Magnetic Properties of GaAs/NiFe Coaxial Core-Shell Structures
Abstract
:1. Introduction
2. Materials and Methods
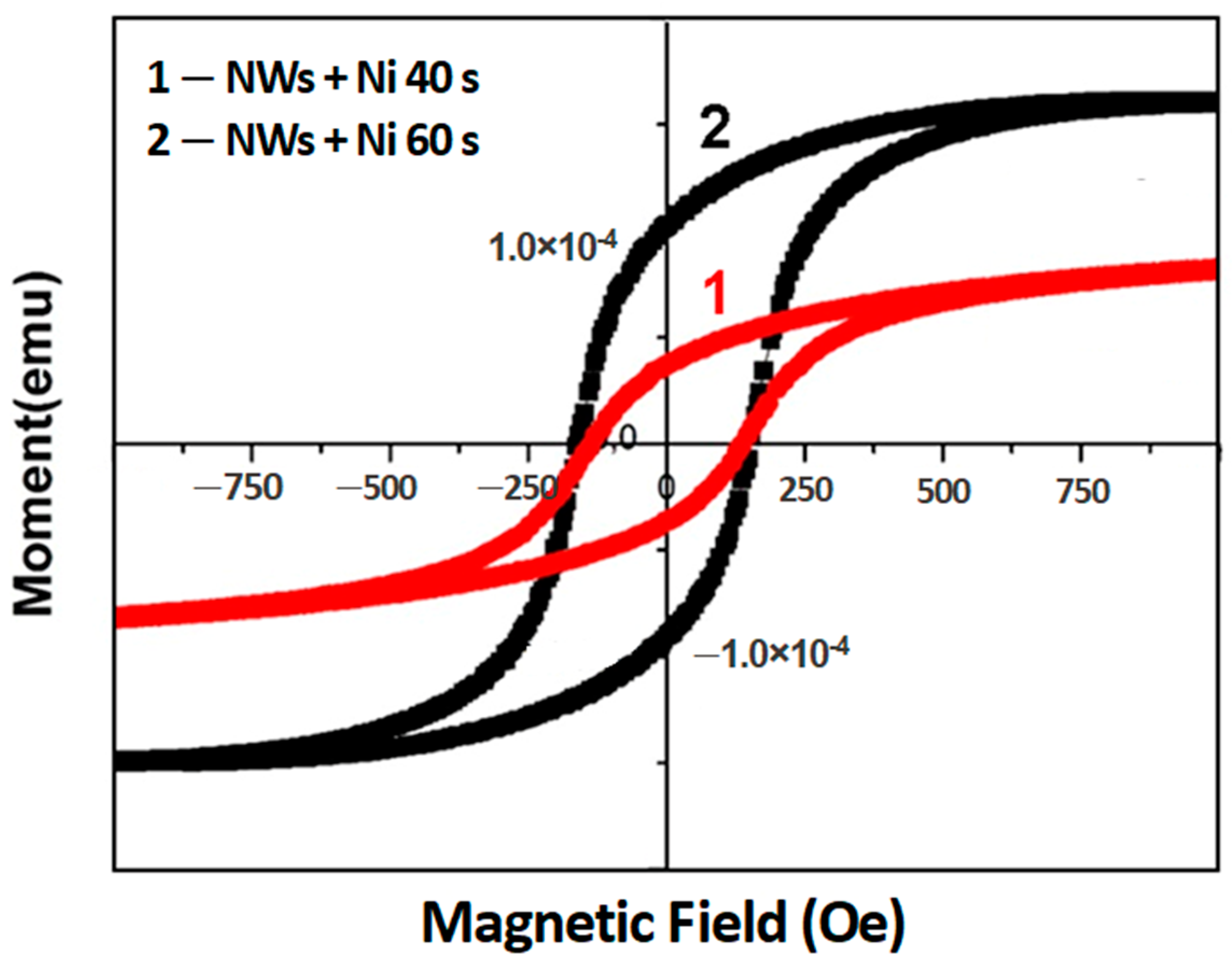
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Asare-Yeboah, K.; Li, Q.; Jiang, C.; He, Z.; Bi, S.; Liu, Y.; Liu, C. High Performance and Efficiency Resonant Photo-Effect-Transistor by Near-Field Nano-Strip-Controlled Organic Light Emitting Diode Gate. J. Phys. Chem. Lett. 2020, 11, 6526–6534. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Jiang, C.; Bi, S.; Asare-Yeboah, K.; He, Z.; Liu, Y. Photo-Triggered Logic Circuits Assembled on Integrated Illuminants and Resonant Nanowires. ACS Appl. Mater. Interfaces 2020, 12, 46501–46508. [Google Scholar] [CrossRef]
- Bi, S.; Li, Q.; He, Z.; Guo, Q.; Asare-Yeboah, K.; Liu, Y.; Jiang, C. Highly enhanced performance of integrated piezo photo-transistor with dual inverted OLED gate and nanowire array channel. Nano Energy 2019, 66, 104101. [Google Scholar] [CrossRef]
- Mohammed, H.; Moreno, J.A.; Kosel, J. Advanced Fabrication and Characterization of Magnetic Nanowires; IntechOpen: London, UK, 2017; ISBN 978-1-78923-679-8. [Google Scholar]
- Piraux, L. Magnetic Nanowires. Appl. Sci. 2020, 10, 1832. [Google Scholar] [CrossRef]
- Proenca, M.P.; Sousa, C.T.; Ventura, J.; Araújo, J.P. Chapter 6—Cylindrical Magnetic Nanotubes: Synthesis, Magnetism and Applications. In Magnetic Nano- and Microwires, 2nd ed.; Vázquez, M., Ed.; Woodhead Publishing Series in Electronic and Optical Materials; Woodhead Publishing: Thorston, UK, 2020; pp. 135–184. ISBN 978-0-08-102832-2. [Google Scholar]
- García, J.; Manterola, A.M.; Méndez, M.; Fernández-Roldán, J.A.; Vega, V.; González, S.; Prida, V.M. Magnetization Reversal Process and Magnetostatic Interactions in Fe56Co44/SiO2/Fe3O4 Core/Shell Ferromagnetic Nanowires with Non-Magnetic Interlayer. Nanomaterials 2021, 11, 2282. [Google Scholar] [CrossRef]
- Staňo, M.; Fruchart, O. Chapter 3—Magnetic Nanowires and Nanotubes. In Handbook of Magnetic Materials; Brück, E., Ed.; Elsevier: Amsterdam, The Netherlands, 2018; Volume 27, pp. 155–267. [Google Scholar]
- Chong, Y.T.; Görlitz, D.; Martens, S.; Yau, M.Y.E.; Allende, S.; Bachmann, J.; Nielsch, K. Multilayered Core/Shell Nanowires Displaying Two Distinct Magnetic Switching Events. Adv. Mater. 2010, 22, 2435–2439. [Google Scholar] [CrossRef]
- Choudhury, S.; Mondal, S.; De, A.; Barman, A. Chapter 21—Tunable Picosecond Magnetization Dynamics in Ferromagnetic Nanostructures. In 21st Century Nanoscience—A Handbook; Routledge Handbooks Online: London, UK, 2020; p. 488. ISBN 978-0-8153-5626-4. [Google Scholar]
- Chou, S.Y.; Krauss, P.R.; Kong, L. Nanolithographically Defined Magnetic Structures and Quantum Magnetic Disk (Invited). J. Appl. Phys. 1996, 79, 6101–6106. [Google Scholar] [CrossRef]
- Moreno, J.A.; Bran, C.; Vazquez, M.; Kosel, J. Cylindrical Magnetic Nanowires Applications. IEEE Trans. Magn. 2021, 57, 800317. [Google Scholar] [CrossRef]
- Zierold, R.; Nielsch, K. (Invited) Tailor-Made, Magnetic Nanotubes by Template-Directed Atomic Layer Deposition. ECS Trans. 2011, 41, 111. [Google Scholar] [CrossRef]
- Tejo, F.; Vidal-Silva, N.; Espejo, A.P.; Escrig, J. Angular Dependence of the Magnetic Properties of Cylindrical Diameter Modulated Ni80Fe20 Nanowires. J. Appl. Phys. 2014, 115, 17D136. [Google Scholar] [CrossRef]
- Escrig, J.; Daub, M.; Landeros, P.; Nielsch, K.; Altbir, D. Angular Dependence of Coercivity in Magnetic Nanotubes. Nanotechnology 2007, 18, 445706. [Google Scholar] [CrossRef]
- Nielsch, K.; Bachmann, J.; Daub, M.; Jing, J.; Knez, M.; Gösele, U.; Barth, S.; Mathur, S.; Escrig, J.; Altbir, D. Ferromagnetic Nanostructures by Atomic Layer Deposition: From Thin Films Towards Core-Shell Nanotubes. ECS Trans. 2007, 11, 139. [Google Scholar] [CrossRef]
- Daub, M.; Knez, M.; Goesele, U.; Nielsch, K. Ferromagnetic Nanotubes by Atomic Layer Deposition in Anodic Alumina Membranes. J. Appl. Phys. 2007, 101, 09J111. [Google Scholar] [CrossRef]
- Salem, M.S.; Tejo, F.; Zierold, R.; Sergelius, P.; Moreno, J.M.M.; Goerlitz, D.; Nielsch, K.; Escrig, J. Composition and Diameter Modulation of Magnetic Nanowire Arrays Fabricated by a Novel Approach. Nanotechnology 2018, 29, 065602. [Google Scholar] [CrossRef] [PubMed]
- Minguez-Bacho, I.; Rodriguez-López, S.; Vázquez, M.; Hernández-Vélez, M.; Nielsch, K. Electrochemical Synthesis and Magnetic Characterization of Periodically Modulated Co Nanowires. Nanotechnology 2014, 25, 145301. [Google Scholar] [CrossRef] [PubMed]
- Bochmann, S.; Döhler, D.; Trapp, B.; Staňo, M.; Fruchart, O.; Bachmann, J. Preparation and Physical Properties of Soft Magnetic Nickel-Cobalt Three-Segmented Nanowires. J. Appl. Phys. 2018, 124, 163907. [Google Scholar] [CrossRef]
- García, J.; Vega, V.; Iglesias, L.; Prida, V.M.; Hernando, B.; Barriga-Castro, E.D.; Mendoza-Reséndez, R.; Luna, C.; Görlitz, D.; Nielsch, K. Template-Assisted Co–Ni Alloys and Multisegmented Nanowires with Tuned Magnetic Anisotropy. Phys. Status Solidi A 2014, 211, 1041–1047. [Google Scholar] [CrossRef]
- Vega, V.; Böhnert, T.; Martens, S.; Waleczek, M.; Montero-Moreno, J.M.; Görlitz, D.; Prida, V.M.; Nielsch, K. Tuning the Magnetic Anisotropy of Co–Ni Nanowires: Comparison between Single Nanowires and Nanowire Arrays in Hard-Anodic Aluminum Oxide Membranes. Nanotechnology 2012, 23, 465709. [Google Scholar] [CrossRef]
- Pirota, K.R.; Navas, D.; Hernández-Vélez, M.; Nielsch, K.; Vázquez, M. Novel Magnetic Materials Prepared by Electrodeposition Techniques: Arrays of Nanowires and Multi-Layered Microwires. J. Alloys Compd. 2004, 369, 18–26. [Google Scholar] [CrossRef]
- Rumpf, K.; Granitzer, P.; Poelt, P.; Michor, H. (Invited) Pore Filling of Porous Silicon with Ferromagnetic Nanostructures. ECS Trans. 2015, 69, 71. [Google Scholar] [CrossRef]
- Salem, M.S.; Nielsch, K. Crossover between Axial and Radial Magnetic Anisotropy in Self-Organized Permalloy Nanowires. Mater. Sci. Eng. B 2017, 223, 120–124. [Google Scholar] [CrossRef]
- Sorop, T.G.; Nielsch, K.; Göring, P.; Kröll, M.; Blau, W.; Wehrspohn, R.B.; Gösele, U.; de Jongh, L.J. Study of the Magnetic Hysteresis in Arrays of Ferromagnetic Fe Nanowires as a Function of the Template Filling Fraction. J. Magn. Magn. Mater. 2004, 272–276, 1656–1657. [Google Scholar] [CrossRef]
- Albrecht, O.; Zierold, R.; Allende, S.; Escrig, J.; Patzig, C.; Rauschenbach, B.; Nielsch, K.; Görlitz, D. Experimental Evidence for an Angular Dependent Transition of Magnetization Reversal Modes in Magnetic Nanotubes. J. Appl. Phys. 2011, 109, 093910. [Google Scholar] [CrossRef]
- Albrecht, O.; Zierold, R.; Patzig, C.; Bachmann, J.; Sturm, C.; Rheinländer, B.; Grundmann, M.; Görlitz, D.; Rauschenbach, B.; Nielsch, K. Tubular Magnetic Nanostructures Based on Glancing Angle Deposited Templates and Atomic Layer Deposition. Phys. Status Solidi B 2010, 247, 1365–1371. [Google Scholar] [CrossRef]
- Monaico, E.I.; Monaico, E.V.; Ursaki, V.V.; Honnali, S.; Postolache, V.; Leistner, K.; Nielsch, K.; Tiginyanu, I.M. Electrochemical Nanostructuring of (111) Oriented GaAs Crystals: From Porous Structures to Nanowires. Beilstein J. Nanotechnol. 2020, 11, 966–975. [Google Scholar] [CrossRef]
- Monaico, E.V.; Morari, V.; Ursaki, V.V.; Nielsch, K.; Tiginyanu, I.M. Core–Shell GaAs-Fe Nanowire Arrays: Fabrication Using Electrochemical Etching and Deposition and Study of Their Magnetic Properties. Nanomaterials 2022, 12, 1506. [Google Scholar] [CrossRef]
- Leistner, K.; Yang, M.; Damm, C.; Oswald, S.; Petr, A.; Kataev, V.; Nielsch, K.; Kavanagh, K.L. Aligned Cuboid Iron Nanoparticles by Epitaxial Electrodeposition. Nanoscale 2017, 9, 5315–5322. [Google Scholar] [CrossRef]
- Leistner, K.; Duschek, K.; Zehner, J.; Yang, M.; Petr, A.; Nielsch, K.; Kavanagh, K.L. Role of Hydrogen Evolution during Epitaxial Electrodeposition of Fe on GaAs. J. Electrochem. Soc. 2017, 165, H3076. [Google Scholar] [CrossRef]
- Kozlovskiy, A.L.; Korolkov, I.V.; Ibragimova, M.A.; Zdorovets, M.V.; Kutuzau, M.D.; Nikolaevich, L.N.; Shumskaya, E.E.; Kaniukov, E.Y. Magnetic Nanostructured System for Biomedical Applications Based on FeNi Nanotubes. Nanotechnol. Russ. 2018, 13, 331–336. [Google Scholar] [CrossRef]
- Uosaki, K.; Kita, H. Effect of the Ru+++ treatment on the electrochemical hydrogen evolution reaction at GaAs electrodes. Chem. Lett. 1984, 13, 953–956. [Google Scholar] [CrossRef]
- Lamrani, S.; Guittoum, A.; Schäfer, R.; Hemmous, M.; Neu, V.; Pofahl, S.; Hadjersi, T.; Benbrahim, N. Morphology, Structure and Magnetic Study of Permalloy Films Electroplated on Silicon Nanowires. J. Magn. Magn. Mater. 2015, 396, 263–267. [Google Scholar] [CrossRef]





| Alloy | Element | Weight % | Atomic % |
|---|---|---|---|
| Permalloy | Ni | 82 | 81 |
| Fe | 18 | 19 | |
| Ni0.65Fe0.35 | Ni | 66 | 65 |
| Fe | 34 | 35 | |
| Ni | Ni | 100 | 100 |
| Sample Description | Deposition Mode | Saturation Moment, Ms (emu) × 10−4 | Remanence, Mr (emu) × 10−4 | Coercive Force, Hc (Oe) |
|---|---|---|---|---|
| Planar/Ni0.65Fe0.35 | Potentiost. 20 s | 2.01 | 1.28 | 107 |
| Planar/Ni0.65Fe0.35 | Potentiost. 40 s | 2.27 | 1.46 | 126 |
| Planar/Ni0.65Fe0.35 | Potentiost. 60 s | 2.61 | 1.75 | 153 |
| Nanowires/Ni0.65Fe0.35 | Potentiost. 20 s | 0.37 | 0.11 | 16 |
| Nanowires/Ni0.65Fe0.35 | Potentiost. 40 s | 1.11 | 0.69 | 84 |
| Nanowires/Ni0.65Fe0.35 | Potentiost. 60 s | 1.94 | 1.34 | 172 |
| Planar/Ni0.8Fe0.2 | Potentiost. 20 s | 0.10 | 0.09 | 72 |
| Planar/Ni0.8Fe0.2 | Potentiost. 40 s | 0.25 | 0.22 | 85 |
| Planar/Ni0.8Fe0.2 | Potentiost. 60 s | 0.36 | 0.32 | 123 |
| Nanowires/Ni0.8Fe0.2 | Potentiost. 20 s | 2.05 | 2.60 | 165 |
| Nanowires/Ni0.8Fe0.2 | Potentiost. 40 s | 2.25 | 1.75 | 188 |
| Nanowires/Ni0.8Fe0.2 | Potentiost. 60 s | 2.45 | 1.98 | 250 |
| Planar/Ni | Potentiost. 60 s | 0.80 | 0.74 | 134 |
| Nanowires/Ni | Potentiost. 60 s | 1.55 | 1.05 | 153 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Monaico, E.V.; Morari, V.; Kutuzau, M.; Ursaki, V.V.; Nielsch, K.; Tiginyanu, I.M. Magnetic Properties of GaAs/NiFe Coaxial Core-Shell Structures. Materials 2022, 15, 6262. https://doi.org/10.3390/ma15186262
Monaico EV, Morari V, Kutuzau M, Ursaki VV, Nielsch K, Tiginyanu IM. Magnetic Properties of GaAs/NiFe Coaxial Core-Shell Structures. Materials. 2022; 15(18):6262. https://doi.org/10.3390/ma15186262
Chicago/Turabian StyleMonaico, Eduard V., Vadim Morari, Maksim Kutuzau, Veaceslav V. Ursaki, Kornelius Nielsch, and Ion M. Tiginyanu. 2022. "Magnetic Properties of GaAs/NiFe Coaxial Core-Shell Structures" Materials 15, no. 18: 6262. https://doi.org/10.3390/ma15186262
APA StyleMonaico, E. V., Morari, V., Kutuzau, M., Ursaki, V. V., Nielsch, K., & Tiginyanu, I. M. (2022). Magnetic Properties of GaAs/NiFe Coaxial Core-Shell Structures. Materials, 15(18), 6262. https://doi.org/10.3390/ma15186262





