Mechanical Characterization of Dental Prostheses Manufactured with PMMA–Graphene Composites
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
- Graphite powder with a purity higher 99.5% (Merck, Darmstadt, Germany);
- Hydrazine hydrate (HH, 80%) (Loba Chemi. Pvt. Ltd., Mumbai, India);
- Methyl methacrylate (MMA) (Acros Chemical Co., Loughborough, UK, 99%);
- Benzoyl peroxide (BP, BDH Chemicals Ltd., East Yorkshire, UK) was used as an initiator;
- Potassium permanganate (KMnO4, >99%) (Merck, Darmstadt, Germany);
- Hydrogen peroxide (H2O2, 30%) (Merck Darmstadt, Germany).
- -
- Three washes with sodium lauryl sulfate 20% and water for 5 min in ultrasound;
- -
- Three washes in distilled water for 5 min in ultrasound;
- -
- Three washes in 96% ethanol for 5 min under ultrasound;
- -
- Drying with nitrogen N2 (g) for 5 min.
2.2. Raman Spectroscopy
2.3. Gel Permeation Chromatography (GPC)
2.4. Shore Hardness (HSD)
2.5. Hardness by Vickers Indentation
2.6. Compression Test
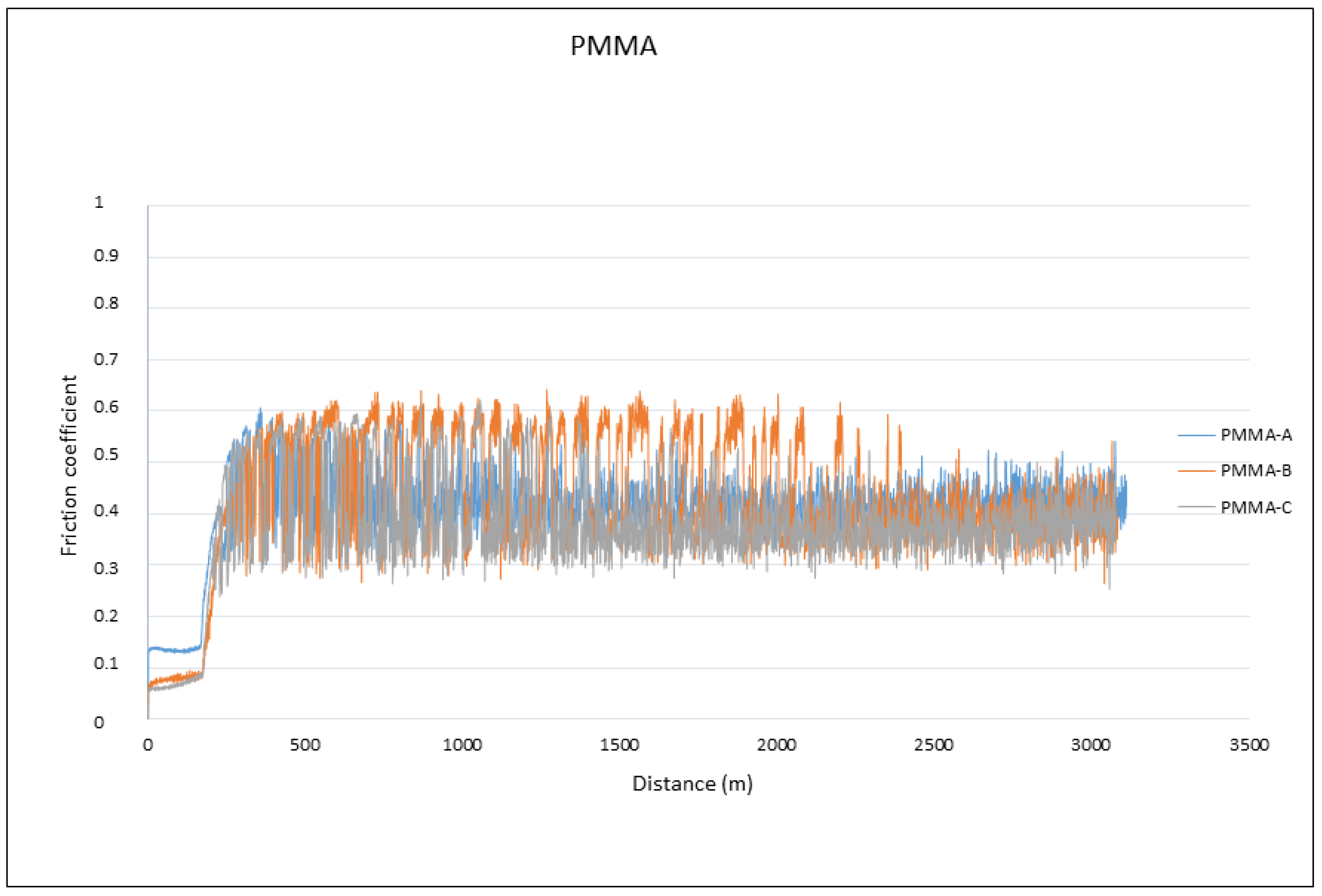
2.7. Surface Wear (“Pin-on-Disc Test”)
2.8. Field Emission Scanning Electron Microscopy (FESEM)
2.9. Statistical Analysis
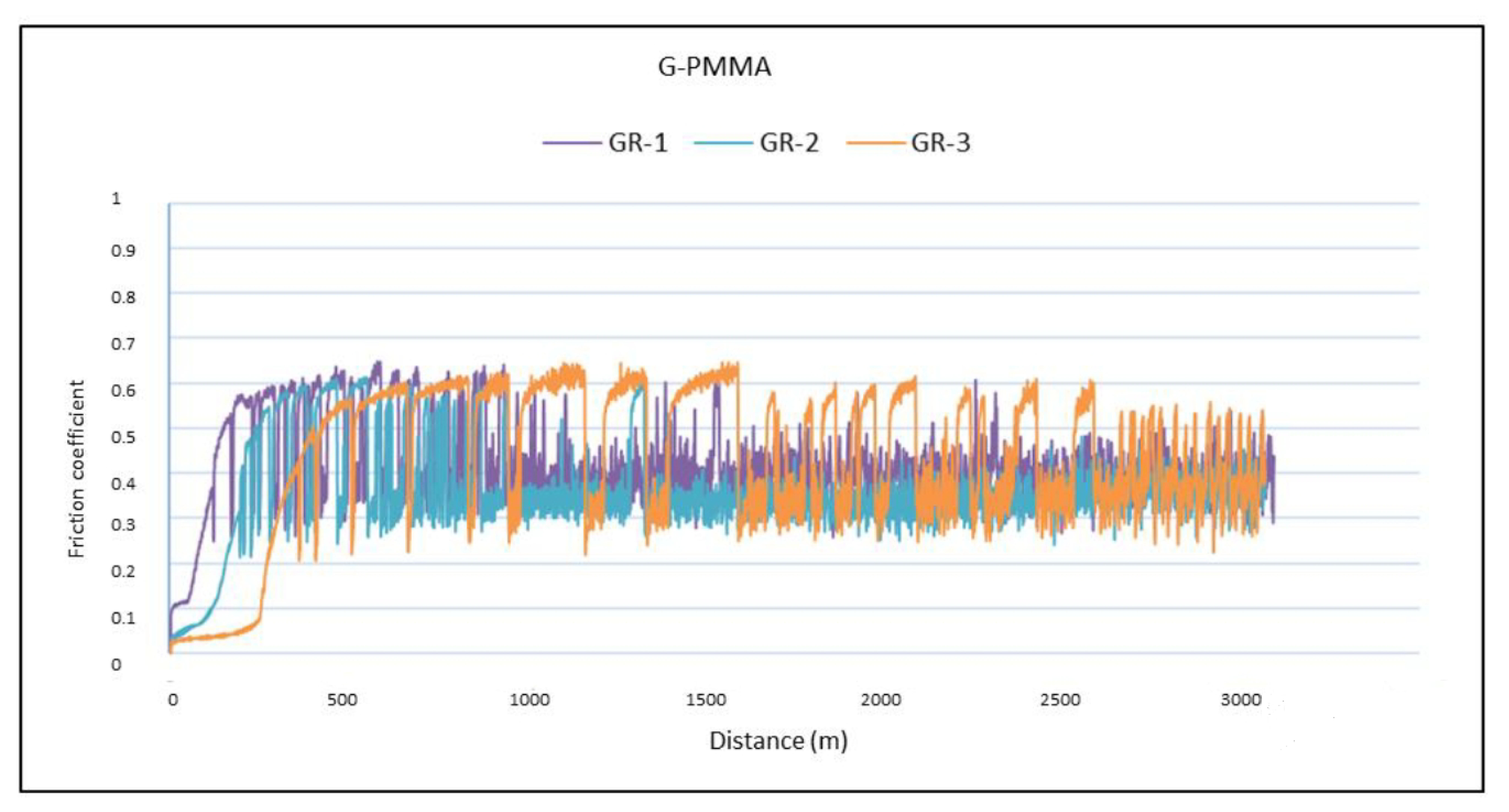
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Denizoğlu, S.; Duymuş, Z.Y.; Akyalçin, S. Evaluation of Ion Release from Two Base-Metal Alloys at Various pH Levels. J. Int. Med. Res. 2004, 32, 33–38. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Benatti, O.F.M.; Miranda, W.G.; Muench, A. In vitro and in vivo corrosion evaluation of nickel-chromium and copper-aluminumbased alloys. J. Prosthet. Dent. 2000, 84, 360–363. [Google Scholar] [CrossRef] [PubMed]
- Wataha, J.C.; Lockwood, P.E.; Khajotia, S.S. Effect of pH on element release from dental casting alloys. J. Prosthet. Dent. 1998, 80, 691–698. [Google Scholar] [CrossRef]
- Huttenhower, C.; Gevers, D.; Knight, R.; Abubucker, S.; Badger, J.H.; Chinwalla, A.T.; Creasy, H.H.; Earl, A.M.; Fitzgerald, M.G.; Fulton, R.S.; et al. Structure, function and diversity of the healthy human microbiome. Nature 2012, 486, 207–214. [Google Scholar] [CrossRef] [Green Version]
- Wade, W.G. The oral microbiome in health and disease. Pharmacol. Res. 2013, 69, 137–143. [Google Scholar] [CrossRef]
- Dewhirst, F.E.; Chen, T.; Izard, J.; Paster, B.J.; Tanner, A.C.R.; Yu, W.H.; Lakshmanan, A.; Wade, W.G. The human oral microbiome. J. Bacteriol. 2010, 192, 5002–5017. [Google Scholar] [CrossRef] [Green Version]
- Padrós, R.; Giner-Tarrida, L.; Herrero-Climent, M.; Punset, M.; Gil, F.J. Corrosion Resistance and Ion Release of Dental Prosthesis of CoCr Obtained by CAD-CAM Milling, Casting and Laser Sintering. Metals 2020, 10, 827. [Google Scholar] [CrossRef]
- Yang, K.; Feng, L.; Hong, H.; Cai, W.; Liu, Z. Preparation and functionalization of graphene nanocomposites for biomedical applications. Nat. Protoc. 2013, 8, 2392–2403. [Google Scholar] [CrossRef] [Green Version]
- Lee, C.; Wei, X.; Kysar, J.W.; Hone, J. Measurement of the elastic properties and intrinsic strength of monolayer graphene. Science 2008, 321, 385–388. [Google Scholar] [CrossRef]
- Lee, J.H.; Jo, J.K.; Kim, D.A.; Patel, K.D.; Kim, H.W.; Lee, H.H. Nano-graphene oxide incorporated into PMMA resin to prevent microbial adhesion. Dent. Mater. 2018, 34, e63–e72. [Google Scholar] [CrossRef]
- Tom, T.N.; Uthappa, M.; Sunny, K.; Begum, F.; Nautiyal, M.; Tamore, S. Provisional restorations: An overview of materials used. J. Adv. Clin. Res. Insights 2016, 3, 212–214. [Google Scholar] [CrossRef]
- Ghaffari, T.; Hamedirad, F.; Ezzati, B. In Vitro Comparison of Compressive and Tensile Strengths ofAcrylic Resins Reinforced by Silver Nanoparticles at 2% and 0.2% Concentrations. J. Dent.Res. Dent.Clin. Dent. Prospect. 2014, 8, 204. [Google Scholar] [CrossRef]
- Gamal, R.; Gomaa, Y.F.; Said, A.M. Incorporating nano graphene oxide to poly-methyl methacrylate; antibacterial effect and thermal expansion. J. Mod. Res. 2019, 1, 19–23. [Google Scholar] [CrossRef] [Green Version]
- Rashahmadi, S.; Hasanzadeh, R.; Mosalman, S. Improving the mechanical properties of poly methyl methacrylate nanocomposites for dentistry applications reinforced with different nanoparticles. Polym. Plast. Technol. Eng. 2017, 56, 1730–1740. [Google Scholar] [CrossRef]
- Azevedo, L.; Antonaya-Martin, J.L.; Molinero-Mourelle, P.; Del Río-Highsmith, J. Improving PMMA resin using graphene oxide for a definitive prosthodontic rehabilitation—A clinical report. J. Clin. Exp. Dent. 2019, 11, e670–e674. [Google Scholar] [CrossRef] [PubMed]
- Ruse, N.D.; Sadoun, M.J. Resin-composite blocks for dental CAD/CAM applications. J. Dent. Res. 2014, 93, 1232–1234. [Google Scholar] [CrossRef] [Green Version]
- Stawarczyk, B.; Ozcan, M.; Trottmann, A.; Schmutz, F.; Roos, M.; Hammerle, C. Two-body wear rate of CAD/CAM resin blocks and their enamel antagonists. J. Prosthet. Dent. 2013, 109, 325–332. [Google Scholar] [CrossRef] [Green Version]
- Stawarczyk, B.; Sener, B.; Trottmann, A.; Roos, M.; Ozcan, M.; Hammerle, C.H. Discoloration of manually fabricated resins and industrially fabricated CAD/CAM blocks versus glass-ceramic: Effect of storage media, duration, and subsequent polishing. Dent. Mater. J. 2012, 31, 377–383. [Google Scholar] [CrossRef] [Green Version]
- Wiegand, A.; Stucki, L.; Hoffmann, R.; Attin, T.; Stawarczyk, B. Repairability of CAD/CAM highdensity PMMA- and composite-based polymers. Clin. Oral. Investig. 2015, 19, 2007–2013. [Google Scholar] [CrossRef]
- Matsuo, H.; Suenaga, H.; Takahashi, M.; Suzuki, O.; Sasaki, K.; Takahashi, N. Deterioration of polymethyl methacrylate dentures in the oral cavity. Dent. Mater. J. 2015, 34, 234–239. [Google Scholar] [CrossRef] [Green Version]
- Rickman, L.J.; Padipatvuthikul, P.; Satterthwaite, J.D. Contemporary denture base resins: Part 1. Dent. Update 2012, 39, 25–30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rawls, H.R. Dental polymers. In Phillips Science of Dental Materials; Anusavice, K.L., Ed.; WB Saunders Co.: St Louis, MO, USA, 2003; pp. 145–146. [Google Scholar]
- El Bahra, S.; Ludwig, K.; Samran, A.; Freitag-Wolf, S.; Kern, M. Linear and volumetric dimensional changes of injection-molded PMMA denture base resins. Dent. Mater. 2013, 29, 1091–1097. [Google Scholar] [CrossRef] [PubMed]
- Cuijpers, V.M.; Jaroszewicz, J.; Anil, S.; Al Farraj Aldosari, A.; Walboomers, X.F.; Jansen, J.A. Resolution, sensitivity, and in vivo application of high-resolution computed tomography for titanium-coated polymethyl methacrylate (PMMA) dental implants. Clin. Oral. Implant. Res. 2014, 25, 359–365. [Google Scholar] [CrossRef]
- Slane, J.; Vivanco, J.; Rose, W.; Ploeg, H.-L.; Squire, M. Mechanical, material, and antimicrobial properties of acrylic bone cement impregnated with silver nanoparticles. Mater. Sci. Eng. C 2015, 48, 188–196. [Google Scholar] [CrossRef]
- Peyton, F.A. History of resins in dentistry. Dent. Clin. N. Am. 1975, 19, 211–212. [Google Scholar] [CrossRef]
- Haselton, D.R.; Diaz-Arnold, A.M.; Vargas, M.A. Flexural strength of provisional crown and fixed partial denture resins. J. Prosthet. Dent. 2002, 87, 225–228. [Google Scholar] [CrossRef] [PubMed]
- Anusavice, K.J.; Shen, C.; Rawls, H.R. Phillips’ Science of Dental Materials; Elsevier Health Sciences: Amsterdam, The Netherlands, 2012; ISBN 978-1-4377-2418-9. [Google Scholar]
- Raj, P.A.; Dentino, A.R. Denture polymers with antimicrobial properties: A review of the development and current status of anionic poly(methyl methacrylate) polymers. Future Med. Chem. 2013, 5, 1635–1645. [Google Scholar] [CrossRef]
- Wen, J.; Jiang, F.; Yeh, C.-K.; Sun, Y. Controlling fungal biofilms with functional drug delivery denture biomaterials. Colloids Surf B Biointerfaces 2016, 140, 19–27. [Google Scholar] [CrossRef] [Green Version]
- Gocke, R.; Gerath, F.; von Schwanewede, H. Quantitative determination of salivary components in the pellicle on PMMA denture base material. Clin. Oral Invest. 2002, 6, 227–235. [Google Scholar] [CrossRef]
- Samaranayake, L.P.; Leung, W.K.; Jin, L. Oral Mucosal Fungal Infection. Periodontol. 2000 2009, 49, 29–59. [Google Scholar] [CrossRef]
- Webb, B.C.; Thomas, C.J.; Willcox, M.D.; Harty, D.W.; Knox, K.W. Candida-associated denture stomatitis. Aetiology and management: A review. Part 3. Treatment of oral candidosis. Aust. Dent. J. 1998, 43, 244–249. [Google Scholar] [CrossRef]
- Busscher, H.J.; Rinastiti, M.; Siswomihardjo, W.; van der Mei, H.C. Biofilm formation on dental restorative and implant materials. J. Dent. Res. 2010, 89, 657–665. [Google Scholar] [CrossRef] [PubMed]
- Flemming, T.; Beikler, T. Control of oral biofilms. Priodontology 2000 2011, 55, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wu, R.; Fan, Y.; Liao, S.; Wang, Y.; Wen, Z.; Xu, X. Antibacterial dental composites with chlorhexidine and mesoporous silica. J. Dent. Res. 2014, 93, 1283–1289. [Google Scholar] [CrossRef] [PubMed]
- Jo, J.-K.; El-Fiqi, A.; Lee, J.-H.; Kim, D.-A.; Kim, H.-W.; Lee, H.-H. Rechargeable microbial anti-adhesive polymethyl methacrylate incorporating silver sulfadiazine-loaded mesoporous silica nanocarriers. Dent. Mater. 2017, 33, e361–e372. [Google Scholar] [CrossRef]
- Lee, J.-H.; El-Fiqi, A.; Jo, J.-K.; Kim, D.-A.; Kim, S.-C.; Jun, S.-K.; Kim, H.-W.; Lee, H.-H. Development of long-term antimicrobial poly(methyl methacrylate) by incorporating mesoporous silica nanocarriers. Dent. Mater. 2016, 32, 1564–1574. [Google Scholar] [CrossRef] [PubMed]
- Beyene, H.D.; Werkneh, A.A.; Bezabh, H.K.; Ambaye, T.G. Synthesis paradigm and applications of silver nanoparticles (AgNPs), a review. Sustainable Mater. Technol. 2017, 13, 18–23. [Google Scholar] [CrossRef]
- Roco, M.C.; Hersam, M.C.; Mirkin, C.A. Nanotechnology Research Directions for Societal Needs in 2020, Retrospective and Outlook, 1st ed.; Springer: Berlin/Heidelberg, Germany, 2011. [Google Scholar] [CrossRef] [Green Version]
- An, Y.; Liu, B.; Yan, X.; Pet, J.; Liu, W. The experimental study on wear resistance of the denture base material reinforced with graphene oxide. Tribology 2013, 33, 222–228. [Google Scholar]
- Song, J.; Zhang, J.; Lin, C. Influence of graphene oxide on the tribological and electrical properties of PMMA composites. J. Nanomater. 2013, 2013, 846102. [Google Scholar] [CrossRef] [Green Version]
- Chen, X.; Mao, S.S. Titanium Dioxide Nanomaterials: Synthesis, Properties, Modifications, and Applications. Chem. Rev. 2007, 107, 2891–2959. [Google Scholar] [CrossRef]
- Tolou, N.B.; Fathi, M.H.; Monshi, A.; Mortazavi, V.S.; Shirani, F.; Sichani, M.M. The Effect of Adding TiO2 Nanoparticles on Dental Amalgam Properties. Iran. J. Mater. Sci. Eng. 2013, 10, 46–56. [Google Scholar]
- Totu, E.E.; Nechifor, A.C.; Nechifor, G.; Aboul-Enein, H.Y.; Cristache, C.M. Poly (Methyl Methacrylate) with TiO2 Nanoparticles Inclusion for Stereolitographic Complete Denture Manufacturing—The Future in Dental Care for Elderly Edentulous Patients. J. Dent. 2017, 59, 68–77. [Google Scholar] [CrossRef] [PubMed]
- El-Zaher, N.A.; Melegy, M.S.; Guirguis, O.W. Thermal and Structural Analyses of PMMA/TiO2 Nanoparticles Composites. Nat. Sci. 2014, 6, 859–870. [Google Scholar] [CrossRef] [Green Version]
- Turagam, N.; Mudrakola, D.P. Effect of Micro-Additions of Carbon Nanotubes to Polymethylmethacrylate on Reduction in Polymerization Shrinkage. J. Prosthodont. 2012, 22, 105–111. [Google Scholar] [CrossRef]
- Devoret, M.H.; Dai, H. Polymethylmethacrylate augmented with carbon nanotubes. Chem. Mater. 2007, 12, 1049–1063. [Google Scholar]
- Kumar, S.; Doshi, H.; Srinivasarao, M.; O Park, J.; A Schiraldi, D. Fibers from polypropylene/nano carbon fiber composites. Polymer 2002, 43, 1701–1703. [Google Scholar] [CrossRef]
- Chen, J.; Peng, H.; Wang, X.; Shao, F.; Yuan, Z.; Han, H. Graphene oxide exhibits broad-spectrum antimicrobial activity against bacterial phytopathogens and fungal conidia by intertwining and membrane perturbation. Nanoscale 2014, 6, 1879–1889. [Google Scholar] [CrossRef]
- Morimune, S.; Nishino, T.; Goto, T. Ecological approach to graphene oxide reinforced poly (methyl methacrylate) nanocomposites. ACS Appl. Mater. Interfaces 2012, 4, 3596–3601. [Google Scholar] [CrossRef]
- Chen, H.; Wang, B.; Gao, D.; Guan, M.; Zheng, L.; Ouyang, H.; Chai, Z.; Zhao, Y.; Feng, W. Broad-spectrum antibacterial activity of carbon nanotubes to human gut bacteria. Small 2013, 9, 2735–2746. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, A.A. Electric Field Effect in Atomically Thin Carbon Films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef] [Green Version]
- Bei, H.P.; Yang, Y.; Zhang, Q.; Tian, Y.; Luo, X.; Yang, M.; Zhao, X. GrapheneBased Nanocomposites for Neural Tissue Engineering. Molecules 2019, 24, 658. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agustín-Panadero, R.; Solá-Ruíz, M.F.; Chust, C.; Ferreiroa, A. Fixed dental prostheses with vertical tooth preparations without finish lines: A report of two patients. J. Prosthet. Dent. 2016, 115, 520–526. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Tao, J.; Yu, B.; Dai, L. Characterization of multiwalled carbon nanotube-polymethyl methacrylate composite resins as denture base materials. J. Prosthet. Dent. 2014, 111, 318–326. [Google Scholar] [CrossRef]
- Fernandez, M.; Mendez, J.A.; Vazquez, B.; Roman, S.; Ginebra, M.P.; Gil, F.J.; Manero, J.M.; Planell, J.A. Acrylic-phosphate glasses composites as self-curing controlled delivery systems of antibiotics. J. Mater. Sci. Mater Med. J. 2002, 13, 1251–1257. [Google Scholar]
- Padovani, G.C.; Feitosa, V.P.; Sauro, S.; Tay, F.R.; Durán, G.; Paula, A.J.; Durán, N. Advances in Dental Materials through Nanotechnology: Facts, Perspectives and Toxicological Aspects. Trends Biotechnol. 2015, 33, 621–636. [Google Scholar] [CrossRef]
- Kim, K.I.; Kim, D.A.; Patel, K.; Shin, U.S.; Kim, H.W.; Lee, J.H.; Lee, H.H. Carbon nanotube incorporation in PMMA to prevent microbial adhesion. Sci. Rep. 2019, 9, 10. [Google Scholar] [CrossRef]
- Vazquez, B.; Ginebra, M.P.; Gil, F.J.; Planell, J.A.; San Roman, J. Acrylic bone cements modified with beta-TCP particles encapsulated with poly(ethylene Glycol). Biomaterials 2005, 26, 4309–4316. [Google Scholar] [CrossRef]
- Banerjee, A.N. Graphene and its derivatives as biomedical materials: Future prospects and challenges. Interface Focus 2018, 8, 20170056. [Google Scholar] [CrossRef]
- Ferrari, A.C.; Meyer, J.C.; Scardaci, V.; Casiraghi, C.; Lazzeri, M.; Mauri, F.; Piscanec, S.; Jiang, D.; Novoselov, K.S.; Roth, S.; et al. Raman spectrum of graphene and graphene layers. Phys. Rev. Lett. 2006, 97, 187401. [Google Scholar] [CrossRef] [Green Version]
- Zhen, Z.; Zhu, H. 1—Structure and Properties of Graphene; Hongwei, Z., Zhiping, X., Dan, X., Ying, F., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 1–12. ISBN 9780128126516. [Google Scholar] [CrossRef]
- Foo, M.E.; Gopinath, S.C. Feasibility of graphene in biomedical applications. Biomed. Pharmacother. 2017, 94, 354–361. [Google Scholar] [CrossRef]
- Zhu, Y.; Murali, S.; Cai, W.; Li, X.; Suk, J.W.; Potts, J.R.; Ruoff, R.S. Graphene and Graphene Oxide: Synthesis, Properties, and Applications. Adv. Mater. 2010, 22, 3906–3924. [Google Scholar] [CrossRef] [PubMed]
- Geim, A.K.; Novoselov, K.S. The rise of graphene. Nanosci. Technol. 2009, 10, 11–19. [Google Scholar] [CrossRef]
- Bolotin, K.; Sikes, K.; Jiang, Z.; Klima, M.; Fudenberg, G.; Hone, J.; Kim, P.; Stormer, H. Stormer, Ultrahigh electron mobility in suspended graphene. Solid State Commun. 2008, 146, 351–355. [Google Scholar] [CrossRef] [Green Version]
- Morozov, S.V.; Novoselov, K.S.; Katsnelson, M.I.; Schedin, F.; Elias, D.C.; Jaszczak, J.A.; Geim, A.K. Giant Intrinsic Carrier Mobilities in Graphene and Its Bilayer. Phys. Rev. Lett. 2008, 100, 016602. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suk, J.W.; Piner, R.D.; An, J.; Ruoff, R.S. Mechanical properties of monolayer graphene oxide. ACS Nano 2010, 4, 6557–6564. [Google Scholar] [CrossRef]
- Balandin, A.A.; Ghosh, S.; Bao, W.; Calizo, I.; Teweldebrhan, D.; Miao, F.; Lau, C.N. Superior Thermal Conductivity of Single-Layer Graphene. Nano Letters 2008, 8, 902–907. [Google Scholar] [CrossRef]
- Dong, R.; Liu, L. Preparation and properties of acrylic resin coating modified by functional graphene oxide. Appl. Surf. Sci. 2016, 368, 378–387. [Google Scholar] [CrossRef]
- Watson, G.; Starost, K.; Bari, P.; Faisal, N.; Mishra, S.; Njuguna, J. Tensile and flexural properties of hybrid graphene oxide/epoxy carbon fibre reinforced composites. IOP Conf. Ser. Mater. Sci. Eng. 2017, 195, 012009. [Google Scholar] [CrossRef] [Green Version]
- Hu, W.; Peng, C.; Luo, W.; Lv, M.; Li, X.; Li, D. Graphene-based antibacterial paper. ACS Nano 2010, 4, 4317–4323. [Google Scholar] [CrossRef]
- Kim, J.; Kim, Y.R.; Kim, Y.; Lim, K.T.; Seonwoo, H.; Park, S. Graphene-incorporated chitosan substrata for adhesion and differentiation of human mesenchymal stem cells. J. Mater. Chem. B 2013, 1, 933–938. [Google Scholar] [CrossRef]
- Park, S.Y.; Park, J.; Sim, S.H.; Sung, M.G.; Kim, K.S.; Hong, B.H. Enhanced differentiation of human neural stem cells into neurons on graphene. Adv. Mater. 2011, 23, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Novoselov, K.S.; Fal, V.; Colombo, L.; Gellert, P.; Schwab, M.; Kim, K. A roadmap for graphene. Nature 2012, 490, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Li, H.; Ding, C.; Zheng, X.; Li, K. Effects of graphene plates’ adoption on the microstructure, mechanical properties, and in vivo biocompatibility of calcium silicate coating. Int. J. Nanomed. 2015, 10, 3855. [Google Scholar] [CrossRef] [Green Version]
- Poot, M.; van der Zant, H.S. Nanomechanical properties of fewlayer graphene membranes. Appl. Phys. Lett. 2008, 92, 063111. [Google Scholar] [CrossRef]
- Harrison, B.S.; Atala, A. Carbon nanotube applications for tissue engineering. Biomaterials 2007, 28, 344–353. [Google Scholar] [CrossRef]
- Park, C.H.; Park, S.; Lee, D.; Choi, K.S.; Lim, H.P.; Kim, J. Graphene as enabling strategy for dental implant and tissue regeneration. Tissue Eng. Regen. Med. 2017, 14, 481–493. [Google Scholar] [CrossRef]
- Segerstrom, S.; Ruyter, I.E. Adhesion properties in systems of laminated pigmented polymers, carbon-graphite fiber composite framework and titanium surfaces in implant suprastructures. Dent. Mater. 2009, 25, 1169–1177. [Google Scholar] [CrossRef]
- Santos, C.M.; Mangadlao, J.; Ahmed, F.; Leon, A.; Advincula, R.C.; Rodrigues, D.F. Graphene nanocomposite for biomedical applications: Fabrication, antimicrobial and cytotoxic investigations. Nanotechnology 2012, 23, 395101. [Google Scholar] [CrossRef]
- Sun, X.; Liu, Z.; Welsher, K.; Robinson, J.T.; Goodwin, A.; Zaric, S. Nano-graphene oxide for cellular imaging and drug delivery. Nano Res. 2008, 1, 203–212. [Google Scholar] [CrossRef] [Green Version]
- Feng, L.; Zhang, S.; Liu, Z. Graphene based gene transfection. Nanoscale 2011, 3, 1252–1257. [Google Scholar] [CrossRef]
- Lee, W.C.; Lim, C.H.Y.; Shi, H.; Tang, L.A.; Wang, Y.; Lim, C.T. Origin of enhanced stem cell growth and differentiation on graphene and graphene oxide. ACS Nano 2011, 5, 7334–7341. [Google Scholar] [CrossRef] [PubMed]
- Hassan, H.; Abdelsayed, V.; Khder, A.; AbouZeid, K.; Terner, J.; El-Shall, M.; Al-Resayes, S.; El-Azhary, A. Microwave synthesis of graphene sheets supporting metal nanocrystals in aqueous and organic media. J. Mater. Chem. 2009, 19, 3832–3837. [Google Scholar] [CrossRef]
- Hummers, W.S.; Offeman, R.E. Preparation of graphitic oxide. J. Am. Chem. Soc. 1958, 80, 1339. [Google Scholar] [CrossRef]
- Aldosari, M.A.; Othman, A.A.; Alsharaeh, E.H. Synthesis and Characterization of the In Situ Bulk Polymerization of PMMA Containing Graphene Sheets Using Microwave Irradiation. Molecules 2013, 18, 3152–3167. [Google Scholar] [CrossRef]
- Aslanyan, E.; Doynikov, A.; Pivovarov, V. VNIIFTRI, Moscow, Russia. In Metrological characteristics of the national shore d scale hardness standard. In Proceedings of the XVII IMEKO World Congress, Dubrovnik, Croatia, 22–27 June 2003. [Google Scholar]
- He, S.; Song, B.; Li, D.; Zhu, C.; Qi, W.; Wen, Y. A graphene nanoprobe for rapid, sensitive, and multicolor fluorescent DNA analysis. Adv Funct. Mater. 2010, 20, 453–459. [Google Scholar] [CrossRef]
- Chicot, D.; Mercier, D.; Roudet, F.; Silva, K.; Staia, M.; Lesage, J. Comparison of instrumented Knoop and Vickers hardness measurements on various soft materials and hard ceramics. J. Eur. Ceram. Soc. 2007, 27, 1905–1911. [Google Scholar] [CrossRef]
- Kato, K. Classification of wear mechanisms/models. Proceedings of the Institution of Mechanical Engineers, Part J. J. Eng. Tribol. 2002, 216, 349–355. [Google Scholar] [CrossRef]
- Fedel, M. Hemocompatibility of Carbon Nanostructures. J. Carbon Res. 2020, 6, 12. [Google Scholar] [CrossRef] [Green Version]
- Brash, J.L. Studies of protein adsorption relevant to blood compatible materials. In Modern Aspects of Protein Adsorption on Biomaterials; Springer: Dordrecht, The Netherlands, 1991; pp. 39–47. ISBN 978-94-011-3752-2. [Google Scholar]
- Pascual, B.; Vázquez, B.; Gurruchaga, M.; Goñi, I.; Ginebra, M.P.; Gil, F.J.; Planell, J.A.; Levenfeld, B.; San Román, J. New aspects of the effect of size and size distribution on the setting parameters and mechanical properties of acrylic bone cements. Biomaterials 1996, 17, 509–516. [Google Scholar] [CrossRef] [PubMed]
- Pascual, B.; Gurruchaga, M.; Ginebra, M.P.; Gil, F.J.; Planell, J.A.; Goñi, I. Influence of the modification of P/L ratio on a new formulation of acrylic bone cement. Biomaterials 1999, 20, 465–474. [Google Scholar] [CrossRef]
- Vazquez, B.; Ginebra, M.P.; Gil, F.J.; Planell, J.A.; Bravo, A.; San Roman, J. Radiopaque acrylic cements prepared with a new acrilic derivate of iodo-quinoline. Biomaterials 1999, 20, 2047–2053. [Google Scholar] [CrossRef]
- Manero, J.M.; Gil, F.J.; Padros, E.; Planell, J.A. Applications of enviromental sacnning electron microscopy (ESEM) in biomaterials field. Microsc. Res. Tech. 2003, 61, 469–480. [Google Scholar] [CrossRef] [PubMed]
- Alamgir, M.; Nayak, G.C.; Mallick, A.; Tiwari, S.K.; Mondal, S.; Gupta, M. Processing of PMMA nanocomposites containing biocompatible GO and TiO2 nanoparticles. Mater. Manuf. Process. 2018, 33, 1291–1298. [Google Scholar] [CrossRef]
- Khan, A.A.; Mirza, E.H.; Mohamed, B.A.; Alharthi, N.H.; Abdo, H.S.; Javed, R.; Alhur, R.S.; Vall itt u, P.K. Physical mechanical, chemical and thermal properties of nanoscale graphene oxide-poly methylmethacrylate composites. J. Compos. Mater. 2018, 52, 2803–2813. [Google Scholar] [CrossRef]
- Ramanathan, T.; Abdala, A.A.; Stankovich, S.; Dikin, D.A.; Herrera-Alonso, M.; Piner, R.D.; Adamson, D.H.; Schniepp, H.C.; Chen, X.; Ruoff, R.S.; et al. Functionalized graphene sheets for polymer nanocomposites. Nat. Nanotech. 2008, 3, 327–331. [Google Scholar] [CrossRef]
- Wang, W.; Liao, S.; Zhu, Y.; Liu, M.; Zhao, Q.; Fu, Y. Recent applications of nanomaterials in prosthodontics. J. Nanomater. 2015, 3, 3. [Google Scholar] [CrossRef] [Green Version]
- Tecco, S.; Grusovin, M.G.; Sciara, S.; Bova, F.; Pantaleo, G.; Capparé, P. The association between three attitude-related indexes of oral hygiene and secondary implant failures: A retrospective longitudinal study. Int. J. Dent. Hyg. 2018, 16, 372–379. [Google Scholar] [CrossRef]
- Cappare, P.; Ferrini, F.; Mariani, G.; Nagni, M.; Cattoni, F. Implant rehabilitation of edentulous jaws with predominantly monolithic zirconia compared to metal-acrylic prostheses: A 2-year retrospective clinical study. J. Biol. Regul. Homeost. Agents 2021, 35 (Suppl. 1), 99–112. [Google Scholar] [CrossRef]
- Manazza, F.; La Rocca, S.; Nagni, M.; Chirico, L.; Cattoni, F. A simplified digital workflow for the prosthetic finishing of implant rehabilitations: A case report. J. Biol. Regul. Homeost. Agents 2021, 35 (Suppl. 1), 87–97. [Google Scholar] [CrossRef]
- Cattoni, F.; Chirico, L.; Cantile, N.; Merlone, A. Traditional prosthetic workflow for implant rehabilitations with a reduced number of fixtures: Proposal of a protocol. J. Biol. Regul. Homeost. Agents 2021, 35 (Suppl. 1), 31–40. [Google Scholar] [CrossRef]
- Joda, T.; Zarone, F.; Ferrari, M. The complete digital workflow in fixed prosthodontics: A systematic review. BMC Oral Health 2017, 17, 124. [Google Scholar] [CrossRef] [PubMed]
- Cattoni, F.; Merlone, A.; Broggi, R.; Manacorda, M.; Vinci, R. Computer-assisted prosthetic planning and implant design with integrated digital bite registration: A treatment protocol. J. Biol. Regul. Homeost. Agents 2021, 35 (Suppl. 1), 11–29. [Google Scholar] [CrossRef] [PubMed]




| Mn [g·mol−1] | Mw [g·mol−1] | Mz [g·mol−1] | Mz [g·mol−1] | PDI [a.u] | |
|---|---|---|---|---|---|
| PMMA | 51,029–54,175 | 104,175–107,231 | 240,674–251,413 | 663,145–698,799 | 1.979–2.041 |
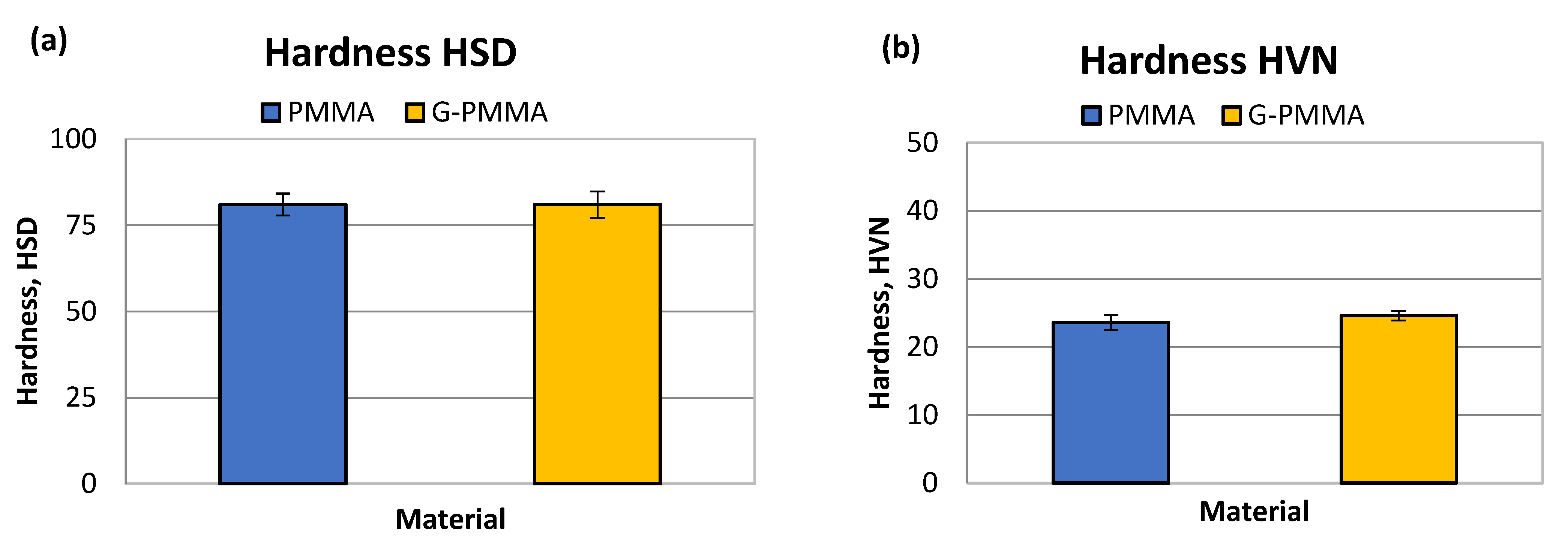
| HSD | HVN | |
|---|---|---|
| PMMA | 81 ± 3.8 | 23.6 ± 1.1 |
| G-PMMA | 81 ± 3.2 | 24.6 ± 0.7 |
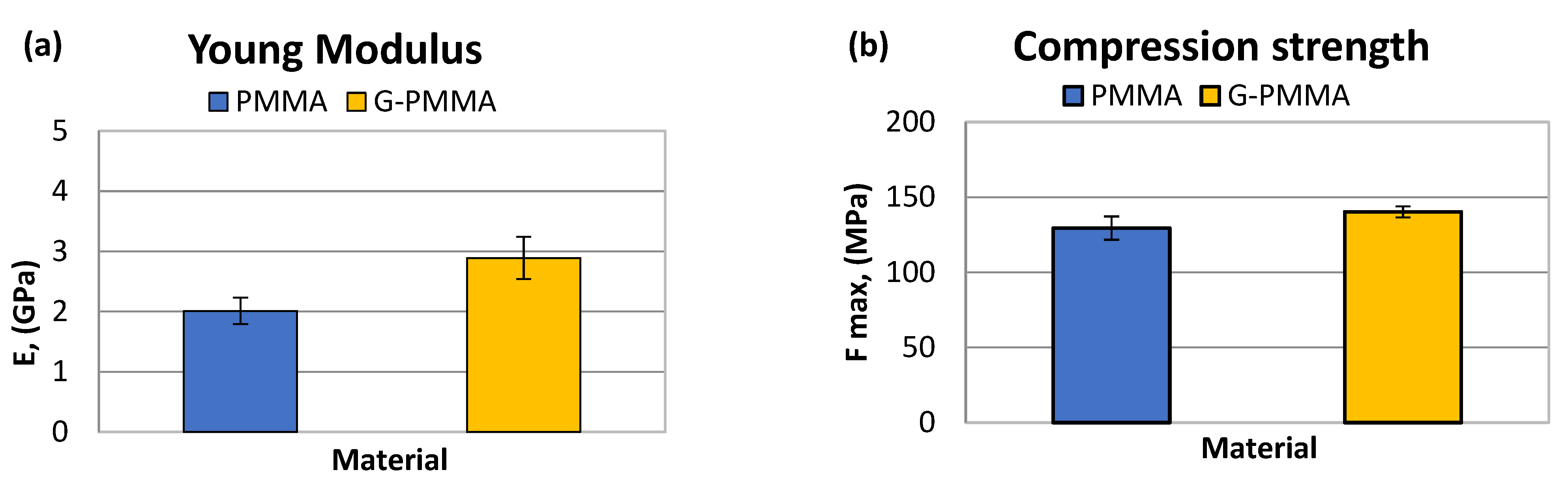
| E (GPa) | Fmax (MPa) | |
|---|---|---|
| PMMA | 2.01 ± 0.22 | 129.43 ± 7.75 |
| G-PMMA | 2.89 ± 0.35 | 140.23 ± 3.69 |
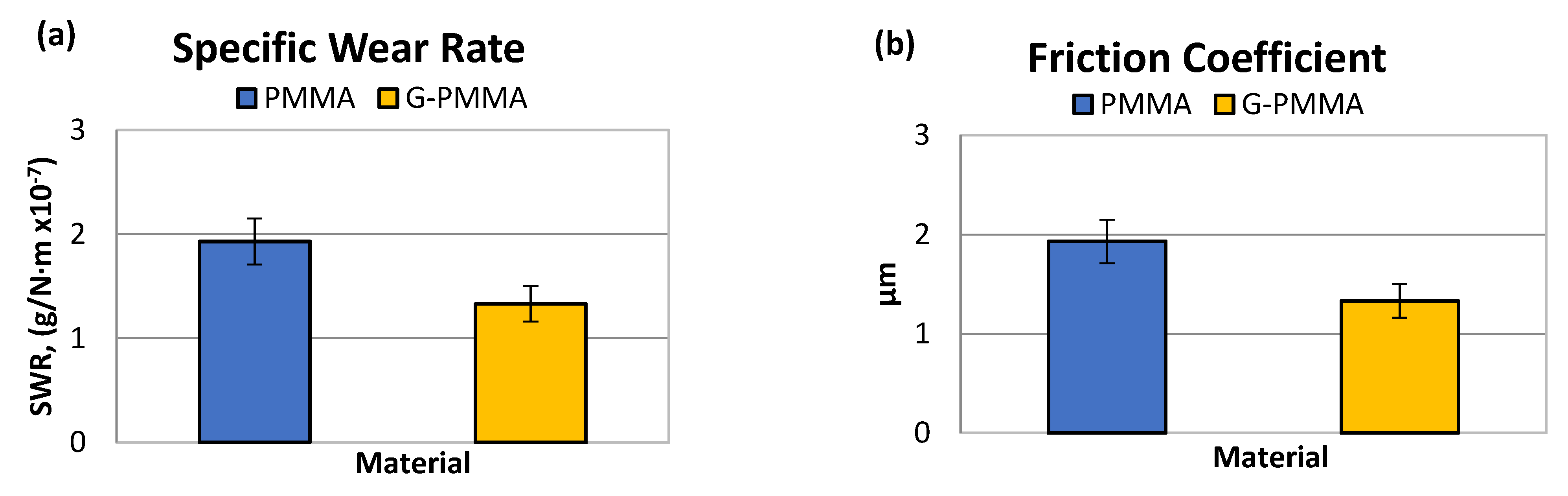
| SWR (g/N·m) | µm (MPa) | |
|---|---|---|
| PMMA | 1.93 × 10−7 ± 0.22 × 10−7 | 0.4032 ± 0.0251 |
| G-PMMA | 1.33 × 10−7 ± 0.17 × 10−7 | 0.4001 ± 0.0109 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Punset, M.; Brizuela, A.; Pérez-Pevida, E.; Herrero-Climent, M.; Manero, J.M.; Gil, J. Mechanical Characterization of Dental Prostheses Manufactured with PMMA–Graphene Composites. Materials 2022, 15, 5391. https://doi.org/10.3390/ma15155391
Punset M, Brizuela A, Pérez-Pevida E, Herrero-Climent M, Manero JM, Gil J. Mechanical Characterization of Dental Prostheses Manufactured with PMMA–Graphene Composites. Materials. 2022; 15(15):5391. https://doi.org/10.3390/ma15155391
Chicago/Turabian StylePunset, Miquel, Aritza Brizuela, Esteban Pérez-Pevida, Mariano Herrero-Climent, José Maria Manero, and Javier Gil. 2022. "Mechanical Characterization of Dental Prostheses Manufactured with PMMA–Graphene Composites" Materials 15, no. 15: 5391. https://doi.org/10.3390/ma15155391
APA StylePunset, M., Brizuela, A., Pérez-Pevida, E., Herrero-Climent, M., Manero, J. M., & Gil, J. (2022). Mechanical Characterization of Dental Prostheses Manufactured with PMMA–Graphene Composites. Materials, 15(15), 5391. https://doi.org/10.3390/ma15155391










