Effect of P2O5 and Na2O on the Solubility of Molybdenum and Structural Features in Borosilicate Glass
Abstract
:1. Introduction
2. Experimental
2.1. Sample Preparation
2.2. Measurements
3. Results and Discussion
3.1. Glass Forming and Crystallization Analysis
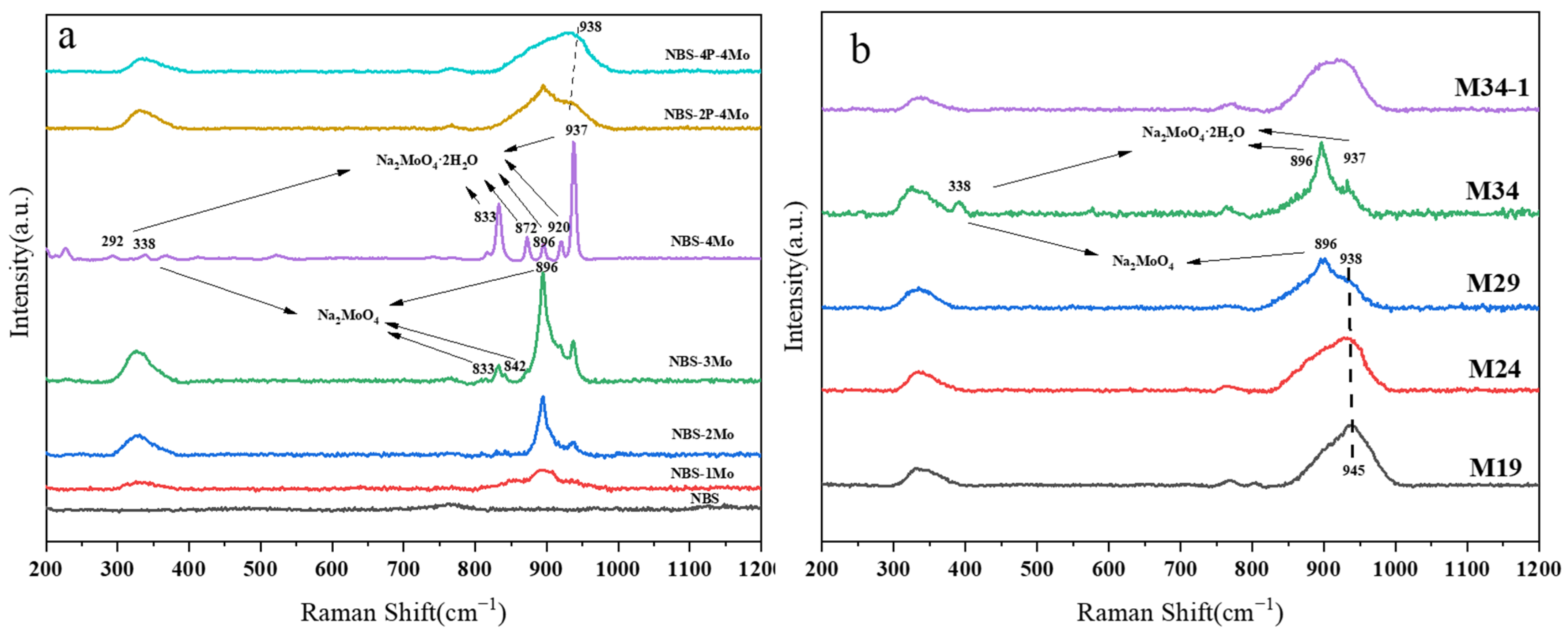
3.2. Raman Spectra
3.3. 31P MAS NMR Spectroscopy
3.4. Thermal Stability and Aqueous Durability
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Caurant, D.; Majérus, O. Glasses and Glass-Ceramics for Nuclear Waste Immobilization. In Encyclopedia of Materials: Technical Ceramics and Glasses; Elsevier: Amsterdam, The Netherlands, 2021; pp. 762–789. [Google Scholar]
- Tan, S.; Ojovan, M.I.; Hyatt, N.C.; Hand, R.J. MoO3 incorporation in magnesium aluminosilicate glasses. J. Nucl. Mater. 2015, 458, 335–342. [Google Scholar] [CrossRef]
- Wang, J. High Level Radioactive Waste Disposal in China: Update 2009. In Proceedings of the International Symposium on Unsaturated Soil Mechanics and Deep Geological Nuclear Waste Disposal (UNSAT-WASTE 2009), Shanghai, China, 24–28 August 2009. [Google Scholar]
- Prakash, A.D.; Mishra, R.K.; Valsala, T.P.; Sharma, V.; Acharya, R.; Tyagi, A.K.; Pujari, P.K.; Kaushik, C.P. Simultaneous quantification of low Z elements in phosphorus containing alkali borosilicate glass samples by in situ Current normalized external (in air) PIGE method utilizing proton beam from FOTIA. J. Radioanal. Nucl. Chem. 2021, 328, 519–526. [Google Scholar] [CrossRef]
- Prakash, A.D.; Singh, M.; Mishra, R.K.; Valsala, T.P.; Tyagi, A.K.; Sarkar, A.; Kaushik, C.P. Studies on modified borosilicate glass for enhancement of solubility of molybdenum. J. Non-Cryst. Solids 2019, 510, 172–178. [Google Scholar] [CrossRef]
- Rodríguez-Penalonga, L.; Soria, B.Y.M. A Review of the Nuclear Fuel Cycle Strategies and the Spent Nuclear Fuel Management Technologies. Energies 2017, 10, 1235. [Google Scholar] [CrossRef] [Green Version]
- Sengupta, P. A review on immobilization of phosphate containing high level nuclear wastes within glass matrix—Present status and future challenge. J. Hazard. Mater. 2012, 235–236, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Pinet, O.; Hollebecque, J.F.; Hugon, I.; Debono, V.; Campayo, L.; Vallat, C.; Lemaitre, V. Glass ceramic for the vitrification of high level waste with a high molybdenum content. J. Nucl. Mater. 2019, 519, 121–127. [Google Scholar] [CrossRef] [Green Version]
- Wang, F.; Wang, Y.; Liao, Q.; Zhang, J.; Zhu, Y. Immobilization of a simulated HLW in phosphate based glasses/glass-ceramics by melt-quenching process. J. Non-Cryst. Solids 2020, 545, 120246. [Google Scholar] [CrossRef]
- Bohre, A.; Avasthi, K.; Pet’kov, V.I. Vitreous and crystalline phosphate high level waste matrices: Present status and future challenges. J. Ind. Eng. Chem. 2017, 50, 1–14. [Google Scholar] [CrossRef]
- Yu, H.; Xin, R.; Zhang, X.; Liu, H.; Zheng, K.; Zhao, J.; Zhan, L.; Xu, C.; Wan, W.; Zhu, Y.; et al. Crystallization behavior, quantitation of Ce3+/Ce4+ and chemical stability analysis of multiple alkaline earths borosilicate glasses for immobilizing simulated tetravalent actinides. J. Non-Cryst. Solids 2021, 558, 1206542. [Google Scholar] [CrossRef]
- Brehault, A.; Patil, D.; Kamat, H.; Youngman, R.E.; Thirion, L.M.; Mauro, J.C.; Corkhill, C.L.; McCloy, J.S.; Goel, A. Compositional Dependence of Solubility/Retention of Molybdenum Oxides in Aluminoborosilicate-Based Model Nuclear Waste Glasses. J. Phys. Chem. B 2018, 122, 1714–1729. [Google Scholar] [CrossRef] [Green Version]
- Chouard, N.; Caurant, D.; Majérus, O.; Dussossoy, J.L.; Klimin, S.; Pytalev, D.; Baddour-Hadjean, R.; Pereira-Ramos, J.P. Effect of MoO3, Nd2O3, and RuO2 on the crystallization of soda–lime aluminoborosilicate glasses. J. Mater. Sci. 2014, 50, 219–241. [Google Scholar] [CrossRef]
- Schuller, S.; Pinet, O.; Penelon, B. Liquid-Liquid Phase Separation Process in Borosilicate Liquids Enriched in Molybdenum and Phosphorus Oxides. J. Am. Ceram. Soc. 2011, 94, 447–454. [Google Scholar] [CrossRef]
- Caurant, D.; Majérus, O.; Fadel, E.; Quintas, A.; Gervais, C.; Charpentier, T.; Neuville, D. Structural investigations of borosilicate glasses containing MoO3 by MAS NMR and Raman spectroscopies. J. Nucl. Mater. 2010, 396, 94–101. [Google Scholar] [CrossRef]
- Achigar, S.; Caurant, D.; Régnier, E.; Majérus, O. Dismantling nuclear waste rich in P2O5, MoO3 and ZrO2: How do these oxides incorporate in aluminoborosilicate glasses? J. Nucl. Mater. 2021, 544, 152731. [Google Scholar] [CrossRef]
- Zhou, J.; Liao, Q.; Wang, F.; Wang, Y.; Zhu, H.; Zhu, Y. Effect of Na2O and CaO on the solubility and crystallization of Mo in borosilicate glasses. J. Non-Cryst. Solids 2021, 557, 120623. [Google Scholar] [CrossRef]
- Krishnamurthy, A.; Kroeker, S. Improving Molybdenum and Sulfur Vitrification in Borosilicate Nuclear Waste Glasses Using Phosphorus: Structural Insights from NMR. Inorg. Chem. 2022, 61, 73–85. [Google Scholar] [CrossRef]
- Eremyashev, V.E.; Korinevskaya, G.G.; Zherebtsov, D.A. Effect of Phosphorus on Crystallization of Alkaline Molybdenum-Containing Borosilicate Glasses. Defect Diffus. Forum 2021, 410, 720–724. [Google Scholar] [CrossRef]
- Caurant, D.; Majerus, O.; Loiseau, P.; Bardez, I.; Baffier, N.; Dussossoy, J.L. Crystallization of neodymium-rich phases in silicate glasses developed for nuclear waste immobilization. J. Nucl. Mater. 2006, 354, 143–162. [Google Scholar] [CrossRef]
- He, Y.; Lü, Y.; Zhang, Q. Characterization of monazite glass–ceramics as wasteform for simulated α-HLLW. J. Nucl. Mater. 2008, 376, 201–206. [Google Scholar] [CrossRef]
- Pinet, O.; Hollebecque, J.; Angeli, F.; Gruber, P.; Naline, S. Vitrification of Molybdenum-Rich High-Level Solutions by the Cold Crucible Melter Process-11502. In Proceedings of the WM2011 Conference, Phoenix, AZ, USA, 28 February–3 March 2011. [Google Scholar]
- Pipiras, V.; Taqqu, M.S. Vitrification of HLW Produced by Uranium/Molybdenum Fuel Reprocessing in COGEMA’s Cold Crucible Melter. Off. Sci. Technol. Inf. 2003, 3, 1585–1591. [Google Scholar]
- Chouard, N.; Caurant, D.; Majérus, O.; Guezi-Hasni, N.; Pereira-Ramos, J.P. Thermal stability of SiO2-B2O3-Al2O3-Na2O-CaO glasses with high Nd2O3 and MoO3 concentrations. J. Alloys Compd. 2016, 671, 84–99. [Google Scholar] [CrossRef]
- Mckeown, D.A.; Gan, H.; Pegg, I.L. X-ray absorption and Raman spectroscopy studies of molybdenum environments in borosilicate waste glasses. J. Nucl. Mater. 2017, 488, 143–149. [Google Scholar] [CrossRef] [Green Version]
- Patil, D.S.; Konale, M.; Gabel, M.; Neill, O.K.; Crum, J.V.; Goel, A.; Stennett, M.C.; Hyatt, N.C.; Mccloy, J.S. Impact of Rare Earth Ion Size on the Phase Evolution of MoO3-Containing Aluminoborosilicate Glass-Ceramics. J. Nucl. Mater. 2018, 510, 539–550. [Google Scholar] [CrossRef]
- Krishnamurthy, A.; Nguyen, T.; Fayek, M.; Shabaga, B.; Kroeker, S. Network Structure and Dissolution Properties of Phosphate-Doped Borosilicate Glasses. J. Phys. Chem. C 2020, 124, 21184–21196. [Google Scholar] [CrossRef]
- Ladislav, K.; Oksana, K.; Petr, K.; Petr, M.; Lionel, M.; Bertrand, R. Crystallization of sodium molybdate-phosphate and tungstate-phosphate glasses. J. Non-Cryst. Solids 2018, 500, 42–48. [Google Scholar]
- Szumera, M. Structural investigations of silicate-phosphate glasses containing MoO3 by FTIR, Raman and 31P MAS NMR spectroscopies. Spectrochim. Acta A—Mol. Biomol. Spectrosc. 2014, 130, 1–6. [Google Scholar] [CrossRef]
- Szumera, M.; Wacławska, I. Effect of molybdenum addition on the thermal properties of silicate–phosphate glasses. J. Therm. Anal. Calorim. 2012, 109, 649–655. [Google Scholar] [CrossRef]
- Cheng, X. Phosphate-Doped Borosilicate Enamel Coating Used to Protect Reinforcing Steel from Corrosion; Missouri University of Science and Technology: Rolla, MO, USA, 2014. [Google Scholar]
- Muñoz, F.; Montagne, L.; Delevoye, L. Influence of phosphorus speciation on the phase separation of Na2O-B2O3-SiO2 glasses. Phys. Chem. Glasses-Eur. J. Glass Sci. Technol. B 2008, 49, 339–345. [Google Scholar]
- Muñoz, F.; Montagne, L.; Delevoye, L.; Durán, A.; Pascual, L.; Cristol, S.; Paul, J.F. Phosphate speciation in sodium borosilicate glasses studied by Nuclear Magnetic Resonance. J. Non-Cryst. Solids 2006, 352, 2958–2968. [Google Scholar] [CrossRef]
- Saiki, K.; Sakida, S.; Benino, Y.; Nanba, T. Phase separation of borosilicate glass containing phosphorus. J.-Ceram. Soc. Jpn. 2010, 118, 603–607. [Google Scholar] [CrossRef] [Green Version]
- Rong, C.; Wong-Moon, K.C.; Li, H.; Hrma, P.; Cho, H. Solid-state NMR investigation of phosphorus in aluminoborosilicate glasses. J. Non-Cryst. Solids 1998, 223, 32–42. [Google Scholar] [CrossRef]
- Oueslati-Omrani, R.; Hamzaoui, A.H.; Chtourou, R.; M’nif, A. thermal and optical properties of phosphate glasses doped with SiO2. J. Non-Cryst. Solids 2018, 481, 10–16. [Google Scholar] [CrossRef]
- Koudelka, L.; MošNer, P. Study of the structure and properties of Pb–Zn borophosphate glasses. J. Non-Cryst. Solids 2001, 293, 635–641. [Google Scholar] [CrossRef]
- Magnin, M.; Schuller, S.; Mercier, C.; Trébosc, J.; Caurant, D.; Majérus, O.; Angéli, F.; Charpentier, T.; Jantzen, C. Modification of Molybdenum Structural Environment in Borosilicate Glasses with Increasing Content of Boron and Calcium Oxide by 95Mo MAS NMR. J. Am. Ceram. Soc. 2011, 94, 4274–4282. [Google Scholar] [CrossRef]
- ASTM C1285-21; Standard Test Methods for Determining Chemical Durability of Nuclear, Hazardous, and Mixed Waste Glasses and Multiphase Glass Ceramics: The Product Consistency Test (PCT). ASTM International: West Conshohocken, PA, USA, 2002.
- Greer, B.J.; Kroeker, S. Characterisation of heterogeneous molybdate and chromate phase assemblages in model nuclear waste glasses by multinuclear magnetic resonance spectroscopy. Phys. Chem. Chem. Phys. 2012, 14, 7375–7383. [Google Scholar] [CrossRef]
- Kamitsos, E.I.; Kapoutsis, J.A.; Jain, H.; Hsieh, C.H. Vibrational study of the role of trivalent ions in sodium trisilicate glass. J. Non-Cryst. Solids 1994, 171, 31–45. [Google Scholar] [CrossRef]
- Chatterjee, S.; Barik, S.K.; Choudhary, R.N.P. Studies of structural, spectroscopic and electrical properties of sodium molybdate ceramics. J. Mater. Sci. Mater. Electron. 2013, 24, 3359–3364. [Google Scholar] [CrossRef]
- Luz-Lima, C.; Saraiva, G.D.; Filho, A.G.S.; Paraguassu, W.; Freire, P.T.C.; Mendes Filho, J. Raman spectroscopy study of Na2MoO4·2H2O and Na2MoO4 under hydrostatic pressure. J. Raman Spectrosc. 2009, 41, 576–581. [Google Scholar] [CrossRef]
- Koudelka, L.; Rösslerová, I.; Holubová, J.; Mošner, P.; Montagne, L.; Revel, B. Structural study of PbO–MoO3–P2O5 glasses by Raman and NMR spectroscopy. J. Non-Cryst. Solids 2011, 357, 2816–2821. [Google Scholar] [CrossRef]
- Deng, L.; Zhang, X.; Li, B.; Jia, X.; Zhang, M.; Ouyang, S. Influence of crucible material on the microstructure and properties of iron rich glass-ceramics. J. Wuhan Univ. Technol.-Mater. Sci. Ed. 2018, 33, 49–55. [Google Scholar] [CrossRef]
- Sawangboon, N.; Nizamutdinova, A.; Uesbeck, T.; Limbach, R.; Meechoowas, E.; Tapasa, K.; Möncke, D.; Wondraczek, L.; Kamitsos, E.I.; Wüllen, L.; et al. Modification of silicophosphate glass composition, structure, and properties via crucible material and melting conditions. Int. J. Appl. Glass Sci. 2019, 11, 46–57. [Google Scholar] [CrossRef]
- Hammad, A.H.; Abdelghany, A.M. Optical and structural investigations of zinc phosphate glasses containing vanadium ions-ScienceDirect. J. Non-Cryst. Solids 2016, 433, 14–19. [Google Scholar] [CrossRef]
- Wu, L.; Li, H.; Wang, X.; Xiao, J.; Teng, Y.; Li, Y.; Vance, E. Effects of Nd Content on Structure and Chemical Durability of Zirconolite–Barium Borosilicate Glass-Ceramics. J. Am. Ceram. Soc. 2016, 99, 4093–4099. [Google Scholar] [CrossRef]






| Samples | SiO2 | B2O3 | Na2O | P2O5 | MoO3 |
|---|---|---|---|---|---|
| NBS | 53.00 | 21.00 | 26.00 | ||
| NBS-1Mo | 52.47 | 20.79 | 25.74 | 1 | |
| NBS-2Mo | 51.94 | 20.58 | 25.48 | 2 | |
| NBS-3Mo | 51.41 | 20.37 | 25.22 | 3 | |
| NBS-4Mo | 50.88 | 20.16 | 24.96 | 4 | |
| NBS-2P-4Mo | 49.82 | 19.74 | 24.44 | 2 | 4 |
| NBS-4P-4Mo | 48.76 | 19.32 | 23.92 | 4 | 4 |
| M19 | 53.76 | 19.32 | 18.92 | 4 | 4 |
| M24 | 48.76 | 19.32 | 23.92 | 4 | 4 |
| M29 | 43.76 | 19.32 | 28.92 | 4 | 4 |
| M34 | 38.76 | 19.32 | 33.92 | 4 | 4 |
| M34-1 | 37.07 | 18.48 | 32.45 | 8 | 4 |
| Assignment | References | |
|---|---|---|
| Broad 750–775 | Six-membered borate rings containing BO4 tetrahedra | [41] |
| Broad ~322 | Bending vibration of [MoO4]2− | [17] |
| Broad ~900 | Symmetric stretching vibration of [MoO4]2− | [15] |
| Broad 938–954 | vibration modes of Mo-O terminal bonds of Mo-O-P | [44] |
| Sharp 338, 833, 842, 896 | Vibration modes of the [MoO4]2− of Na2MoO4 | [42] |
| Sharp 292, 833, 872, 896, 920, 937 | Vibration modes of the [MoO4]2− of Na2MoO4 2H2O | [43] |
| Samples | SiO2 | Na2O | Al2O3 | P2O5 | MoO3 |
|---|---|---|---|---|---|
| M19 | 66.63(64.92) | 23.45(21.79) | 0(2.72) | 4.96(4.64) | 4.96(5.91) |
| M24 | 60.44(58.83) | 29.65(27.77) | 0(2.68) | 4.96(4.62) | 4.96(6.09) |
| M29 | 54.24(51.86) | 35.85(34.54) | 0(2.76) | 4.96(4.60) | 4.96(6.21) |
| M34 | 48.04(45.94) | 42.04(40.36) | 0(2.70) | 4.96(4.67) | 4.96(6.24) |
| M34-1 | 45.47(43.32) | 39.84(36.15) | 0(5.01) | 9.81(9.23) | 4.91(6.27) |
| Sample | Chemical Shift (ppm) | Phosphate Units | A (%) | Ref. |
|---|---|---|---|---|
| M19 | +5.5 | P0 | 27.40 | [18,27,31,33] |
| +1.5 | P1 | 61.35 | ||
| −4.4 | P21B/Al | 11.25 | ||
| M24 | +5.1 | P0 | 28.38 | |
| +1.5 | P1 | 61.99 | ||
| −6.4 | P21B/Al | 9.63 | ||
| M29 | +4.6 | P0 | 29.16 | |
| +1.5 | P1 | 67.62 | ||
| −6.9 | P21B/Al | 3.22 | ||
| M34 | +4.5 | P0 | 24.95 | |
| +1.3 | P1 | 75.05 |
| Samples | M19 | M24 | M29 | M34 | M34-1 |
|---|---|---|---|---|---|
| Tg (°C) | 460 | 466 | 473 | 475 | 475 |
| Tc (°C) | 590 | 578 | 581 | 573 | 579 |
| Tc−Tg (°C) | 130 | 112 | 108 | 98 | 104 |
| Na (LRNa)/ × 10−4 g·m−2·d−1 (28 d) | 6.04 | 6.59 | / | / | 6.65 |
| Mo (LRMo)/ × 10−4 g·m−2·d−1 (28 d) | 1.80 | 1.29 | / | / | 3.73 |
| P (LRP)/ × 10−4 g·m−2·d−1 (28 d) | 2.21 | 3.08 | / | / | 5.83 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, H.; Zhu, Y.; Huo, J.; Cui, Z.; Zhang, X.; Jiang, Q.; Yang, D.; Meng, B. Effect of P2O5 and Na2O on the Solubility of Molybdenum and Structural Features in Borosilicate Glass. Materials 2022, 15, 5464. https://doi.org/10.3390/ma15155464
Liu H, Zhu Y, Huo J, Cui Z, Zhang X, Jiang Q, Yang D, Meng B. Effect of P2O5 and Na2O on the Solubility of Molybdenum and Structural Features in Borosilicate Glass. Materials. 2022; 15(15):5464. https://doi.org/10.3390/ma15155464
Chicago/Turabian StyleLiu, Hao, Yongchang Zhu, Jichuan Huo, Zhu Cui, Xingquan Zhang, Qin Jiang, Debo Yang, and Baojian Meng. 2022. "Effect of P2O5 and Na2O on the Solubility of Molybdenum and Structural Features in Borosilicate Glass" Materials 15, no. 15: 5464. https://doi.org/10.3390/ma15155464





