Activation of Waste Materials with Carbon(IV) Oxide as an Effective Method of Obtaining Biochars of Attractive Sorption Properties towards Liquid and Gas Pollutants
Abstract
1. Introduction
2. Material and Method
2.1. Materials and Chemical Reagents
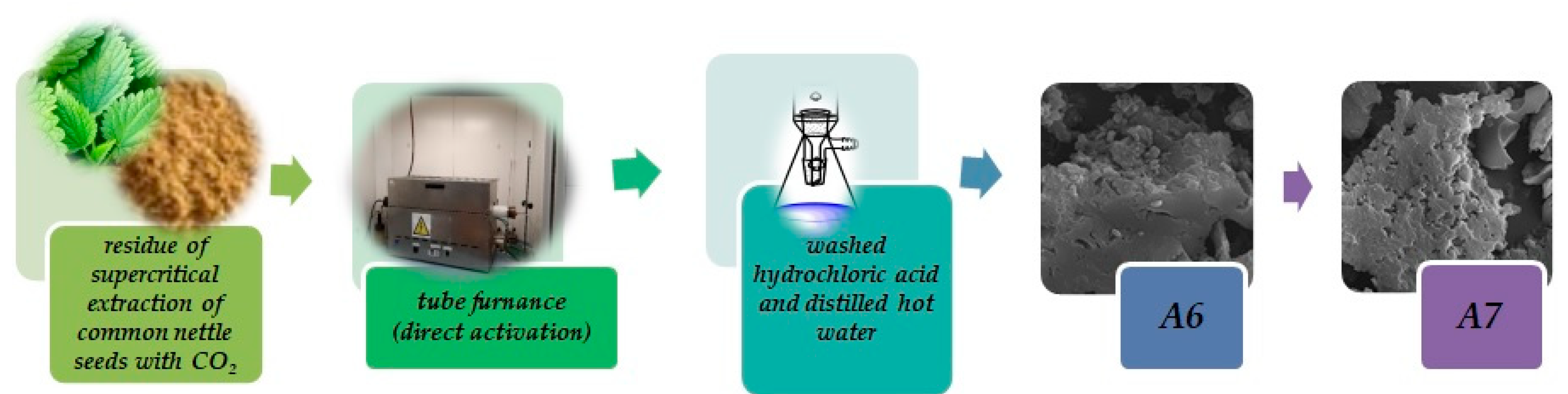
2.2. Preparation of Biochars
2.3. Characterization of Precursor and Biochars
2.3.1. Elemental Analysis
2.3.2. Low-Temperature Nitrogen Sorption
2.3.3. Scanning Electron Microscope (SEM)
2.3.4. Acidic and Basic Character of the Precursor and Biochar Surfaces
2.4. Adsorption Studies
2.4.1. NO2Adsorption
2.4.2. Methylene Blue (MB) Adsorption
- C0—initial concentration of MB solution (mg/L);
- Ce—MB concentration remaining in solution at equilibrium(mg/L);
- M—mass of biochar(g);
- V—volume of the solution (L).
- Ce—concentrations of MB in the solution (mg/L);
- qe—adsorption capacity (mg/g);
- qmax—MB monolayer adsorption capacity (mg/g);
- KL—Langmuir adsorption constant (L/mg);
- nF—Freundlich adsorption constant related to adsorption capacity;
- KF—Freundlich adsorption constant related to sorption intensity (mg/g (mg/L)1/n).
- qt—amount of MB adsorbed at time t (mg/g);
- k1—pseudo-first-order model rate constant of adsorption (1/min);
- k2—pseudo-second-order model rate constant of adsorption (g/mg×min).
- T—sorption temperature (K);
- R—gas constant (83,144 J/K×mol);
- KC—thermodynamic equilibrium constant expressed by qe/Ce.
3. Results and Discussion
3.1. Characterization of the Precursor and Biochars
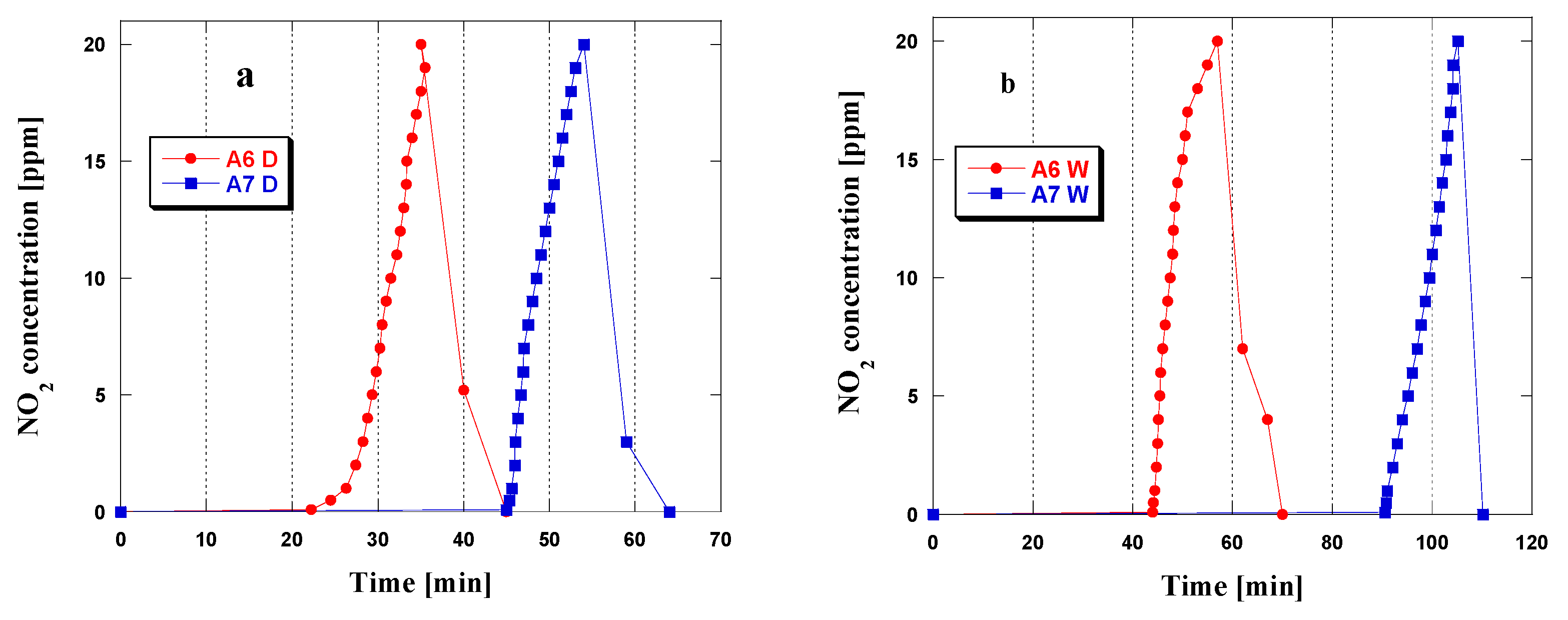
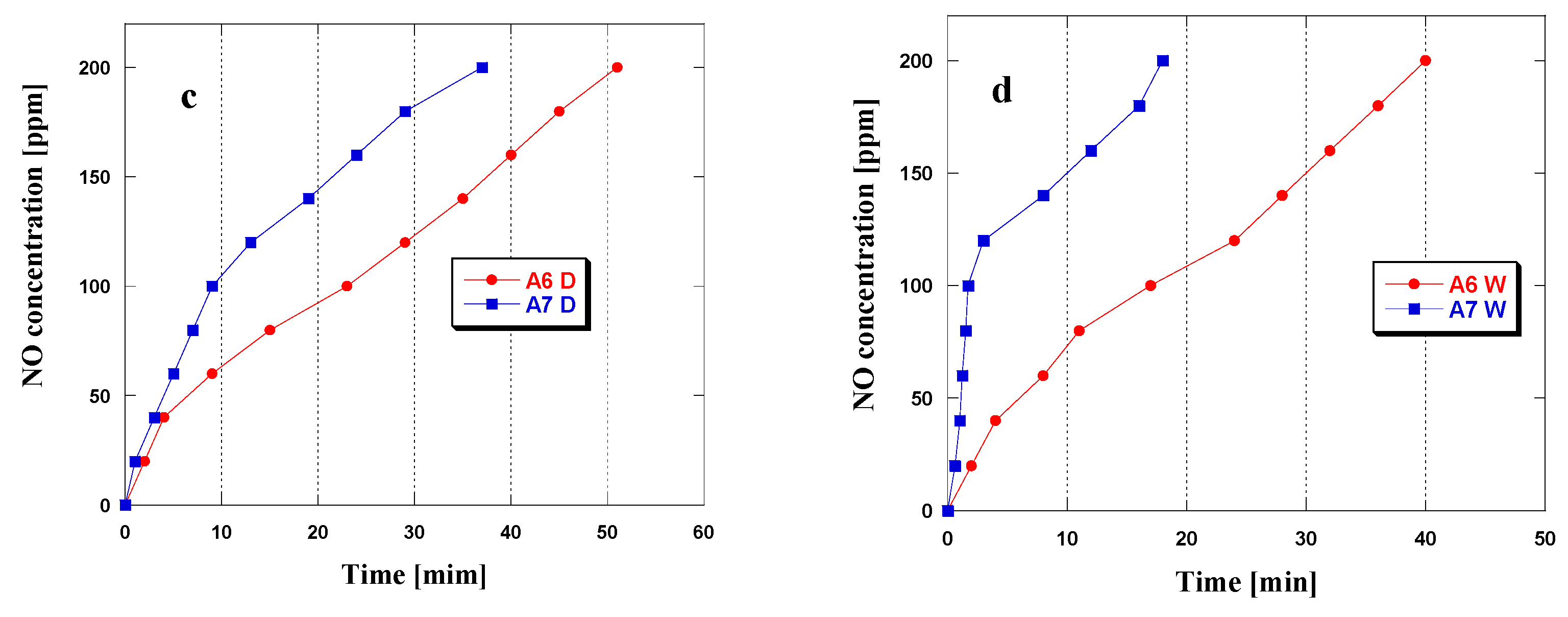
3.2. NO2Adsorption
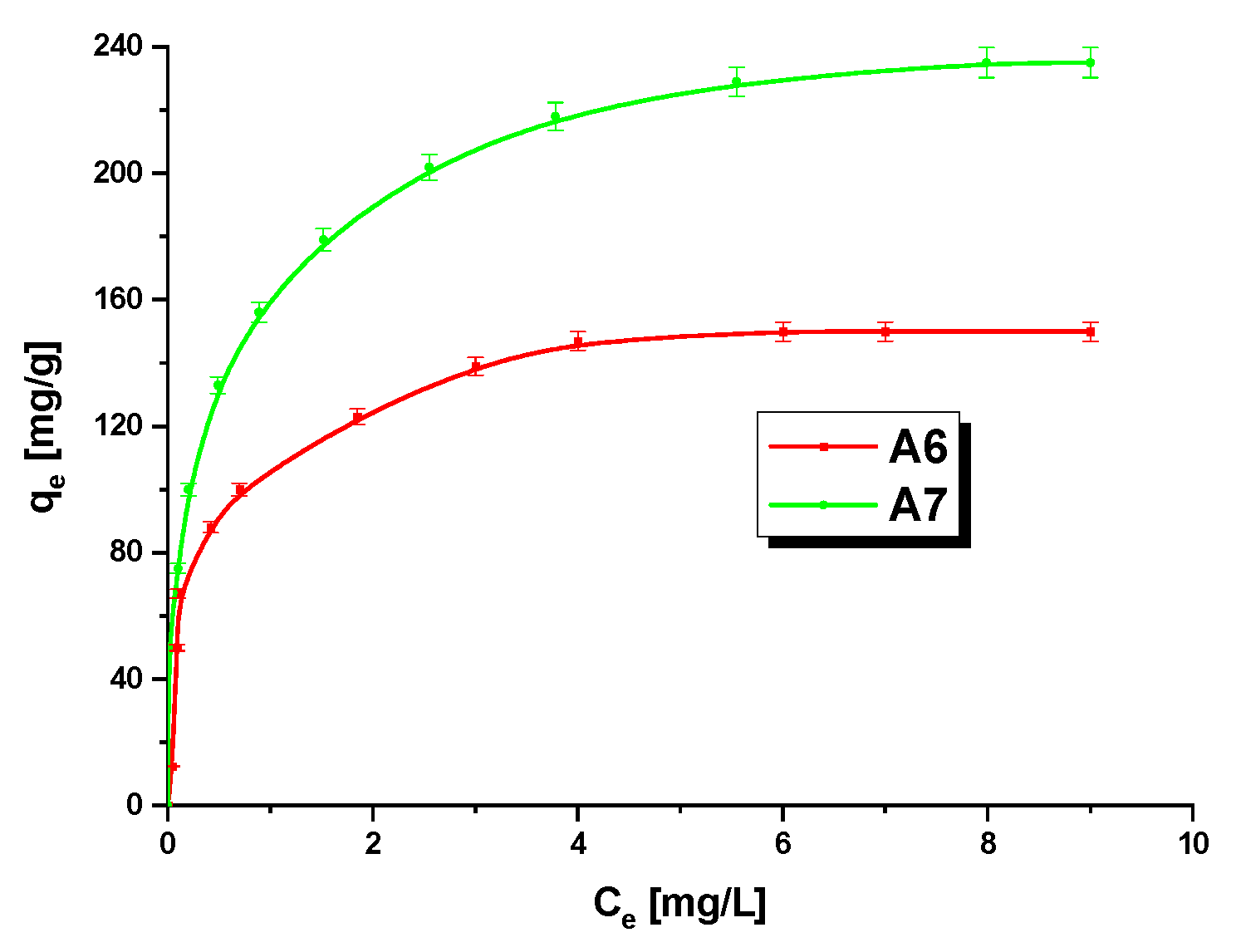
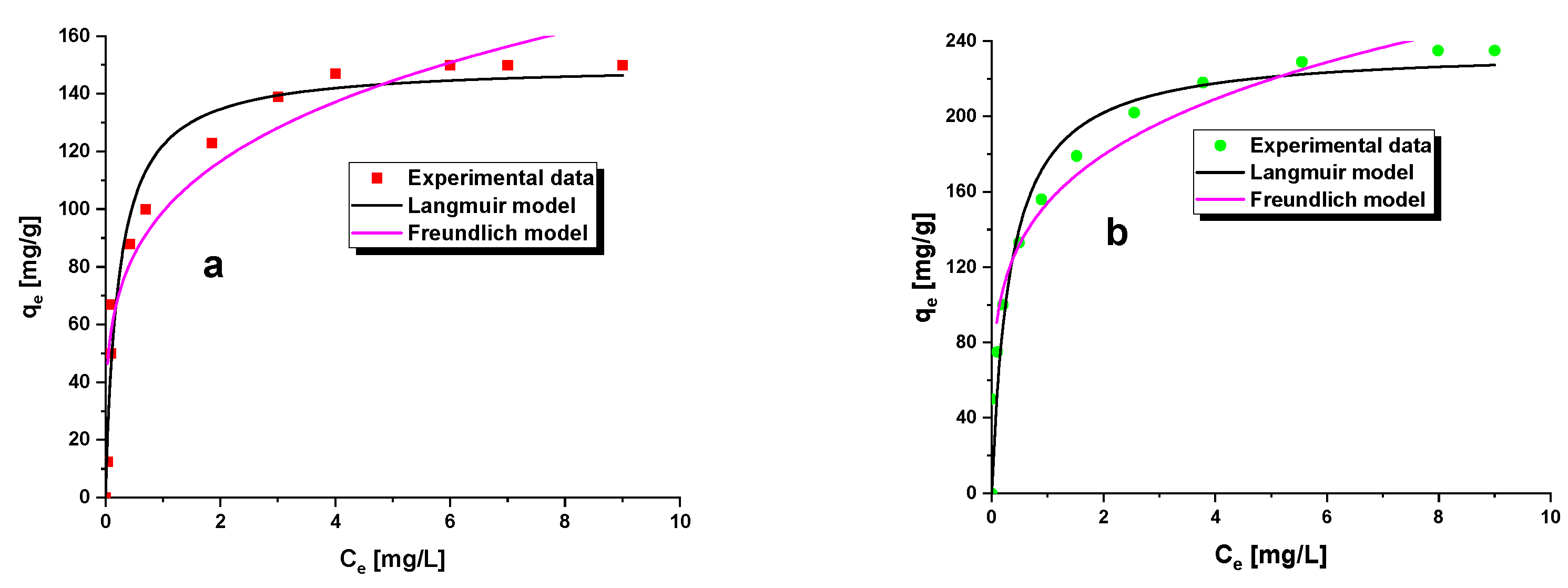
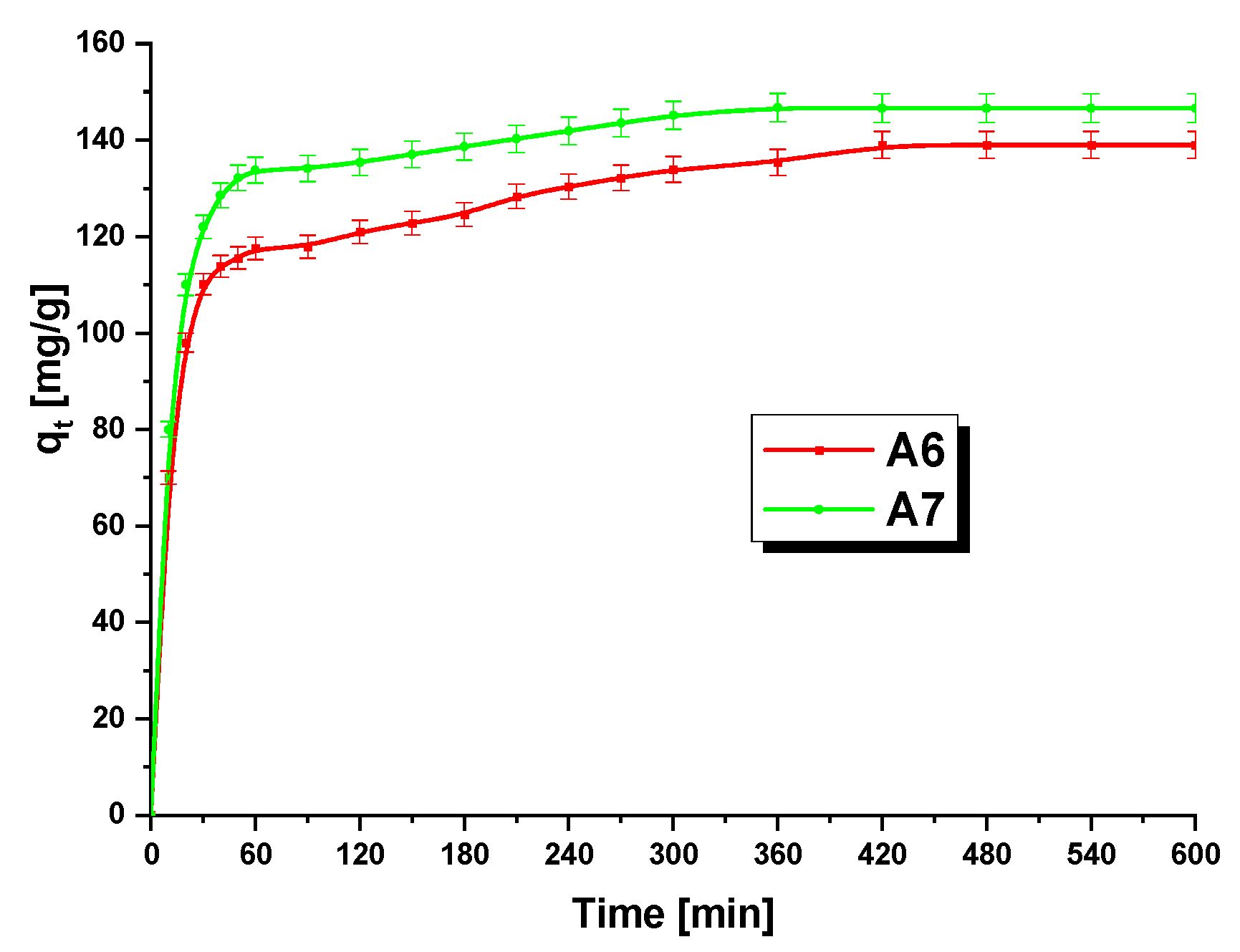
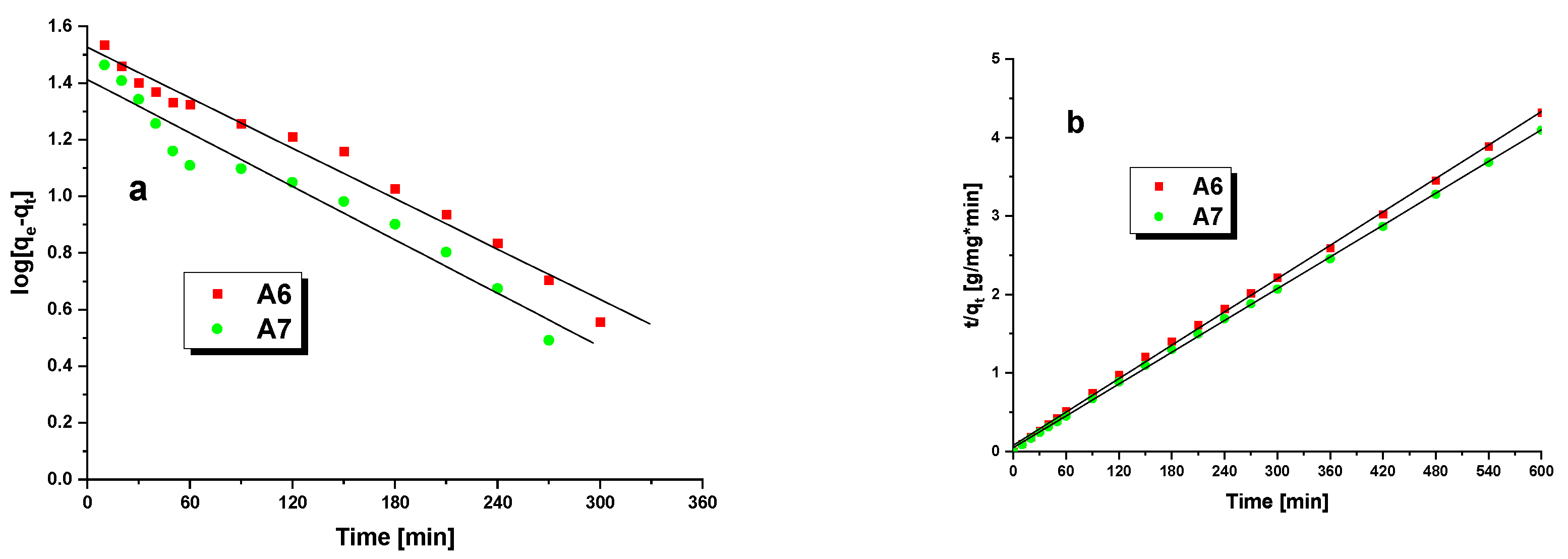
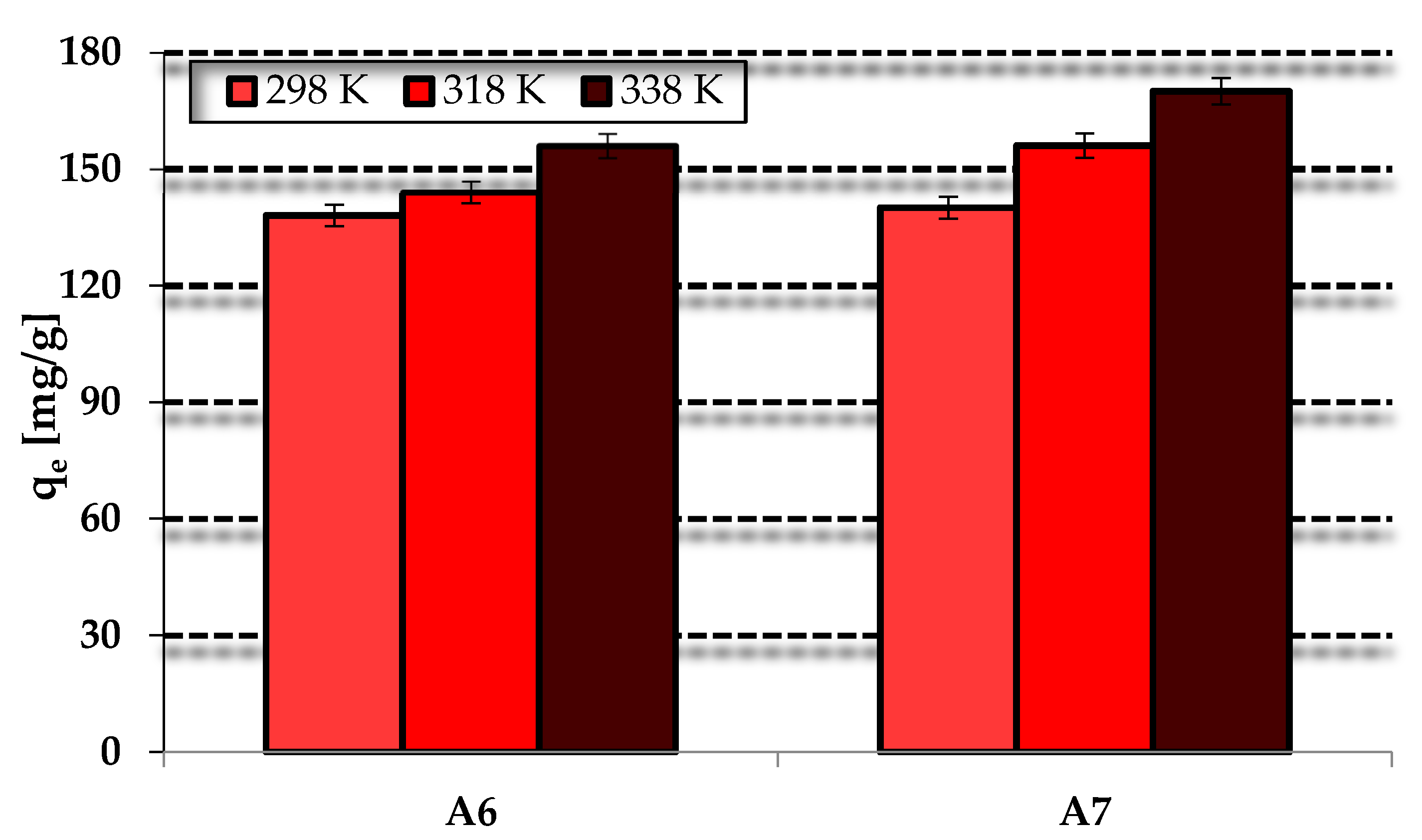
3.3. Methylene BlueAdsorption
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Crominski da Silva Medeirosa, D.C.; Chelme-Ayala, P.; Benally, C.; Al-Anzi, B.S.; El-Din, M.G. Review on carbon-based adsorbents from organic feedstocks for removal of organic contaminants from oil and gas industry process water: Production, adsorption performance and research gaps. J. Environ. Manag. 2022, 320, 115739. [Google Scholar]
- Gabruś, E.; Tabero, P.; Aleksandrzak, T. A study of the thermal regeneration of carbon and zeolite adsorbents after adsorption of 1-hexene vapor. Appl. Therm. Eng. 2022, 216, 119065. [Google Scholar] [CrossRef]
- Gayathiri, M.; Pulingam, T.; Lee, K.T.; Sudesh, K. Activated carbon from biomass waste precursors: Factors affecting production and adsorption mechanism. Chemosphere 2022, 294, 133764. [Google Scholar] [CrossRef] [PubMed]
- Zakaria, R.; Jamalluddin, N.A.; Abu Bakar, M.Z. Effect of impregnation ratio and activation temperature on the yield and adsorption performance of mangrove based activated carbon for methylene blue removal. Results Mat. 2021, 10, 100183. [Google Scholar] [CrossRef]
- Kumar, A.; Jena, H.M. Adsorption of Cr(VI) from aqueous solution by prepared high surface area activated carbon from Fox nutshell by chemical activation with H3PO4. J. Environ. Chem. Eng. 2017, 5, 2032–2041. [Google Scholar] [CrossRef]
- Yan, L.; Dong, F.-X.; Li, Y.; Guo, P.-R.; Kong, L.-J.; Chu, W.; Diao, Z.-H. Synchronous removal of Cr(VI) and phosphates by a novel crayfish shell biochar-Fe composite from aqueous solution: Reactivity and mechanism. J. Environ. Chem. Eng. 2022, 10, 107396. [Google Scholar] [CrossRef]
- Dong, F.-X.; Yan, L.; Zhou, X.-H.; Huang, S.-T.; Liang, J.-Y.; Zhang, W.-X.; Guo, Z.-W.; Guo, P.-R.; Qian, W.; Kong, L.-J.; et al. Simultaneous adsorption of Cr(VI) and phenol by biochar-based iron oxide composites in water: Performance, kinetics and mechanism. J. Hazard. Mater. 2021, 416, 125930. [Google Scholar] [CrossRef]
- Diao, Z.-H.; Dong, F.-X.; Yan, L.; Chen, Z.-L.; Qian, W.; Kong, L.-J.; Zhang, Z.-W.; Zhang, T.; Tao, X.-Q.; Du, J.-L.; et al. Synergistic oxidation of Bisphenol A in a heterogeneous ultrasound-enhanced sludge biochar catalyst/persulfate process: Reactivity and mechanism. J. Hazard. Mater. 2020, 384, 121385. [Google Scholar] [CrossRef]
- Chen, D.; Sawut, A.; Wang, T. Synthesis of new functionalized magnetic nano adsorbents and adsorption performance for Hg(II) ions. Heliyon 2022, 8, e10528. [Google Scholar] [CrossRef]
- Prasety, N.; Li, K. Synthesis of defective MOF-801 via an environmentally benign approach for diclofenac removal from water streams. Sep. Purif. Technol. 2022, 301, 122024. [Google Scholar] [CrossRef]
- Bazan, A.; Nowicki, P.; Pietrzak, R. Removal of NO2 by carbonaceous adsorbents obtained from residue after supercritical extraction of marigold. Adsorption 2016, 22, 465–471. [Google Scholar] [CrossRef][Green Version]
- Nowicki, P.; Pietrzak, R.; Wachowska, H. Comparison of physicochemical properties of nitrogen-enriched activated carbons prepared by physical and chemical activation of brown coal. Energy Fuels 2008, 22, 4133–4138. [Google Scholar] [CrossRef]
- Ghouma, I.; Jeguirim, M.; Dorge, S.; Limousy, L.; Matei Ghimbeu, C.; Ouederni, A. Activated carbon prepared by physical activation of olive stones for the removal of NO2 at ambient temperature. C. R. Chim. 2015, 18, 63–74. [Google Scholar] [CrossRef]
- Bergna, D.; Varila, T.; Romar, H.; Lassi, U. Comparison of the properties of activated carbons produced in one-stage and two-stage processes. C 2018, 4, 41. [Google Scholar] [CrossRef]
- Ahmadpour, A.; Do, D.D. The preparation of active carbons from coal by chemical and physical activation. Carbon 1996, 34, 471–479. [Google Scholar] [CrossRef]
- Tonoya, T.; Matsui, Y.; Hinago, H.; Ishikawa, M. Microporous activated carbon derived from azulmic acid precursor with high sulfur loading and its application to lithium-sulfur battery cathode. Electrochem. Commun. 2022, 140, 10733. [Google Scholar] [CrossRef]
- Bazan-Wozniak, A.; Cielecka-Piontek, J.; Nosal-Wiercińnska, A.; Pietrzak, R. Adsorption of Organic Compounds on Adsorbents Obtained with the Use of Microwave Heating. Materials 2022, 15, 5664. [Google Scholar] [CrossRef]
- Reza, M.S.; Hasan, A.K.; Afroze, S.; Bakar, M.S.A.; Taweekun, J.; Azad, A.K. Analysis on preparation, application, and recycling of activated carbon to aid in COVID-19 protection. Int. J. Integr. Eng. 2020, 12, 233–244. [Google Scholar] [CrossRef]
- Kazmierczak-Razna, J.; Nowicki, P.; Pietrzak, R. Characterization and application of bio-activated carbons prepared by direct activation of hay with the use of microwave radiation. Powder Technol. 2017, 319, 302–312. [Google Scholar] [CrossRef]
- Heidarinejad, Z.; Dehghani, M.H.; Heidari, M.; Javedan, G.; Ali, I.; Sillanpää, M. Methods for preparation and activation of activated carbon: A review. Environ. Chem. Lett. 2020, 18, 393–415. [Google Scholar] [CrossRef]
- Puchana-Rosero, M.J.; Adebayo, M.A.; Lima, E.C.; Machado, F.M.; Thue, P.S.; Vaghetti, J.C.P.; Gutterres, M. Microwave-assisted activated carbon obtained from the sludge of tannery-treatment effluent plant for removal of leather dyes. Colloids Surf. A Physicochem. Eng. Asp. 2016, 504, 105–115. [Google Scholar] [CrossRef]
- Jing, J.; Zhao, Z.; Zhang, X.; Feng, J.; Li, W. CO2 capture over activated crbon derived from pulverized semi-coke. Separations 2022, 9, 174. [Google Scholar] [CrossRef]
- Hoang, A.T.; Kumar, S.; Lichtfouse, E.; Cheng, C.K.; Varma, R.S.; Senthilkumar, N.; Nguyen, P.Q.P.; Nguyen, X.P. Remediation of heavy metal polluted waters using activated carbon from lignocellulosic biomass: An update of recent trends. Chemosphere 2022, 302, 134825. [Google Scholar] [CrossRef] [PubMed]
- Hoang, A.T.; Goldfarb, J.L.; Foley, A.M.; Lichtfouse, E.; Kumar, M.; Xiao, L.; Ahmed, S.F.; Said, Z.; Luque, R.; Bui, V.G.; et al. Production of biochar from crop residues and its application for anaerobic digestion. Bioresour. Technol. 2022, 363, 127970. [Google Scholar] [CrossRef] [PubMed]
- Bazan-Wozniak, A.; Nowicki, P.; Pietrzak, R. Production of new activated bio-carbons by chemical activation of residue left after supercritical extraction of hops. Environ. Res. 2018, 161, 456–463. [Google Scholar] [CrossRef]
- Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef]
- Freundlich, H.M.F. Over the Adsorption in Solution. J. Phys. Chem. 1906, 57, 385–470. [Google Scholar]
- Revellame, E.D.; Fortela, D.L.; Sharp, W.; Hernandez, R.; Zappi, M.E. Adsorption kinetic modeling using pseudo-first order and pseudo-second order rate laws: A review. Clean. Eng. Technol. 2020, 1, 100032. [Google Scholar] [CrossRef]
- Marciniak, M.; Goscianska, J.; Norman, M.; Jesionowski, T.; Bazan-Wozniak, A.; Pietrzak, R. Equilibrium, kinetic, and thermodynamic studies on adsorption of rhodamine B from aqueous solutions using oxidized mesoporous carbons. Materials 2022, 15, 5573. [Google Scholar] [CrossRef]
- Bansal, R.C.H.; Goyal, M. Activated Carbon Adsorption, 1st ed.; Taylor & Francis Group: Boca Raton, FL, USA, 2005; pp. 263–277. [Google Scholar]
- Martínez-Mendoza, K.L.; Barraza Burgos, J.M.; Marriaga-Cabrales, N.; Machuca-Martinez, F.; Barajas, M.; Romero, M. Production and characterization of activated carbon from coal for gold adsorption in cyanide solutions. Ing. Investig. 2020, 40, 34–44. [Google Scholar] [CrossRef]
- Bazan-Wozniak, A.; Nowicki, P.; Pietrzak, R. The influence of activation procedure on the physicochemical and sorption properties of activated carbons prepared from pistachio nutshells for removal of NO2/H2S gases and dyes. J. Clean. Prod. 2017, 152, 211–222. [Google Scholar] [CrossRef]
- Neme, I.; Gonfa, G.; Mas, C. Preparation and characterization of activated carbon from castor seed hull by chemical activation with H3PO4. Results Mat. 2022, 15, 100303. [Google Scholar] [CrossRef]
- Zubrik, A.; Matik, M.; Hredzák, S.; Lovás, M.; Danková, Z.; Kováčová, M.; Briančin, J. Preparation of chemically activated carbon from waste biomass by single-stage and two-stage pyrolysis. J. Clean. Prod. 2017, 143, 643–653. [Google Scholar] [CrossRef]
- Khalil, S.H.; Aroua, M.K.; Daud, W.M.A.W. Study on the improvement of the capacity of amine-impregnated commercial activated carbon beds for CO2 adsorbing. Chem. Eng. J. 2012, 183, 15–20. [Google Scholar] [CrossRef]
- Djilani, C.; Zaghdoudi, R.; Djazi, F.; Bouchekima, B.; Lallam, A.; Modarressi, A.; Rogalski, M. Adsorption of dyes on activated carbon prepared from apricot stones and commercial activated carbon. J. Taiwan Inst. Chem. Eng. 2015, 53, 112–121. [Google Scholar] [CrossRef]
- Nowicki, P.; Skrzypczak, M.; Pietrzak, R. Effect of activation method on the physicochemical properties and NO2 removal abilities of sorbents obtained from plum stones (Prunus domestica). Chem. Eng. J. 2010, 162, 723–729. [Google Scholar] [CrossRef]
- Jeguirim, M.; Tschamber, V.; Brilhac, J.F.; Ehrburger, P. Oxidation mechanism of carbon black by NO2: Effect of water vapour. Fuel 2005, 84, 1949–1956. [Google Scholar] [CrossRef]
- Pietrzak, R.; Bandosz, T.J. Activated carbons modified with sewage sludge derived phase and their application in the process of NO2 removal. Carbon 2007, 45, 2537–2546. [Google Scholar] [CrossRef]
- Ebrahim, A.M.; Bandosz, T.J. Effect of amine modification on the properties of zirconium–carboxylic acid based materials andtheir applications as NO2 adsorbents at ambient conditions. Microporous Mesoporous Mater. 2014, 188, 149–162. [Google Scholar] [CrossRef]
- Kazmierczak-Razna, J.; Gralak-Podemska, B.; Nowicki, P.; Pietrzak, R. The use of microwave radiation for obtaining activated carbons from sawdust and their potential application in removal of NO2 and H2S. Chem. Eng. J. 2015, 269, 352–358. [Google Scholar] [CrossRef]
- Hofman, M.; Pietrzak, R. Adsorbents obtained from waste tires forNO2 removal under dry conditions at room temperature. Chem. Eng. J. 2011, 170, 202–208. [Google Scholar] [CrossRef]
- Mittal, H.; Alili, A.A.; Morajkar, P.P.; Alhassan, S.M. Graphene oxide crosslinked hydrogel nanocomposites of xanthan gum for the adsorption of crystal violet dye. J. Mol. Liq. 2021, 321, 115034. [Google Scholar] [CrossRef]
- Medhat, A.; El-Maghrabi, H.H.; Abdelghany, A.; Abdel Menem, N.M.; Raynaud, P.; Moustafa, Y.M.; Nada, A.A. Efficiently activated carbons from corn cob for methylene blue adsorption. Apply. Surf Sci. Advan. 2021, 3, 100037. [Google Scholar] [CrossRef]
- Yao, X.; Ji, L.; Guo, J.; Ge, S.; Lu, W.; Chen, Y.; Song, W. An abundant porous biochar material derived from wakame (Undaria pinnatifida) with high adsorption performance for three organic dyes. Bioresour. Technol. 2020, 318, 124082. [Google Scholar] [CrossRef]
- Alswieleh, A.M. Efficient Removal of Dyes from Aqueous Solution by Adsorption on L-Arginine-Modified Mesoporous Silica Nanoparticles. Processes 2022, 10, 1079. [Google Scholar] [CrossRef]
- AlOthman, Z.A.; Habila, M.A.; Ali, R.; Abdel Ghafar, A.; El-din Hassouna, M.S. Valorization of two waste streams into activated carbon and studying its adsorption kinetics, equilibrium isotherms and thermodynamics for methylene blue removal. Arab. J. Chem. 2014, 7, 1148–1158. [Google Scholar] [CrossRef]
- Ho, Y.S.; McKay, G. Sorption of dye from aqueous solution by peat. Chem. Eng. J. 1998, 70, 115–124. [Google Scholar] [CrossRef]
- Su, L.; Zhang, H.; Oh, K.; Liu, N.; Luo, Y.; Cheng, H.; Zhang, G.; He, X. Activated biochar derived from spent Auricularia auricula substrate for the efficient adsorption of cationic azo dyes from single and binary adsorptive systems. Water Sci. Technol. 2021, 84, 101–121. [Google Scholar] [CrossRef]
- Soldatkina, L.; Yanar, M. Equilibrium, Kinetic, and Thermodynamic Studies of Cationic Dyes Adsorption on Corn Stalks Modified by Citric Acid. Colloids Interfaces 2021, 5, 52. [Google Scholar] [CrossRef]
- Zhang, H.; Peng, B.; Liu, Q.; Wu, C.; Li, W. Preparation of porous biochar from heavy bio-oil for adsorption of methylene blue in wastewater. Fuel Process. Technol. 2022, 238, 107485. [Google Scholar] [CrossRef]
- Patawat, C.; Silakate, K.; Chuan-Udom, S.; Supanchaiyamat, N.; Hunt, Y.A.; Ngernyen, J. Preparation of activated carbon from dipterocarpusalatus fruit and its application for methylene blue adsorption. RSC Adv. 2020, 10, 21082–21091. [Google Scholar] [CrossRef] [PubMed]
- Lv, B.; Dong, B.; Zhang, C.; Chen, Z.; Zhao, Z.; Deng, Z.; Fang, C. Effective adsorption of methylene blue from aqueous solution by coal gangue-based zeolite granules in a fluidized bed: Fluidization characteristics and continuous adsorption. Powder Technol. 2022, 408, 117764. [Google Scholar] [CrossRef]
- Soury, R.; Jabli, M.; Latif, S.; Alenezi, K.M.; El Oudi, M.; Abdulaziz, F.; Teka, S.; El Moll, H.; Haque, A. Synthesis and characterization of a new meso-tetrakis (2,4,6-trimethylphenyl) porphyrinto) zinc(II) supported sodium alginate gel beads for improved adsorption of methylene blue dye. Int. J. Biol. Macromol. 2022, 202, 161–176. [Google Scholar] [CrossRef] [PubMed]
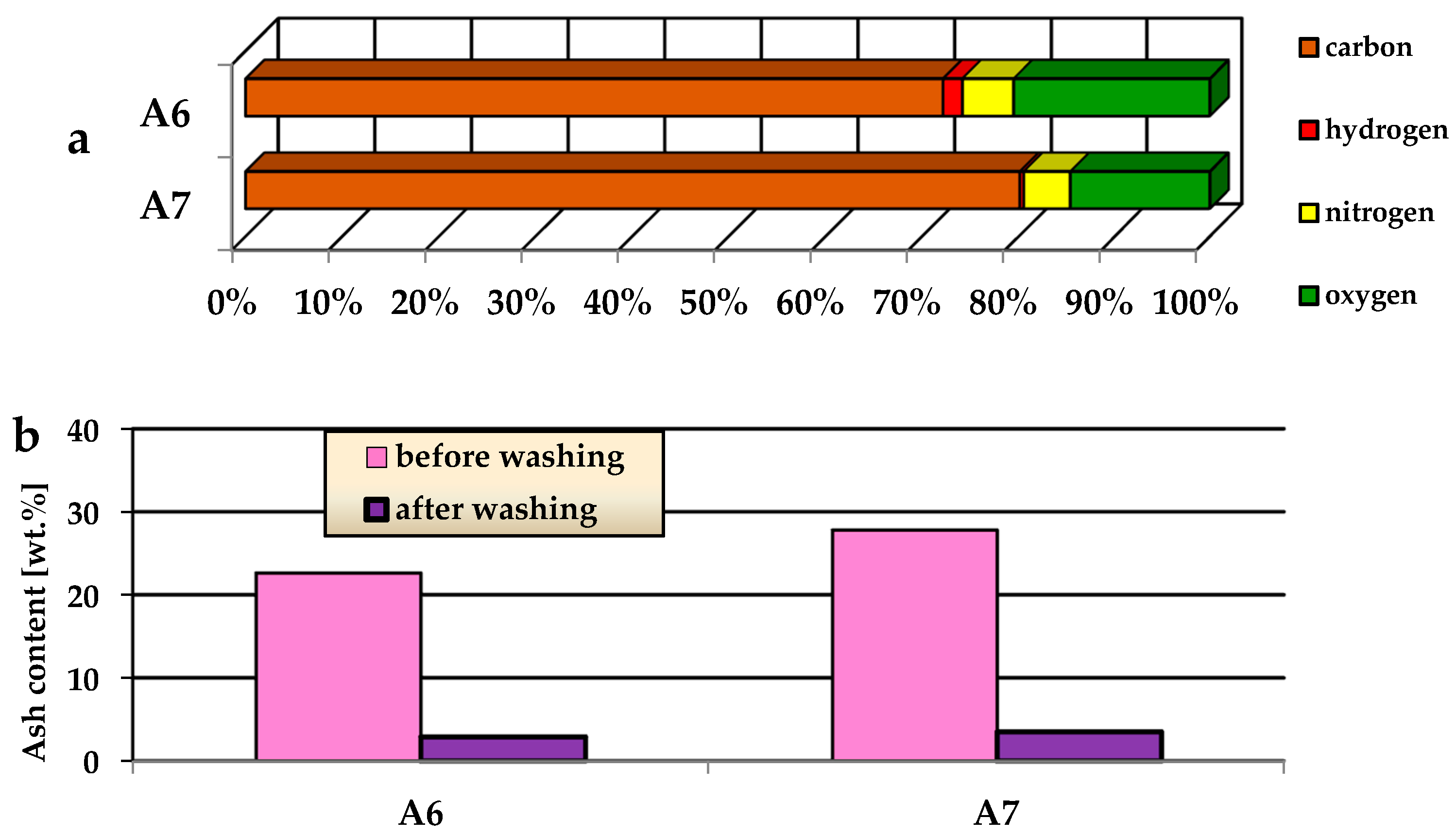

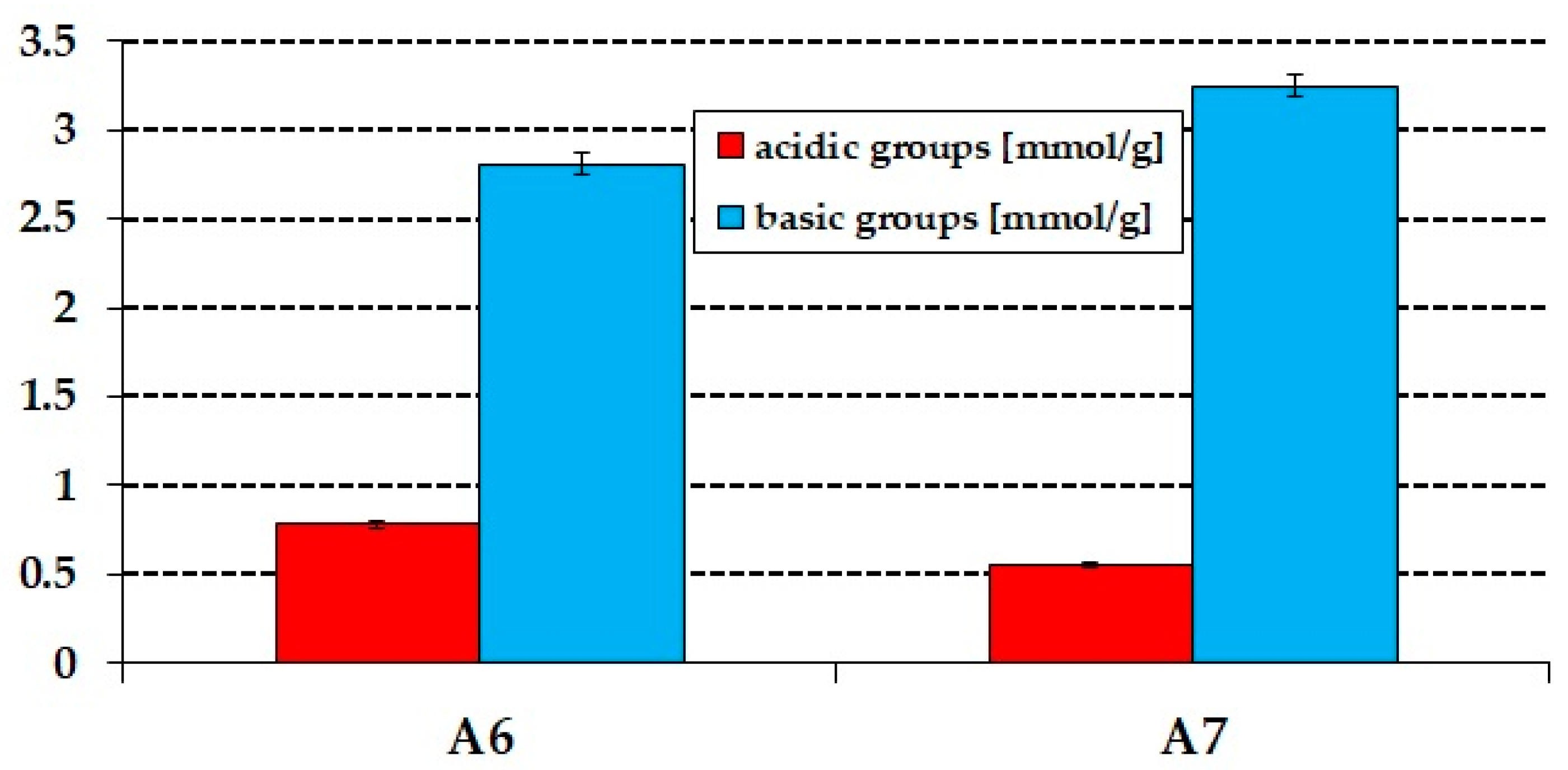
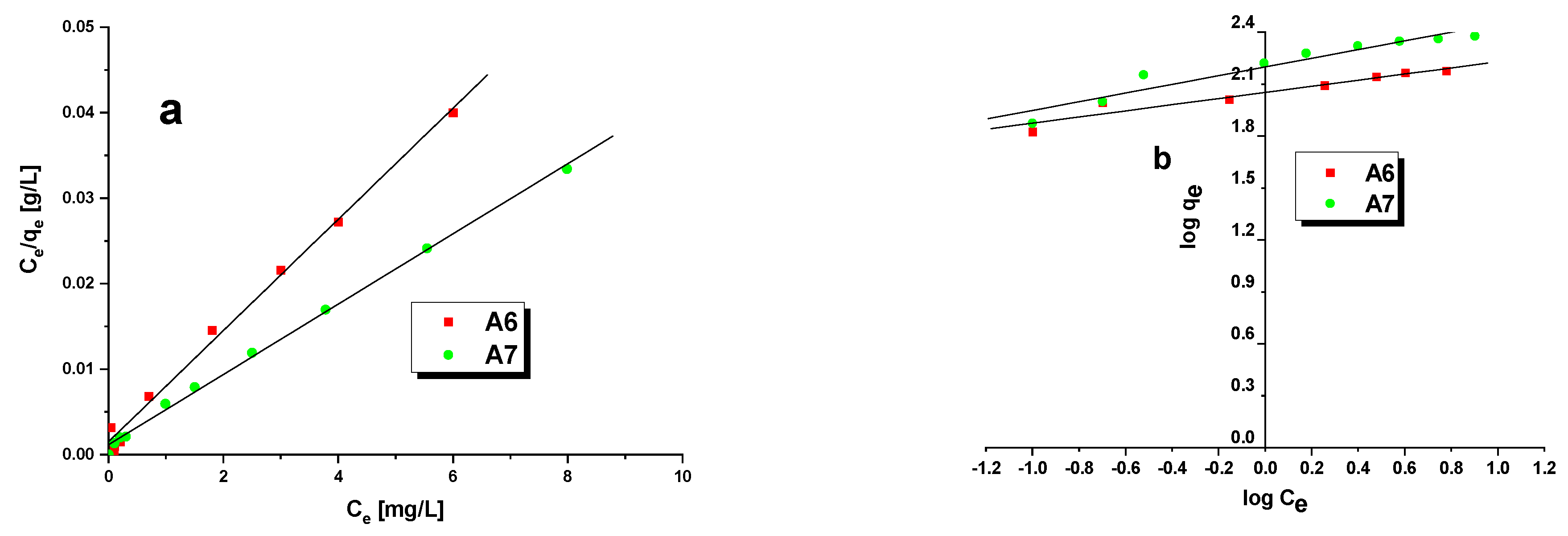
| Sample | Ash | Cdaf,1 | Hdaf | Ndaf | Sdaf | Odaf,2 | Acidic Groups (mmol/g) | Basic Groups (mmol/g) |
|---|---|---|---|---|---|---|---|---|
| precursor | 3.2 | 49.4 | 9.9 | 4.6 | 0.1 | 36.0 | 2.01 | 0.48 |
| Biochar | Surface Area 1 (m2/g) | Total Pore Volume (cm3/g) | Average Pore Diameter (nm) | Micropore Area (m2/g) |
|---|---|---|---|---|
| A6 | 888 | 0.55 | 4.1 | 689 |
| A7 | 1024 | 0.73 | 3.5 | 903 |
| Biochar | Dry Conditions [mg/g] | Wet Conditions [mg/g] |
|---|---|---|
| A6 | 20.1 | 42.8 |
| A7 | 28.7 | 59.1 |
| Material/Sample | Adsorption Capacity (mg/g) | References |
|---|---|---|
| marigold | 102.1 | [11] |
| hops | 155.3 | [25] |
| zirconium—carboxylic ligand | 154 | [40] |
| sawdust pellets | 54.7 | [41] |
| waste tires | 11.4 | [42] |
| A7 | 59.1 | (This study) |
| Isotherms | Parameters | A6 | A7 |
|---|---|---|---|
| Langmuir | R2 | 0.9950 | 0.9967 |
| qm(mg/g) | 153.85 | 243.90 | |
| KL(L/mg) | 0.0281 | 0.0168 | |
| RL | 0.3086–0.8772 | 0.3511–0.7485 | |
| Freundlich | R2 | 0.9215 | 0.9240 |
| KF(mg/g(L/mg)1/n) | 112.90 | 158.45 | |
| 1/n | 0.1771 | 0.2512 |
| Isotherms | Parameters | A6 | A7 |
|---|---|---|---|
| Langmuir | R2 | 0.9677 | 0.9631 |
| qm(mg/g) | 152.07 | 240.73 | |
| KL(L/mg) | 4.3612 | 2.9817 | |
| Freundlich | R2 | 0.9117 | 0.9458 |
| KF(mg/g(L/mg)1/n) | 99.01 | 154.12 | |
| 1/n | 0.2351 | 0.2204 |
| Isotherms | Parameters | A6 | A7 |
|---|---|---|---|
| qe,exp(mg/g) | 138.99 | 146.60 | |
| Pseudo-first-order | R2 | 0.9769 | 0.9358 |
| k1(1/min) | 0.0059 | 0.0083 | |
| qe,cal(mg/g) | 35.61 | 27.69 | |
| Pseudo-second-order | R2 | 0.9993 | 0.9998 |
| k2(g/mg×min) | 0.0007 | 0.0009 | |
| qe,cal(mg/g) | 140.85 | 149.25 |
| Sample | Temperature (K) | ΔG (kJ/mol) | ΔH (kJ/mol) | ΔS (J/mol × K) |
|---|---|---|---|---|
| A6 | 298 | −8.77 | 16.99 | 86.13 |
| 318 | −10.23 | |||
| 338 | −12.24 | |||
| A7 | 298 | −11.59 | 21.91 | 111.91 |
| 318 | −13.35 | |||
| 338 | −16.11 |
| Material/Sample | Adsorption Capacity (mg/g) | References |
|---|---|---|
| heavy bio-oil | 411 | [51] |
| Dipterocarpus alatus fruit | 269 | [52] |
| coal gangue-based zeolite granules | 108 | [53] |
| meso-tetrakis(2,4,6-trimethylphenyl) porphyrinto) zinc(II) supported sodium alginate gel beads | 52 | [54] |
| A7 | 239 | (This study) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bazan-Wozniak, A.; Cielecka-Piontek, J.; Nosal-Wiercińska, A.; Pietrzak, R. Activation of Waste Materials with Carbon(IV) Oxide as an Effective Method of Obtaining Biochars of Attractive Sorption Properties towards Liquid and Gas Pollutants. Materials 2022, 15, 8000. https://doi.org/10.3390/ma15228000
Bazan-Wozniak A, Cielecka-Piontek J, Nosal-Wiercińska A, Pietrzak R. Activation of Waste Materials with Carbon(IV) Oxide as an Effective Method of Obtaining Biochars of Attractive Sorption Properties towards Liquid and Gas Pollutants. Materials. 2022; 15(22):8000. https://doi.org/10.3390/ma15228000
Chicago/Turabian StyleBazan-Wozniak, Aleksandra, Judyta Cielecka-Piontek, Agnieszka Nosal-Wiercińska, and Robert Pietrzak. 2022. "Activation of Waste Materials with Carbon(IV) Oxide as an Effective Method of Obtaining Biochars of Attractive Sorption Properties towards Liquid and Gas Pollutants" Materials 15, no. 22: 8000. https://doi.org/10.3390/ma15228000
APA StyleBazan-Wozniak, A., Cielecka-Piontek, J., Nosal-Wiercińska, A., & Pietrzak, R. (2022). Activation of Waste Materials with Carbon(IV) Oxide as an Effective Method of Obtaining Biochars of Attractive Sorption Properties towards Liquid and Gas Pollutants. Materials, 15(22), 8000. https://doi.org/10.3390/ma15228000







