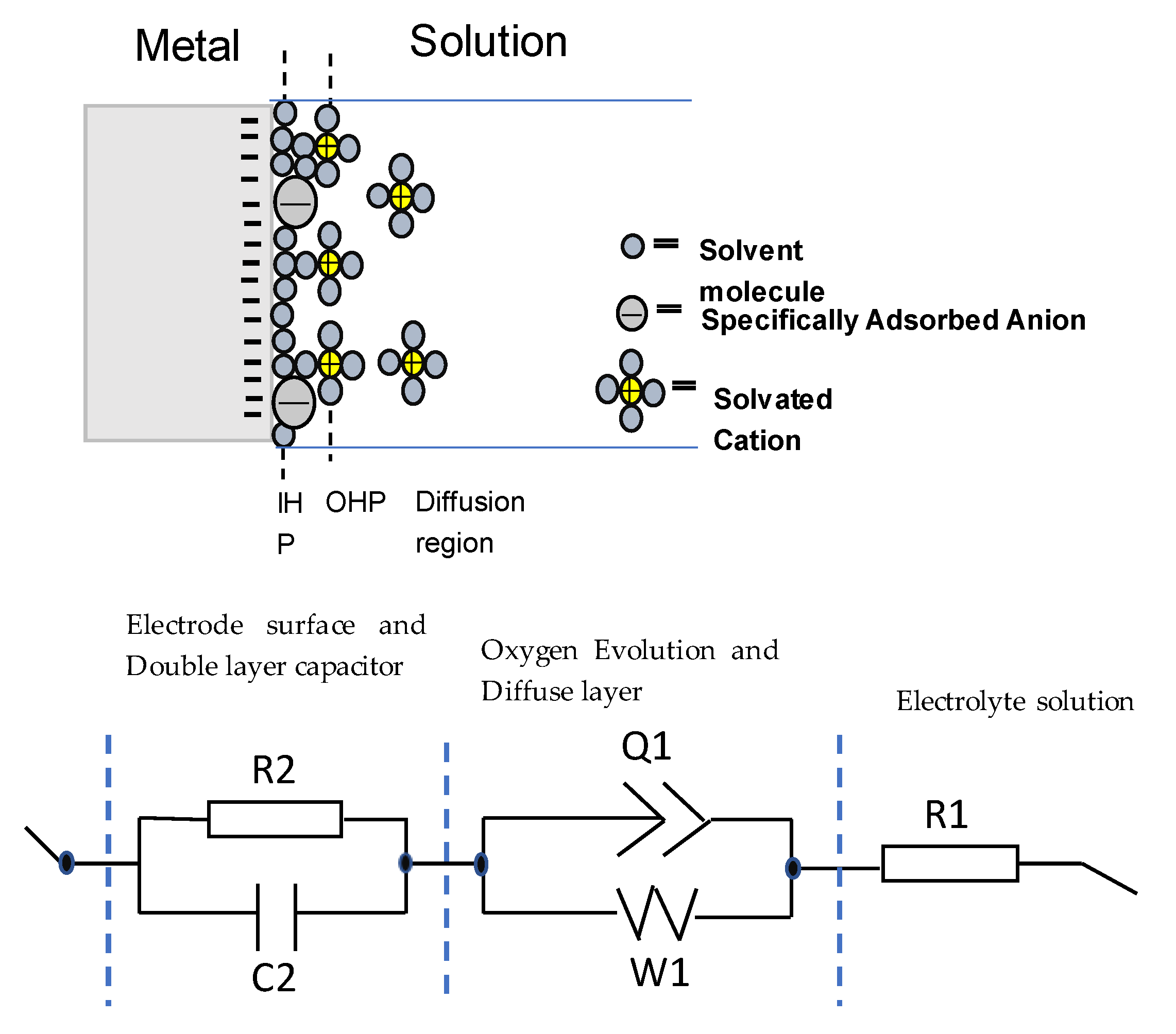
2.3. Electrochemical Impedance Spectroscopy
2.4. Surface Characterization
3. Results and Discussion
3.1. Electrochemical Measurements in NaCl Solution
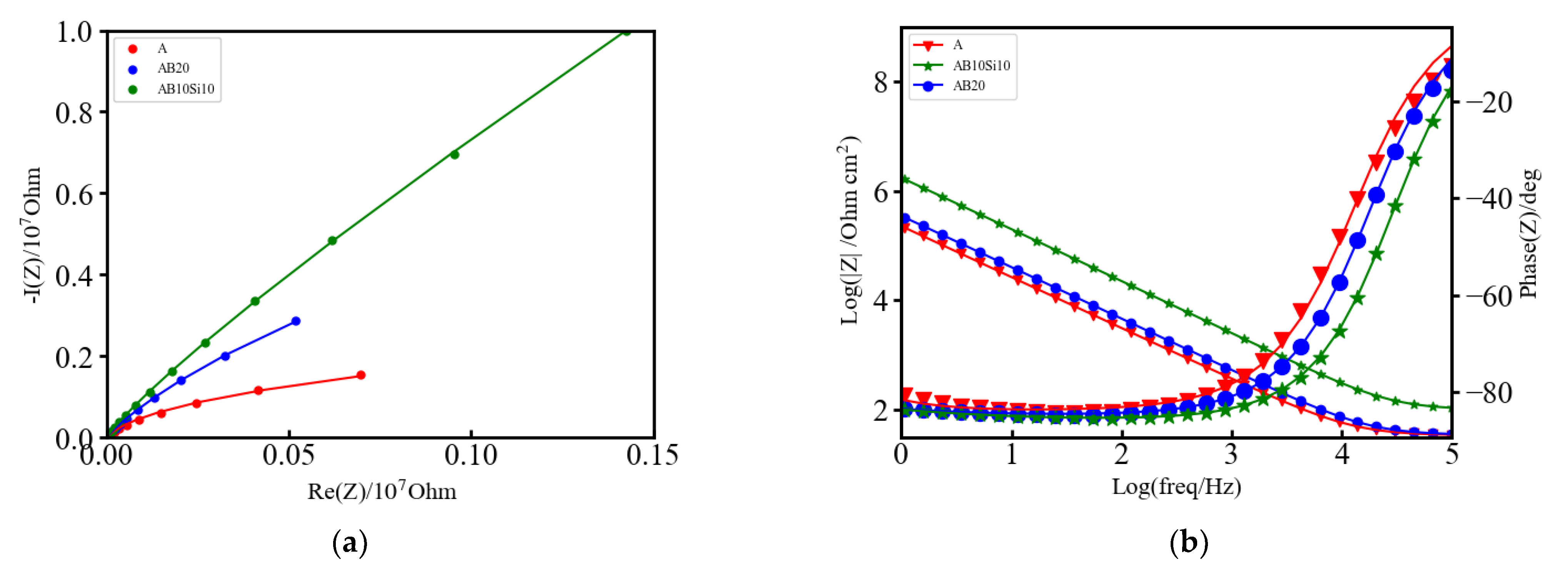
3.2. Electrochemical Impedance Spectroscopy (EIS) Measurements
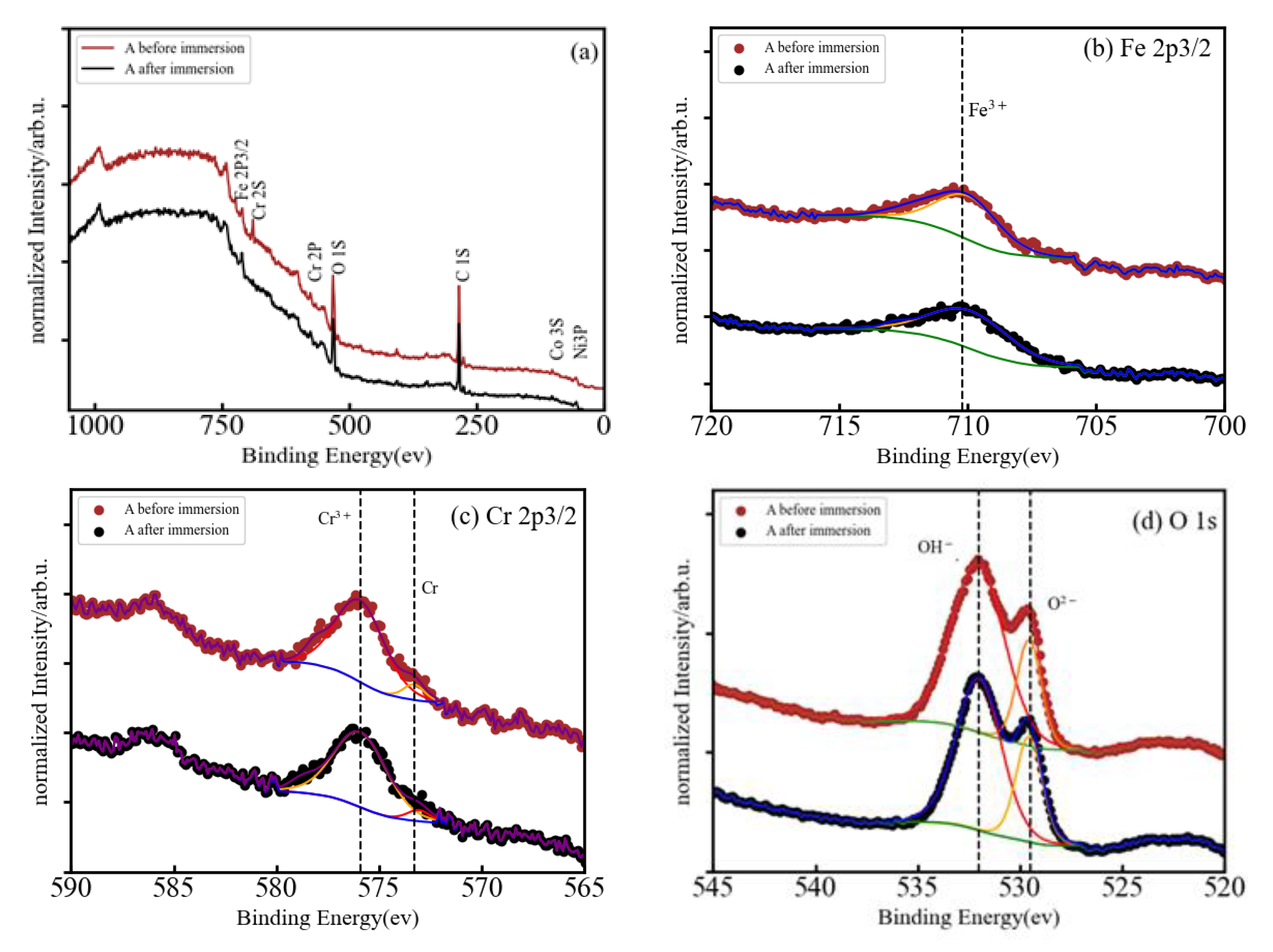
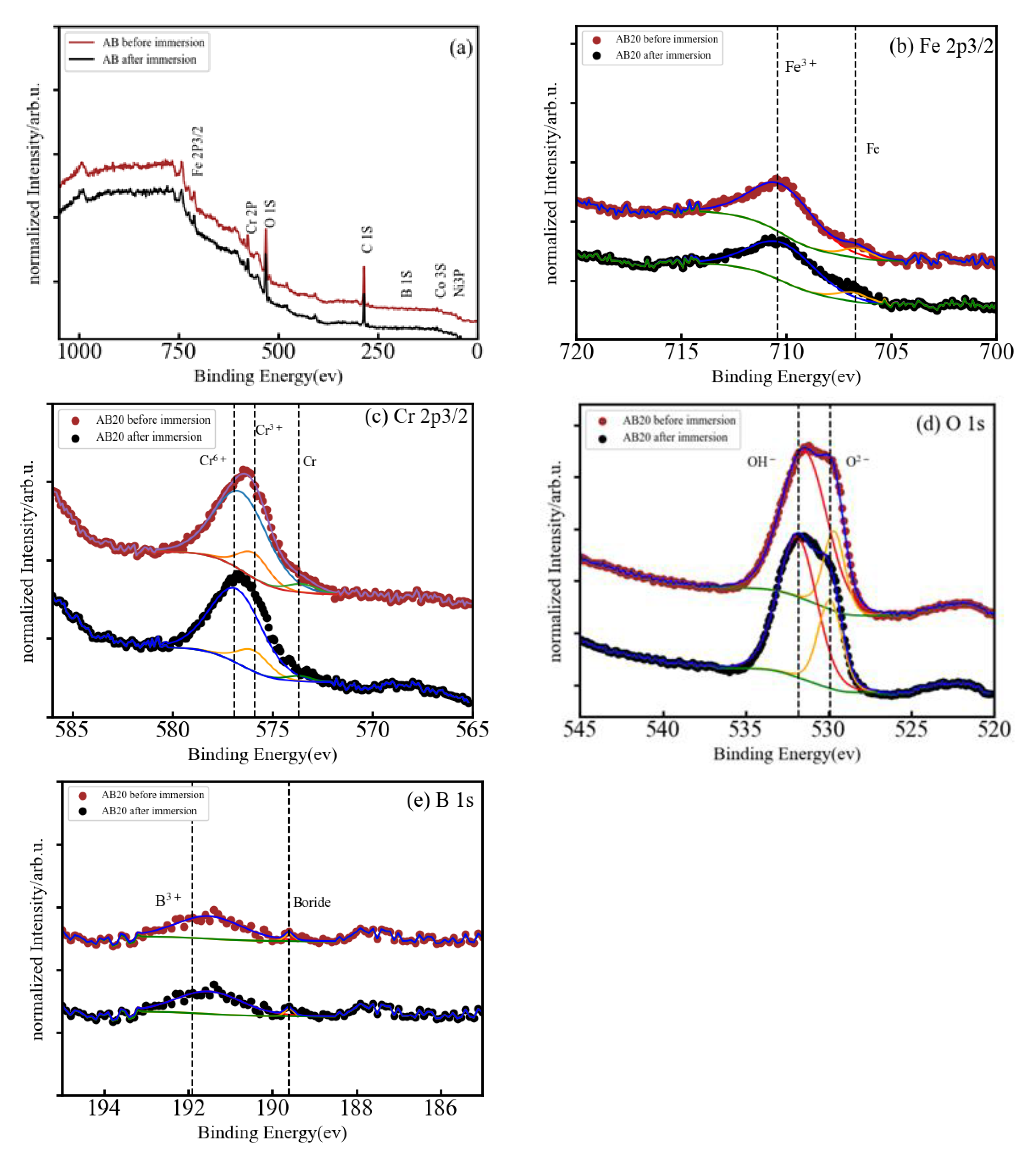
3.3. XPS Analysis of A, AB20, and AB10Si10 Alloys Immersed in NaCl
3.4. Microstructure Characterization after Immersion in NaCl
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Mudali, U.K.; Scudino, S.; Kühn, U.; Eckert, J.; Gebert, A. Polarisation behaviour of the Zr57Ti8Nb 2.5Cu13.9Ni11.1Al7.5 alloy in different microstructural states in acid solutions. Scr. Mater. 2004, 50, 1379–1384. [Google Scholar] [CrossRef]
- Pang, S.J.; Zhang, T.; Asami, K.; Inoue, A. Synthesis of Fe-Cr-Mo-C-B-P bulk metallic glasses with high corrosion resistance. Acta Mater. 2002, 50, 489–497. [Google Scholar] [CrossRef]
- Kim, J.H.; Akiyama, E.; Habazaki, H.; Kawashima, A.; Asami, K.; Hashimoto, K. The corrosion behavior of sputter-deposited amorphous chromium-zirconium alloys in 6 M HCl solution. Corros. Sci. 1993, 34, 1817–1827. [Google Scholar] [CrossRef]
- Wang, A.P.; Chang, X.C.; Hou, W.L.; Wang, J.Q. Corrosion behavior of Ni-based amorphous alloys and their crystalline counterparts. Corros. Sci. 2007, 49, 2628–2635. [Google Scholar] [CrossRef]
- Yang, S.; Liu, Z.; Pi, J. Microstructure and wear behavior of the AlCrFeCoNi high-entropy alloy fabricated by additive manufacturing. Mater. Lett. 2020, 261, 127004. [Google Scholar] [CrossRef]
- Zhang, H.; Zhou, J.; Muhammad, Y.; Tang, R.; Liu, K.; Zhu, Y.; Tong, Z. Citric acid modified bentonite for congo red adsorption. Front. Mater. 2019, 6, 5. [Google Scholar] [CrossRef]
- Jadhav, M.; Singh, S.; Srivastava, M.; Chethan, R.P.S.; Panigrahi, B.B. Effect of minute element addition on the oxidation resistance of FeCoCrNiAl and FeCoCrNi2Al high entropy alloy. Adv. Powder Technol. 2022, 33, 103410. [Google Scholar] [CrossRef]
- Nascimento, C.B.; Donatus, U.; Ríos, C.T.; De Oliveira, M.C.L.; Antunes, R.A. A review on Corrosion of High Entropy Alloys: Exploring the Interplay between Corrosion Properties, Alloy Composition, Passive Film Stability and Materials Selection. Mater. Res. 2022, 25, e20210442. [Google Scholar] [CrossRef]
- Masumoto, T.; Hashimoto, K. Corrosion properties of amorphous metals. J. Phys. Colloq. 1980, 41, C8–894. [Google Scholar] [CrossRef]
- Ren, B.; Zhao, R.F.; Liu, Z.X.; Guan, S.K.; Zhang, H.S. Microstructure and properties of Al0.3CrFe 1.5MnNi0.5Ti x and Al0.3CrFe 1.5MnNi0.5Si x high-entropy alloys. Rare Met. 2014, 33, 149–154. [Google Scholar] [CrossRef]
- Li, Y.; Wang, S.; Wang, X.; Yin, M.; Zhang, W. New FeNiCrMo(P, C, B) high-entropy bulk metallic glasses with unusual thermal stability and corrosion resistance. J. Mater. Sci. Technol. 2020, 43, 32–39. [Google Scholar] [CrossRef]
- Wang, F.; Inoue, A.; Kong, F.L.; Han, Y.; Zhu, S.L.; Shalaan, E.; Al-Marouki, F. Formation, thermal stability and mechanical properties of high entropy (Fe,Co,Ni,Cr,Mo)-B amorphous alloys. J. Alloys Compd. 2018, 732, 637–645. [Google Scholar] [CrossRef]
- Gong, P.; Wang, D.; Zhang, C.; Wang, Y.; Jamili-Shirvan, Z.; Yao, K.; Wang, X. Corrosion behavior of TiZrHfBeCu(Ni) high-entropy bulk metallic glasses in 3.5 wt. % NaCl. npj Mater. Degrad. 2022, 6, 1–14. [Google Scholar] [CrossRef]
- Panahi, S.L.; Fornell, J.; Popescu, C.; Pineda, E.; Sort, J.; Bruna, P. Structure, mechanical properties and nanocrystallization of (FeCoCrNi)-(B,Si) high-entropy metallic glasses. Intermetallics 2022, 141, 107432. [Google Scholar] [CrossRef]
- Leila Panahi, S.; Garcia-Ramón, M.; Pineda, E.; Bruna, P. New (FeCoCrNi)-(B,Si) high-entropy metallic glasses, study of the crystallization processes by X-ray diffraction and Mössbauer spectroscopy. J. Non. Cryst. Solids 2020, 547. [Google Scholar] [CrossRef]
- Ding, J.; Inoue, A.; Han, Y.; Kong, F.L.; Zhu, S.L.; Wang, Z.; Shalaan, E.; Al-Marzouki, F. High entropy effect on structure and properties of (Fe,Co,Ni,Cr)-B amorphous alloys. J. Alloys Compd. 2017, 696, 345–352. [Google Scholar] [CrossRef]
- Li, M.; Chen, Q.; Cui, X.; Peng, X.; Huang, G. Evaluation of corrosion resistance of the single-phase light refractory high entropy alloy TiCrVNb0.5Al0.5 in chloride environment. J. Alloys Compd. 2021, 857, 158278. [Google Scholar] [CrossRef]
- Shang, X.L.; Wang, Z.J.; Wu, Q.F.; Wang, J.C.; Li, J.J.; Yu, J.K. Effect of Mo Addition on Corrosion Behavior of High-Entropy Alloys CoCrFeNiMo x in Aqueous Environments. Acta Metall. Sin. Engl. Lett. 2019, 32, 41–51. [Google Scholar] [CrossRef]
- Zhang, H.; Li, W.; Xu, H.; Chen, L.; Zeng, J.; Ding, Z.; Guo, W.; Liu, B. Microstructure and Corrosion Behavior of Laser Cladding FeCoNiCrBSi Based High-Entropy Alloy Coatings. Coatings 2022, 12, 628. [Google Scholar] [CrossRef]
- Bijalwan, P.; Kumar, A.; Nayak, S.K.; Banerjee, A.; Dutta, M.; Laha, T. Microstructure and corrosion behavior of Fe-based amorphous composite coatings developed by atmospheric plasma spraying. J. Alloys Compd. 2019, 796, 47–54. [Google Scholar] [CrossRef]
- Huang, F.; Kang, J.J.; Yue, W.; Fu, Z.Q.; Zhu, L.N.; She, D.S.; Liang, J.; Wang, C.B. biao Corrosion Behavior of FeCrMoCBY Amorphous Coating Fabricated by High-Velocity Air Fuel Spraying. J. Therm. Spray Technol. 2019, 28, 842–850. [Google Scholar] [CrossRef]
- Cui, P.; Bao, Z.; Liu, Y.; Zhou, F.; Lai, Z.; Zhou, Y.; Zhu, J. Corrosion behavior and mechanism of dual phase Fe1.125Ni1.06CrAl high entropy alloy. Corros. Sci. 2022, 201, 110276. [Google Scholar] [CrossRef]
- Takeuchi, A.; Amiya, K.; Wada, T.; Yubuta, K.; Zhang, W.; Makino, A. Entropies in alloy design for high-entropy and bulk glassy alloys. Entropy 2013, 15, 3810–3821. [Google Scholar] [CrossRef]
- Kumar, N.; Fusco, M.; Komarasamy, M.; Mishra, R.S.; Bourham, M.; Murty, K.L. Understanding effect of 3.5 wt.% NaCl on the corrosion of Al0.1CoCrFeNi high-entropy alloy. J. Nucl. Mater. 2017, 495, 154–163. [Google Scholar] [CrossRef]
- Córdoba-Torres, P. Relationship between constant-phase element (CPE) parameters and physical properties of films with a distributed resistivity. Electrochim. Acta 2017, 225, 592–604. [Google Scholar] [CrossRef]
- Ulum, M.F.; Caesarendra, W.; Alavi, R.; Hermawan, H. In-vivo corrosion characterization and assessment of absorbable metal implants. Coatings 2019, 9, 282. [Google Scholar] [CrossRef]
- Sun, Y.P.; Wang, Z.; Yang, H.J.; Lan, A.D.; Qiao, J.W. Effects of the element La on the corrosion properties of CrMnFeNi high entropy alloys. J. Alloys Compd. 2020, 842, 155825. [Google Scholar] [CrossRef]
- Bar-Cohen, Y. High Temperature Materials and Mechanisms; CRC Press: Boca Raton, FL, USA, 2017; ISBN 9781138071544. [Google Scholar]
- Beke, D.L.; Erdélyi, G. On the diffusion in high-entropy alloys. Mater. Lett. 2016, 164, 111–113. [Google Scholar] [CrossRef]
- Bredar, A.R.C.; Chown, A.L.; Burton, A.R.; Farnum, B.H. Electrochemical Impedance Spectroscopy of Metal Oxide Electrodes for Energy Applications. ACS Appl. Energy Mater. 2020, 3, 66–98. [Google Scholar] [CrossRef]
- Qiu, Y.; Thomas, S.; Gibson, M.A.; Fraser, H.L.; Pohl, K.; Birbilis, N. Microstructure and corrosion properties of the low-density single-phase compositionally complex alloy AlTiVCr. Corros. Sci. 2018, 133, 386–396. [Google Scholar] [CrossRef]
- Qiu, Y.; Thomas, S.; Fabijanic, D.; Barlow, A.J.; Fraser, H.L.; Birbilis, N. Microstructural evolution, electrochemical and corrosion properties of Al x CoCrFeNiTi y high entropy alloys. Mater. Des. 2019, 170, 107698. [Google Scholar] [CrossRef]
- Koga, G.Y.; Otani, L.B.; Silva, A.M.B.; Roche, V.; Nogueira, R.P.; Jorge, A.M.; Bolfarini, C.; Kiminami, C.S.; Botta, W.J. Characterization and corrosion resistance of boron-containing-austenitic stainless steels produced by rapid solidification techniques. Materials 2018, 11, 2189. [Google Scholar] [CrossRef]
- Muangtong, P.; Rodchanarowan, A.; Chaysuwan, D.; Chanlek, N.; Goodall, R. The corrosion behaviour of CoCrFeNi-x (x = Cu, Al, Sn) high entropy alloy systems in chloride solution. Corros. Sci. 2020, 172, 108740. [Google Scholar] [CrossRef]
- Shi, Y.; Yang, B.; Liaw, P.K. Corrosion-resistant high-entropy alloys: A review. Metals 2017, 7, 43. [Google Scholar] [CrossRef]
- Hsu, Y.J.; Chiang, W.C.; Wu, J.K. Corrosion behavior of FeCoNiCrCux high-entropy alloys in 3.5% sodium chloride solution. Mater. Chem. Phys. 2005, 92, 112–117. [Google Scholar] [CrossRef]
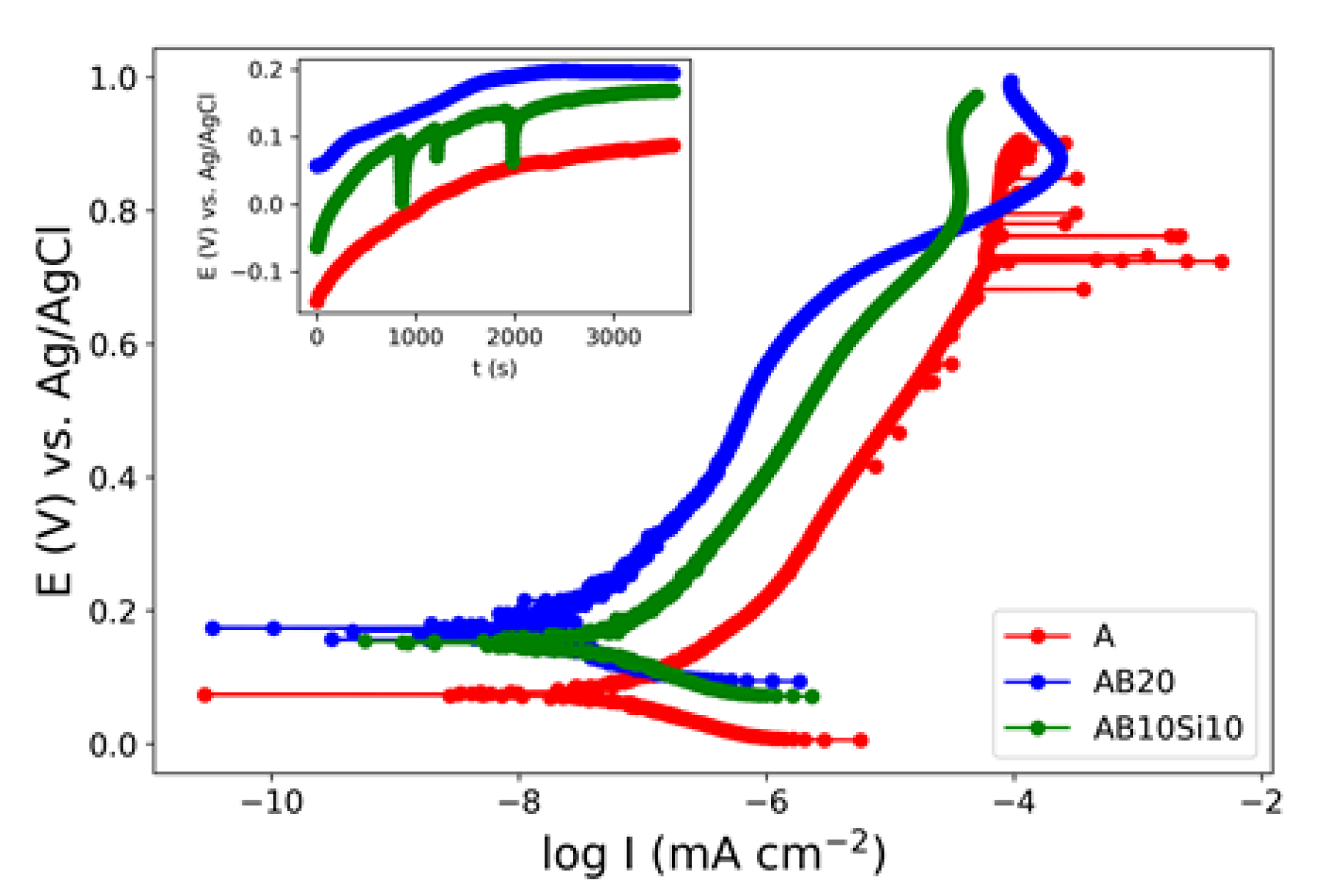



| Alloy | Ecorr (mV) | Icorr (nA cm−2) |
|---|---|---|
| A | 70 | 0.09 |
| AB20 | 170 | 0.06 |
| AB10Si10 | 150 | 0.06 |
| Alloy | R1 (Ω) | R2 (KΩ) | C2 (F) | Q1 (F s(a−1)) | a | W1 (Ω s−1/2) |
|---|---|---|---|---|---|---|
| A | 33.45 | 1149 | 1.697 × 10−6 | 1.335 × 10−6 | 0.906 | 3.219 × 106 |
| AB20 | 34.41 | 21,310 | 0.854 × 10−6 | 0.991 × 10−6 | 0.913 | 1.844 × 106 |
| AB10Si10 | 100.4 | 258,800 | 0.156 × 10−6 | 0.200 × 10−6 | 0.927 | 5.543 × 106 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Panahi, S.L.; Bruna, P.; Pineda, E. Effect of Si and B on the Electrochemical Behavior of FeCoNiCr-Based High-Entropy Amorphous Alloys. Materials 2022, 15, 8897. https://doi.org/10.3390/ma15248897
Panahi SL, Bruna P, Pineda E. Effect of Si and B on the Electrochemical Behavior of FeCoNiCr-Based High-Entropy Amorphous Alloys. Materials. 2022; 15(24):8897. https://doi.org/10.3390/ma15248897
Chicago/Turabian StylePanahi, S. Leila, Pere Bruna, and Eloi Pineda. 2022. "Effect of Si and B on the Electrochemical Behavior of FeCoNiCr-Based High-Entropy Amorphous Alloys" Materials 15, no. 24: 8897. https://doi.org/10.3390/ma15248897
APA StylePanahi, S. L., Bruna, P., & Pineda, E. (2022). Effect of Si and B on the Electrochemical Behavior of FeCoNiCr-Based High-Entropy Amorphous Alloys. Materials, 15(24), 8897. https://doi.org/10.3390/ma15248897







