A Novel Dual-Emission Fluorescence Probe Based on CDs and Eu3+ Functionalized UiO-66-(COOH)2 Hybrid for Visual Monitoring of Cu2+
Abstract
1. Introduction
2. Experimental Details
2.1. Reagents and Instruments
2.2. Synthesis of UiO-66-(COOH)2 (Denoted as F1)
2.3. One-Pot Synthesis of CDs@Eu-UiO-66(COOH)2 (CDs-F2)
2.4. Luminescence Sensing Experiments
2.5. Preparation of Fluorescent Films
3. Result and Discussion
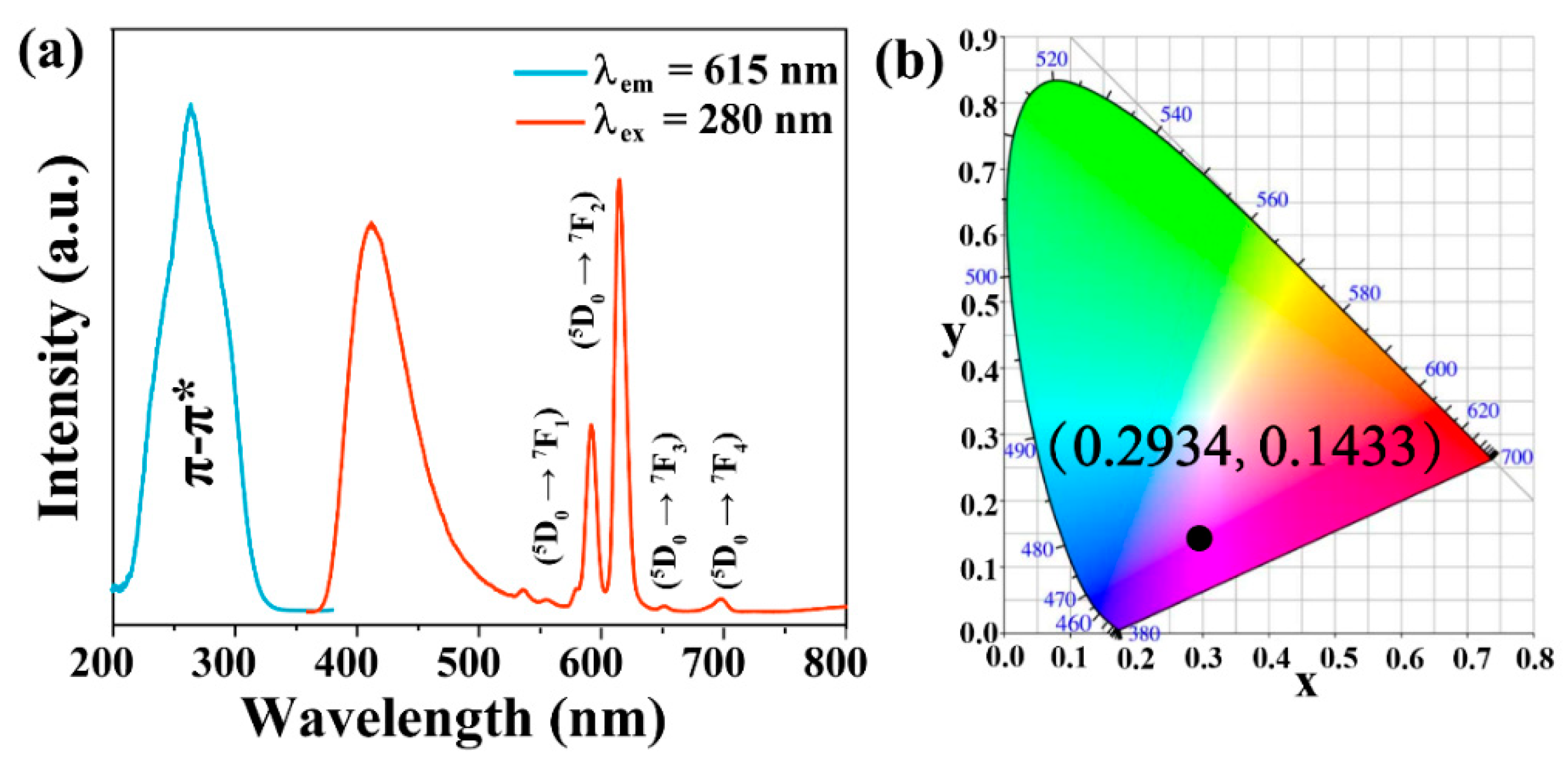
3.1. Optimization of the Fluorescence Properties
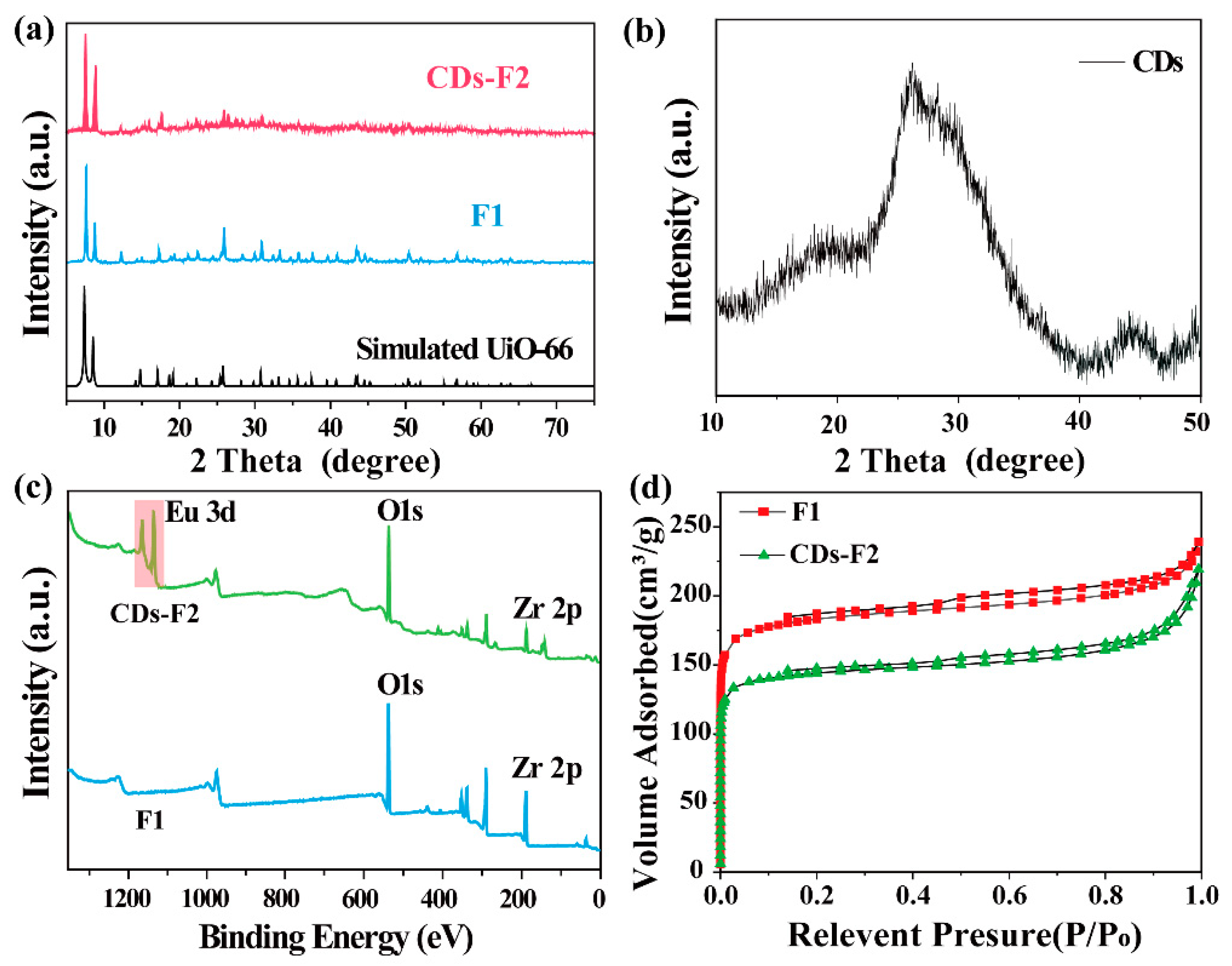
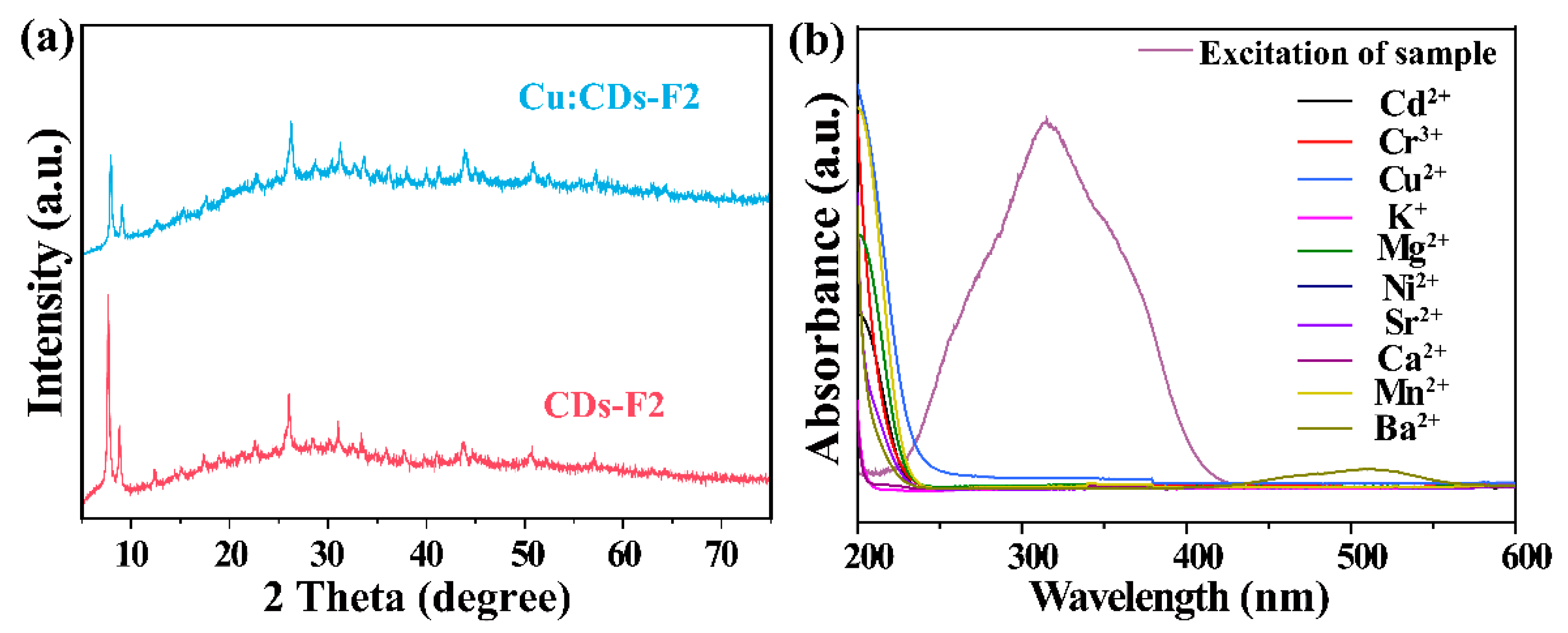
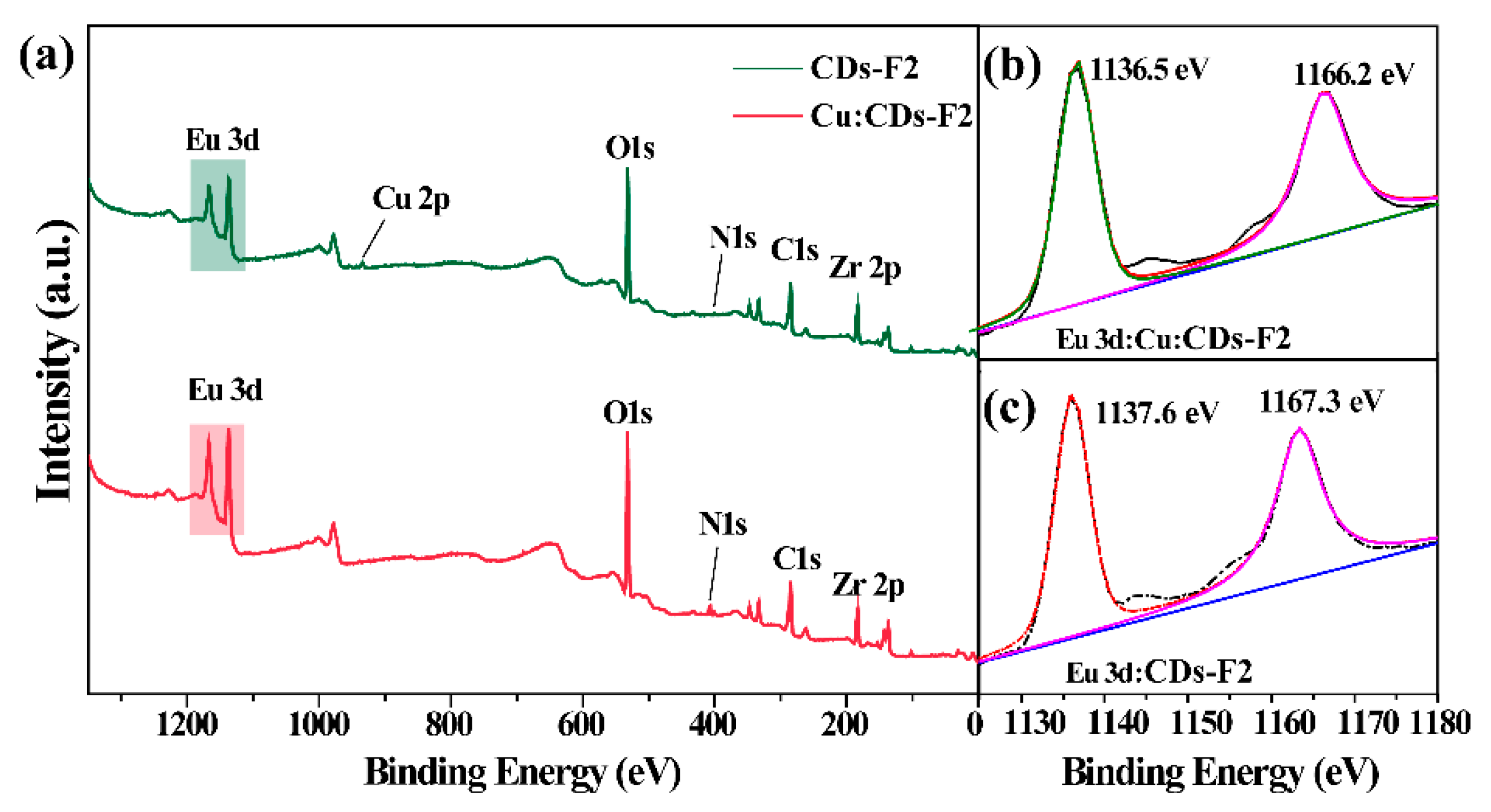
3.2. Characterizations
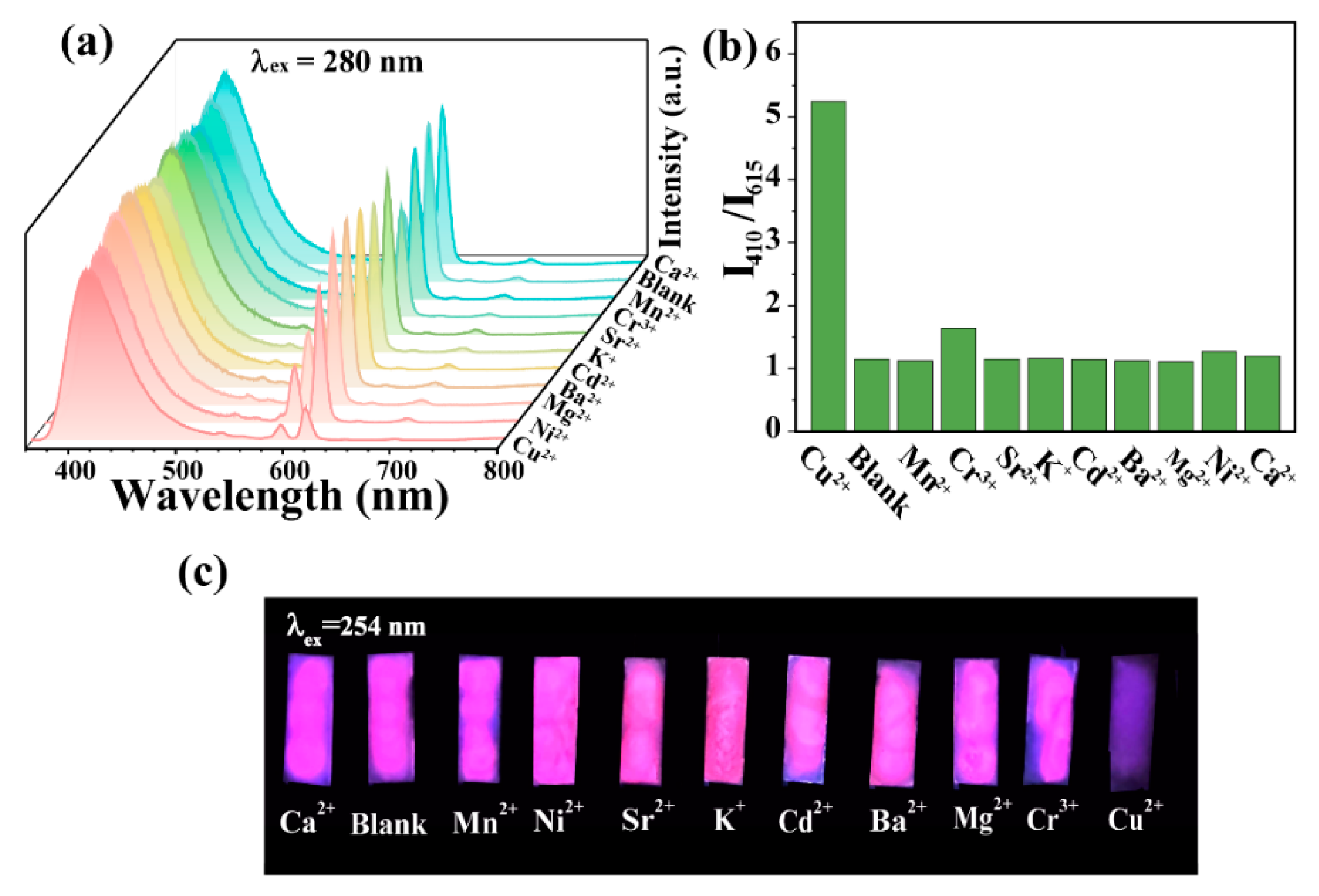
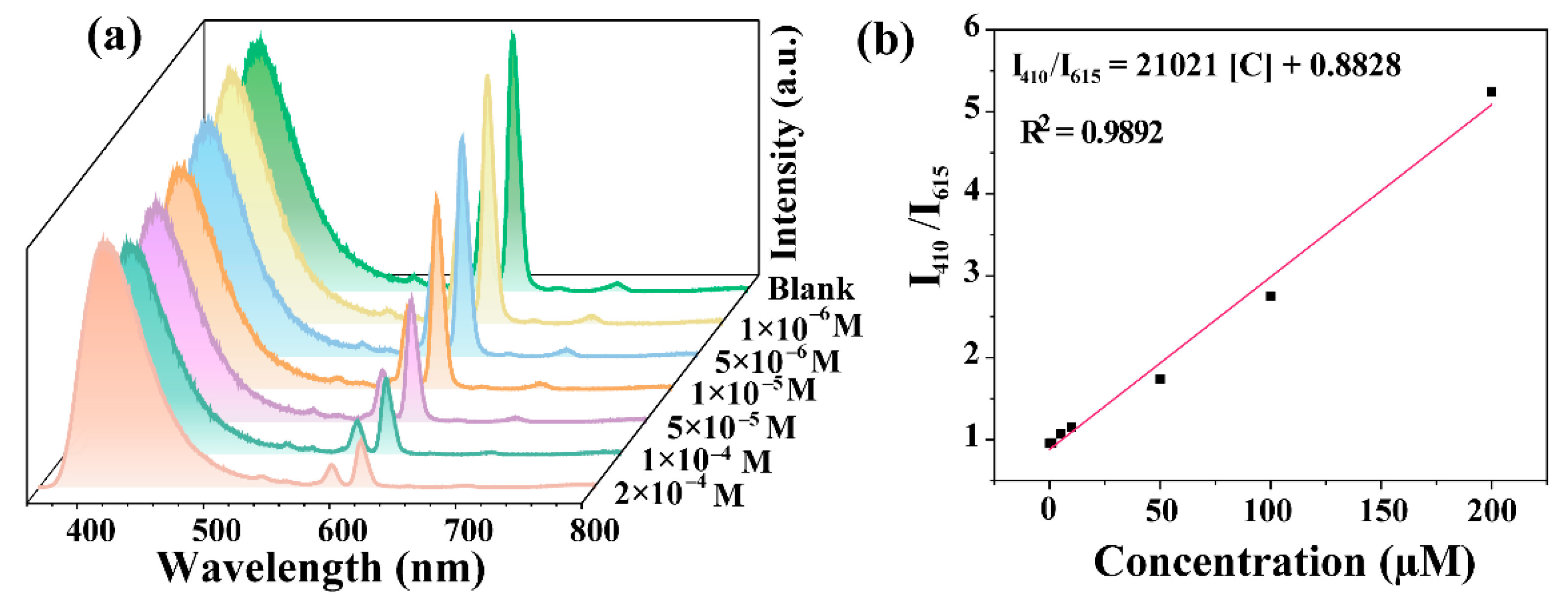
3.3. Fluorescence Sensing for Cu2+
3.4. Possible Sensing Mechanism of CDs-F2 for Cu2+
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Erdemir, S.; Malkondu, S. Dual-emissive fluorescent probe based on phenolphthalein appended diaminomaleonitrile for Al3+ and the colorimetric recognition of Cu2+. Dye. Pigm. 2019, 163, 330–336. [Google Scholar] [CrossRef]
- Liu, H.; Cui, S.; Shi, F.; Pu, S. A diarylethene based multi-functional sensor for fluorescent detection of Cd2+ and colorimetric detection of Cu2+. Dye. Pigm. 2019, 161, 34–43. [Google Scholar] [CrossRef]
- Wu, F.; Wang, J.; Pu, C.; Qiao, L.; Jiang, C. Wilson’s Disease: A Comprehensive Review of the Molecular Mechanisms. Int. J. Mol. Sci. 2015, 16, 6419–6431. [Google Scholar] [CrossRef] [PubMed]
- Roy, D.; Chakraborty, A.; Ghosh, R. Perimidine based selective colorimetric and fluorescent turn-off chemosensor of aqueous Cu2+: Studies on its antioxidant property along with its interaction with calf thymus-DNA. RSC Adv. 2017, 7, 40563–40570. [Google Scholar] [CrossRef]
- Arain, S.A.; Kazi, T.G.; Afridi, H.I.; Abbasi, A.R.; Panhwar, A.H.; Naeemullah; Shanker, B.; Arain, M.B. Application of dual-cloud point extraction for the trace levels of copper in serum of different viral hepatitis patients by flame atomic absorption spectrometry: A multivariate study. Spectrochim. Acta A 2014, 133, 651–656. [Google Scholar] [CrossRef]
- Li, J.-J.; Ji, C.-H.; Hou, C.-J.; Huo, D.-Q.; Zhang, S.-Y.; Luo, X.-G.; Yang, M.; Fa, H.-B.; Deng, B. High efficient adsorption and colorimetric detection of trace copper ions with a functional filter paper. Sens. Actuators B 2016, 223, 853–860. [Google Scholar] [CrossRef]
- Xie, Y.-L.; Zhao, S.-Q.; Ye, H.-L.; Yuan, J.; Song, P.; Hu, S.-Q. Graphene/CeO2 hybrid materials for the simultaneous electrochemical detection of cadmium(II), lead(II), copper(II), and mercury(II). J. Electroanal. Chem. 2015, 757, 235–242. [Google Scholar] [CrossRef]
- Zhao, L.; Zhong, S.; Fang, K.; Qian, Z.; Chen, J. Determination of cadmium(II), cobalt(II), nickel(II), lead(II), zinc(II), and copper(II) in water samples using dual-cloud point extraction and inductively coupled plasma emission spectrometry. J. Hazard. Mater. 2012, 239–240, 206–212. [Google Scholar] [CrossRef]
- He, L.; Dong, B.; Liu, Y.; Lin, W. Fluorescent chemosensors manipulated by dual/triple interplaying sensing mechanisms. Chem. Soc. Rev. 2016, 45, 6449–6461. [Google Scholar] [CrossRef]
- Yang, J.; Che, J.; Jiang, X.; Fan, Y.; Gao, D.; Bi, J.; Ning, Z. A Novel Turn-On Fluorescence Probe Based on Cu(II) Functionalized Metal–Organic Frameworks for Visual Detection of Uric Acid. Molecules 2022, 27, 4803. [Google Scholar] [CrossRef]
- Zeng, X.; Zhou, Q.; Wang, L.; Zhu, X.; Cui, K.; Peng, X.; Steele, T.W.J.; Chen, H.; Xu, H.; Zhou, Y. A Fluorescence Kinetic-Based Aptasensor Employing Stilbene Isomerization for Detection of Thrombin. Materials 2021, 14, 6927. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Pei, Y.; Qian, X.; An, X. Eu-metal organic framework@TEMPO-oxidized cellulose nanofibrils photoluminescence film for detecting copper ions. Carbohydr. Polym. 2020, 236, 116030. [Google Scholar] [CrossRef] [PubMed]
- Qian, X.; Deng, S.; Chen, X.; Gao, Q.; Hou, Y.-L.; Wang, A.; Chen, L. A highly stable, luminescent and layered zinc(II)-MOF: Iron(III)/copper(II) dual sensing and guest-assisted exfoliation. Chin. Chem. Lett. 2020, 31, 2211–2214. [Google Scholar] [CrossRef]
- Yin, Y.; Yang, J.; Pan, Y.; Gao, Y.; Huang, L.; Luan, X.; Lin, Z.; Zhu, W.; Li, Y.; Song, Y. Mesopore to Macropore Transformation of Metal–Organic Framework for Drug Delivery in Inflammatory Bowel Disease. Adv. Healthc. Mater. 2021, 10, 2000973. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Zhang, X.; Huang, H.; Zhang, Z.; Yildirim, T.; Zhou, W.; Xiang, S.; Chen, B. A microporous aluminum-based metal-organic framework for high methane, hydrogen, and carbon dioxide storage. Nano Res. 2021, 14, 507–511. [Google Scholar] [CrossRef]
- Feng, L.; Dong, C.; Li, M.; Li, L.; Jiang, X.; Gao, R.; Wang, R.; Zhang, L.; Ning, Z.; Gao, D.; et al. Terbium-based metal-organic frameworks: Highly selective and fast respond sensor for styrene detection and construction of molecular logic gate. J. Hazard. Mater. 2020, 388, 121816. [Google Scholar] [CrossRef]
- Zhou, X.; Liu, L.; Niu, Y.; Song, M.; Feng, Y.; Lu, J.; Tai, X. A Water-Stable Zn-MOF Used as Multiresponsive Luminescent Probe for Sensing Fe3+/Cu2+, Trinitrophenol and Colchicine in Aqueous Medium. Materials 2022, 15, 7006. [Google Scholar] [CrossRef]
- Wang, K.; Qian, M.; Qi, H.; Gao, Q.; Zhang, C. Single Particle-Based Confocal Laser Scanning Microscopy for Visual Detection of Copper Ions in Confined Space. Chin. J. Chem. 2021, 39, 1804–1810. [Google Scholar] [CrossRef]
- Jiang, Q.; Wang, Z.; Li, M.; Song, J.; Yang, Y.; Xu, X.; Xu, H.; Wang, S. A nopinone based multi-functional probe for colorimetric detection of Cu2+ and ratiometric detection of Ag+. Photochem. Photobiol. Sci. 2020, 19, 49–55. [Google Scholar] [CrossRef]
- Li, Y.; Qi, S.; Xia, C.; Xu, Y.; Duan, G.; Ge, Y. A FRET ratiometric fluorescent probe for detection of Hg2+ based on an imidazo[1,2-a]pyridine-rhodamine system. Anal. Chim. Acta 2019, 1077, 243–248. [Google Scholar] [CrossRef]
- Lohar, S.; Dhara, K.; Roy, P.; Sinha Babu, S.P.; Chattopadhyay, P. Highly Sensitive Ratiometric Chemosensor and Biomarker for Cyanide Ions in the Aqueous Medium. ACS Omega 2018, 3, 10145–10153. [Google Scholar] [CrossRef] [PubMed]
- Raj, P.; Lee, S.-y.; Lee, T.Y. Carbon Dot/Naphthalimide Based Ratiometric Fluorescence Biosensor for Hyaluronidase Detection. Materials 2021, 14, 1313. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Dong, C.; Yang, J.; Yang, T.; Bai, F.; Ning, Z.; Gao, D.; Bi, J. Solvothermal synthesis of La-based metal-organic frameworks and their color-tunable photoluminescence properties. J. Mater. Sci. Mater. Electron. 2021, 32, 9903–9911. [Google Scholar] [CrossRef]
- Li, Z.; Sun, W.; Chen, C.; Guo, Q.; Li, X.; Gu, M.; Feng, N.; Ding, J.; Wan, H.; Guan, G. Deep eutectic solvents appended to UiO-66 type metal organic frameworks: Preserved open metal sites and extra adsorption sites for CO2 capture. Appl. Surf. Sci. 2019, 480, 770–778. [Google Scholar] [CrossRef]
- Huang, J.; Tian, B.; Wang, J.; Wang, Y.; Lu, W.; Li, Q.; Jin, L.; Li, C.; Wang, Z. Controlled synthesis of 3D flower-like MgWO4:Eu3+ hierarchical structures and fluorescence enhancement through introduction of carbon dots. CrystEngComm 2018, 20, 608–614. [Google Scholar] [CrossRef]
- Zhang, Y.; Yan, B. A portable self-calibrating logic detector for gradient detection of formaldehyde based on luminescent metal organic frameworks. J. Mater. Chem. C 2019, 7, 5652–5657. [Google Scholar] [CrossRef]
- Wang, H.; Li, Y.; Ning, Z.; Huang, L.; Zhong, C.; Wang, C.; Liu, M.; Lai, X.; Gao, D.; Bi, J. A novel red phosphor LixNa1-xEu(WO4)2 solid solution: Influences of Li/Na ratio on the microstructures and luminescence properties. J. Lumin. 2018, 201, 364–371. [Google Scholar] [CrossRef]
- Singhal, P.; Vats, B.G.; Jha, S.K.; Neogy, S. Green, Water-Dispersible Photoluminescent On–Off–On Probe for Selective Detection of Fluoride Ions. ACS Appl. Mater. 2017, 9, 20536–20544. [Google Scholar] [CrossRef]
- Zhang, M.; Zhai, X.; Sun, M.; Ma, T.; Huang, Y.; Huang, B.; Du, Y.; Yan, C. When rare earth meets carbon nanodots: Mechanisms, applications and outlook. Chem. Soc. Rev. 2020, 49, 9220–9248. [Google Scholar] [CrossRef]
- Wang, Y.; Hong, F.; Yu, L.; Xu, H.; Liu, G.; Dong, X.; Yu, W.; Wang, J. Construction, energy transfer, tunable multicolor and luminescence enhancement of YF3:RE3+(RE = Eu, Tb)/carbon dots nanocomposites. J. Lumin. 2020, 221, 117072. [Google Scholar] [CrossRef]
- Xiaoxiong, Z.; Wenjun, Z.; Cuiliu, L.; Xiaohong, Q.; Chengyu, Z. Eu3+-Postdoped UIO-66-Type Metal–Organic Framework as a Luminescent Sensor for Hg2+ Detection in Aqueous Media. Inorg. Chem. 2019, 58, 3910–3915. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.; Lv, R.; Su, J.; Li, H.; Yang, B.; Gu, W.; Liu, X. A dual-emission nano-rod MOF equipped with carbon dots for visual detection of doxycycline and sensitive sensing of MnO4−. Rsc. Adv. 2018, 8, 4766–4772. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Luo, J.; Deng, L.; Ma, F.; Yang, M. In Situ Incorporation of Fluorophores in Zeolitic Imidazolate Framework-8 (ZIF-8) for Ratio-Dependent Detecting a Biomarker of Anthrax Spores. Anal. Chem. 2020, 92, 7114–7122. [Google Scholar] [CrossRef]
- Lan, S.; Wang, X.; Liu, Q.; Bao, J.; Yang, M.; Fa, H.; Hou, C.; Huo, D. Fluorescent sensor for indirect measurement of methyl parathion based on alkaline-induced hydrolysis using N-doped carbon dots. Talanta 2019, 192, 368–373. [Google Scholar] [CrossRef]
- Huo, Q.; Tu, W.; Guo, L. Enhanced photoluminescence property and broad color emission of ZnGa2O4 phosphor due to the synergistic role of Eu3+ and carbon dots. Opt. Mater. 2017, 72, 305–312. [Google Scholar] [CrossRef]
- Xia, C.; Xu, Y.; Cao, M.-M.; Liu, Y.-P.; Xia, J.-F.; Jiang, D.-Y.; Zhou, G.-H.; Xie, R.-J.; Zhang, D.-F.; Li, H.-L. A selective and sensitive fluorescent probe for bilirubin in human serum based on europium(III) post-functionalized Zr(IV)-Based MOFs. Talanta 2020, 212, 120795. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.-X.; Bao, G.-M.; Zhong, Y.-F.; He, J.-X.; Zeng, L.; Yuan, H.-Q. Highly selective detection of Cu2+ in aqueous media based on Tb3+-functionalized metal-organic framework. Spectrochim. Acta A 2020, 240, 118621. [Google Scholar] [CrossRef] [PubMed]
- Qu, X.-L.; Yan, B. Stable Tb(III)-Based Metal–Organic Framework: Structure, Photoluminescence, and Chemical Sensing of 2-Thiazolidinethione-4-carboxylic Acid as a Biomarker of CS2. Inorg. Chem. 2019, 58, 524–534. [Google Scholar] [CrossRef]
- Wang, B.; Yan, B. A turn-on fluorescence probe Eu3+ functionalized Ga-MOF integrated with logic gate operation for detecting ppm-level ciprofloxacin (CIP) in urine. Talanta 2020, 208, 120438. [Google Scholar] [CrossRef]
- Fu, Y.; Pang, X.-X.; Wang, Z.-Q.; Chai, Q.; Ye, F. A highly sensitive and selective fluorescent probe for determination of Cu (II) and application in live cell imaging. Spectrochim. Acta A. 2019, 208, 198–205. [Google Scholar] [CrossRef]
- Dong, C.-L.; Li, M.-F.; Yang, T.; Feng, L.; Ai, Y.-W.; Ning, Z.-L.; Liu, M.-J.; Lai, X.; Gao, D.-J. Controllable synthesis of Tb-based metal–organic frameworks as an efficient fluorescent sensor for Cu2+ detection. Rare Met. 2021, 40, 505–512. [Google Scholar] [CrossRef]
- Guo, Y.; Wang, L.; Zhuo, J.; Xu, B.; Li, X.; Zhang, J.; Zhang, Z.; Chi, H.; Dong, Y.; Lu, G. A pyrene-based dual chemosensor for colorimetric detection of Cu2+ and fluorescent detection of Fe3+. Tetrahedron Lett. 2017, 58, 3951–3956. [Google Scholar] [CrossRef]
- Cheah, P.W.; Heng, M.P.; Saad, H.M.; Sim, K.S.; Tan, K.W. Specific detection of Cu2+ by a pH-independent colorimetric rhodamine based chemosensor. Opt. Mater. 2021, 114, 110990. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, G.; Xu, T.; Wu, Y.; Dong, C.; Shuang, S. Silk Fibroin-Confined Star-Shaped Decahedral Silver Nanoparticles as Fluorescent Probe for Detection of Cu2+ and Pyrophosphate. Acs Biomater. Sci. Eng. 2020, 6, 2770–2777. [Google Scholar] [CrossRef] [PubMed]
- Sağırlı, A.; Bozkurt, E. Rhodamine-Based Arylpropenone Azo Dyes as Dual Chemosensor for Cu2+/Fe3+ Detection. J. Photochem. Photobiol. A Chem. 2020, 403, 112836. [Google Scholar] [CrossRef]
- Su, S.; Mo, Z.; Tan, G.; Wen, H.; Chen, X.; Hakeem, D.A. PAA Modified Upconversion Nanoparticles for Highly Selective and Sensitive Detection of Cu2+ Ions. Front. Chem. 2021, 8, 619764. [Google Scholar] [CrossRef]
- Zhang, J.; Chen, M.-Y.; Bai, C.-B.; Qiao, R.; Wei, B.; Zhang, L.; Li, R.-Q.; Qu, C.-Q. A Coumarin-Based Fluorescent Probe for Ratiometric Detection of Cu2+ and Its Application in Bioimaging. Front. Chem. 2020, 8, 00800. [Google Scholar] [CrossRef]
- Nie, K.; Dong, B.; Shi, H.; Chao, L.; Duan, X.; Jiang, X.-F.; Liu, Z.; Liang, B. N-alkylated diketopyrrolopyrrole-based ratiometric/fluorescent probes for Cu2+ detection via radical process. Dye. Pigm. 2019, 160, 814–822. [Google Scholar] [CrossRef]
- He, C.; Zhou, H.; Yang, N.; Niu, N.; Hussain, E.; Li, Y.; Yu, C. A turn-on fluorescent BOPHY probe for Cu2+ ion detection. New J. Chem. 2018, 42, 2520–2525. [Google Scholar] [CrossRef]
- Han, Z.; Nan, D.; Yang, H.; Sun, Q.; Pan, S.; Liu, H.; Hu, X. Carbon quantum dots based ratiometric fluorescence probe for sensitive and selective detection of Cu2+ and glutathione. Sens. Actuators B 2019, 298, 126842. [Google Scholar] [CrossRef]
- Hao, J.-N.; Xu, X.-Y.; Lian, X.; Zhang, C.; Yan, B. A Luminescent 3d-4f-4d MOF Nanoprobe as a Diagnosis Platform for Human Occupational Exposure to Vinyl Chloride Carcinogen. Inorg. Chem. 2017, 56, 11176–11183. [Google Scholar] [CrossRef] [PubMed]
- Hao, J.-N.; Yan, B. Determination of Urinary 1-Hydroxypyrene for Biomonitoring of Human Exposure to Polycyclic Aromatic Hydrocarbons Carcinogens by a Lanthanide-functionalized Metal-Organic Framework Sensor. Adv. Funct. Mater. 2017, 27, 1603856. [Google Scholar] [CrossRef]
| Material | LOD (μM) | Line Range | Ref. |
|---|---|---|---|
| {[Mg3(ndc)2.5(HCO2)2(H2O)][NH2Me2] 2H2O·DMF} | 0.56 | 10–45 µM | [19] |
| Eu(FBPT) (H2O) (DMF) | 8.5 | 0–17 equiv | [42] |
| 2,4,6-trihydroxybenzaldehyde rhodamine B hydrazone | 0.48 | 0–12 µM | [43] |
| SF@AgNPs | 0.333 | 1–6 µM | [44] |
| APA-Rh | 1.04 | 0–40 µM | [45] |
| Na(Yb,Nd)F4@Na(Yb,Gd)F4:Tm@NaGdF4 | 0.1 | 0.125–3.125 µM | [46] |
| MOF-525 NPs | 3.5 | 1.0–250 nM | [47] |
| FDPP-C8; TDPP-C8 | 65 × 103 127 × 103 | 0–4 µM; 0–8 µM | [48] |
| BOPHY-PTZ | ---- | 0–2 µM | [49] |
| Tb-MOFs | 10 | 1–5 × 103 µM | [41] |
| CDs-F2 | 0.409 | 0–200 µM | This work |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Che, J.; Jiang, X.; Fan, Y.; Li, M.; Zhang, X.; Gao, D.; Ning, Z.; Li, H. A Novel Dual-Emission Fluorescence Probe Based on CDs and Eu3+ Functionalized UiO-66-(COOH)2 Hybrid for Visual Monitoring of Cu2+. Materials 2022, 15, 7933. https://doi.org/10.3390/ma15227933
Che J, Jiang X, Fan Y, Li M, Zhang X, Gao D, Ning Z, Li H. A Novel Dual-Emission Fluorescence Probe Based on CDs and Eu3+ Functionalized UiO-66-(COOH)2 Hybrid for Visual Monitoring of Cu2+. Materials. 2022; 15(22):7933. https://doi.org/10.3390/ma15227933
Chicago/Turabian StyleChe, Jie, Xin Jiang, Yangchun Fan, Mingfeng Li, Xuejuan Zhang, Daojiang Gao, Zhanglei Ning, and Hongda Li. 2022. "A Novel Dual-Emission Fluorescence Probe Based on CDs and Eu3+ Functionalized UiO-66-(COOH)2 Hybrid for Visual Monitoring of Cu2+" Materials 15, no. 22: 7933. https://doi.org/10.3390/ma15227933
APA StyleChe, J., Jiang, X., Fan, Y., Li, M., Zhang, X., Gao, D., Ning, Z., & Li, H. (2022). A Novel Dual-Emission Fluorescence Probe Based on CDs and Eu3+ Functionalized UiO-66-(COOH)2 Hybrid for Visual Monitoring of Cu2+. Materials, 15(22), 7933. https://doi.org/10.3390/ma15227933







