Rheological Properties of Different Graphene Nanomaterials in Biological Media
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Dispersions Preparation
2.3. Characterization of Samples
3. Results and Discussion
3.1. Transmission and Scanning Electron Microscopy Characterization
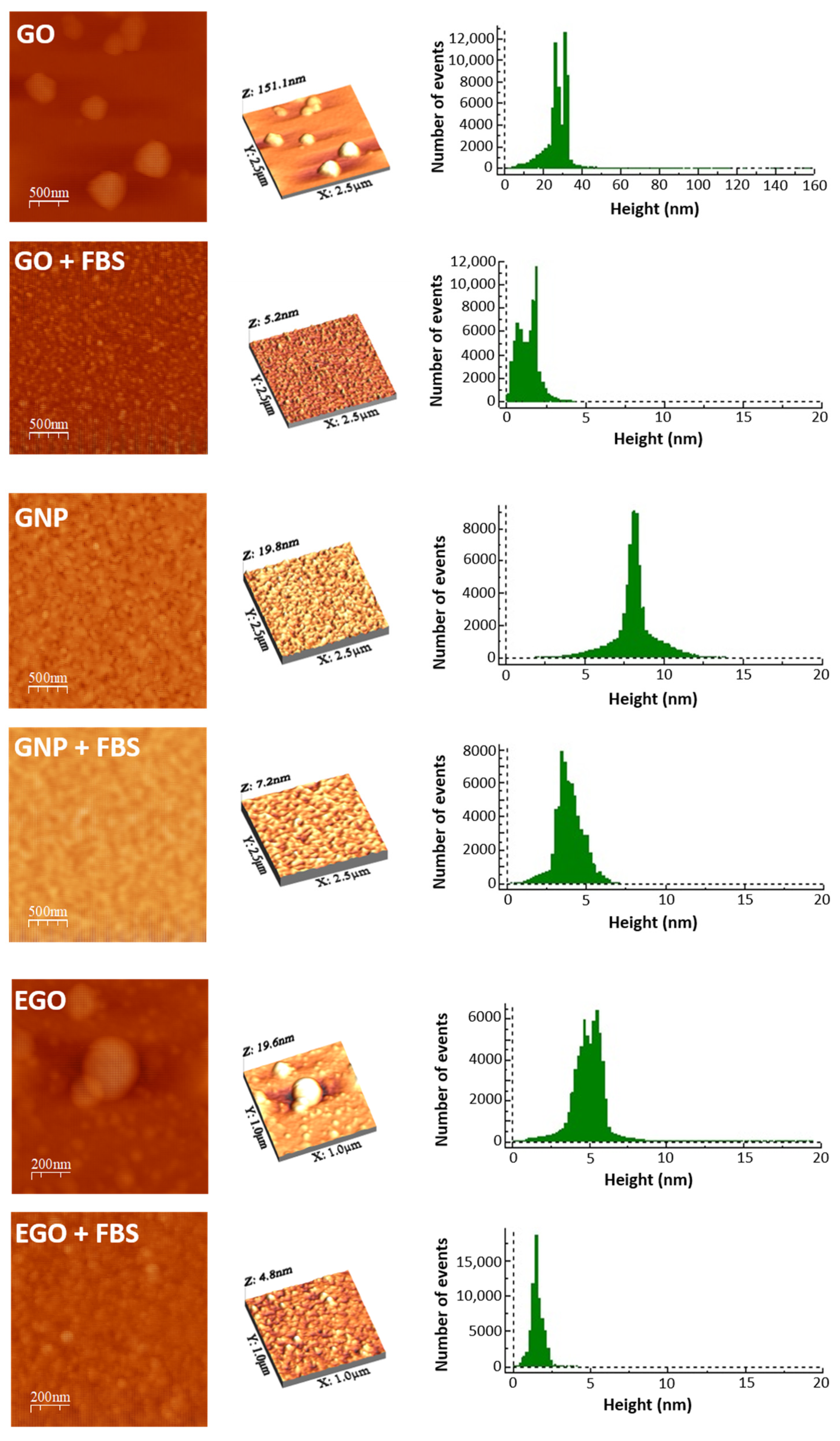
3.2. Atomic Forces Microscopy Characterization
3.3. Zeta Potential Measurements
3.4. Rheological Studies
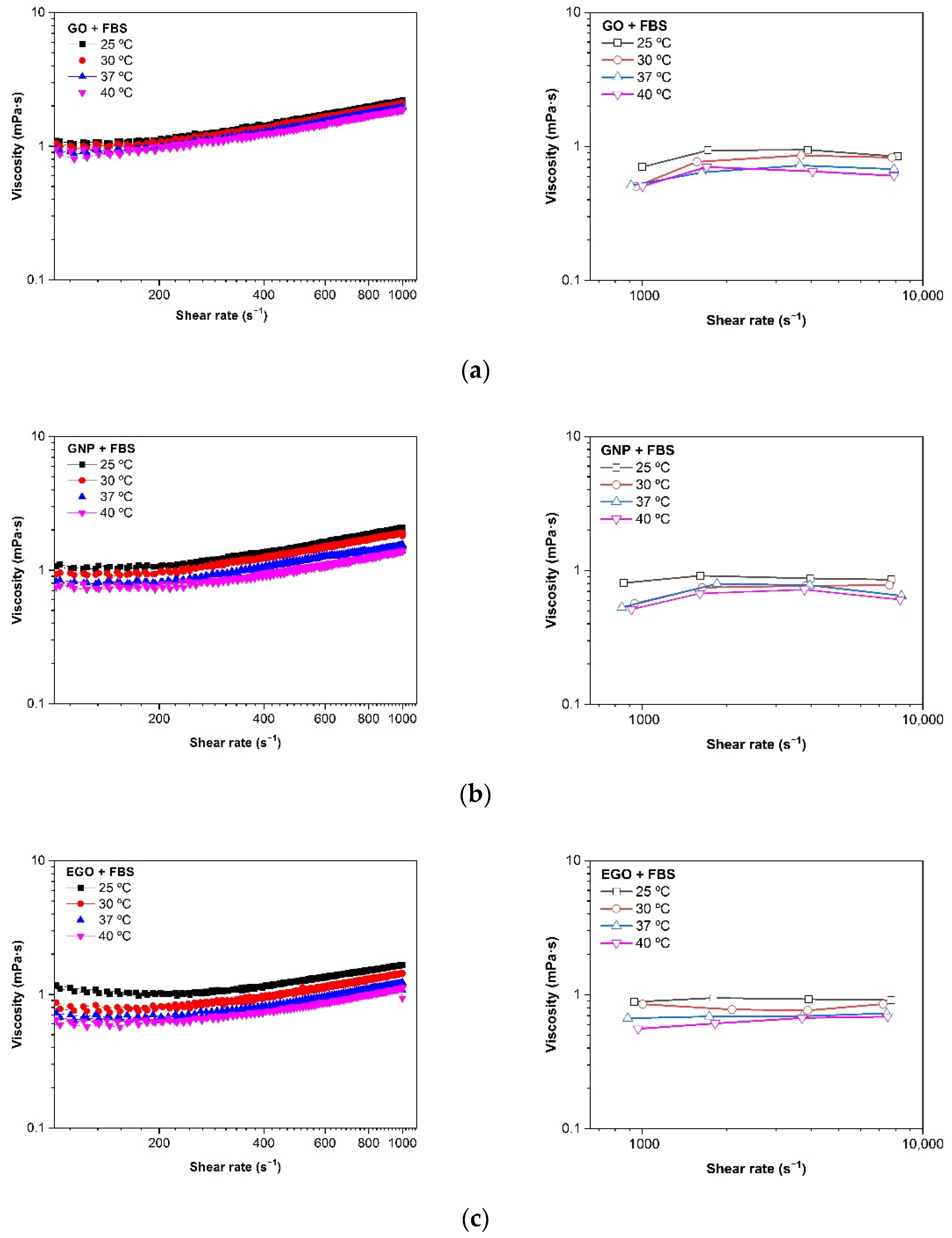
3.4.1. Type of Graphene-Based Nanomaterial
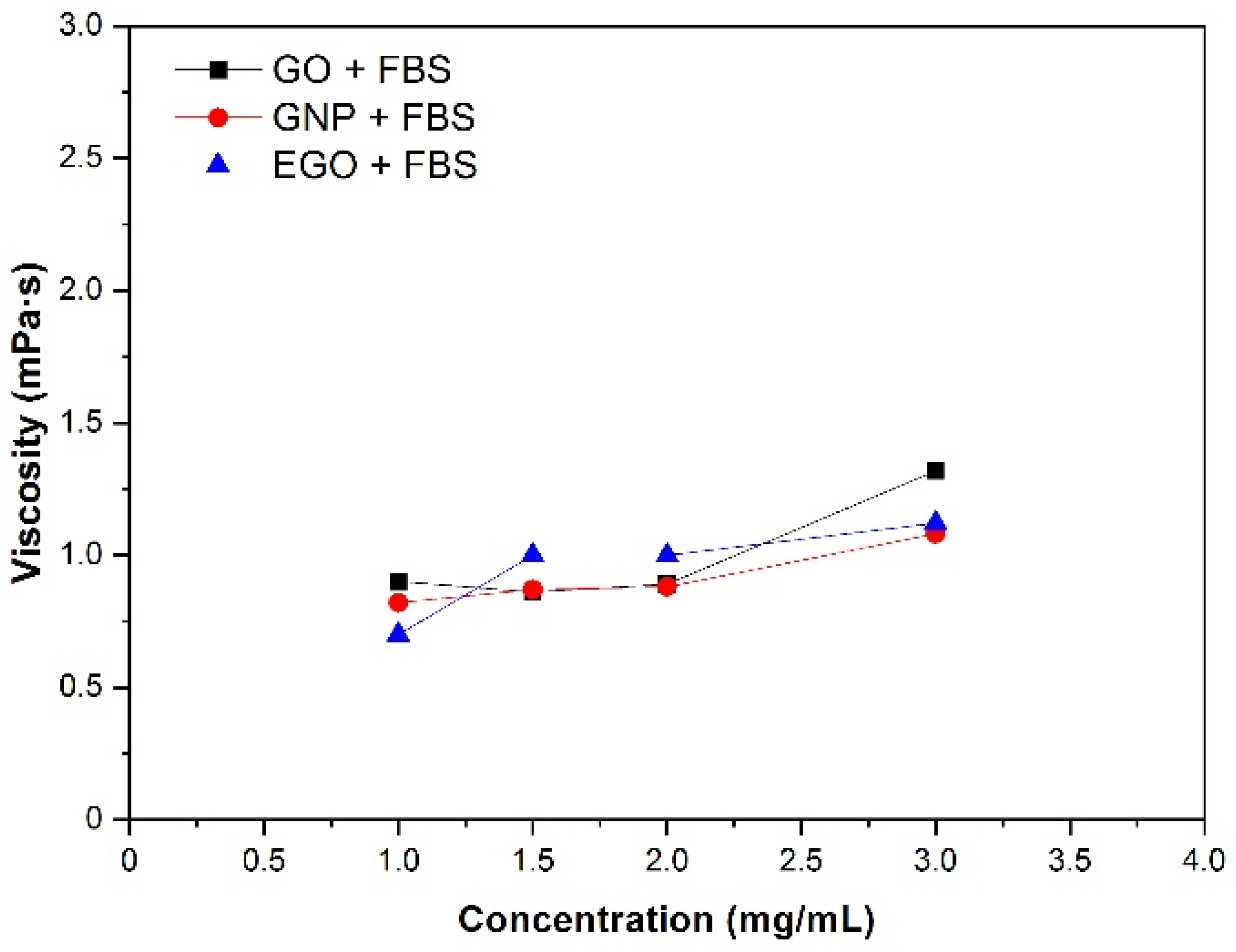
3.4.2. Nanomaterial Concentration
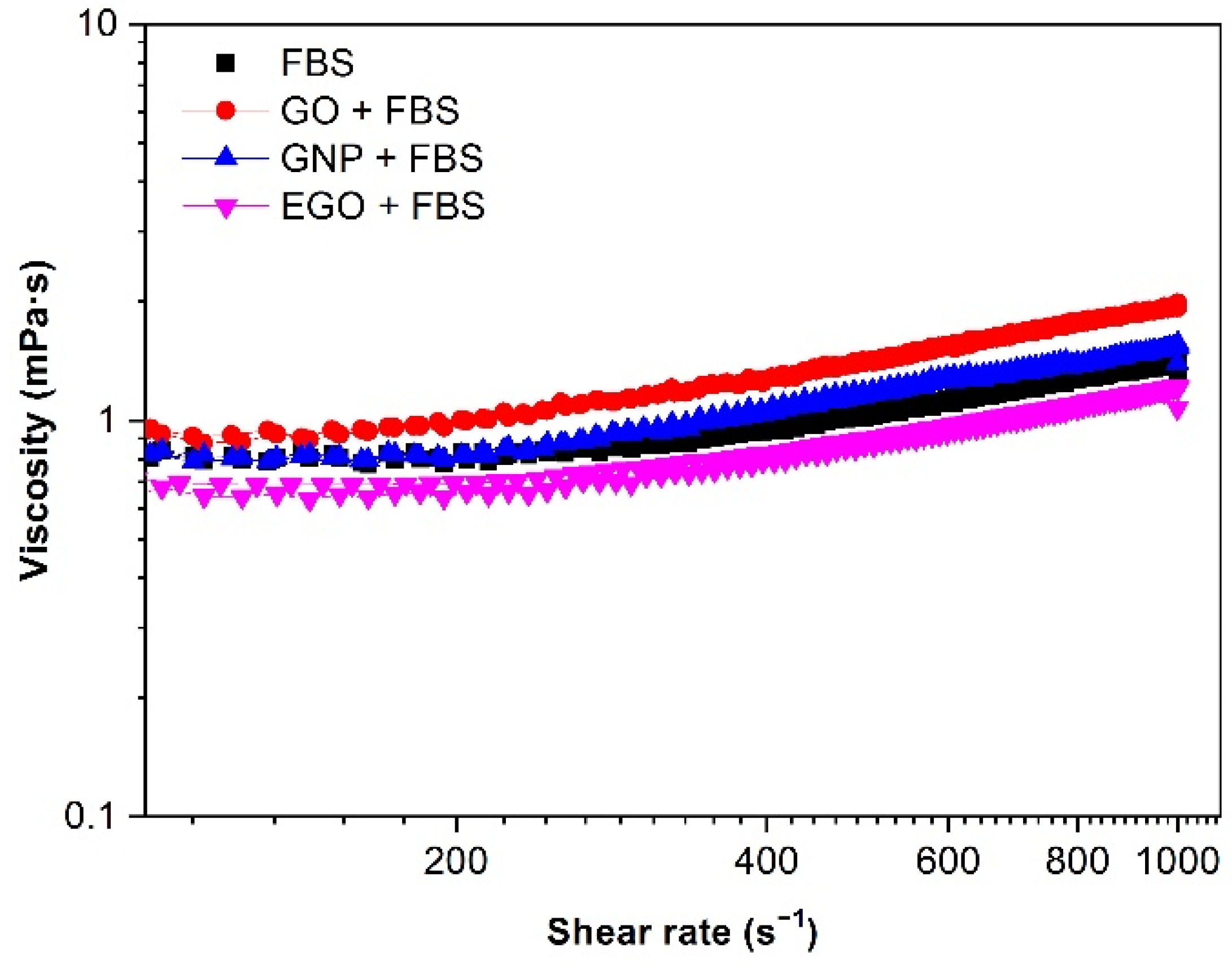
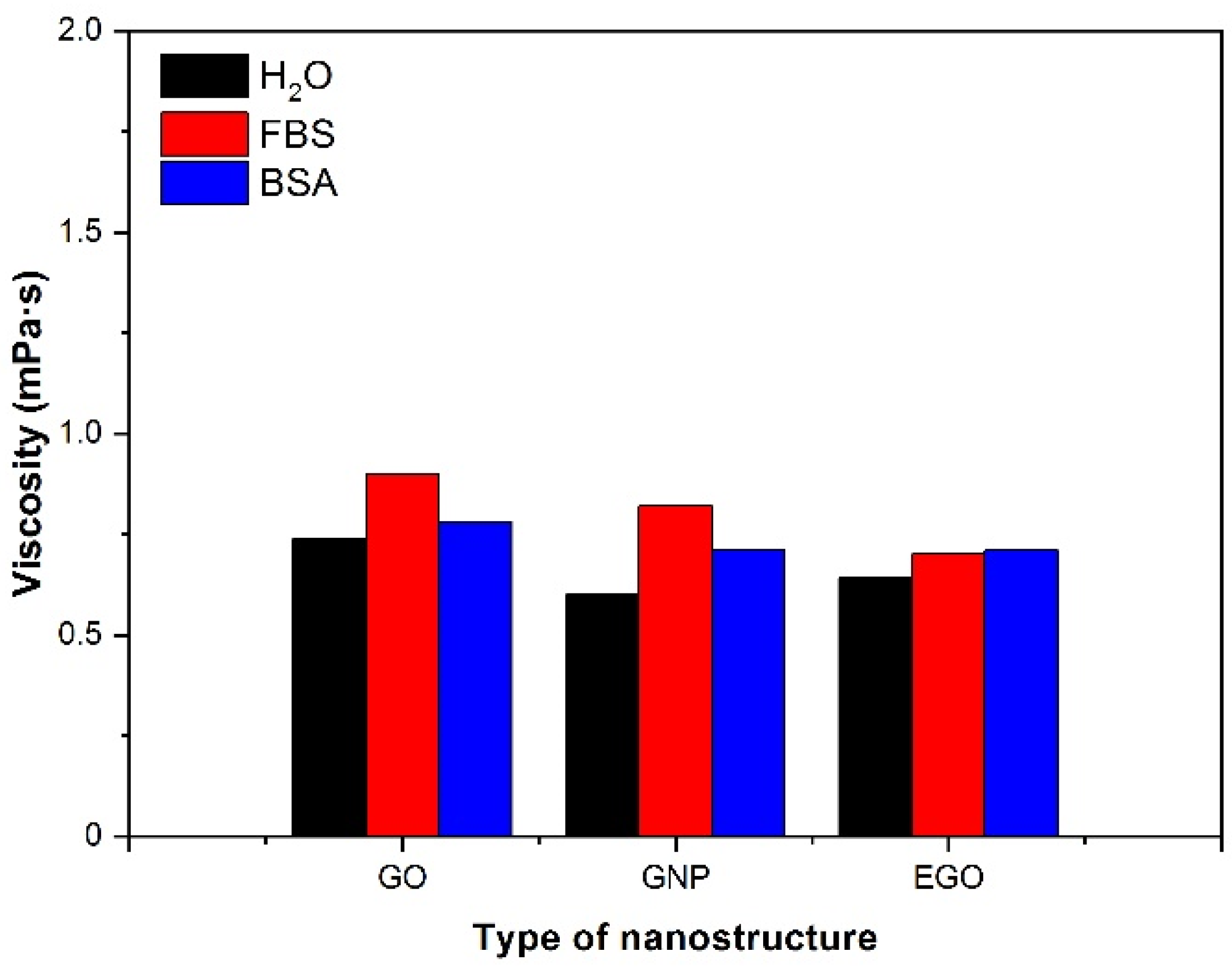
3.4.3. Type of Fluid
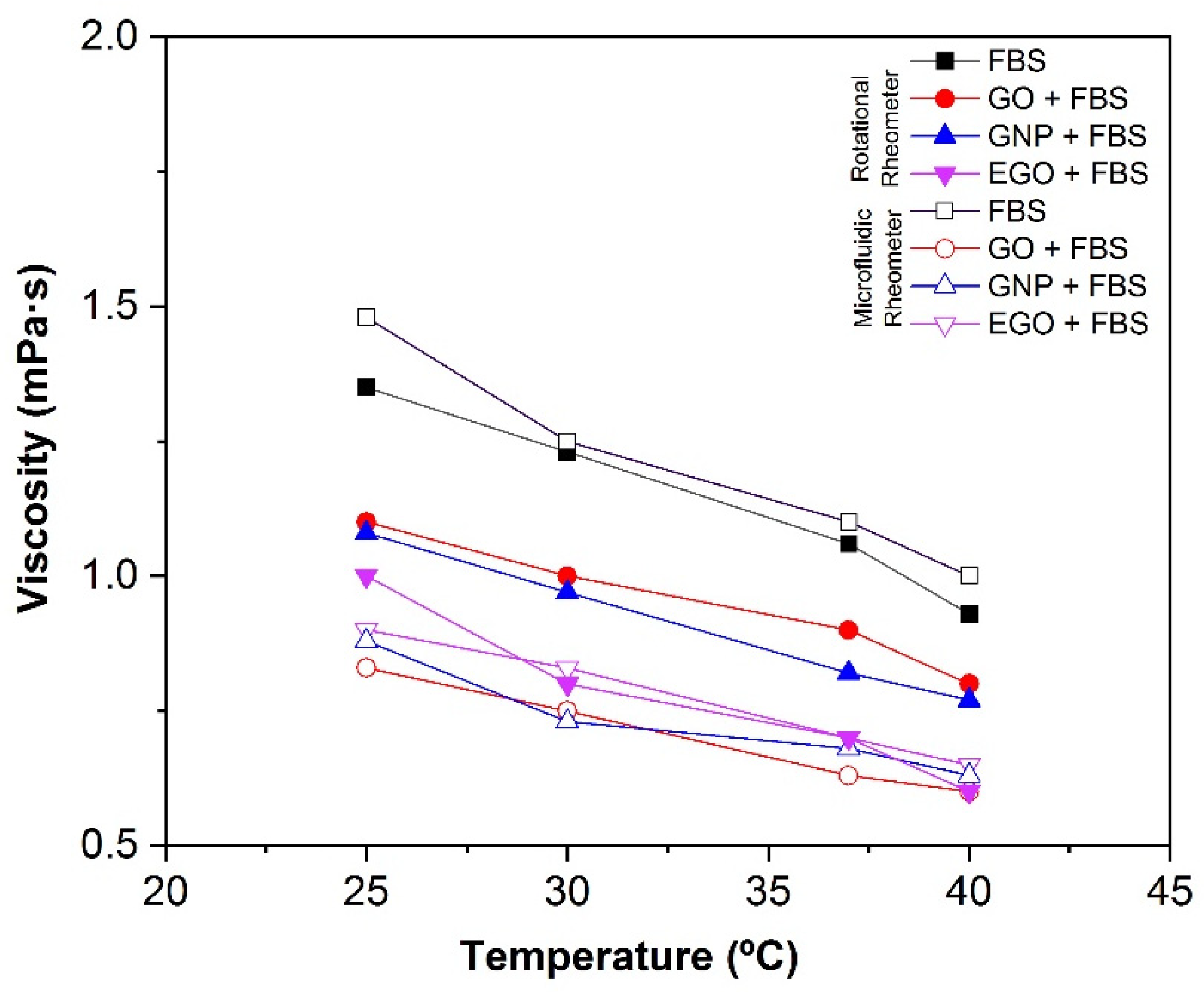
3.4.4. Effect of Temperature
4. Conclusions
- SEM images show a higher tendency to compaction and aggregation of GO, GNP and EGO when the FBS is used as the dispersion medium. The images obtained by AFM show the change in the surface texture, which indicates the nanomaterial–protein interaction.
- The dispersions of GO + FBS, GNP + FBS and EGO + FBS present Newtonian behavior in all the studied ranges of concentration, although measurements performed with rotational rheometers give an apparent shear thickening. The microfluidic chip rheometer is a much more reliable technique for these systems.
- The viscosity values obtained are between 0.6 and 1.3 mPa·s for the concentration range of 1–3 mg/mL at a temperature of 37 °C. These values tend to slightly increase when the concentrations increase, although these changes are not significative.
- The dispersions of nanomaterials in FBS lead to slightly higher viscosity values than those in BSA and water. The viscosity values obtained for GO, GNP and EGO dispersed in FBS at a temperature of 37 °C and a concentration of 1 mg/mL are between 0.6 and 0.9 mPa·s.
- The viscosity of the dispersions decreases with the temperature from 1.1 to 0.6 mPa·s in the temperature range of 25–40 °C at 1 mg/mL.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, A.A. Electric field effect in atomically thin carbon films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, Y.; Asiri, A.M.; Tang, Z.; Du, D.; Lin, Y. Graphene based materials for biomedical applications. Mater. Today 2013, 16, 365–373. [Google Scholar] [CrossRef]
- Mkhoyan, K.A.; Contryman, A.W.; Silcox, J.; Stewart, D.A.; Eda, G.; Mattevi, C.; Miller, S.; Chhowalla, M. Atomic and electronic structure of graphene-oxide. Nanoletters 2009, 9, 1058–1063. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.; Xia, J.; Zhao, Q.; Liu, L.; Zhang, Z. Functional graphene oxide as a nanocarrier for controlled loading and targeted delivery of mixed anticancer drugs. Small 2010, 6, 537–544. [Google Scholar] [CrossRef] [PubMed]
- Yin, D.; Li, Y.; Lin, H.; Guo, B.; Du, Y.; Li, X.; Jia, H.; Zhao, X.; Tang, J.; Zhang, L. Functional graphene oxide as a plasmid-based Stat3 siRNA carrier inhibits mouse malignant melanoma growth in vivo. Nanotechnology 2013, 24, 105102. [Google Scholar] [CrossRef] [PubMed]
- Bolibok, P.; Szymczak, B.; Roszek, K.; Terzyk, A.P.; Wisniewski, M. A new approach to obtaining nano-sized graphene oxide for biomedical applications. Materials 2021, 14, 1327. [Google Scholar] [CrossRef]
- Matalkah, F.; Soroushian, P. Graphene nanoplatelet for enhancement the mechanical properties and durability characteristics of alkali activated binder. Constr. Build. Mater. 2020, 249, 118773. [Google Scholar] [CrossRef]
- Cho, J.; Lee, H.; Nam, K.H.; Yeo, H.; Yang, C.M.; Seong, D.G.; Lee, D.; Kim, S.Y. Enhanced electrical conductivity of polymer nanocomposite based on edge-selectivity functionalized graphene nanoplatelets. Compos. Sci. Technol. 2020, 189, 108001. [Google Scholar] [CrossRef]
- Park, C.S.; Yoon, H.; Kwon, O.S. Graphene-based nanoelectronic biosensors. J. Ind. Eng. Chem. 2016, 38, 13–22. [Google Scholar] [CrossRef]
- Kargar, S.; Elhamifar, D.; Zarnegaryan, A. Ionic liquid modified graphene oxide supported Mo-complex: A novel, efficient and highly stable catalyst. Surf. Interfaces 2021, 23, 100946. [Google Scholar] [CrossRef]
- Liu, S.; Zeng, T.H.; Hofmann, M.; Burcombe, E.; Wei, J.; Jiang, R.; Kong, J.; Chen, Y. Antibacterial activity of graphite, graphite oxide, graphene oxide, and reduced graphene oxide: Membrane and oxidative stress. ACS Nano 2011, 5, 6971–6980. [Google Scholar] [CrossRef] [PubMed]
- Kuropka, P.; Dobrzynski, M.; Bazanow, B.; Stygar, D.; Gebarowski, T.; Leskow, A.; Tarnowska, M.; Szyszka, K.; Malecka, M.; Nowak, N.; et al. A Study of the Impact of Graphene Oxide on Viral Infection Related to A549 and TC28a2 Human Cell Lines. Materials 2021, 14, 7788. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, K.; Mukherjee, S.P.; Gallud, A.; Burkert, S.C.; Bistarelli, S.; Bellucci, S.; Bottini, M.; Star, A.; Fadeel, B. Biological interactions of carbon-based nanomaterials: From coronation to degradation. Nanomed. Nanotechnol. Biol. Med. 2016, 12, 333–351. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, C.; Xi, Y.; Weng, Y. Progress in the Development of Graphene-Based Biomaterials for Tissue Engineering and Regeneration. Materials 2022, 15, 2164. [Google Scholar] [CrossRef]
- Trusek, A.; Kijak, E. Drug Carriers Based on Graphene Oxide and Hydrogel: Opportunities and Challenges in Infection Control Tested by Amoxicillin Release. Materials 2021, 14, 3182. [Google Scholar] [CrossRef]
- Singh, D.P.; Herrera, C.E.; Singh, B.; Shing, S.; Shing, R.K.; Kumar, R. Graphene oxide: An efficient material and recent approach for biotechnological and biomedical applications. Mater. Sci. Eng. C 2018, 86, 173–197. [Google Scholar] [CrossRef]
- Kopac, T. Protein corona, understanding the nanoparticle-protein interactions and future perspectives: A critical review. Int. J. Biol. Macromol. 2021, 169, 290–301. [Google Scholar] [CrossRef]
- Sopotnik, M.; Leonardi, A.; Krizaj, I.; Dusak, P.; Makovec, D.; Mesaric, T.; Ulrih, N.P.; Junkar, I.; Sepcic, K.; Drobne, D. Comparative study of serum protein binding to three different carbon-based nanomaterials. Carbon 2015, 95, 560–572. [Google Scholar] [CrossRef]
- Mahmoudi, M.; Lynch, I.; Ejtehadi, M.R.; Monopoli, M.P.; Bombelli, F.B.; Laurent, S. Protein-nanoparticle interactions: Opportunities and challenges. Chem. Rev. 2011, 111, 5610–5637. [Google Scholar] [CrossRef]
- Wu, Z.; Zhang, B.; Yan, B. Regulation of enzyme activity through interactions with nanoparticles. Int. J. Mol. Sci. 2009, 10, 4198–4209. [Google Scholar] [CrossRef] [Green Version]
- Sheng, A.; Liu, F.; Xie, N.; Liu, J. Impact of proteins on aggregation kinetics and adsorption ability of hematite nanoparticles in aqueous dispersions. Environ. Sci. Technol. 2016, 50, 2228–2235. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Zou, X.; Ye, L. Controlled pH-and glucose-responsive drug release behavior of cationic chitosan based nano-composite hydrogels by using graphene oxide as drug nanocarrier. J. Ind. Eng. Chem. 2017, 49, 36–45. [Google Scholar] [CrossRef]
- Wu, C.; He, Q.; Zhu, A.; Yang, H.; Liu, Y. Probing the protein conformation and adsorption behaviors in nanographene oxide-protein complexes. J. Nanosci. Nanotechnol. 2014, 14, 2591–2598. [Google Scholar] [CrossRef] [PubMed]
- Moghassemi, S.; Hadjizadeh, A.; Omidfar, K. Formulation and Characterization of Bovine Serum Albumin-Loaded Niosome. AAPS PharmSciTech 2017, 18, 27–33. [Google Scholar] [CrossRef] [Green Version]
- Zheng, X.; Baker, H.; Hancock, W.S.; Fawaz, F.; McCaman, M.; Pungor, E., Jr. Proteomic analysis for the assessment of different lots of fetal bovine serum as a raw material for cell culture. Part IV. Application of proteomics to the manufacture of biological drugs. Biotechnol. Prog. 2006, 22, 1294–1300. [Google Scholar] [CrossRef]
- Punyiczki, M.; Rosenberg, A. The effect of viscosity on the accessibility of the single tryptophan in human serum albumin. Biophys. Chem. 1992, 42, 93–100. [Google Scholar] [CrossRef]
- Zidar, M.; Rozman, P.; Belko-Parkel, K.; Ravnik, M. Control of viscosity in biopharmaceutical protein formulations. J. Colloid Interface Sci. 2020, 580, 308–317. [Google Scholar] [CrossRef]
- Wonerow, T.; Uhler, M.; Nuppnau, J.; Kretzer, J.P.; Mantwill, F. Rheologic Behavior of Bovine Calf Serum. Materials 2021, 14, 2538. [Google Scholar] [CrossRef]
- Vallejo, J.P.; Gómez-Barreiro, S.; Cabaleiro, D.; Gracia-Fernández, C.; Fernández-Seara, J.; Lugo, L. Flow behaviour of suspensions of functionalized graphene nanoplatelets in propylene glycol–water mixtures. Int. Commun. Heat Mass Transf. 2018, 91, 150–157. [Google Scholar] [CrossRef]
- Alyamac, E.; Fidan, T.; Turgut, A.; Ozgur, M. Stability, rheology, and thermophysical properties of surfactant free aqueous single-walled carbon nanotubes and graphene nanoplatelets nanofluids: A comparative study. J. Dispers. Sci. Technol. 2021, 1–10. [Google Scholar] [CrossRef]
- Cerpa, A.; Lado, I.; Quiroga, O.; Moreno, R.; García, R.; Cerdán, S.; Abu-Lail, N.I. Colloidal and rheological characterization of SWCNT in biological media. Int. J. Smart Nano Mater. 2019, 10, 300–315. [Google Scholar] [CrossRef]
- Cerpa, A.; Ibañez, B.; Lado, I.; Arce, M.P.; Pérez, J.; Barrios, N.; Moreno, R.; Cerdán, S. Rheological behaviour of carbon nanotubes suspensions with biomedical applications. In Estudos em Biociencias e Biotecnologia, Chapter 2; Editora Artemis: Curitiba, Brazil, 2021; pp. 16–27. [Google Scholar]
- Lavin-Lopez, M.P.; Paton-Carrero, A.; Sanchez-Silva, L.; Valverde, J.L.; Romero, A. Influence of the reduction strategy in the synthesis of reduced graphene oxide. Adv. Powder Technol. 2017, 28, 3195–3203. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.; Eom, S.H.; Chung, J.S.; Hur, S.H. Large-scale production of high-quality reduced graphene oxide. Chem. Eng. J. 2013, 233, 297–304. [Google Scholar] [CrossRef]
- Cheong, Y.K.; Arce, M.; Benito, A.; Chen, D.; Luengo, N.; Kerai, L.; Rodríguez, G.; Valverde, J.L.; Vadalia, M.; Cerpa, A.; et al. Synergistic antifungal study of PEGylated graphene oxides and copper nanoparticles against Candida albicans. Nanomaterials 2020, 10, 819. [Google Scholar] [CrossRef]
- Franqui, L.S.; De Farias, M.A.; Portugal, R.V.; Costa, C.A.; Domingues, R.R.; Souza-Filho, A.G.; Coluci, V.R.; Leme, A.F.P.; Martinez, D.S.T. Interaction of graphene oxide with cell culture medium: Evaluating the fetal bovine serum protein corona formation towards in vitro nanotoxicity assessment and nanobiointeractions. Mater. Sci. Eng. C 2019, 100, 363–377. [Google Scholar] [CrossRef]
- Zhu, Y.; Li, W.; Li, Q.; Li, Y.; Li, Y.; Zhang, X.; Huang, Q. Effects of serum proteins on intracellular uptake and cytotoxicity of carbon nanoparticles. Carbon 2009, 47, 1351–1358. [Google Scholar] [CrossRef]
- Park, M.; Nguyen, T.P.; Choi, K.S.; Park, J.; Ozturk, A.; Kim, S.Y. MoS2-nanosheet/graphene-oxide composite hole injection layer in organic light-emitting diodes. Electron. Mater. Lett. 2017, 13, 344–350. [Google Scholar] [CrossRef]
- Palmieri, V.; Bugli, F.; Cacaci, M.; Perini, G.; De Maio, F.; Delogu, G.; Torelli, R.; Conti, C.; Sanguinetti, M.; De Spirito, M.; et al. Graphene oxide coatings prevent Candida albicans biofilm formation with a controlled release of curcumin-loaded nanocomposites. Nanomedicine 2018, 13, 2867–2879. [Google Scholar] [CrossRef]
- Sapsford, K.E.; Tyner, K.M.; Dair, B.J.; Deschamps, J.R.; Medintz, I.L. Analyzing nanomaterial bioconjugates: A review of current and emerging purification and characterization techniques. Anal. Chem. 2011, 83, 4453–4488. [Google Scholar] [CrossRef]
- Carnicer, V.; Alcázar, C.; Orts, M.J.; Sánchez, E.; Moreno, R. Microfluidic rheology: A new approach to measure viscosity of ceramic suspensions at extremely high shear rates. Open Ceram. 2021, 5, 100052. [Google Scholar] [CrossRef]
- Raslan, A.; Saenz, L.; Espona-Noguera, A.; Ochoa, A.M.; Sanjuán, M.L.; Cañibano-Hernández, A.; Gálvez-Martín, P.; Ciriza, J.; Pedraz, J.L. BSA- and Elastin-coated GO, but no collagen-coated GO, enhance the biological performance of alginate hydrogels. Pharmaceutics 2020, 12, 543. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Wang, W.S.; Vanapalli, S.A. Microfluidic viscometers for shear rheology of complex fluids and biofluids. Biomicrofluidics 2016, 10, 043402. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rothammer, B.; Marian, M.; Rummel, F.; Schroeder, S.; Uhler, M.; Kretzer, J.P.; Tremmel, S.; Wartzack, S. Rheological behavior of an artificial synovial fluid—Influence of temperature, shear rate and pressure. J. Mech. Behav. Biomed. Mater. 2021, 115, s104278. [Google Scholar] [CrossRef] [PubMed]
- Bortel, E.; Charbonnier, B.; Heuberger, R. Development of a Synthetic Synovial Fluid for Tribological Testing. Lubricants 2015, 3, 664–686. [Google Scholar] [CrossRef] [Green Version]
- Mazzucco, D.; McKinley, G.; Scott, R.D.; Spector, M. Rheology of joint fluid in total knee arthroplasty patients. J. Orthop. Res. 2002, 20, 1157–1163. [Google Scholar] [CrossRef]



| Sample | ZP | Mobility | pH |
|---|---|---|---|
| (mV) | (μm·cm/V·s) | ||
| GO + FBS | −25.7 ± 1.6 | −2.0 ± 0.1 | 6.6 |
| GNP + FBS | −18.1 ± 0.9 | −1.4 ± 0.1 | 7.0 |
| EGO + FBS | −26.6 ± 1.7 | −2.1 ± 0.1 | 6.7 |
| Viscosity (mPa·s) | ||||||
|---|---|---|---|---|---|---|
| Sample | Rheometer | Shear Rate (s−1) | 25 °C | 30 °C | 37 °C | 40 °C |
| FBS | Fluidicam | 1000–10,000 | 1.5 | 1.3 | 1.1 | 1.0 |
| Rotational | 200 | 1.8 | 1.5 | 1.4 | 1.3 | |
| GO + FBS | Fluidicam | 1000–10,000 | 0.8 | 0.8 | 0.6 | 0.6 |
| Rotational | 200 | 1.1 | 1.0 | 0.9 | 0.8 | |
| GNP + FBS | Fluidicam | 1000–10,000 | 0.9 | 0.7 | 0.7 | 0.6 |
| Rotational | 200 | 1.0 | 1.0 | 0.8 | 0.8 | |
| EGO + FBS | Fluidicam | 1000–10,000 | 0.9 | 0.8 | 0.7 | 0.7 |
| Rotational | 200 | 1.0 | 0.8 | 0.7 | 0.6 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cerpa-Naranjo, A.; Pérez-Piñeiro, J.; Navajas-Chocarro, P.; Arce, M.P.; Lado-Touriño, I.; Barrios-Bermúdez, N.; Moreno, R.; Rojas-Cervantes, M.L. Rheological Properties of Different Graphene Nanomaterials in Biological Media. Materials 2022, 15, 3593. https://doi.org/10.3390/ma15103593
Cerpa-Naranjo A, Pérez-Piñeiro J, Navajas-Chocarro P, Arce MP, Lado-Touriño I, Barrios-Bermúdez N, Moreno R, Rojas-Cervantes ML. Rheological Properties of Different Graphene Nanomaterials in Biological Media. Materials. 2022; 15(10):3593. https://doi.org/10.3390/ma15103593
Chicago/Turabian StyleCerpa-Naranjo, Arisbel, Javier Pérez-Piñeiro, Pablo Navajas-Chocarro, Mariana P. Arce, Isabel Lado-Touriño, Niurka Barrios-Bermúdez, Rodrigo Moreno, and María Luisa Rojas-Cervantes. 2022. "Rheological Properties of Different Graphene Nanomaterials in Biological Media" Materials 15, no. 10: 3593. https://doi.org/10.3390/ma15103593
APA StyleCerpa-Naranjo, A., Pérez-Piñeiro, J., Navajas-Chocarro, P., Arce, M. P., Lado-Touriño, I., Barrios-Bermúdez, N., Moreno, R., & Rojas-Cervantes, M. L. (2022). Rheological Properties of Different Graphene Nanomaterials in Biological Media. Materials, 15(10), 3593. https://doi.org/10.3390/ma15103593










