Additively Manufactured Scaffolds with Optimized Thickness Based on Triply Periodic Minimal Surface
Abstract
1. Introduction
2. Methodology
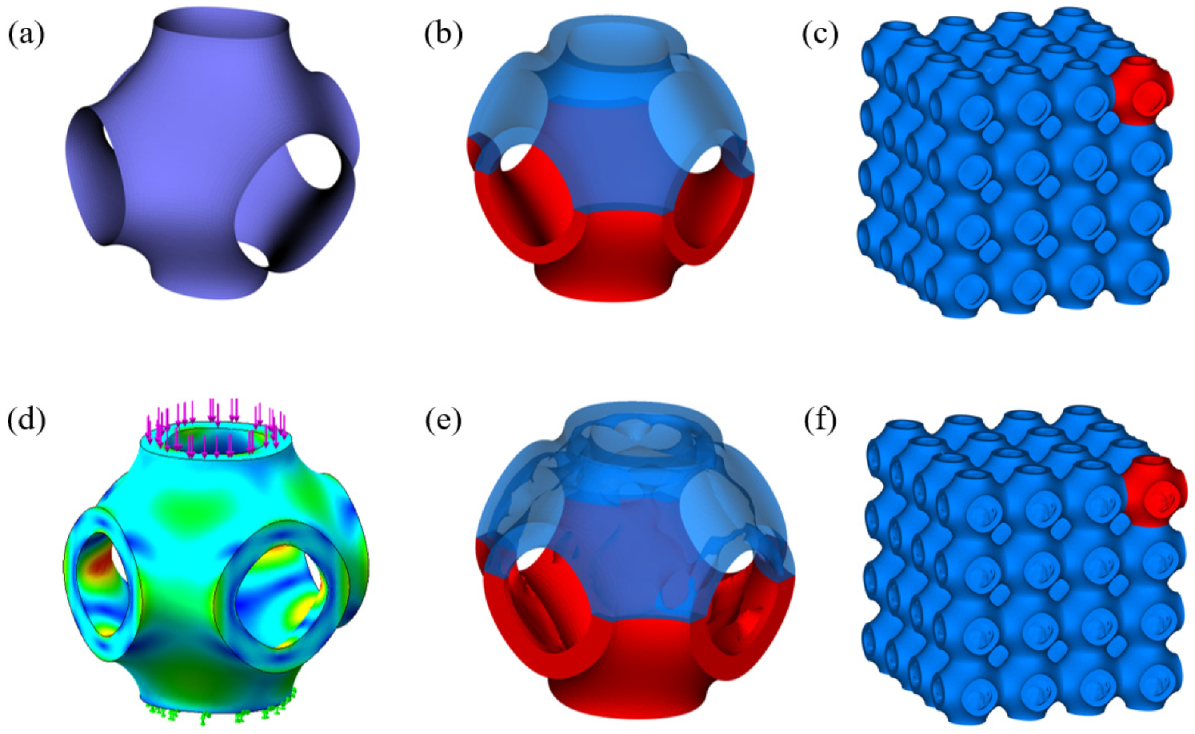
2.1. Design of P-TPMS Scaffolds
2.2. Additive Manufacturing of Scaffolds
2.3. Morphological Characterization
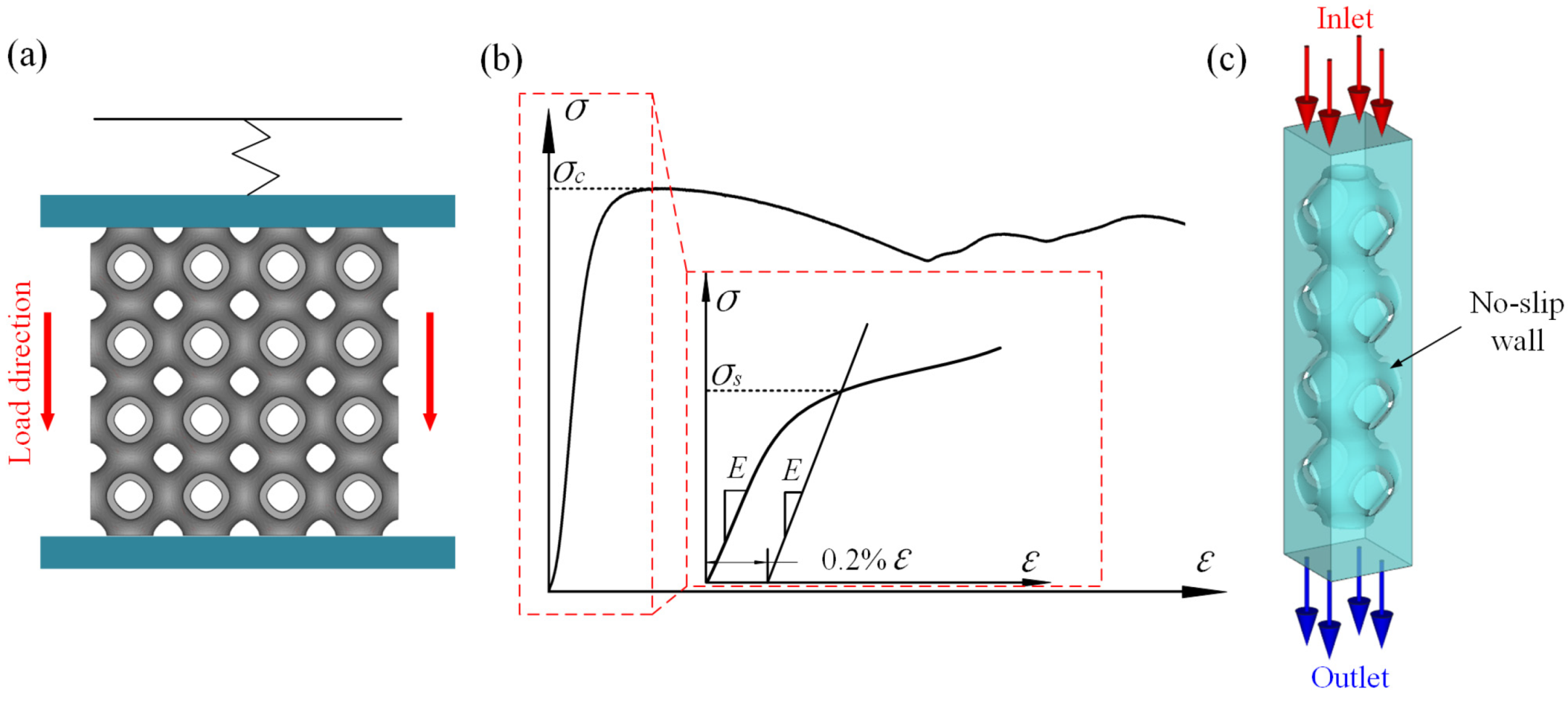
2.4. Quasi-Static Compression Testing
2.5. CFD Analysis
3. Results
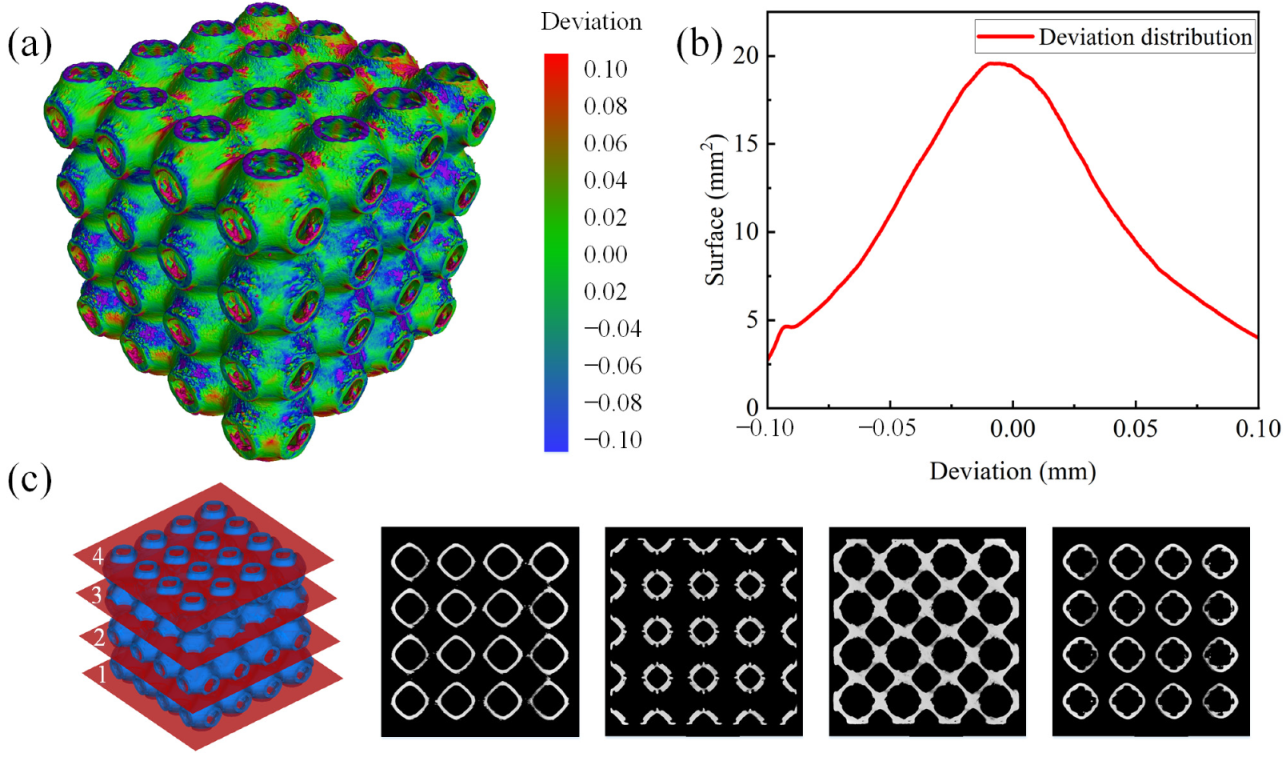
3.1. Manufacturing Fidelity and Morphological Analysis
3.2. Mechanical Properties
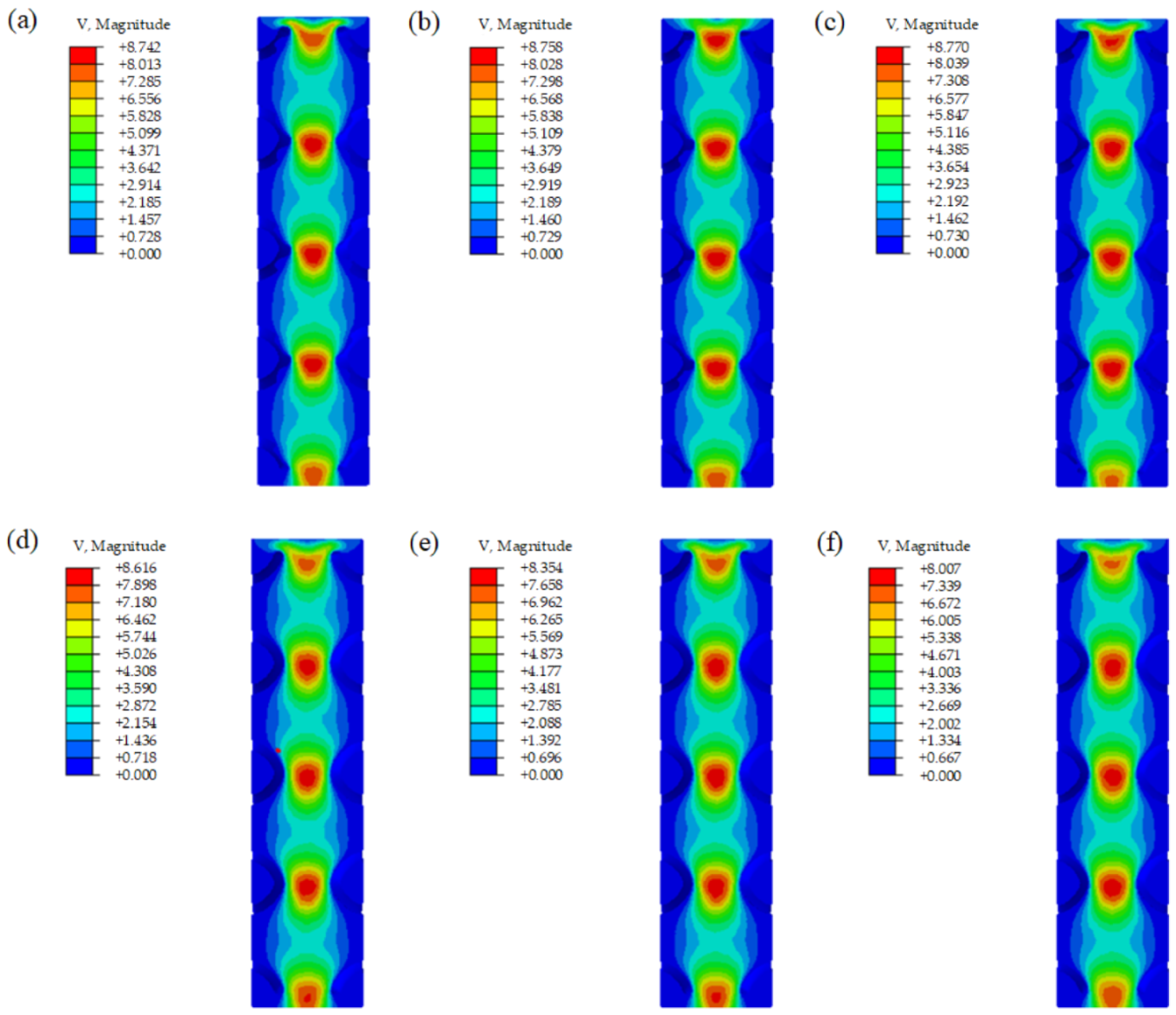
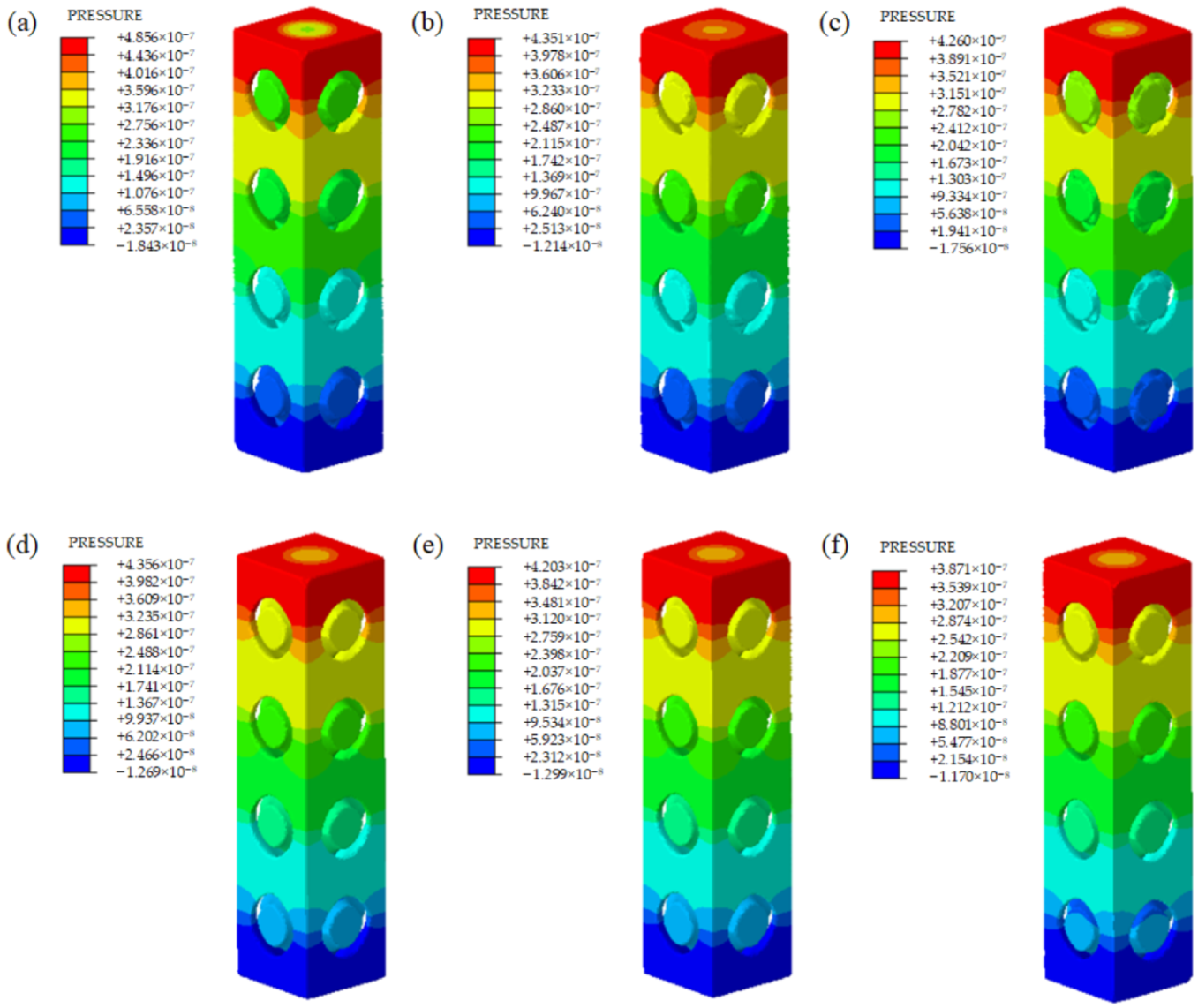
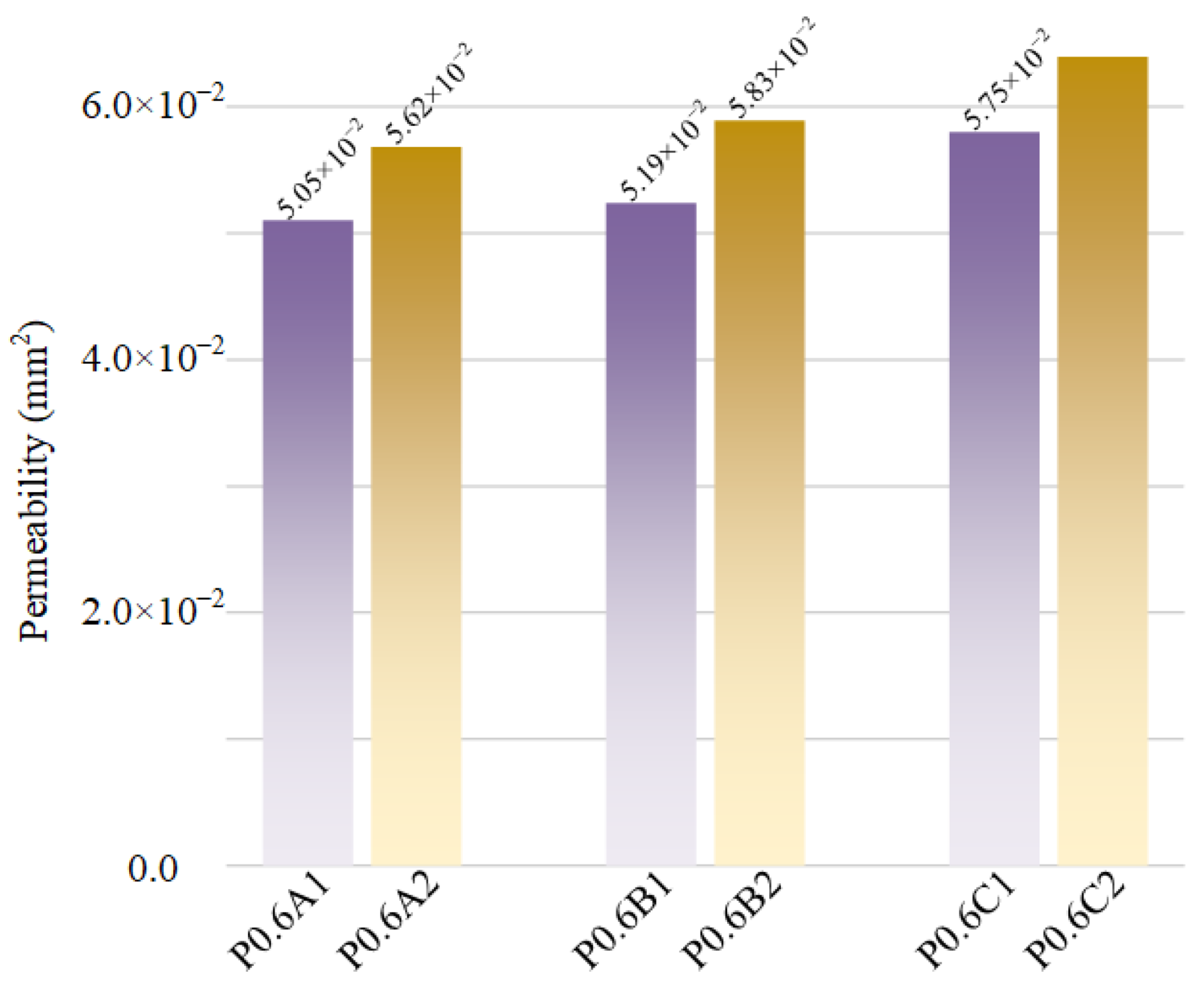
3.3. Fluid Permeability
4. Conclusions
- (1)
- Combining the micro-CT reconstructed models and captured SEM images, no macro-defects were observed, only some particles adhered to the surface and some microstructural defects in the form of micro-voids and cracks. Samples with optimized sheet thickness had larger volume deviation and a rougher inner surface than the samples with uniform sheet thickness. The majority of the deviations of the optimized scaffolds were around 0 mm. Therefore, the designed scaffolds could be additively fabricated by SLM successfully with acceptable deviations.
- (2)
- In terms of mechanical behavior, the elastic modulus, yield strength, and compressive strength of the samples with optimized sheet thickness were greater than the samples with uniform sheet thickness. However, the degree of optimization of the scaffolds had no linear relationship with the performance improvement. In addition, all samples exhibited similar failure modes. The mechanical properties of all the samples increased with increasing relative density, which is consistent with expectations of the Gibson–Ashby model.
- (3)
- The CFD simulation demonstrated that all of the samples had similar velocity distribution and pressure distribution. The velocity showed a slight upward trend with the improvement of the optimization degree, and the optimized unit cells had higher velocity compared to the same volume unit cells. However, the permeability of scaffolds with optimized sheet thickness was slightly smaller than that of the uniform sheet thickness ones.
Author Contributions
Funding
Conflicts of Interest
References
- Yang, L.; Yan, C.; Fan, H.; Li, Z.; Cai, C.; Chen, P.; Shi, Y.; Yang, S. Investigation on the Orientation Dependence of Elastic Response in Gyroid Cellular Structures. J. Mech. Behav. Biomed. Mater. 2019, 90, 73–85. [Google Scholar] [CrossRef] [PubMed]
- Kader, M.A.; Hazell, P.J.; Brown, A.D.; Tahtali, M.; Ahmed, S.; Escobedo, J.P.; Saadatfar, M. Novel Design of Closed-Cell Foam Structures for Property Enhancement. Addit. Manuf. 2020, 31, 100976. [Google Scholar] [CrossRef]
- Maggi, A.; Li, H.; Greer, J.R. Three-Dimensional Nano-Architected Scaffolds with Tunable Stiffness for Efficient Bone Tissue Growth. Acta Biomater. 2017, 63, 294–305. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Song, B.; Yang, L.; Shi, Y. Tailored Mechanical Response and Mass Transport Characteristic of Selective Laser Melted Porous Metallic Biomaterials for Bone Scaffolds. Acta Biomater. 2020, 112, 298–315. [Google Scholar] [CrossRef] [PubMed]
- Diloksumpan, P.; De Ruijter, M.; Castilho, M.; Gbureck, U.; Vermonden, T.; Van Weeren, P.R.; Malda, J.; Levato, R. Combining Multi-Scale 3D Printing Technologies to Engineer Reinforced Hydrogel-Ceramic Interfaces. Biofabrication 2020, 12, 025014. [Google Scholar] [CrossRef] [PubMed]
- Paré, A.; Charbonnier, B.; Tournier, P.; Vignes, C.; Veziers, J.; Lesoeur, J.; Laure, B.; Bertin, H.; De Pinieux, G.; Cherrier, G.; et al. Tailored Three-Dimensionally Printed Triply Periodic Calcium Phosphate Implants: A Preclinical Study for Craniofacial Bone Repair. ACS Biomater. Sci. Eng. 2020, 6, 553–563. [Google Scholar] [CrossRef]
- Parthasarathy, J.; Starly, B.; Raman, S. A Design for the Additive Manufacture of Functionally Graded Porous Structures with Tailored Mechanical Properties for Biomedical Applications. J. Manuf. Process. 2011, 13, 160–170. [Google Scholar] [CrossRef]
- Yang, N.; Gao, L.; Zhou, K. Simple Method to Generate and Fabricate Stochastic Porous Scaffolds. Mater. Sci. Eng. C 2015, 56, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Kadkhodapour, J.; Montazerian, H.; Darabi, A.C.; Zargarian, A.; Schmauder, S. The Relationships between Deformation Mechanisms and Mechanical Properties of Additively Manufactured Porous Biomaterials. J. Mech. Behav. Biomed. Mater. 2017, 70, 28–42. [Google Scholar] [CrossRef]
- Cheng, X.Y.; Li, S.J.; Murr, L.E.; Zhang, Z.B.; Hao, Y.L.; Yang, R.; Medina, F.; Wicker, R.B. Compression Deformation Behavior of Ti-6Al-4V Alloy with Cellular Structures Fabricated by Electron Beam Melting. J. Mech. Behav. Biomed. Mater. 2012, 16, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Hollister, S.J. Porous Scaffold Design for Tissue Engineering. Nat. Mater. 2005, 4, 518–524. [Google Scholar] [CrossRef] [PubMed]
- Bose, S.; Vahabzadeh, S.; Bandyopadhyay, A. Bone Tissue Engineering Using 3D Printing. Mater. Today 2013, 16, 496–504. [Google Scholar] [CrossRef]
- Davoodi, E.; Montazerian, H.; Mirhakimi, A.S.; Zhianmanesh, M.; Ibhadode, O.; Shahabad, S.I.; Esmaeilizadeh, R.; Sarikhani, E.; Toorandaz, S.; Sarabi, S.A.; et al. Additively Manufactured Metallic Biomaterials; KeAi Communications Co., Ltd.: Beijing, China, 2022; Volume 15, ISBN 8415683111. [Google Scholar]
- Ma, S.; Tang, Q.; Han, X.; Feng, Q.; Song, J.; Setchi, R.; Liu, Y.; Liu, Y.; Goulas, A.; Engstrøm, D.S.; et al. Manufacturability, Mechanical Properties, Mass-Transport Properties and Biocompatibility of Triply Periodic Minimal Surface (TPMS) Porous Scaffolds Fabricated by Selective Laser Melting. Mater. Des. 2020, 195, 109034. [Google Scholar] [CrossRef]
- Bobbert, F.S.L.; Lietaert, K.; Eftekhari, A.A.; Pouran, B.; Ahmadi, S.M.; Weinans, H.; Zadpoor, A.A. Additively Manufactured Metallic Porous Biomaterials Based on Minimal Surfaces: A Unique Combination of Topological, Mechanical, and Mass Transport Properties. Acta Biomater. 2017, 53, 572–584. [Google Scholar] [CrossRef] [PubMed]
- Kapfer, S.C.; Hyde, S.T.; Mecke, K.; Arns, C.H.; Schröder-Turk, G.E. Minimal Surface Scaffold Designs for Tissue Engineering. Biomaterials 2011, 32, 6875–6882. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.; Quan, Z.; Zhang, D.; Tian, Y. Multi-Morphology Transition Hybridization CAD Design of Minimal Surface Porous Structures for Use in Tissue Engineering. CAD Comput. Aided Des. 2014, 56, 11–21. [Google Scholar] [CrossRef]
- Poltue, T.; Karuna, C.; Khrueaduangkham, S.; Seehanam, S.; Promoppatum, P. Design Exploration of 3D-Printed Triply Periodic Minimal Surface Scaffolds for Bone Implants. Int. J. Mech. Sci. 2021, 211, 106762. [Google Scholar] [CrossRef]
- Blanquer, S.B.G.; Werner, M.; Hannula, M.; Sharifi, S.; Lajoinie, G.P.R.; Eglin, D.; Hyttinen, J.; Poot, A.A.; Grijpma, D.W. Surface Curvature in Triply-Periodic Minimal Surface Architectures as a Distinct Design Parameter in Preparing Advanced Tissue Engineering Scaffolds. Biofabrication 2017, 9, 025001. [Google Scholar] [CrossRef]
- Wang, S.; Shi, Z.; Liu, L.; Zhou, X.; Zhu, L.; Hao, Y. The Design of Ti6Al4V Primitive Surface Structure with Symmetrical Gradient of Pore Size in Biomimetic Bone Scaffold. Mater. Des. 2020, 193, 108830. [Google Scholar] [CrossRef]
- Al-Ketan, O.; Rowshan, R.; Abu Al-Rub, R.K. Topology-Mechanical Property Relationship of 3D Printed Strut, Skeletal, and Sheet Based Periodic Metallic Cellular Materials. Addit. Manuf. 2018, 19, 167–183. [Google Scholar] [CrossRef]
- Sun, Q.; Sun, J.; Guo, K.; Wang, L. Compressive Mechanical Properties and Energy Absorption Characteristics of SLM Fabricated Ti6Al4V Triply Periodic Minimal Surface Cellular Structures. Mech. Mater. 2022, 166, 104241. [Google Scholar] [CrossRef]
- Novak, N.; Al-Ketan, O.; Krstulović-Opara, L.; Rowshan, R.; Abu Al-Rub, R.K.; Vesenjak, M.; Ren, Z. Quasi-Static and Dynamic Compressive Behaviour of Sheet TPMS Cellular Structures. Compos. Struct. 2021, 266, 113801. [Google Scholar] [CrossRef]
- Li, X.; Xiao, L.; Song, W. Compressive Behavior of Selective Laser Melting Printed Gyroid Structures under Dynamic Loading. Addit. Manuf. 2021, 46, 102054. [Google Scholar] [CrossRef]
- Li, J.; Chen, D.; Fan, Y. Evaluation and Prediction of Mass Transport Properties for Porous Implant with Different Unit Cells: A Numerical Study. Biomed Res. Int. 2019, 2019, 3610785. [Google Scholar] [CrossRef]
- Santos, J.; Pires, T.; Gouveia, B.P.; Castro, A.P.G.; Fernandes, P.R. On the Permeability of TPMS Scaffolds. J. Mech. Behav. Biomed. Mater. 2020, 110, 103932. [Google Scholar] [CrossRef]
- Castro, A.P.G.; Pires, T.; Santos, J.E.; Gouveia, B.P.; Fernandes, P.R. Permeability versus Design in TPMS Scaffolds. Materials 2019, 12, 1313. [Google Scholar] [CrossRef]
- Gómez, S.; Vlad, M.D.; López, J.; Fernández, E. Design and Properties of 3D Scaffolds for Bone Tissue Engineering. Acta Biomater. 2016, 42, 341–350. [Google Scholar] [CrossRef]
- Ali, D.; Ozalp, M.; Blanquer, S.B.G.; Onel, S. Permeability and Fluid Flow-Induced Wall Shear Stress in Bone Scaffolds with TPMS and Lattice Architectures: A CFD Analysis. Eur. J. Mech. B/Fluids 2020, 79, 376–385. [Google Scholar] [CrossRef]
- Asbai-ghoudan, R.; Ruiz, S.G.; Rodriguez-florez, N. Analytical Model for the Prediction of Permeability of Triply Periodic Minimal Surfaces. J. Mech. Behav. Biomed. Mater. 2021, 124, 104804. [Google Scholar] [CrossRef]
- Jia, H.; Lei, H.; Wang, P.; Meng, J.; Li, C.; Zhou, H.; Zhang, X.; Fang, D. An Experimental and Numerical Investigation of Compressive Response of Designed Schwarz Primitive Triply Periodic Minimal Surface with Non-Uniform Shell Thickness. Extrem. Mech. Lett. 2020, 37, 100671. [Google Scholar] [CrossRef]
- Wang, H.; Tan, D.; Liu, Z.; Yin, H.; Wen, G. On Crashworthiness of Novel Porous Structure Based on Composite TPMS Structures. Eng. Struct. 2022, 252, 113640. [Google Scholar] [CrossRef]
- Liu, B.; Liu, M.; Cheng, H.; Cao, W.; Lu, P. A New Stress-Driven Composite Porous Structure Design Method Based on Triply Periodic Minimal Surfaces. Thin-Walled Struct. 2022, 181, 109974. [Google Scholar] [CrossRef]
- Jin, Y.; Zou, S.; Pan, B.; Li, G.; Shao, L.; Du, J. Biomechanical Properties of Cylindrical and Twisted Triply Periodic Minimal Surface Scaffolds Fabricated by Laser Powder Bed Fusion. Addit. Manuf. 2022, 56, 102899. [Google Scholar] [CrossRef]
- AlMahri, S.; Santiago, R.; Lee, D.W.; Ramos, H.; Alabdouli, H.; Alteneiji, M.; Guan, Z.; Cantwell, W.; Alves, M. Evaluation of the Dynamic Response of Triply Periodic Minimal Surfaces Subjected to High Strain-Rate Compression. Addit. Manuf. 2021, 46, 102220. [Google Scholar] [CrossRef]
- Maconachie, T.; Leary, M.; Lozanovski, B.; Zhang, X.; Qian, M.; Faruque, O.; Brandt, M. SLM Lattice Structures: Properties, Performance, Applications and Challenges. Mater. Des. 2019, 183, 108137. [Google Scholar] [CrossRef]




| Element | Fe | Si | Cr | Ni | Mn | C | O | S | P |
|---|---|---|---|---|---|---|---|---|---|
| wt% | Bal. | 0.43 | 19.35 | 9.53 | 0.71 | 0.006 | 0.056 | 0.005 | 0.010 |
| Symbol | Description | Unit |
|---|---|---|
| K | the permeability coefficient | mm2 |
| L | the length of the model | mm |
| ΔP | pressure difference | MPa |
| μ | the dynamic viscosity coefficient of the fluid | MPa·s |
| v | the velocity of the fluid | mm/s |
| Type | Actual Volume (mm3) | Theoretical Volume (mm3) | Mass (g) | Average Relative Density (%) | Deviation (%) |
|---|---|---|---|---|---|
| P0.4A1 | 2169.62 ± 15.29 | 1988.59 | 17.14 ± 0.12 | 14.71 ± 0.10 | 9.45 ± 0.77 |
| P0.4A2 | 2153.59 ± 1.58 | 1988.59 | 17.01 ± 0.01 | 14.60 ± 0.01 | 7.95 ± 0.08 |
| P0.4B1 | 2117.72 ± 12.70 | 1875.28 | 16.73 ± 0.10 | 14.36 ± 0.09 | 13.45 ± 0.68 |
| P0.4B2 | 2021.94 ± 4.18 | 1875.28 | 15.97 ± 0.03 | 13.71 ± 0.03 | 7.33 ± 0.22 |
| P0.4C1 | 2015.19 ± 9.47 | 1760.67 | 15.92 ± 0.07 | 13.66 ± 0.06 | 15.12 ± 0.54 |
| P0.4C2 | 1956.96 ± 7.80 | 1760.67 | 15.46 ± 0.06 | 13.27 ± 0.05 | 10.51 ± 0.44 |
| P0.6A1 | 3118.57 ± 2.15 | 2933.09 | 24.64 ± 0.02 | 21.14 ± 0.01 | 6.32 ± 0.07 |
| P0.6A2 | 3032.49 ± 7.33 | 2933.09 | 23.96 ± 0.06 | 20.56 ± 0.05 | 3.39 ± 0.25 |
| P0.6B1 | 2940.93 ± 10.20 | 2744.79 | 23.23 ± 0.08 | 19.94 ± 0.07 | 7.15 ± 0.37 |
| P0.6B2 | 2899.58 ± 4.18 | 2744.79 | 22.91 ± 0.03 | 19.66 ± 0.03 | 5.64 ± 0.15 |
| P0.6C1 | 2735.44 ± 8.20 | 2493.18 | 21.61 ± 0.06 | 18.55 ± 0.06 | 9.72 ± 0.33 |
| P0.6C2 | 2618.99 ± 19.64 | 2493.18 | 20.69 ± 0.16 | 17.76 ± 0.13 | 5.05 ± 0.79 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, J.; Zou, S.; Mu, Y.; Wang, J.; Jin, Y. Additively Manufactured Scaffolds with Optimized Thickness Based on Triply Periodic Minimal Surface. Materials 2022, 15, 7084. https://doi.org/10.3390/ma15207084
Zhu J, Zou S, Mu Y, Wang J, Jin Y. Additively Manufactured Scaffolds with Optimized Thickness Based on Triply Periodic Minimal Surface. Materials. 2022; 15(20):7084. https://doi.org/10.3390/ma15207084
Chicago/Turabian StyleZhu, Junjie, Sijia Zou, Yanru Mu, Junhua Wang, and Yuan Jin. 2022. "Additively Manufactured Scaffolds with Optimized Thickness Based on Triply Periodic Minimal Surface" Materials 15, no. 20: 7084. https://doi.org/10.3390/ma15207084
APA StyleZhu, J., Zou, S., Mu, Y., Wang, J., & Jin, Y. (2022). Additively Manufactured Scaffolds with Optimized Thickness Based on Triply Periodic Minimal Surface. Materials, 15(20), 7084. https://doi.org/10.3390/ma15207084





