Effect of Alkali Salts on the Hydration Process of Belite Clinker
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Hydration Process
3.1.1. Initial Period
3.1.2. Induction Period
3.1.3. Acceleration Period
3.1.4. Deceleration Period
3.1.5. Final Slow Reaction Period
3.2. X-Ray Diffraction Analysis
3.2.1. BC
3.2.2. BC-CS
3.2.3. BC-NS
3.2.4. BC-NC
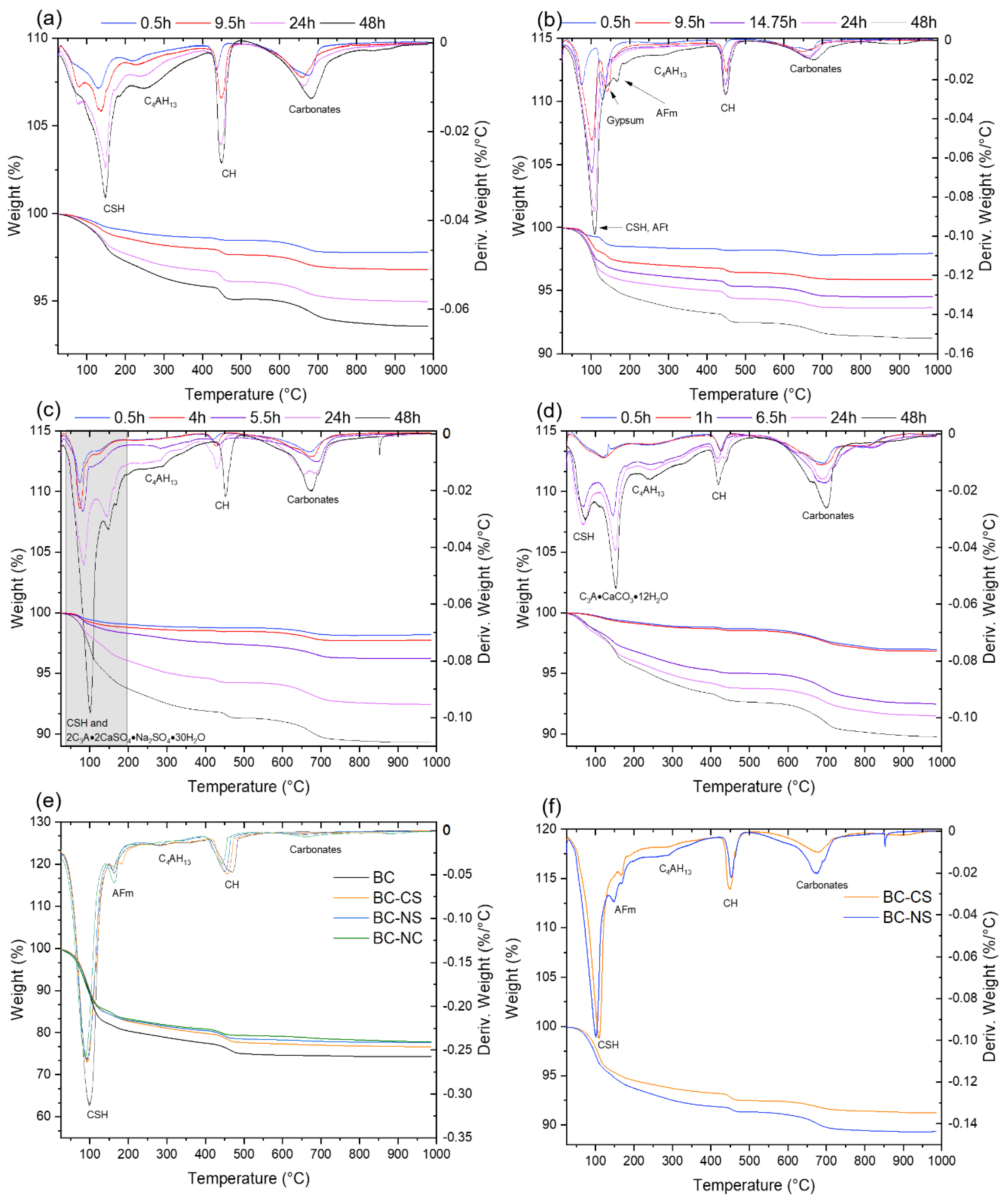
3.3. Differential Thermal Analysis
3.3.1. BC
3.3.2. BC-CS
3.3.3. BC-NS
3.3.4. BC-NC
3.4. SEM-EDS Analysis
4. Conclusions
- The initial stage of hydration in BC is affected by the absence of SO42−, which disables ettringite formation; thus, the aluminate phases hydrate very quickly, followed by the formation of stable aluminous phases, such as C4AH13. The amount of hydraulic nonactive γ-C2S remained almost unchanged throughout the hydration period (max. 28 days). The lowest amount of C–S–H phase formed for BC, in contrast to the others, after 48 h.
- The main effect of the activation of belite-rich clinker by CaSO4 was the earliest occurrence of ettringite compared to other activators. Moreover, the highest amount of portlandite was detected after 48 h.
- Na2SO4 showed the most noticeable modification of shape of the silicate peak compared to the other activators. Due to the presence of SO42, together with Na+, the formation of ettringite and ettringite-like structures was observed. BC-NS showed the greatest formation of C–S–H phase after 48 h, as well as the highest total weight loss.
- The silicate peak accelerated the most in the case of BC-NC due to the presence of CO32−, which has the highest dissolution enthalpy. Moreover, the absence of SO42− suppressed the formation of ettringite.
- Based on the measured data, the most appropriate alkaline activator is Na2SO4, where Na+ accelerates the reaction of hydration at the early stage; however, at the same time, SO42− can regulate the undesired fast setting reaction caused by C3A, leading to the formation of stable products.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Schneider, M. The cement industry on the way to a low-carbon future. Cem. Concr. Res. 2019, 124, 105792. [Google Scholar] [CrossRef]
- Andrew, R.M. Global co2 emissions from cement production, 1928–2018. Earth Syst. Sci. Data 2019, 11, 1675–1710. [Google Scholar] [CrossRef] [Green Version]
- Gartner, E.; Sui, T.B. Alternative cement clinkers. Cem. Concr. Res. 2018, 114, 27–39. [Google Scholar] [CrossRef]
- Odler, I. Special Inorganic Cements; E & FN Spon: New York, NY, USA, 2000; 395p. [Google Scholar]
- Jiang, C.M.; Jiang, L.H.; Li, S.X.; Tang, X.J.; Zhang, L. Impact of cation type and fly ash on deterioration process of high belite cement pastes exposed to sulfate attack. Constr. Build. Mater. 2021, 286, 122961. [Google Scholar] [CrossRef]
- Dan, E.; Janotka, I. Chemical resistance of portland cement, blast-furnace slag portland cement and sulphoaluminate-belite cement in acid, chloride and sulphate solution: Some preliminary results. Ceram.-Silik. 2003, 47, 141–148. [Google Scholar]
- Ludwig, U.; Pohlmann, R. Production of low-lime portland cements. Zem.-Kalk-Gips 1985, 38, 595–598. [Google Scholar]
- Cheng, X.; Chang, J.; Lu, L.C.; Liu, F.T.; Teng, B. Study of ba-bearing calcium sulphoaluminate minerals and cement. Cem. Concr. Res. 2000, 30, 77–81. [Google Scholar]
- Bohac, M.; Stanek, T.; Rybova, A.; Zezulova, A.; Bellmann, F.; Ludwig, H.M. Early hydration of C2S doped with combination of s and li. SN Appl. Sci. 2020, 2, 429. [Google Scholar] [CrossRef] [Green Version]
- Morsli, K.; de la Torre, A.G.; Cuberos, A.J.M.; Zahir, M.; Aranda, M.A.G. Preparation and characterization of alkali-activated white belite cements. Mater. Constr. 2009, 59, 19–29. [Google Scholar]
- Morsli, K.; de la Torre, A.G.; Stober, S.; Cuberos, A.J.M.; Zahir, M.; Aranda, M.A.G. Quantitative phase analysis of laboratory-active belite clinkers by synchrotron powder diffraction. J. Am. Ceram. Soc. 2007, 90, 3205–3212. [Google Scholar] [CrossRef]
- Sanchez-Herrero, M.J.; Fernandez-Jimenez, A.; Palomo, A. Alkaline hydration of C2S and C3S. J. Am. Ceram. Soc. 2016, 99, 604–611. [Google Scholar] [CrossRef]
- Sanchez-Herrero, M.J.; Fernandez-Jimenez, A.; Palomo, A. C3S and C2S hydration in the presence of Na2Co3 and Na2So4. J. Am. Ceram. Soc. 2017, 100, 3188–3198. [Google Scholar] [CrossRef]
- Bohac, M.; Stanek, T.; Zezulova, A.; Rybova, A.; Kubatova, D.; Novotny, R. Early hydration of activated belite-rich cement. Adv. Mater. Res. 2019, 1151, 23–27. [Google Scholar] [CrossRef]
- Ashraf, W. Microstructure of chemically activated gamma-dicalcium silicate paste. Constr. Build. Mater. 2018, 185, 617–627. [Google Scholar] [CrossRef]
- Bensted, J.; Barnes, P. Structure and Performance of Cements, 2nd ed.; Spon Press: London, UK; New York, NY, USA, 2002; 565p. [Google Scholar]
- Bullard, J.W.; Jennings, M.; Livingston, R.A.; Nonat, A.; Sherer, G.W.; Schweizer, J.S.; Scrivener, K.; Thomas, J. Mechanisms of cement hydration. Cem. Concr. Res. 2011, 41, 1208–1223. [Google Scholar] [CrossRef]
- Breval, E. Hydration products of tricalcium aluminate. Scand. J. Metall. 1977, 6, 21. [Google Scholar]
- Black, L.; Breen, C.; Yarwood, J.; Deng, C.S.; Phipps, J.; Maitland, G. Hydration of tricalcium aluminate (c(3)a) in the presence and absence of gypsum—Studied by raman spectroscopy and X-ray diffraction. J. Mater. Chem. 2006, 16, 1263–1272. [Google Scholar] [CrossRef]
- Lea, F.M.; Hewlett, P.C. Lea’s Chemistry of Cement and Concrete, 4th ed.; Arnold; Copublished in North, Central, and South America; J. Wiley: London, UK; New York, NY, USA, 1998; 1053p. [Google Scholar]
- Aïtcin, P.-C.; Flatt, R.J. Science and Technology of Concrete Admixtures, 1st ed.; Elsevier Science & Technology: Cambridge, UK, 2015. [Google Scholar]
- Taylor, H.F.W. Cement Chemistry, 2nd ed.; T. Telford: London, UK, 1997; 459p. [Google Scholar]
- Collepardi, M.; Baldini, G.; Pauri, M.; Corradi, M. Tricalcium aluminate hydration in presence of lime, gypsum or sodium sulfate. Cem. Concr. Res. 1978, 8, 571–580. [Google Scholar] [CrossRef]
- Minard, H.; Garrault, S.; Regnaud, L.; Nonat, A. Mechanisms and parameters controlling the tricalcium aluminate reactivity in the presence of gypsum. Cem. Concr. Res. 2007, 37, 1418–1426. [Google Scholar] [CrossRef]
- Liu, X.; Feng, P.; Lyu, C.; Ye, S.X. The role of sulfate ions in tricalcium aluminate hydration: New insights. Cem. Concr. Res. 2020, 130, 105973. [Google Scholar] [CrossRef]
- Corti, H.R.; Fernandezprini, R. Thermodynamics of solution of gypsum and anhydrite in water over a wide temperature-range. Can. J. Chem. 1984, 62, 484–488. [Google Scholar] [CrossRef]
- Marliacy, P.; Bourdet, J.B.; Schuffenecker, L.; Solimando, R. Dissolution enthalpy of anhydrous sodium sulfate in water. Experimental measurements and treatment with the electrolyte-nrtl model. J. Chem. Thermodyn. 2002, 34, 579–591. [Google Scholar] [CrossRef]
- Thomas, J.J.; Jennings, H.M.; Chen, J.J. Influence of nucleation seeding on the hydration mechanisms of tricalcium silicate and cement. J. Phys. Chem. C 2009, 113, 4327–4334. [Google Scholar] [CrossRef] [Green Version]
- Mehta, P.K.; Monteiro, P.J.M. Concrete: Microstructure, Properties, and Materials, 3rd ed.; McGraw-Hill: New York, NY, USA, 2006; 659p. [Google Scholar]
- Tong, H.S.; Young, J.F. Composition of solutions in contact with hydrating beta-dicalcium silicate. J. Am. Ceram. Soc. 1977, 60, 321–323. [Google Scholar] [CrossRef]
- Scrivener, K.L.; Juilland, P.; Monteiro, P.J.M. Advances in understanding hydration of portland cement. Cem. Concr. Res. 2015, 78, 38–56. [Google Scholar] [CrossRef]
- Gong, Y.F.; Liu, C.; Chen, Y.L. Properties and mechanism of hydration of fly ash belite cement prepared from low-quality fly ash. Appl. Sci. 2020, 10, 7026. [Google Scholar] [CrossRef]
- Borstnar, M.; Daneu, N.; Dolenec, S. Phase development and hydration kinetics of belite-calcium sulfoaluminate cements at different curing temperatures. Ceram. Int. 2020, 46, 29421–29428. [Google Scholar] [CrossRef]
- Donatello, S.; Fernandez-Jimenez, A.; Palomo, A. Very high volume fly ash cements. Early age hydration study using na2so4 as an activator. J. Am. Ceram. Soc. 2013, 96, 900–906. [Google Scholar] [CrossRef]
- Bohac, M.; Palou, M.; Novotny, R.; Masilko, J.; Soukal, F.; Opravil, T. Influence of temperature on early hydration of portland cement-metakaolin-slag system. J. Therm. Anal. Calorim. 2017, 127, 309–318. [Google Scholar] [CrossRef]
- Oliveira, A.D.; Dweck, J.; Fairbairn, E.D.R.; Gomes, O.D.M.; Toledo, R.D.T. Crystalline admixture effects on crystal formation phenomena during cement pastes’ hydration. J. Therm. Anal. Calorim. 2020, 139, 3361–3375. [Google Scholar] [CrossRef]
- Kurdowski, W. Cement and Concrete Chemistry; Springer: Dordrecht, The Netherlands; New York, NY, USA, 2014; 700p. [Google Scholar]



| Raw Mix | Chemical Composition (wt. %) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| CaO | SiO2 | Al2O3 | Fe2O3 | Na2O | K2O | TiO2 | MgO | SO3 | |
| 69.2 | 19.5 | 5.7 | 2.4 | 0.3 | 1.6 | 0.3 | 0.7 | 0.3 | |
| Constituents (wt. %) | |||||
|---|---|---|---|---|---|
| Belite Clinker | Water | CaSO4·2H2O | Na2SO4 | Na2CO3 | |
| BC | 66.6 | 33.3 | - | - | - |
| BC-CS | 64.5 | 32.3 | 3.2 | - | - |
| BC-NS | 64.9 | 32.4 | - | 2.7 | - |
| BC-NC | 65.3 | 32.7 | - | - | 2.0 |
| Weight Loss (wt. %) | |||||
|---|---|---|---|---|---|
| C–S–H | Portlandite | Carbonates | Total wt. Loss | ||
| Sample | Hydration Time | 0–400 °C | 400–500 °C | 500–1000 °C | 0–1000 °C |
| BC | 0.5 | 1.325 | 0.150 | 0.688 | 2.21 |
| 9.5 | 1.961 | 0.357 | 0.839 | 3.20 | |
| 24.0 | 3.190 | 0.647 | 1.128 | 5.02 | |
| 48.0 | 4.089 | 0.754 | 1.544 | 6.43 | |
| BC-CS | 0.5 | 1.63 | 0.106 | 0.272 | 2.06 |
| 9.5 | 3.14 | 0.378 | 0.565 | 4.12 | |
| 14.8 | 4.091 | 0.528 | 0.798 | 5.47 | |
| 24.0 | 4.904 | 0.688 | 0.716 | 6.36 | |
| 48.0 | 6.683 | 0.782 | 1.256 | 8.78 | |
| BC-NC | 0.5 | 1.149 | 0.14 | 1.695 | 3.02 |
| 1.0 | 1.271 | 0.152 | 1.702 | 3.16 | |
| 6.5 | 4.62 | 0.275 | 2.518 | 7.52 | |
| 24.0 | 5.686 | 0.442 | 2.264 | 8.51 | |
| 48.0 | 6.612 | 0.694 | 2.859 | 10.23 | |
| BC-NS | 0.5 | 1.166 | 0.037 | 0.58 | 1.81 |
| 4.0 | 1.424 | 0.078 | 0.716 | 2.26 | |
| 5.5 | 2.358 | 0.207 | 1.144 | 3.77 | |
| 24.0 | 5.309 | 0.377 | 1.788 | 7.56 | |
| 48.0 | 8.028 | 0.588 | 1.991 | 10.68 | |
| Element (at. %) | C | O | Na | Al | Si | S | Ca | |
|---|---|---|---|---|---|---|---|---|
| Point | ||||||||
| #1 | 6.57 | 62.90 | - | 4.37 | 6.42 | - | 19.74 | |
| #2 | 6.62 | 62.64 | - | 0.64 | 10.02 | 0.26 | 19.82 | |
| #3 | 5.95 | 75.64 | - | 4.86 | 1.17 | 2.06 | 10.32 | |
| #4 | 5.46 | 58.44 | 3.00 | 2.45 | 7.22 | 3.14 | 20.29 | |
| #5 | 8.42 | 66.29 | 1.59 | 4.36 | 3.91 | - | 14.79 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iliushchenko, V.; Kalina, L.; Sedlačík, M.; Cába, V.; Másilko, J.; Novotný, R. Effect of Alkali Salts on the Hydration Process of Belite Clinker. Materials 2022, 15, 3424. https://doi.org/10.3390/ma15103424
Iliushchenko V, Kalina L, Sedlačík M, Cába V, Másilko J, Novotný R. Effect of Alkali Salts on the Hydration Process of Belite Clinker. Materials. 2022; 15(10):3424. https://doi.org/10.3390/ma15103424
Chicago/Turabian StyleIliushchenko, Valeriia, Lukáš Kalina, Martin Sedlačík, Vladislav Cába, Jiří Másilko, and Radoslav Novotný. 2022. "Effect of Alkali Salts on the Hydration Process of Belite Clinker" Materials 15, no. 10: 3424. https://doi.org/10.3390/ma15103424
APA StyleIliushchenko, V., Kalina, L., Sedlačík, M., Cába, V., Másilko, J., & Novotný, R. (2022). Effect of Alkali Salts on the Hydration Process of Belite Clinker. Materials, 15(10), 3424. https://doi.org/10.3390/ma15103424






