Thermal Energy Storage (TES) Prototype Based on Geopolymer Concrete for High-Temperature Applications
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
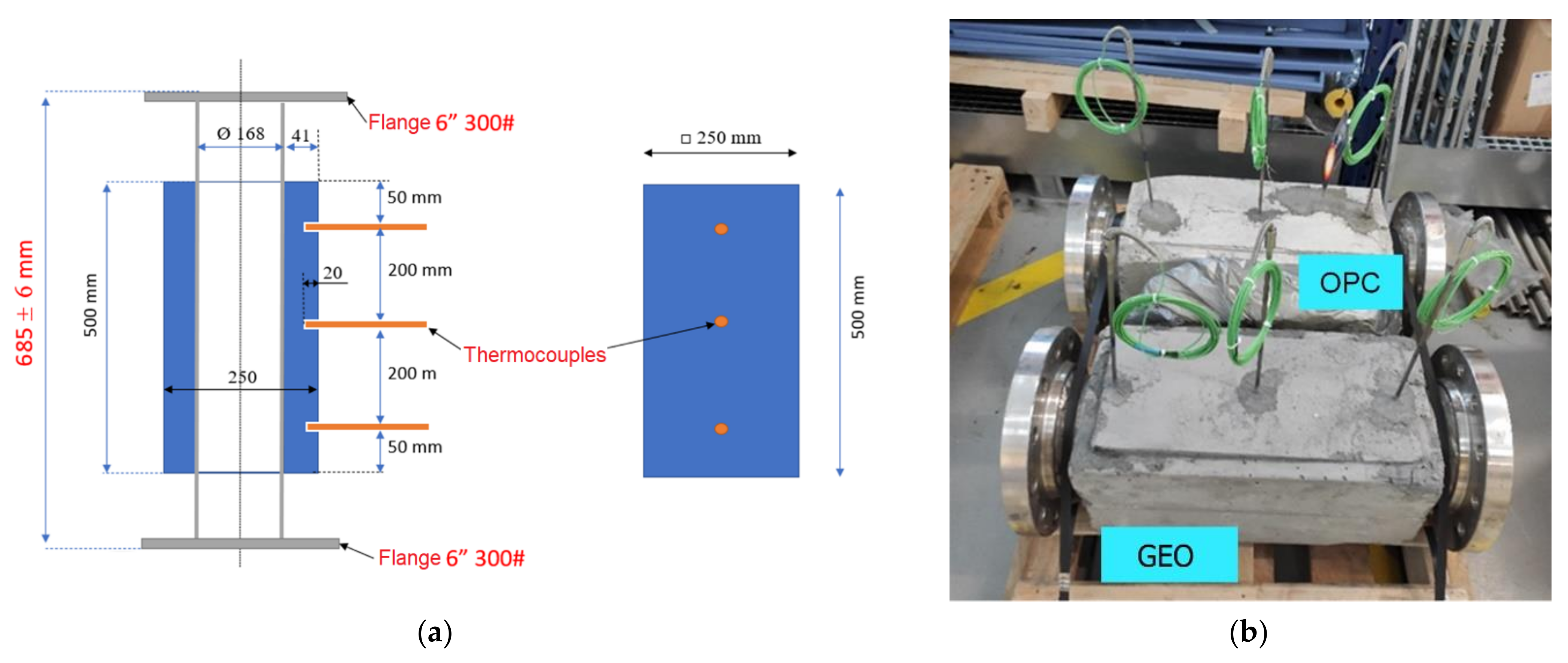
2.2. Module Description
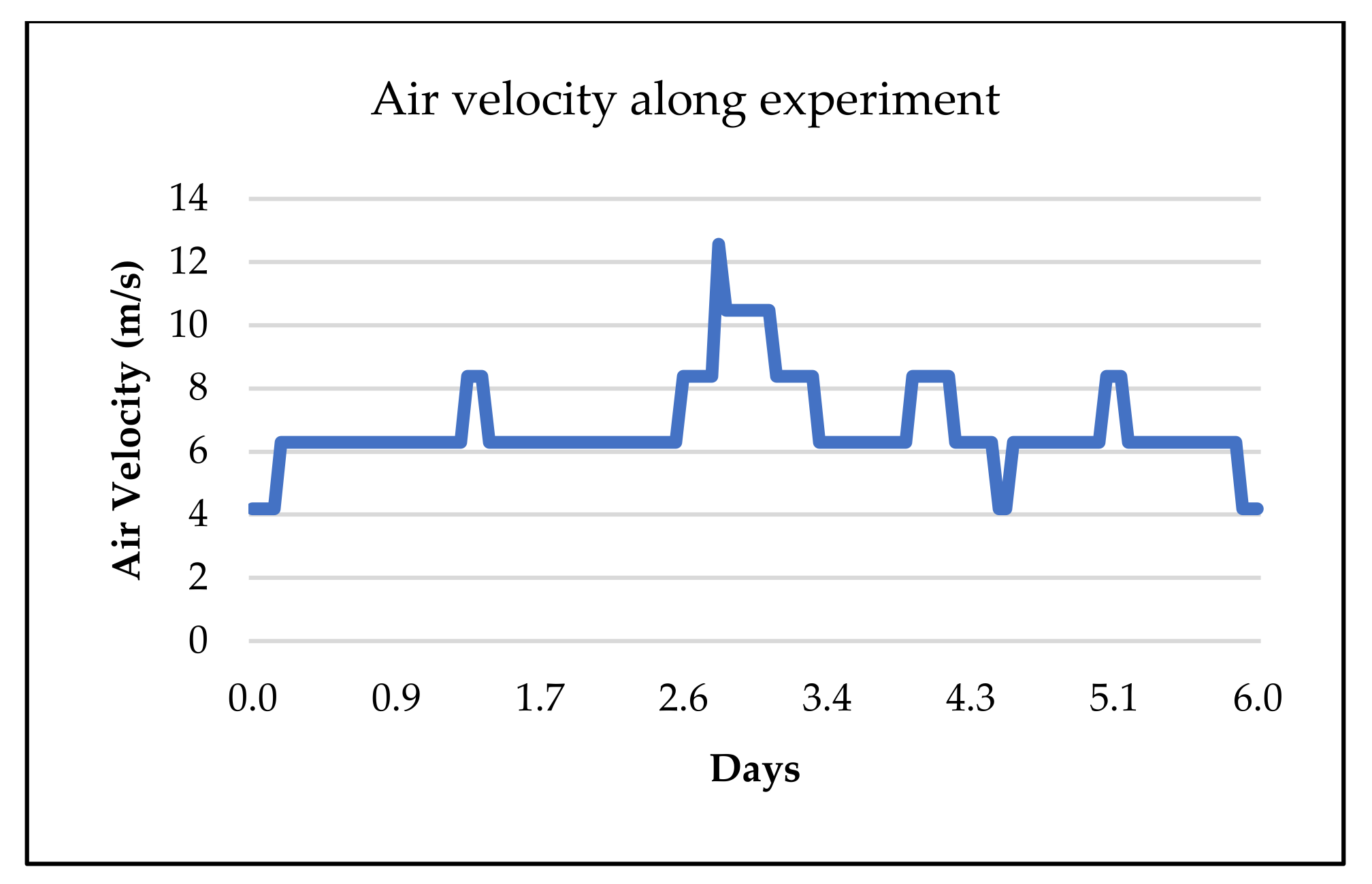
2.3. Thermal Cycles Setup
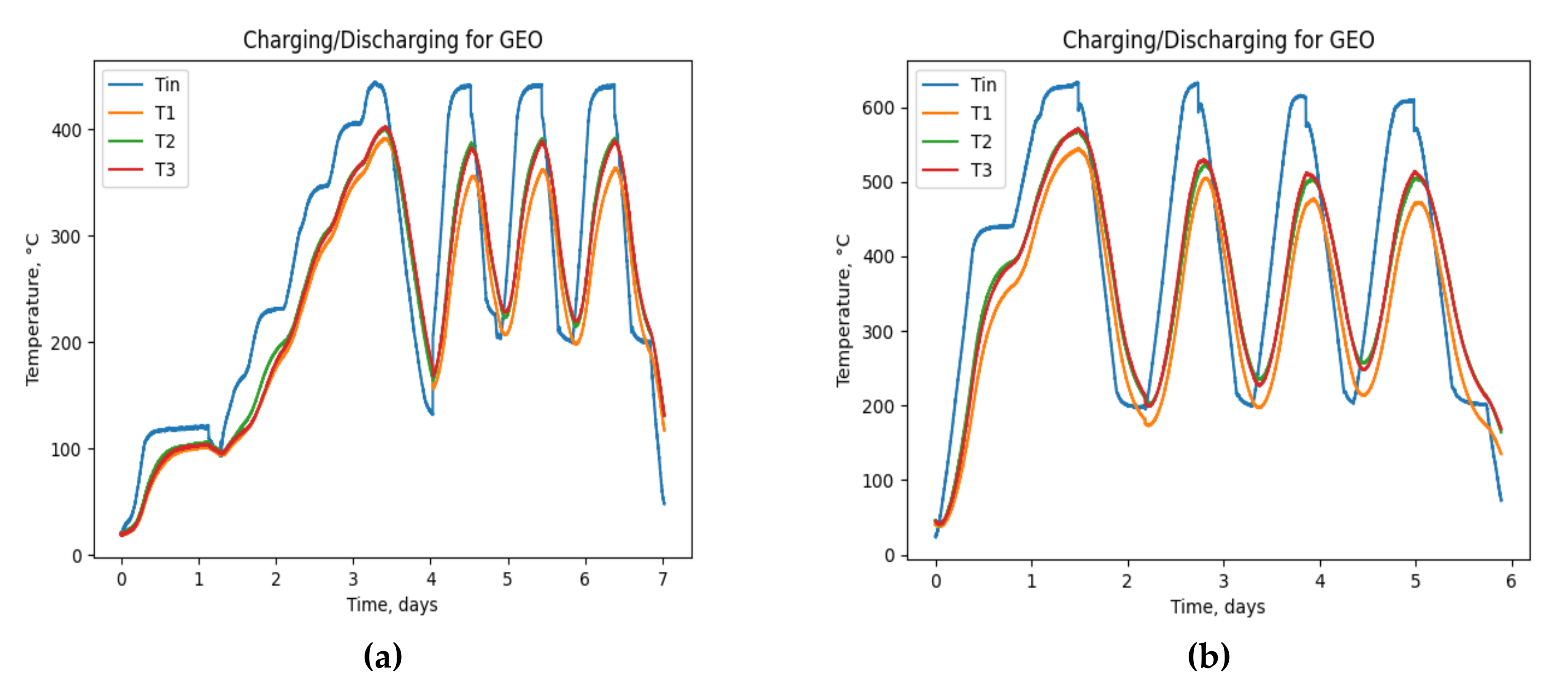
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Cycle | Time, h | Tmin, °C | Tmax, °C | ΔT, °C | Qvol, MJ/m3 |
|---|---|---|---|---|---|
| 1 | 7.5 | 260 | 350 | 90 | 260 |
| 2 | 9 | 262 | 352 | 90 | 260 |
| 3 | 9 | 265 | 357 | 92 | 265 |
| 4 | 9 | 270 | 360 | 90 | 260 |
References
- Keith, L.; Steinmann, W.-D. Concentrating Solar Power Technology: Principles, Developments and Applications; Woodhead Publishing Limited: Sawston, UK, 2012. [Google Scholar]
- Dincer, I.; Rosen, M.A. Thermal Energy Storage Systems and Applications; John Wiley & Sons: New York, NY, USA, 2021. [Google Scholar]
- Crespo, L. The double role of CSP plants on the future Electrical Systems. In Proceedings of the World Bank’s MENA CSP KIP Capstone, Online, 1–2 April 2020. [Google Scholar]
- Laing, D.; Bahl, C.; Bauer, T.; Fiss, M.; Breidenbach, N.; Hempel, M. High-Temperature Solid-Media Thermal Energy Storage for Solar Thermal Power Plants. Proc. IEEE 2012, 100, 516–524. [Google Scholar] [CrossRef]
- Skinner, J.E.; Strasser, M.N.; Brown, B.M.; Selvam, R.P. Testing of high-performance concrete as a thermal energy storage medium at high temperatures’ solar energy. J. Sol. Energy Eng. 2014, 136, 2. [Google Scholar]
- Hoivik, N.; Greiner, C.; Barragan, J.; Iniesta, A.C.; Skeie, G. Long-term performance results of concrete-based modular thermal energy storage system. Energy Storage 2019, 74, 100735. [Google Scholar] [CrossRef]
- Laing, D.; Steinmann, W.-D.; Fiß, M.; Tamme, R.; Brand, T.; Bahl, C. Solid Media Thermal Storage Development and Analysis of Modular Storage Operation Concepts for Parabolic Trough Power Plants. Sol. Energy Eng. 2008, 130, 11006. [Google Scholar] [CrossRef]
- Laing, D.; Bahl, C.; Bauer, T.; Lehmann, D.; Steinmann, W.-D. Thermal energy storage for direct steam generation. Sol. Energy 2011, 85, 627–633. [Google Scholar] [CrossRef]
- Bai, F.; Chao, X. Performance analysis of a two-stage thermal energy storage system using concrete and steam accumulator. Appl. Therm. Eng. 2011, 31, 2764–2771. [Google Scholar] [CrossRef]
- Salomoni, V.A.; Majorana, C.E.; Giannuzzi, G.M.; Miliozzi, A.; Maggio, R.D. Thermal storage of sensible heat using concrete modules in solar power plants. Sol. Energy 2014, 103, 303–315. [Google Scholar] [CrossRef]
- Ndiaye, K.; Ginestet, S.; Cyr, M. Thermal energy storage based on cementitious materials: A review. AIMS Energy 2018, 6, 97–120. [Google Scholar] [CrossRef]
- Emerson, J.; Hale, M.; Selvam, P. Concrete as a thermal energy storage medium for thermocline solar energy storage systems. Sol. Energy 2013, 96, 194–204. [Google Scholar]
- Özge, A.Ç.; Oğuzhan, Ç.; Kambiz, R. Effect of high temperature on mechanical and microstructural properties of cement mortar. In Proceedings of the 11th International Conference on Durability of Building Materials and Components, Istanbul, Turkey, 11 May 2008; pp. 11–14. [Google Scholar]
- John, E.; Hale, W.; Selvam, P. Effect of high temperatures and heating rates on high strength concrete for use as thermal energy storage. Energy Sustain. 2010, 43956, 709–713. [Google Scholar]
- Ndiaye, K.; Cyr, M.; Ginestet, S. Development of a cementitious material for thermal energy storage at low temperature. Constr. Build. Mater. 2020, 242, 118130. [Google Scholar] [CrossRef]
- Manzano, H.; Ayuela, A.; Telesca, A.; Monteiro, P.; Dolado, J. Ettringite strengthening at high pressures induced by the densification of the hydrogen bond network. J. Phys. Chem. 2010, 116, 16138–16143. [Google Scholar] [CrossRef]
- Cuesta, A.; Aranda, M. Belite cements and their activation. Cem. Concr. Res. 2021, 140, 106319. [Google Scholar] [CrossRef]
- Guerrero, S.; Moragues, A.; Dolado, J. Microstructure and mechanical performance of belite cements from high calcium coal. J. Am. Ceram. Soc. 2005, 88, 1845–1853. [Google Scholar] [CrossRef]
- Ukrainczyk, N.; Matusinović, T. Thermal properties of hydrating calcium aluminate cement pastes. Cem. Concr. Res. 2010, 40, 128–136. [Google Scholar] [CrossRef]
- Palomo, M.; Blanco, M. Alkali-activated fly ashes: A cement for the future. Cem. Concr. Res. 1999, 29, 1323–1329. [Google Scholar] [CrossRef]
- Duxson, P.; Fernández-Jiménez, A.; Provis, J.; Lukey, G.; Palomo, A.; Deventer, J.v. Geopolymer technology: The current state of the art. J. Mater. Sci. 2007, 42, 2917–2933. [Google Scholar] [CrossRef]
- Provis, J.; Lukey, G.; Deventer, J.V. Do geopolymers actually contain nanocrystalline zeolites? A reexamination of existing results. Chem. Mater. 2005, 17, 3075–3085. [Google Scholar]
- Sakulich, A.R. Reinforced geopolymer composites for enhanced material greenness and durability. Sustain. Cities Soc. 2011, 1, 195–210. [Google Scholar] [CrossRef]
- Ferone, F.; Frattini, D.; Roviello, G.; Cioff, R.; Maggio, R. Finite Element Method Modeling of Sensible Heat Thermal Energy Storage with Innovative Concretes and Comparative Analysis with Literature Benchmarks. Energies 2014, 7, 5291–5316. [Google Scholar] [CrossRef]
- Occhicone, A.; Frattini, D.; Ferone, C.; Cioffi, R. Fibre-Reinforced Geopolymer Concretes for Sensible Heat Thermal Energy Storage: Simulations and Environmental. Materials 2021, 14, 414. [Google Scholar]
- He, R.; Nan, D.; Wang, Z. Thermal and mechanical properties of geopolymers exposed to high temperature: A literature review. Adv. Civ. Eng. 2020, 2020, 7532703. [Google Scholar] [CrossRef]
- Rivera, O.; Long, W.; Weiss, C., Jr.; Moser, R.; Williams, B.; Torres-Cancel, K.; Gore, E.; Allison, P. Effect of elevated temperature on alkali-activated geopolymeric binders compared to portland cement-based binders. Cem. Concr. Res. 2016, 90, 43–51. [Google Scholar] [CrossRef]
- Rahjoo, M.; Goracci, G.; Martauz, P.; Rojas, E.; Dolado, J. Geopolymer Concrete Performance Study for High-Temperature Thermal Energy Storage (TES) Applications. Sustainbility 2022, 14, 1937. [Google Scholar] [CrossRef]
- Martauz, P.; Janotka, I.; Strigác, J.; Bačuvčík, M. Fundamental properties of industrial hybrid cement: Utilization in ready-mixed concretes and shrinkage-reducing applications. Mater. Constr. 2016, 66, 322. [Google Scholar]



| Material | GEO | OPC |
|---|---|---|
| Cement | 9.90 [kg] | 9.00 [kg] |
| Limestone aggregates 0/12 1 | - | 35.25 [kg] |
| Limestone aggregates 4/12 2 | - | 24.75 [kg] |
| SLK aggregates 0/8 3 | 66.00 [kg] | - |
| Water | 5.78 [kg] | 5.25 [kg] |
| Viscocrete | 70.80 [g] | 28.00 [g] |
| Sikament | 104.50 [g] | 95.00 [g] |
| TES Sample | GEO (°C) | OPC (°C) |
|---|---|---|
| Inlet = 450 °C | 397.7 | 366 |
| 358 | 352 | |
| 380 | 350 | |
| 380 | 349 | |
| Inlet = 650 °C | 561 | 500 |
| 518 | 457 | |
| 503 | 463 | |
| 503 | 463 |
| Properties | GEO [28] | Heatcrete 1 [6] | |
|---|---|---|---|
| Density | kg/m3 | 2890.0 | 2253 |
| Thermal conductivity | W/m °K | 1.2 | 1.78 |
| Heat capacity | J/kg °K | 1000 | 1280 |
| Thermal diffusivity | ×10−7 m2/s | 5.58 | 6.01 |
| Scenario | Cycle | Time, h | Tmin, °C | Tmax, °C | ΔT, °C | Qvol, MJ/m3 |
|---|---|---|---|---|---|---|
| Tinlet 450 °C | 1 | 81.6 | 20 | 397.7 | 377.7 | 1091.6 |
| 2 | 14.9 | 163 | 358 | 195 | 563.6 | |
| 3 | 12.0 | 219 | 380 | 161 | 465.3 | |
| 4 | 12.2 | 211 | 380 | 169 | 488.4 | |
| Tinlet 650 °C | 1 | 35.3 | 44 | 561 | 517 | 1494.1 |
| 2 | 14.9 | 192 | 518 | 326 | 942.1 | |
| 3 | 14.9 | 219 | 503 | 284 | 820.8 | |
| 4 | 13.2 | 240 | 503 | 263 | 760.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rahjoo, M.; Goracci, G.; Gaitero, J.J.; Martauz, P.; Rojas, E.; Dolado, J.S. Thermal Energy Storage (TES) Prototype Based on Geopolymer Concrete for High-Temperature Applications. Materials 2022, 15, 7086. https://doi.org/10.3390/ma15207086
Rahjoo M, Goracci G, Gaitero JJ, Martauz P, Rojas E, Dolado JS. Thermal Energy Storage (TES) Prototype Based on Geopolymer Concrete for High-Temperature Applications. Materials. 2022; 15(20):7086. https://doi.org/10.3390/ma15207086
Chicago/Turabian StyleRahjoo, Mohammad, Guido Goracci, Juan J. Gaitero, Pavel Martauz, Esther Rojas, and Jorge S. Dolado. 2022. "Thermal Energy Storage (TES) Prototype Based on Geopolymer Concrete for High-Temperature Applications" Materials 15, no. 20: 7086. https://doi.org/10.3390/ma15207086
APA StyleRahjoo, M., Goracci, G., Gaitero, J. J., Martauz, P., Rojas, E., & Dolado, J. S. (2022). Thermal Energy Storage (TES) Prototype Based on Geopolymer Concrete for High-Temperature Applications. Materials, 15(20), 7086. https://doi.org/10.3390/ma15207086










